Submitted:
04 March 2026
Posted:
04 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
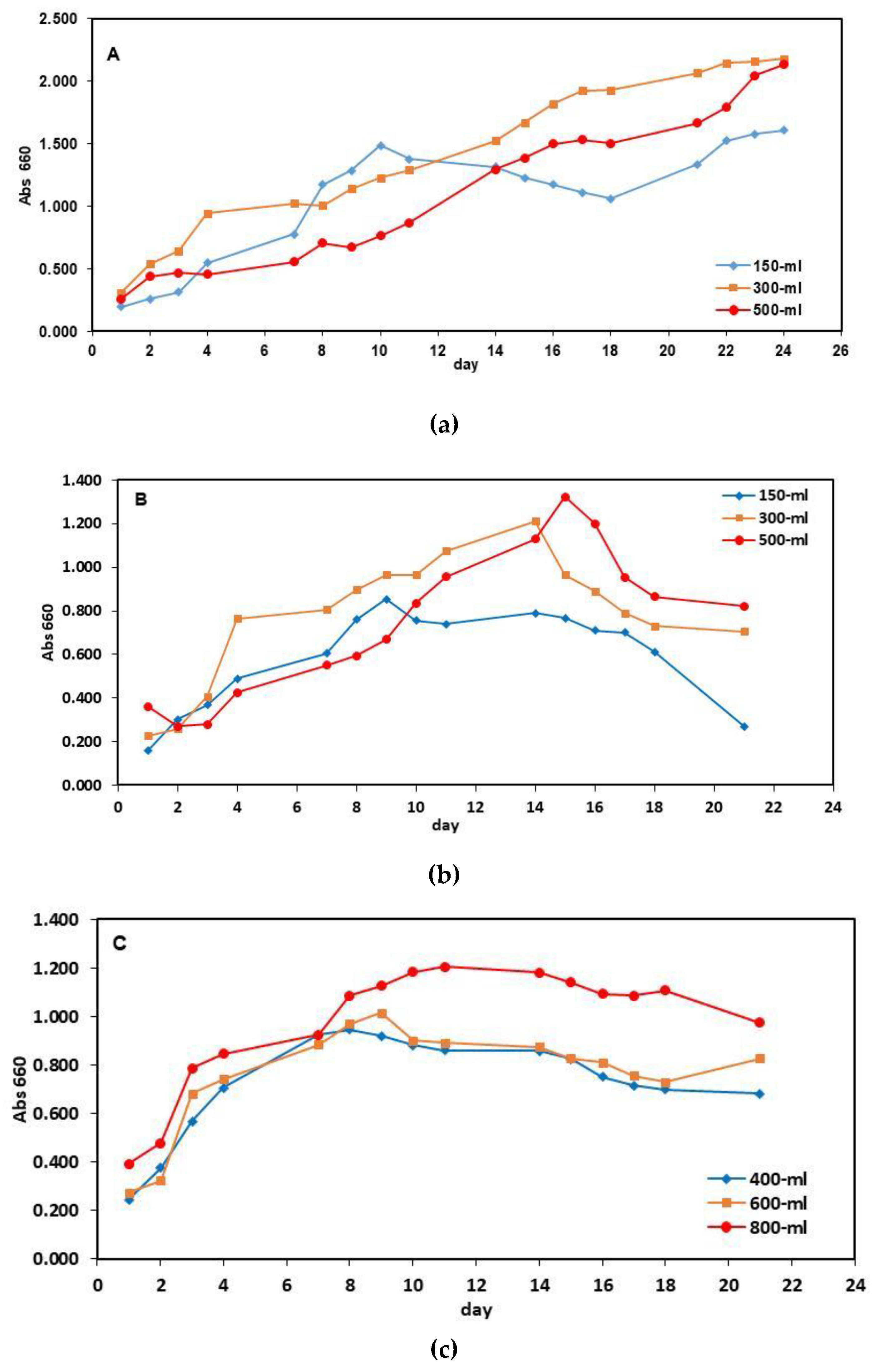
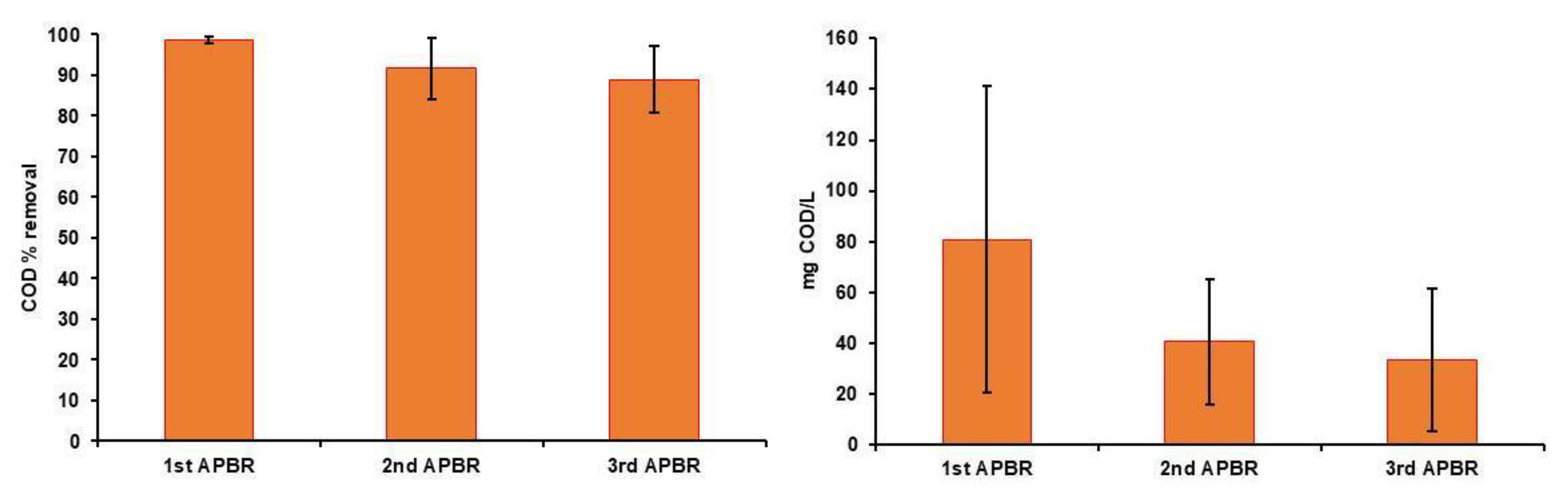
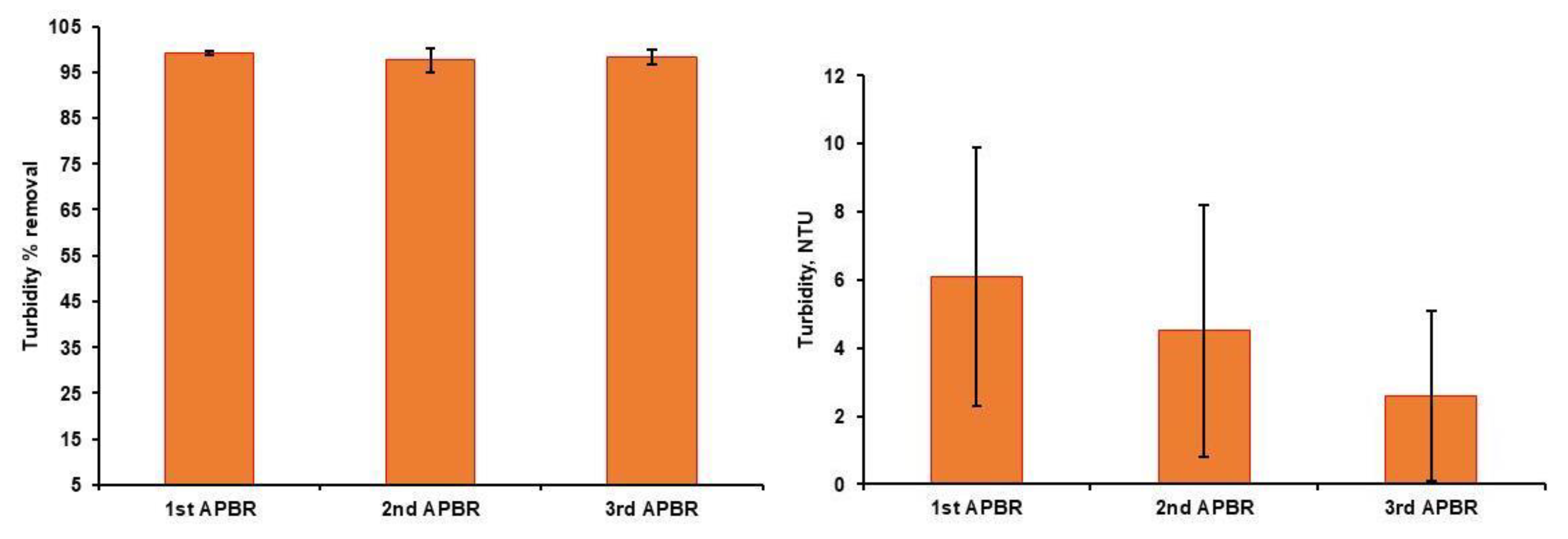
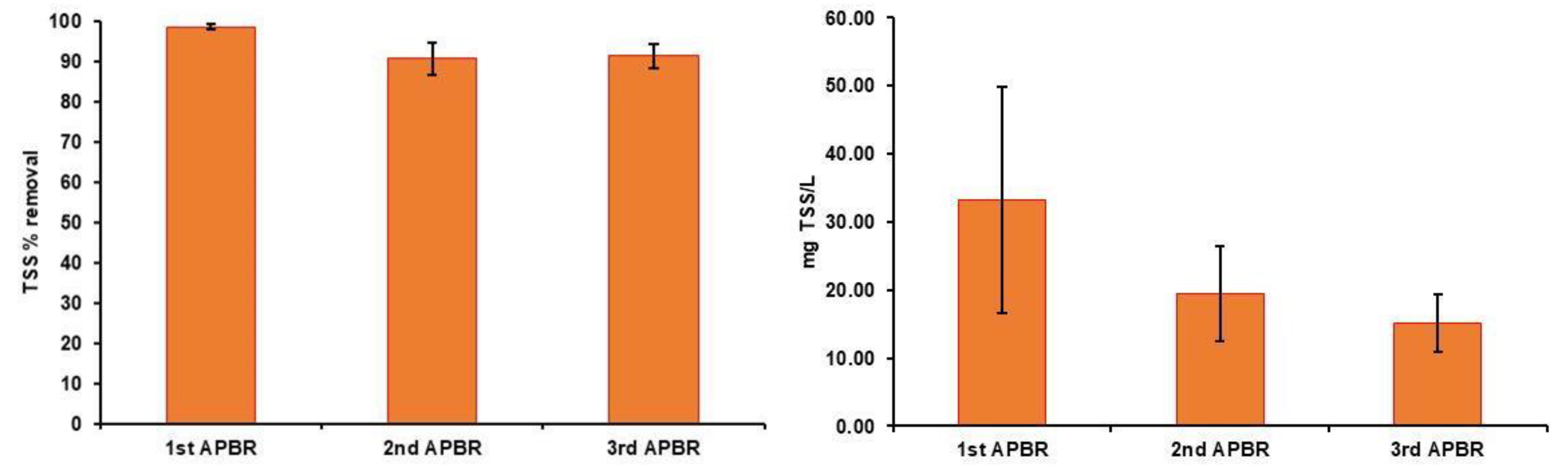
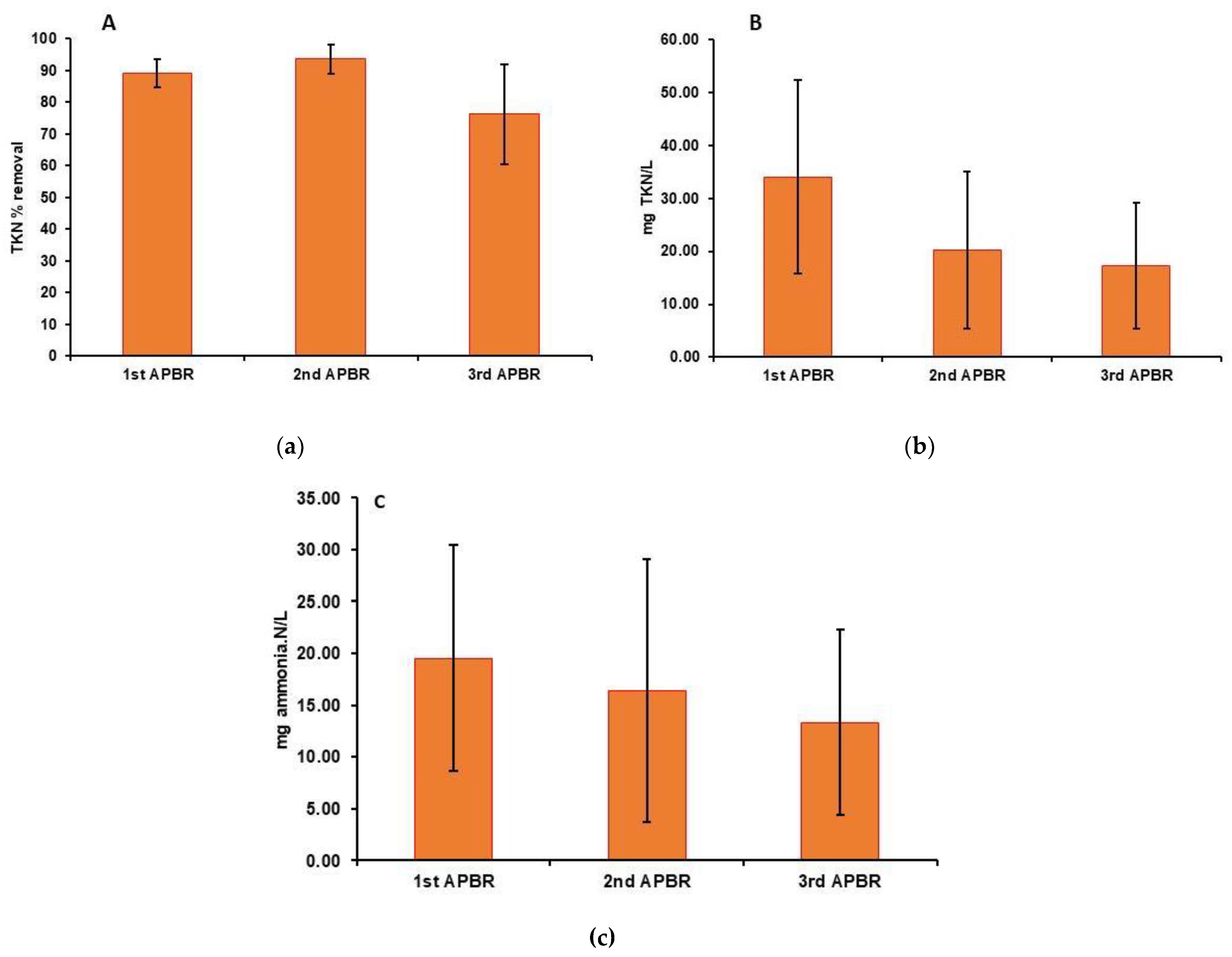
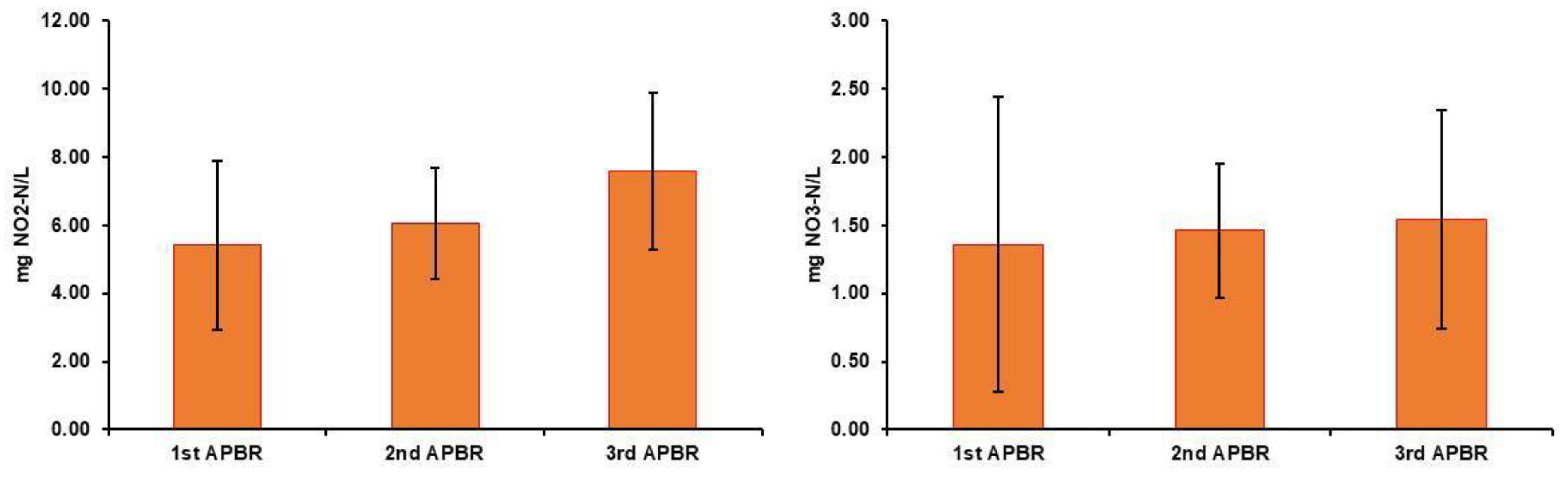
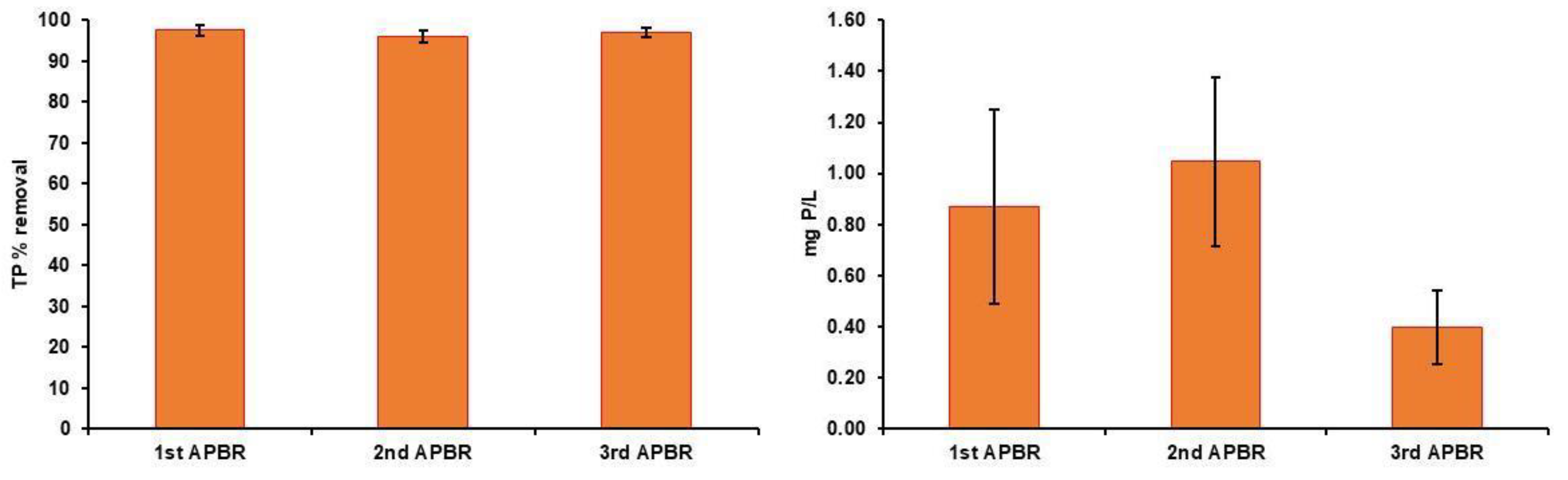
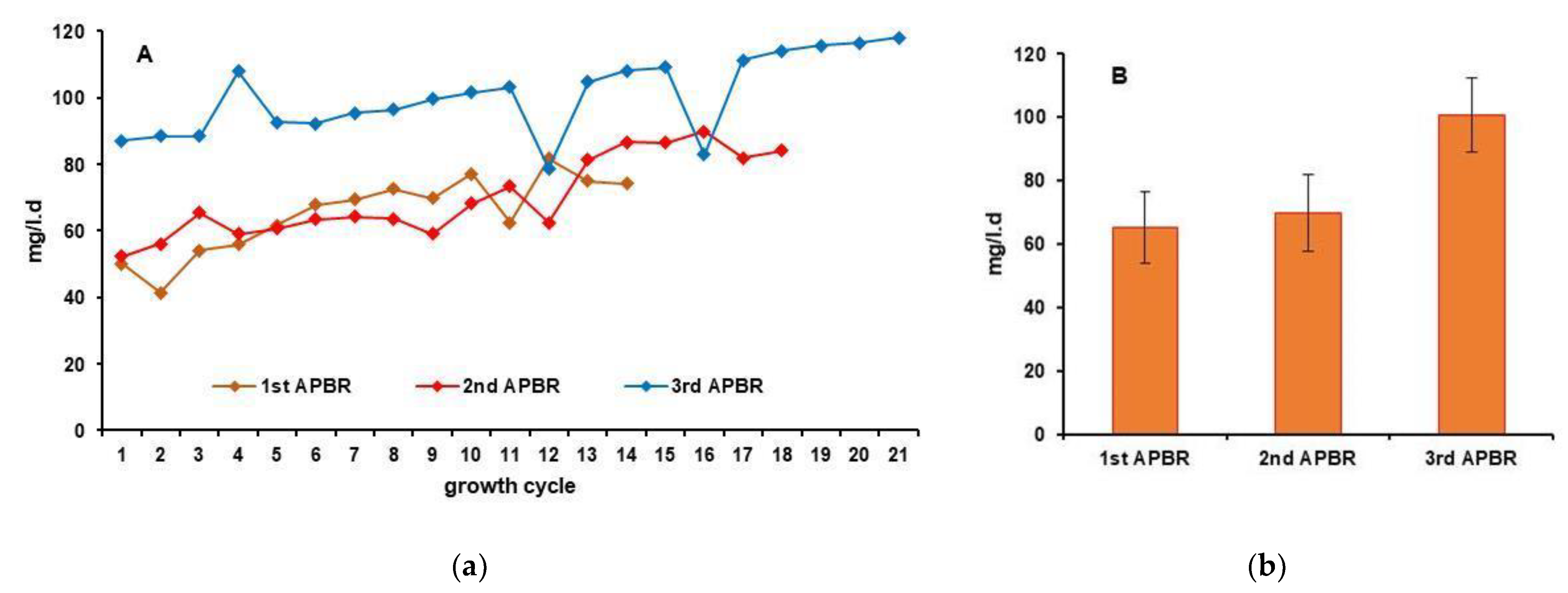
3. Results
3.1. Characteristics of Raw Wastewaters
4. Conclusions
- The APBR has the potential to treat PCP wastewater with different qualities
- Characteristics of PCP wastewater influences quantity and quality of algal biomass.
- Nutrients recovery by algal biomass is the main route of nutrients removal in the APBR
- Non-mediate algal nitrogen removal depends ammonia/TKN ratio of the feed
- Algal biomass from APBR treating PCP wastewater has substantial amount of protein
- Acknowledgement: The author would like to present their sincere thanks to the STDF for funding this study. Also, the authors extend their sincere thanks to chemist Ahmed A. Nasr for supporting sampling program during this study.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nagarajan, D.; Kusmayadi, A.; Yen, H.-W.; Dong, C.-D.; Lee, D.-J.; Chang, J.-S. Current advances in biological swine wastewater treatment using microalgae-based processes. Bioresour. Technol. 2019, vol. 289, 121718. [Google Scholar] [CrossRef]
- Meneses-Jácome; Diaz-Chavez, R.; Velásquez-Arredondo, H. I.; Cárdenas-Chávez, D. L.; Parra, R.; Ruiz-Colorado, A. A. Sustainable Energy from agro-industrial wastewaters in Latin-America. Renew. Sustain. Energy Rev. 2016, vol. 56, 1249–1262. [Google Scholar] [CrossRef]
- Yaashikaa, P. R.; Kumar, P. S.; Varjani, S. Valorization of agro-industrial wastes for biorefinery process and circular bioeconomy: A critical review. Bioresour. Technol. 2022, vol. 343, 126126. [Google Scholar] [CrossRef] [PubMed]
- Qin, S.; Wang, K.; Gao, F.; Ge, B.; Cui, H.; Li, W. Biotechnologies for bulk production of microalgal biomass: from mass cultivation to dried biomass acquisition. Biotechnol. biofuels Bioprod. 2023, vol. 16(no. 1), 131. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Pawar, S. B. An integrated approach for microalgae cultivation using raw and anaerobic digested wastewaters from food processing industry. Bioresour. Technol. 2018, vol. 269, 571–576. [Google Scholar] [CrossRef]
- Qin, L. , Cultivation of Chlorella vulgaris in Dairy Wastewater Pretreated by UV Irradiation and Sodium Hypochlorite. Appl. Biochem. Biotechnol. 2014, vol. 172(no. 2), 1121–1130. [Google Scholar] [CrossRef]
- Lu, W.; Wang, Z.; Wang, X.; Yuan, Z. Cultivation of Chlorella sp. using raw dairy wastewater for nutrient removal and biodiesel production: Characteristics comparison of indoor bench-scale and outdoor pilot-scale cultures. Bioresour. Technol. 2015, vol. 192, 382–388. [Google Scholar] [CrossRef]
- Gupta, S.; Pawar, S. B.; Pandey, R. A. Current practices and challenges in using microalgae for treatment of nutrient rich wastewater from agro-based industries. Sci. Total Environ. 2019, vol. 687, 1107–1126. [Google Scholar] [CrossRef]
- Roostaei, J.; Zhang, Y.; Gopalakrishnan, K.; Ochocki, A. J. Mixotrophic Microalgae Biofilm: A Novel Algae Cultivation Strategy for Improved Productivity and Cost-efficiency of Biofuel Feedstock Production. Sci. Rep. 2018, vol. 8(no. 1), 12528. [Google Scholar] [CrossRef]
- Passos, F.; Uggetti, E.; Carrère, H.; Ferrer, I. Pretreatment of microalgae to improve biogas production: A review. Bioresour. Technol. 2014, vol. 172, 403–412. [Google Scholar] [CrossRef]
- Xu, Z. Development of integrated culture systems and harvesting methods for improved algal biomass productivity and wastewater resource recovery–a review. Sci. Total Environ. 2020, vol. 746, 141039. [Google Scholar]
- Ray; Nayak, M.; Ghosh, A. A review on co-culturing of microalgae: A greener strategy towards sustainable biofuels production. Sci. Total Environ. 2022, vol. 802, 149765. [Google Scholar] [CrossRef]
- Pandey, S.; Narayanan, I.; Vinayagam, R.; Selvaraj, R.; Varadavenkatesan, T.; Pugazhendhi, A. A review on the effect of blue green 11 medium and its constituents on microalgal growth and lipid production. J. Environ. Chem. Eng. 2023, vol. 11(no. 3), 109984. [Google Scholar] [CrossRef]
- Pleissner, D.; Smetana, S. Estimation of the economy of heterotrophic microalgae- and insect-based food waste utilization processes. Waste Manag. 2020, vol. 102, 198–203. [Google Scholar] [CrossRef] [PubMed]
- Spiller, M.; Muys, M.; Papini, G.; Sakarika, M.; Buyle, M.; Vlaeminck, S. E. Environmental impact of microbial protein from potato wastewater as feed ingredient: Comparative consequential life cycle assessment of three production systems and soybean meal. Water Res. 2020, vol. 171, 115406. [Google Scholar] [CrossRef] [PubMed]
- Kalra, R.; Gaur, S.; Goel, M. Microalgae bioremediation: A perspective towards wastewater treatment along with industrial carotenoids production. J. Water Process Eng. 2021, vol. 40, 101794. [Google Scholar] [CrossRef]
- Castillo, T.; Ramos, D.; García-Beltrán, T.; Brito-Bazan, M.; Galindo, E. Mixotrophic cultivation of microalgae: An alternative to produce high-value metabolites. Biochem. Eng. J. 2021, vol. 176, 108183. [Google Scholar]
- Wang, Y. Perspectives on the feasibility of using microalgae for industrial wastewater treatment. 2016. [Google Scholar] [CrossRef]
- Norsker, N.-H.; Barbosa, M. J.; Vermuë, M. H.; Wijffels, R. H. Microalgal production — A close look at the economics. Biotechnol. Adv. 2011, vol. 29(no. 1), 24–27. [Google Scholar] [CrossRef]
- Acién, F. G.; Gómez-Serrano, C.; Morales-Amaral, M. del M.; Fernández-Sevilla, J. M.; Molina-Grima, E. Wastewater treatment using microalgae: how realistic a contribution might it be to significant urban wastewater treatment? Appl. Microbiol. Biotechnol. 2016, vol. 100(no. 21), 9013–9022. [Google Scholar] [CrossRef]
- Oswald, W. J. My sixty years in applied algology. J. Appl. Phycol. 2003, vol. 15(no. 2), 99–106. [Google Scholar] [CrossRef]
- Singh, S. P.; Singh, P. Effect of temperature and light on the growth of algae species: A review. Renew. Sustain. energy Rev. 2015, vol. 50, 431–444. [Google Scholar] [CrossRef]
- Mohsenpour, S. F.; Hennige, S.; Willoughby, N.; Adeloye, A.; Gutierrez, T. Integrating micro-algae into wastewater treatment: A review. Sci. Total Environ. 2021, vol. 752, 142168. [Google Scholar] [CrossRef] [PubMed]
- Almomani, F.; Judd, S.; Bhosale, R. R.; Shurair, M.; Aljaml, K.; Khraisheh, M. Intergraded wastewater treatment and carbon bio-fixation from flue gases using Spirulina platensis and mixed algal culture. Process Saf. Environ. Prot. 2019, vol. 124, 240–250. [Google Scholar] [CrossRef]
- Molazadeh, M.; Ahmadzadeh, H.; Pourianfar, H. R.; Lyon, S.; Rampelotto, P. H. The use of microalgae for coupling wastewater treatment with CO2 biofixation. Front. Bioeng. Biotechnol. 2019, vol. 7, 42. [Google Scholar] [CrossRef]
- Maurya, R. , Advances in microalgal research for valorization of industrial wastewater. Bioresour. Technol. 2022, vol. 343, 126128. [Google Scholar] [CrossRef]
- Proietti Tocca, G.; Agostino, V.; Menin, B.; Tommasi, T.; Fino, D.; Di Caprio, F. Mixotrophic and heterotrophic growth of microalgae using acetate from different production processes. Rev. Environ. Sci. Bio/Technology 2024, vol. 23(no. 1), 93–132. [Google Scholar] [CrossRef]
- Calderini, M. L.; Salmi, P.; Rigaud, C.; Peltomaa, E.; Taipale, S. J. Metabolic plasticity of mixotrophic algae is key for their persistence in browning environments. Mol. Ecol. 2022, vol. 31(no. 18), 4726–4738. [Google Scholar] [CrossRef]
- El Semary, N.; Abd El-Sattar, A. M.; Ahmed, E. Z.; Aldayel, M. Mixotrophy of algae: more algal biomass and more biofertilization for plants. Sustainability 2023, vol. 15(no. 7), 5815. [Google Scholar]
- Hernández, D.; Riaño, B.; Coca, M.; García-González, M. C. Treatment of agro-industrial wastewater using microalgae–bacteria consortium combined with anaerobic digestion of the produced biomass. Bioresour. Technol. 2013, vol. 135, 598–603. [Google Scholar] [CrossRef]
- Posadas, E.; Bochon, S.; Coca, M.; García-González, M. C.; García-Encina, P. A.; Muñoz, R. Microalgae-based agro-industrial wastewater treatment: a preliminary screening of biodegradability. J. Appl. Phycol. 2014, vol. 26(no. 6), 2335–2345. [Google Scholar] [CrossRef]
- Khoo, K. S.; Chia, W. Y.; Chew, K. W.; Show, P. L. Microalgal-bacterial consortia as future prospect in wastewater bioremediation, environmental management and bioenergy production. Indian J. Microbiol. 2021, vol. 61(no. 3), 262–269. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Li, X.; Yu, J.; Wang, Q. Increased hydrogen production in co-culture of Chlamydomonas reinhardtii and Bradyrhizobium japonicum. Bioresour. Technol. 2012, vol. 123, 184–188. [Google Scholar] [CrossRef] [PubMed]
- Do Nascimento, M.; Dublan, M. de los A.; Ortiz-Marquez, J. C. F.; Curatti, L. High lipid productivity of an Ankistrodesmus – Rhizobium artificial consortium. Bioresour. Technol. 2013, vol. 146, 400–407. [Google Scholar] [CrossRef]
- Feng, S. , Performance of a microalgal-bacterial consortium system for the treatment of dairy-derived liquid digestate and biomass production. Bioresour. Technol. 2020, vol. 306, 123101. [Google Scholar] [CrossRef]
- Zhou, X. Enhancement of productivity of Chlorella pyrenoidosa lipids for biodiesel using co-culture with ammonia-oxidizing bacteria in municipal wastewater. Renew. Energy 2020, vol. 151, 598–603. [Google Scholar] [CrossRef]
- Wang, L. Interaction of Chlorella vulgaris and bacteria when co-cultivated in anaerobically digested swine manure. Bioresour. Technol. 2021, vol. 320, 124250. [Google Scholar] [CrossRef]
- Stephenson, L.; Kazamia, E.; Dennis, J. S.; Howe, C. J.; Scott, S. A.; Smith, A. G. Life-cycle assessment of potential algal biodiesel production in the United Kingdom: a comparison of raceways and air-lift tubular bioreactors. Energy & Fuels 2010, vol. 24(no. 7), 4062–4077. [Google Scholar]
- Jorquera; Kiperstok, A.; Sales, E. A.; Embiruçu, M.; Ghirardi, M. L. Comparative energy life-cycle analyses of microalgal biomass production in open ponds and photobioreactors. Bioresour. Technol. 2010, vol. 101(no. 4), 1406–1413. [Google Scholar] [CrossRef]
- Kohlheb, N. Assessing the life-cycle sustainability of algae and bacteria-based wastewater treatment systems: High-rate algae pond and sequencing batch reactor. J. Environ. Manage. 2020, vol. 264, 110459. [Google Scholar]
- Scott, S. A. Biodiesel from algae: challenges and prospects. Curr. Opin. Biotechnol. 2010, vol. 21(no. 3), 277–286. [Google Scholar] [CrossRef]
- Carvalho, P.; Meireles, L. A.; Malcata, F. X. Microalgal reactors: a review of enclosed system designs and performances. Biotechnol. Prog. 2006, vol. 22(no. 6), 1490–1506. [Google Scholar] [CrossRef]
- Kadir, W. N. A.; Lam, M. K.; Uemura, Y.; Lim, J. W.; Lee, K. T. Harvesting and pre-treatment of microalgae cultivated in wastewater for biodiesel production: a review. Energy Convers. Manag. 2018, vol. 171, 1416–1429. [Google Scholar] [CrossRef]
- Barros; Gonçalves, A. L.; Simões, M.; Pires, J. C. M. Harvesting techniques applied to microalgae: a review. Renew. Sustain. energy Rev. 2015, vol. 41, 1489–1500. [Google Scholar] [CrossRef]
- Ruiz-Marin; Mendoza-Espinosa, L. G.; Stephenson, T. Growth and nutrient removal in free and immobilized green algae in batch and semi-continuous cultures treating real wastewater. Bioresour. Technol. 2010, vol. 101(no. 1), 58–64. [Google Scholar] [CrossRef]
- Patsialou, S.; Politou, E.; Nousis, S.; Liakopoulou, P.; Vayenas, D. V; Tekerlekopoulou, A. G. Hybrid treatment of confectionery wastewater using a biofilter and a cyanobacteria-based system with simultaneous valuable metabolic compounds production. Algal Res. 2024, vol. 79, 103483. [Google Scholar] [CrossRef]
- El-Shafai, S. A.; El-Gohary, F. A.; Naser, F. A.; Van der Steen, P.; Gijzen, H. J. Nitrogen recovery in an integrated system for wastewater treatment and tilapia production. Environmentalist 2007, vol. 27(no. 2), 287–302. [Google Scholar] [CrossRef]
- Al-Mallahi, J.; Ishii, K. Attempts to alleviate inhibitory factors of anaerobic digestate for enhanced microalgae cultivation and nutrients removal: A review. J. Environ. Manage. 2022, vol. 304, 114266. [Google Scholar] [CrossRef]
- Mujtaba, G.; Lee, K. Treatment of real wastewater using co-culture of immobilized Chlorella vulgaris and suspended activated sludge. Water Res. 2017, vol. 120, 174–184. [Google Scholar] [CrossRef]
- Michael; del Ninno, M.; Gross, M.; Wen, Z. Use of wavelength-selective optical light filters for enhanced microalgal growth in different algal cultivation systems. Bioresour. Technol. 2015, vol. 179, 473–482. [Google Scholar] [CrossRef]
- Jiang, L.-X.; Cui, Y.-W.; Mi, Y.-N.; Zhou, D.-X.; Li, M.-T.; Yang, R.-C. Recovery of volatile ethanol gas via microalgal-bacterial consortium: Ethanol-to-acetate conversion pathway boosts lipid production. J. Environ. Manage. 2025, vol. 381, 125210. [Google Scholar] [CrossRef]
- Fischer, R.; Andersen, T.; Hillebrand, H.; Ptacnik, R. The exponentially fed batch culture as a reliable alternative to conventional chemostats. Limnol. Oceanogr. Methods 2014, vol. 12(no. 7), 432–440. [Google Scholar] [CrossRef]
- APHA, Standard Methods for the Examination of Water and Wastewater, 24th ed.; American Public Health Association, American Water Works Association, Water Environment Federation, 2023.
- Xia; Murphy, J. D. Microalgal Cultivation in Treating Liquid Digestate from Biogas Systems. Trends Biotechnol. 2016, vol. 34(no. 4), 264–275. [Google Scholar] [CrossRef]
- Cheng, P. Tribonema sp. and Chlorella zofingiensis co-culture to treat swine wastewater diluted with fishery wastewater to facilitate harvest. Bioresour. Technol. 2020, vol. 297, 122516. [Google Scholar] [CrossRef]
- Uggetti, E.; Sialve, B.; Latrille, E.; Steyer, J.-P. Anaerobic digestate as substrate for microalgae culture: The role of ammonium concentration on the microalgae productivity. Bioresour. Technol. 2014, vol. 152, 437–443. [Google Scholar] [CrossRef]
- Park, J.; Jin, H.-F.; Lim, B.-R.; Park, K.-Y.; Lee, K. Ammonia removal from anaerobic digestion effluent of livestock waste using green alga Scenedesmus sp. Bioresour. Technol. 2010, vol. 101(no. 22), 8649–8657. [Google Scholar] [CrossRef]
- Praveen, P.; Guo, Y.; Kang, H.; Lefebvre, C.; Loh, K.-C. Enhancing microalgae cultivation in anaerobic digestate through nitrification. Chem. Eng. J. 2018, vol. 354, 905–912. [Google Scholar] [CrossRef]
- Li, J.; Wang, L.; Lu, Q.; Zhou, W. Toxicity alleviation for microalgae cultivation by cationic starch addition and ammonia stripping and study on the cost assessment. RSC Adv. 2019, vol. 9(no. 65), 38235–38245. [Google Scholar] [CrossRef]
- Wang, Q.; Hyman, M.; Higgins, B. T. Factors impacting the effectiveness of biological pretreatment for the alleviation of algal growth inhibition on anaerobic digestate. Algal Res. 2021, vol. 53, 102129. [Google Scholar] [CrossRef]
- Wang, Q.; Prasad, R.; Higgins, B. T. Aerobic bacterial pretreatment to overcome algal growth inhibition on high-strength anaerobic digestates. Water Res. 2019, vol. 162, 420–426. [Google Scholar] [CrossRef]
- Zahid, W. M.; El-Shafai, S. A. Impacts of alum addition on the treatment efficiency of cloth-media MBR. Desalination 2012, vol. 301, 53–58. [Google Scholar] [CrossRef]
- AlMomani, F. A.; Örmeci, B. Performance of Chlorella vulgaris, Neochloris oleoabundans, and mixed indigenous microalgae for treatment of primary effluent, secondary effluent and centrate. Ecol. Eng. 2016, vol. 95, 280–289. [Google Scholar] [CrossRef]
- Woertz; Feffer, A.; Lundquist, T.; Nelson, Y. Algae grown on dairy and municipal wastewater for simultaneous nutrient removal and lipid production for biofuel feedstock. J. Environ. Eng. 2009, vol. 135(no. 11), 1115–1122. [Google Scholar] [CrossRef]
- Li, Y.; Horsman, M.; Wang, B.; Wu, N.; Lan, C. Q. Effects of nitrogen sources on cell growth and lipid accumulation of green alga Neochloris oleoabundans. Appl. Microbiol. Biotechnol. 2008, vol. 81(no. 4), 629–636. [Google Scholar] [CrossRef]
- El-Shafai, S. A.; Zahid, W. M. Performance of aerated submerged biofilm reactor packed with local scoria for carbon and nitrogen removal from municipal wastewater. Bioresour. Technol. 2013, vol. 143, 476–482. [Google Scholar] [CrossRef]
- Zahid, W. M.; El-Qelish, M.; El-Shafai, S. A. Effect of operating regime on the performance of biofilm reactor treating municipal wastewater. Int. J. Environ. Sci. Technol. 2023, vol. 20(no. 8), 8799–8812. [Google Scholar] [CrossRef]
- Mayers, J.; Ekman Nilsson, A.; Svensson, E.; Albers, E. Integrating Microalgal Production with Industrial Outputs—Reducing Process Inputs and Quantifying the Benefits. Ind. Biotechnol. 2016, vol. 12(no. 4), 219–234. [Google Scholar] [CrossRef]
- Santiago, B.; Lovell, R. T. Amino Acid Requirements for Growth of Nile Tilapia. J. Nutr. 1988, vol. 118(no. 12), 1540–1546. [Google Scholar] [CrossRef]
- Furuya, W. M.; da Cruz, T. P.; Gatlin, D. M., III. Amino acid requirements for nile tilapia: An update. Animals 2023, vol. 13(no. 5), 900. [Google Scholar] [CrossRef]
- Choi, S.-H.; Kozukue, N.; Kim, H.-J.; Friedman, M. Analysis of protein amino acids, non-protein amino acids and metabolites, dietary protein, glucose, fructose, sucrose, phenolic, and flavonoid content and antioxidative properties of potato tubers, peels, and cortexes (pulps). J. Food Compos. Anal. 2016, vol. 50, 77–87. [Google Scholar] [CrossRef]
- Pęksa; Miedzianka, J.; Nemś, A. Amino acid composition of flesh-coloured potatoes as affected by storage conditions. Food Chem. 2018, vol. 266, 335–342. [Google Scholar] [CrossRef]
| Source of PCP wastewater | S effluent | UASB effluent | SBR effluent | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Feed volume, ml | 150 | 300 | 500 | 150 | 300 | 500 | 400 | 600 | 800 |
| Pre-enriched algal culture, ml | 200 | 200 | 200 | 200 | 200 | 200 | 200 | 200 | 200 |
| Demi-water, ml | 650 | 500 | 300 | 650 | 500 | 300 | 400 | 200 | 0 |
| Effective reactor volume, ml | 1000 | 1000 | 1000 | 1000 | 1000 | 1000 | 1000 | 1000 | 1000 |
| Parameter | Unit | After screening | After UASB | After SBR |
|---|---|---|---|---|
| pH | - | 5.3-6.3 | 8.0-8.4 | 7.6-8.3 |
| Turbidity | NTU | 743±96 | 201±23 | 172±18 |
| TSS | mg/L | 2424±215 | 225±36 | 180±18 |
| COD | mg O2/L | 6189±1113 | 544±178 | 292±31 |
| BOD | mg O2/L | 4300±841 | 272±77 | 158±16 |
| Ammonia. N | mg N/L | 102±21 | 231±18 | 13.5±3.1 |
| TKN | mg N/L | 302±53 | 313±12 | 72.5±5.0 |
| NO3 | mg N/l | Not detected | Not detected | 2.5±1.2 |
| Total P | mg P/L | 36.2±5.5 | 27.1±4.4 | 13.5±1.2 |
| Amino acid | 1st APBR | 2nd APBR | 3rd APBR | |||
|---|---|---|---|---|---|---|
| % of DM | % of Protein | % of DM | % of Protein | % of DM | % of Protein | |
| Aspartate | 3.27 | 10.66 | 3.50 | 10.76 | 3.69 | 10.36 |
| Glutamate | 3.86 | 12.59 | 4.58 | 14.08 | 5.21 | 14.61 |
| Serine | 1.49 | 4.87 | 1.66 | 5.10 | 1.74 | 4.88 |
| Histidine | 0.59 | 1.91 | 0.55 | 1.69 | 0.61 | 1.72 |
| Glycine | 2.08 | 6.79 | 2.22 | 6.84 | 2.34 | 6.56 |
| Threonine | 1.85 | 6.03 | 1.73 | 5.30 | 1.86 | 5.21 |
| Arginine | 1.64 | 5.35 | 2.33 | 7.15 | 2.67 | 7.50 |
| Alanine | 2.35 | 7.67 | 3.03 | 9.31 | 3.42 | 9.60 |
| Tyrosine | 2.17 | 7.10 | 1.26 | 3.86 | 1.53 | 4.28 |
| Valine | 1.78 | 5.82 | 1.87 | 5.74 | 2.15 | 6.02 |
| Methionine | 0.66 | 2.15 | 0.54 | 1.66 | 0.49 | 1.37 |
| Phenylalanine | 1.77 | 5.78 | 1.67 | 5.14 | 1.79 | 5.02 |
| Isoleucine | 1.53 | 5.01 | 1.58 | 4.86 | 1.70 | 4.77 |
| Leucine | 2.73 | 8.90 | 2.92 | 8.99 | 3.14 | 8.82 |
| Lysine | 1.57 | 5.14 | 1.57 | 4.83 | 1.83 | 5.12 |
| Proline | 1.30 | 4.24 | 1.54 | 4.72 | 1.49 | 4.17 |
| Total Protein | 30.64 | 100.00 | 32.53 | 100.00 | 35.65 | 100.00 |
| Item | 1st APBR | 2nd APBR | 3rd APBR |
|---|---|---|---|
| Number of batches | 14 | 18 | 21 |
| Feed volume | 150 | 150 | 500 |
| Number of days | 154 | 144 | 126 |
| TN loading rate, mg/L 1* | 634 | 845 | 852 |
| Average yield, mg/l.d | 65.25 | 69.91 | 100.62 |
| Total yield, mg/l | 10049 | 10067 | 12678 |
| Protein content, % | 30.64 | 32.53 | 35.65 |
| N recovery, mg/l | 492.6 | 524.0 | 723.2 |
| N recovery, % | 77.7 | 62.0 | 84.9 |
| Residual nitrogen, mg/l 2* | 34.1 | 20.2 | 17.3 |
| Residual nitrogen, % | 5.4 | 2.4 | 2.0 |
| N removal, % | 16.9 | 35.6 | 13.1 |
| Actual %N removal 3* | 94.6 | 97.6 | 98.0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





