Submitted:
02 March 2026
Posted:
04 March 2026
You are already at the latest version
Abstract
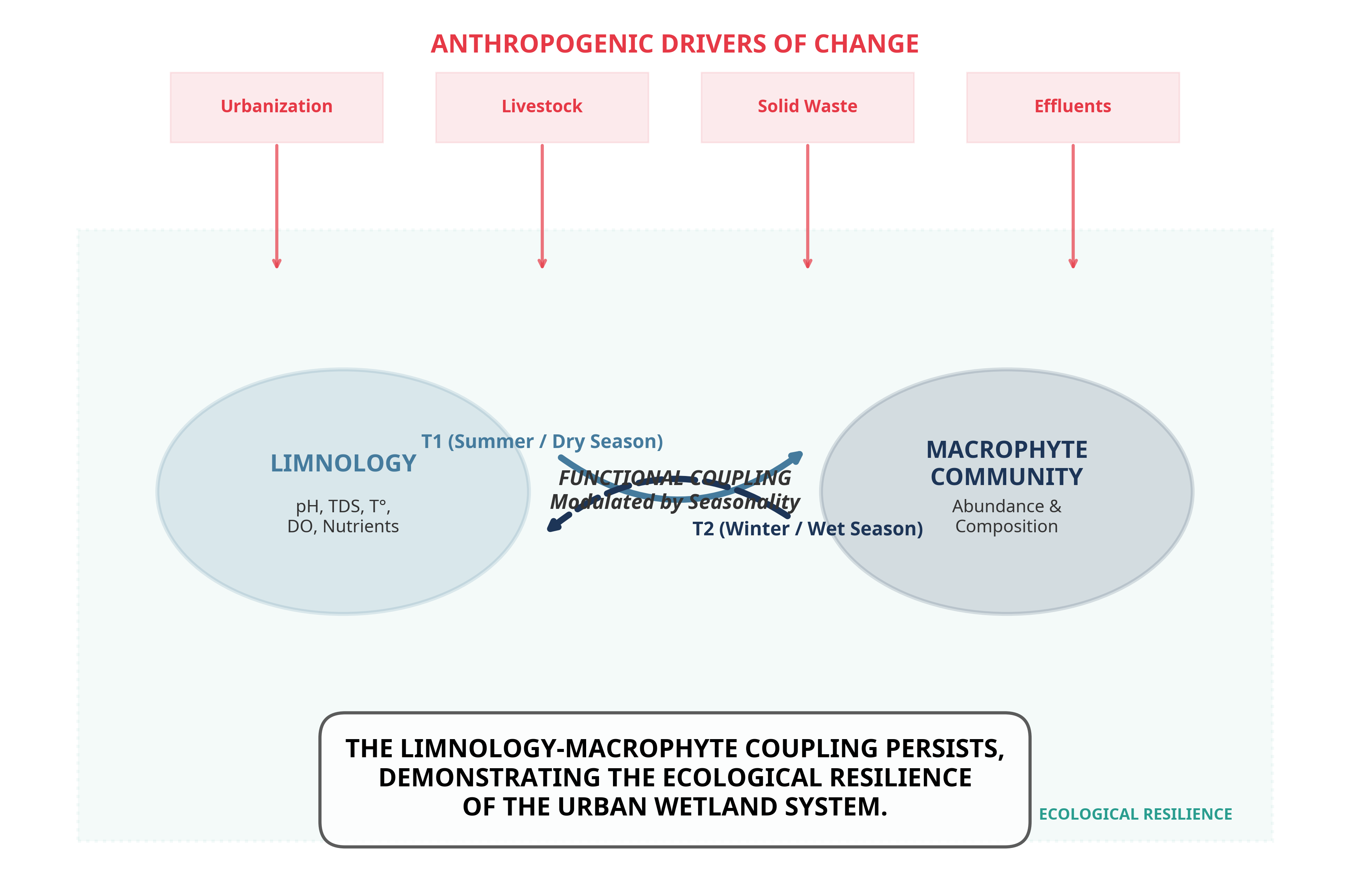
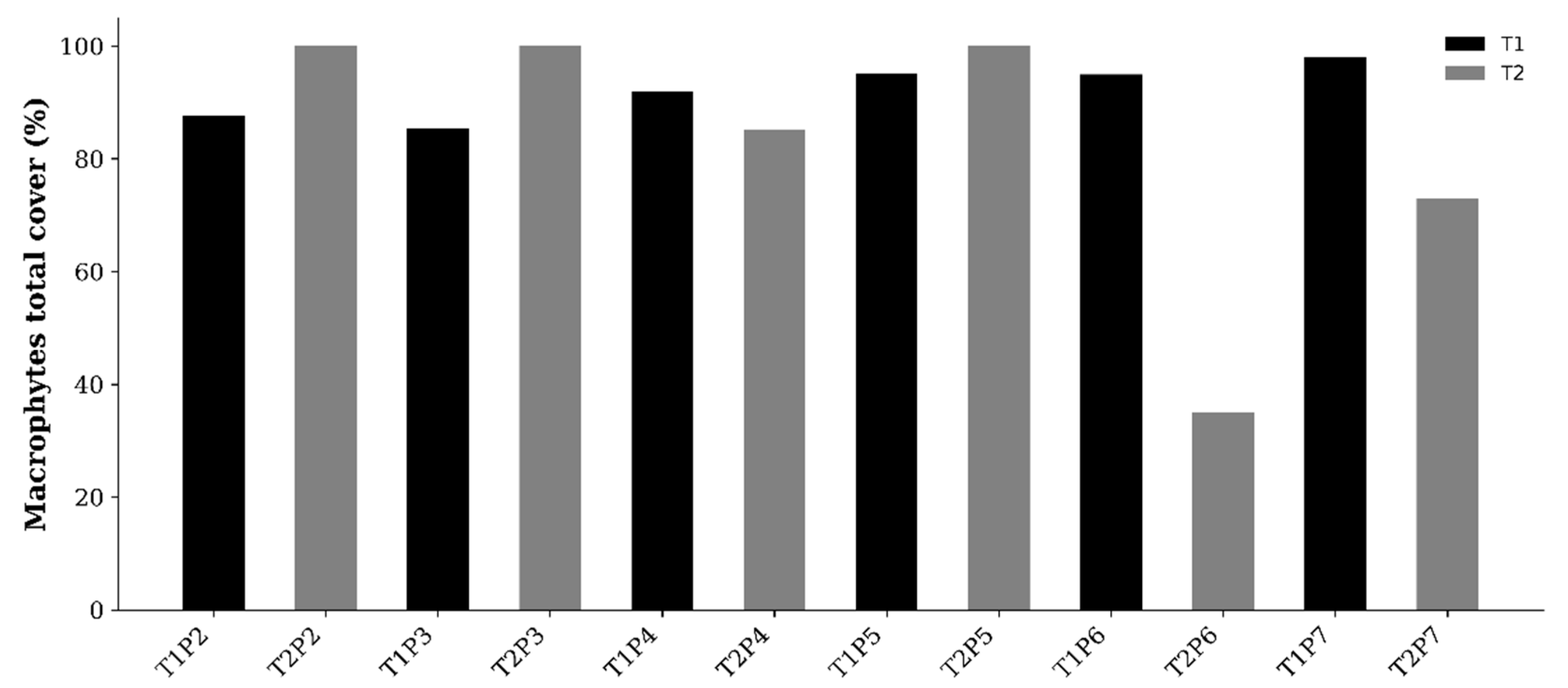
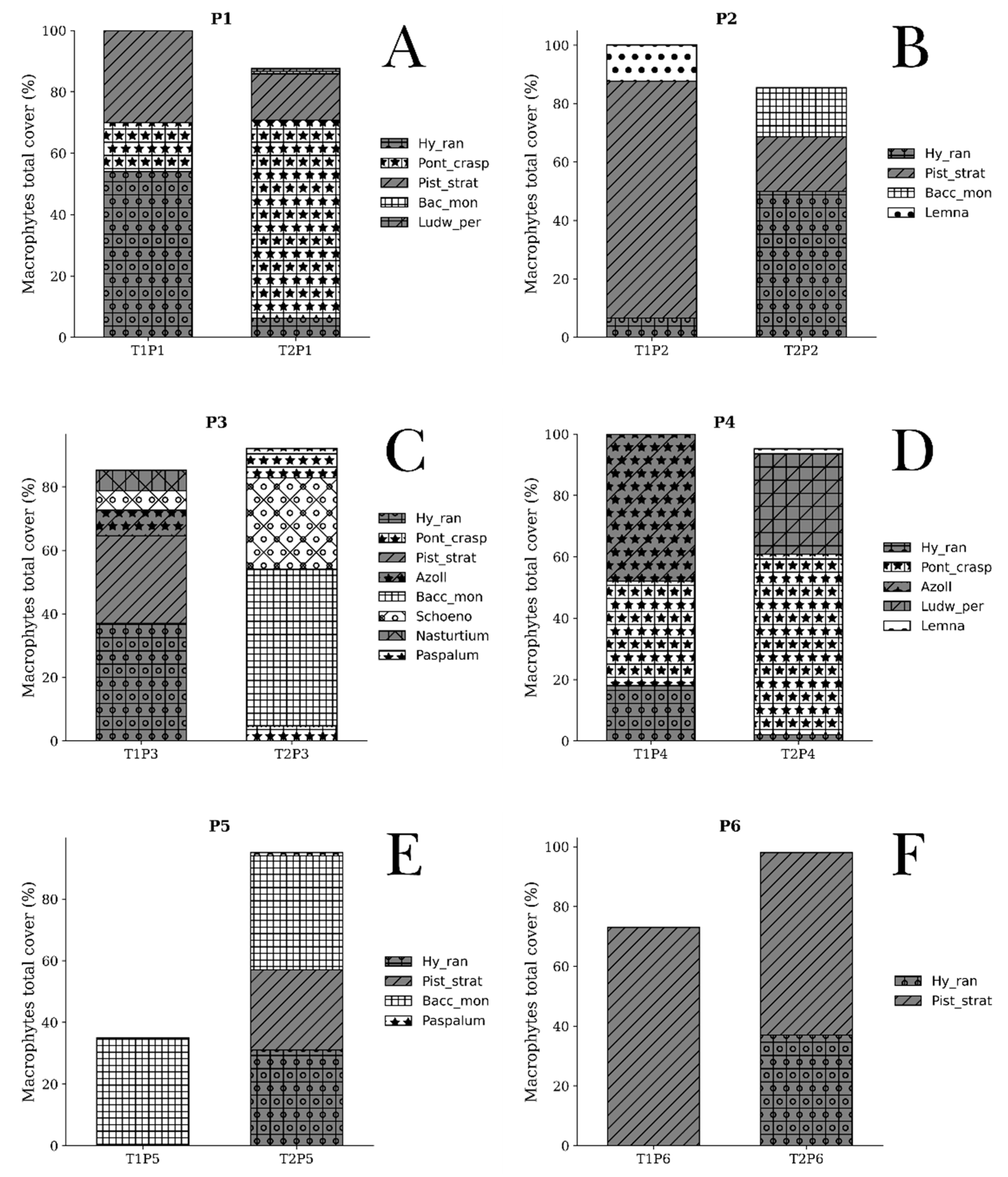
Urban coastal wetlands along the Peruvian Pacific coast are increasingly affected by urban expansion, pollution, and hydrological alterations, compromising their ecological integrity. In this context, the spatiotemporal variation of the aquatic macrophyte community and its relationship with limnological conditions and drivers of change were evaluated in the Santa Rosa wetland (Chancay, Lima). The objective is to evaluate the spatiotemporal variation of the aquatic macrophyte community in the Santa Rosa wetland and analyze its relationship with physicochemical limnological variables and drivers of change. Sampling was conducted during two contrasting hydrological seasons in 2022: T1 (summer) and T2 (winter), at six sampling points (P1–P6). Physicochemical variables (water depth, temperature, pH, conductivity, TDS, TSS, dissolved oxygen, turbidity, nitrate, ammonium, phosphorus, and dissolved organic matter) were measured, and the relative abundance of aquatic macrophytes was evaluated. Drivers of change were identified through direct observation and a structured matrix, with a PCoA performed to summarize spatiotemporal trends. Data were analyzed using Principal Component Analysis (PCA), Co-inertia analysis, and Multi-Response Permutation Procedures (MRPP). Significant spatiotemporal variation was observed in physicochemical parameters (p < 0.05), with moderate covariation between the two matrices (RV = 0.47). A total of ten aquatic macrophyte species were recorded, with higher abundance of Pontederia crassipes and Pistia stratiotes in T1, and Hydrocotyle ranunculoides and Bacopa monnieri in T2. The most relevant drivers of change were solid waste, livestock grazing, organic contamination, and urban expansion. Spatial heterogeneity was observed in the drivers of change affecting the Santa Rosa wetland, forming a mosaic of areas with different impact profiles. Despite multiple anthropogenic pressures, the Santa Rosa wetland maintains a limnological structure and a functionally coupled macrophyte community, evidencing ecological resilience to environmental degradation. The observed covariation between physicochemical conditions and vegetation confirms the persistence of essential ecological processes, even within an altered urban context. This study demonstrates that integrating biotic components, limnological variables, and drivers of change is fundamental to understanding and monitoring the ecological dynamics of urban wetlands along the Peruvian coast.
Keywords:
1. Introduction
2. Materials and Methods
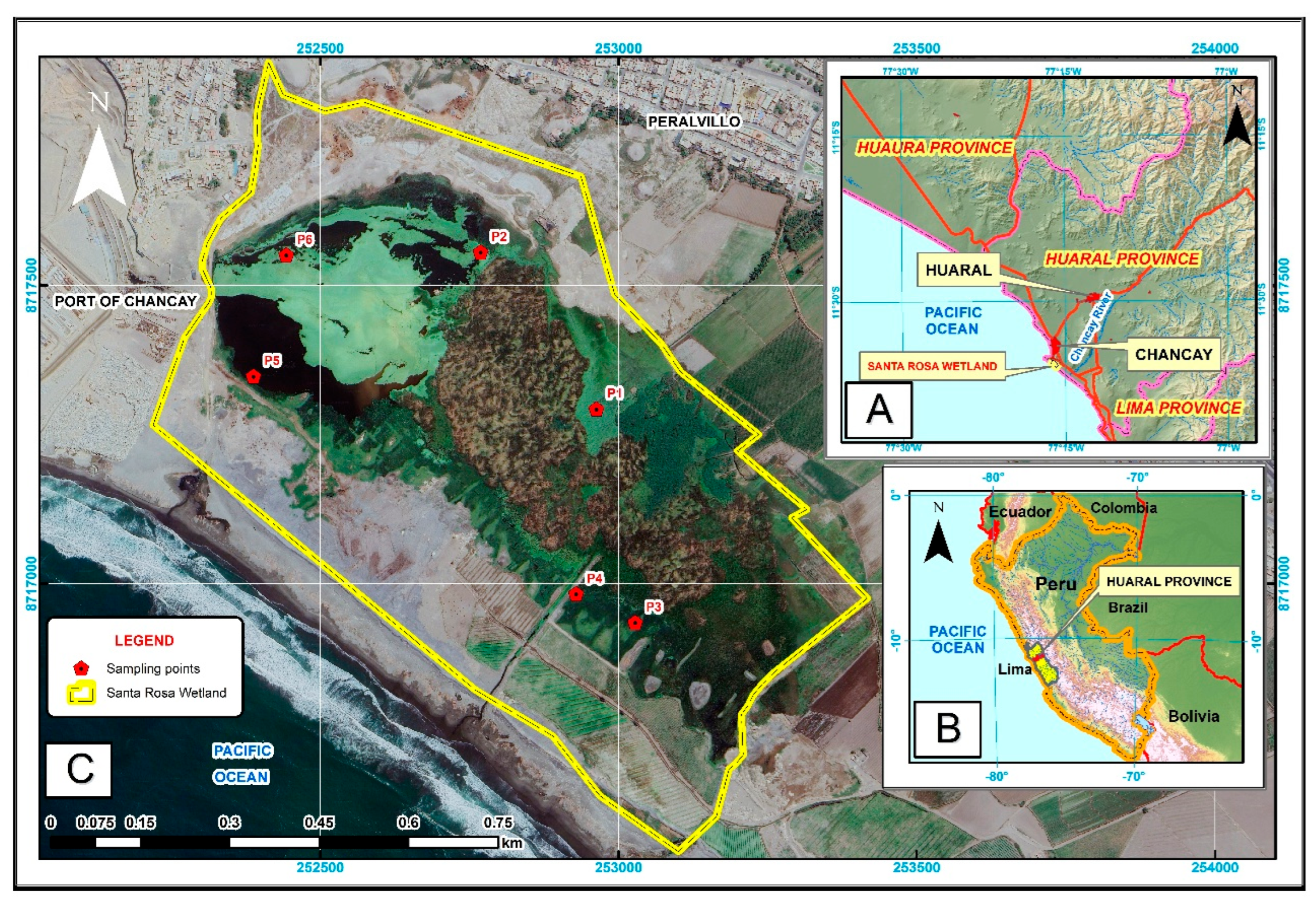
2.1. Study Area
2.2. Sampling Design
2.3. In situ Analysis of Physicochemical Parameters
2.4. Aquatic Macrophytes Abundance Determination and Sample Collection
2.4.1. Determination of the Relative Abundance of Aquatic Macrophytes
2.4.2. Species Identification of Aquatic Macrophytes
2.5. Identification and Classification of Drivers of Change (DOC)
2.6. Data Analysis
3. Results
3.1. Spatiotemporal Variation
3.1.1. Macrophytes
3.1.2. Physicochemical Parameters
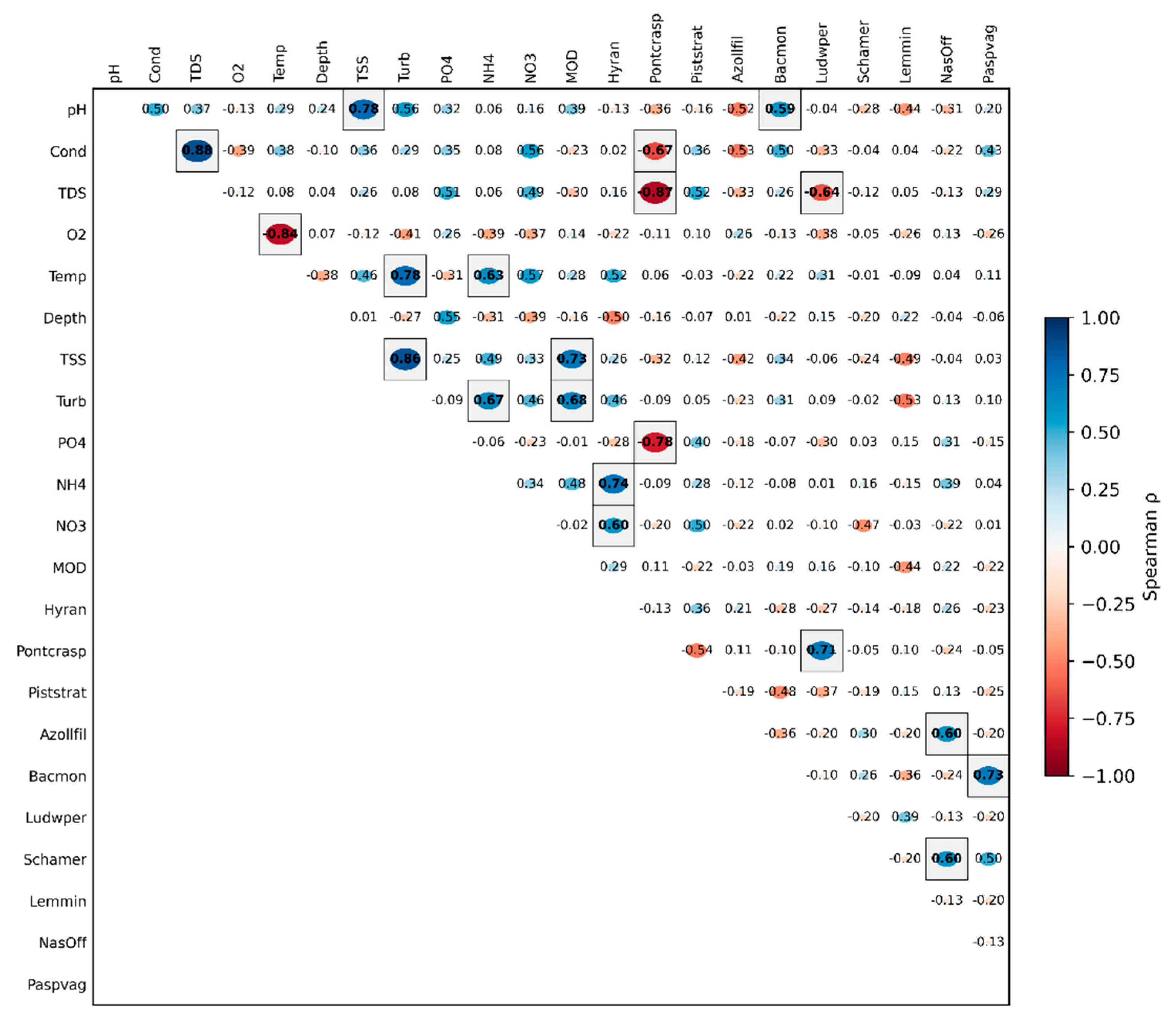
3.2. Spearman's Correlation Matrix and Drivers of Change
3.2.1. Spearman’s Correlation Matrix
3.2.2. Drivers of Change
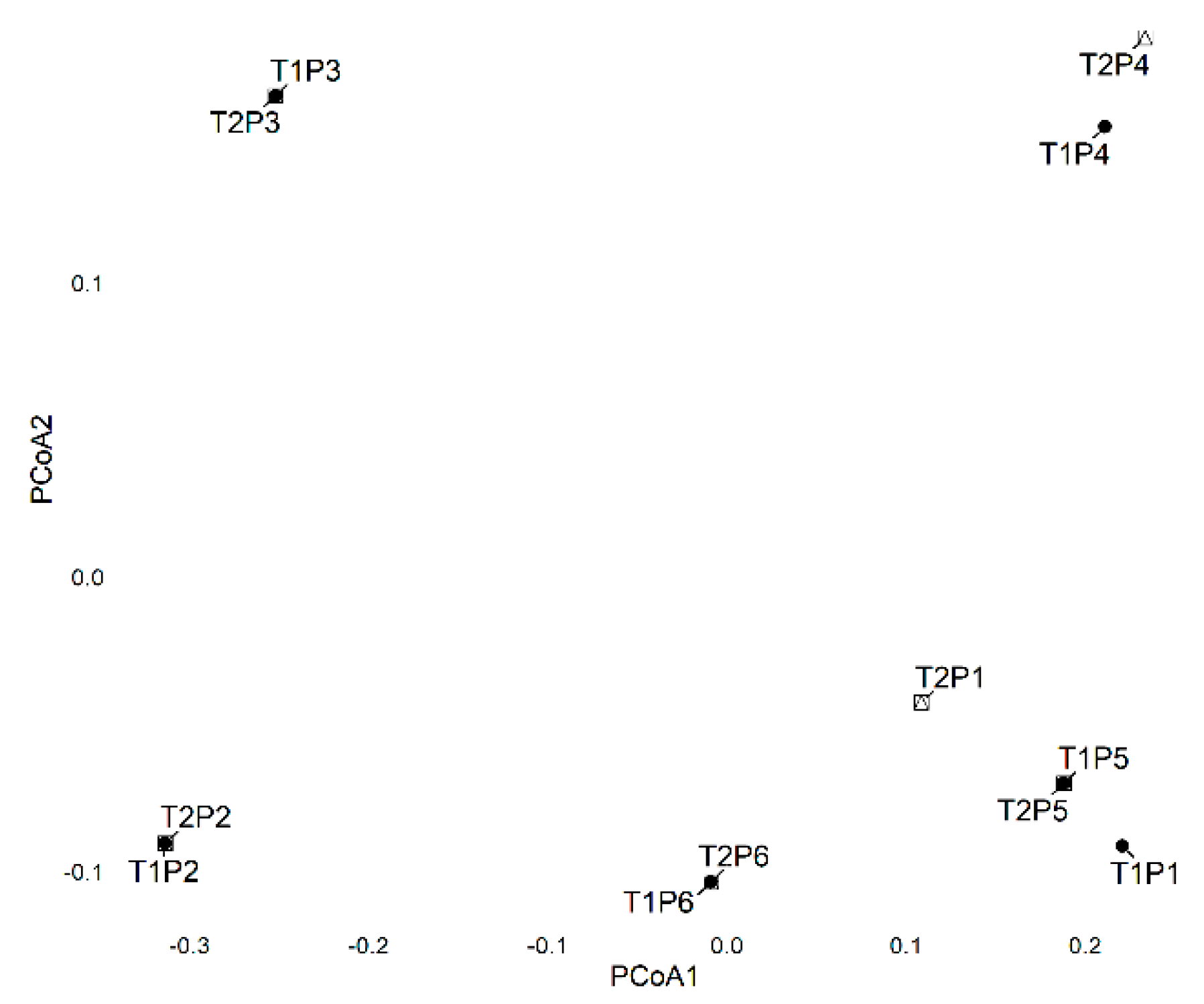
3.2.3. Principal Coordinates Analysis (PCoA)
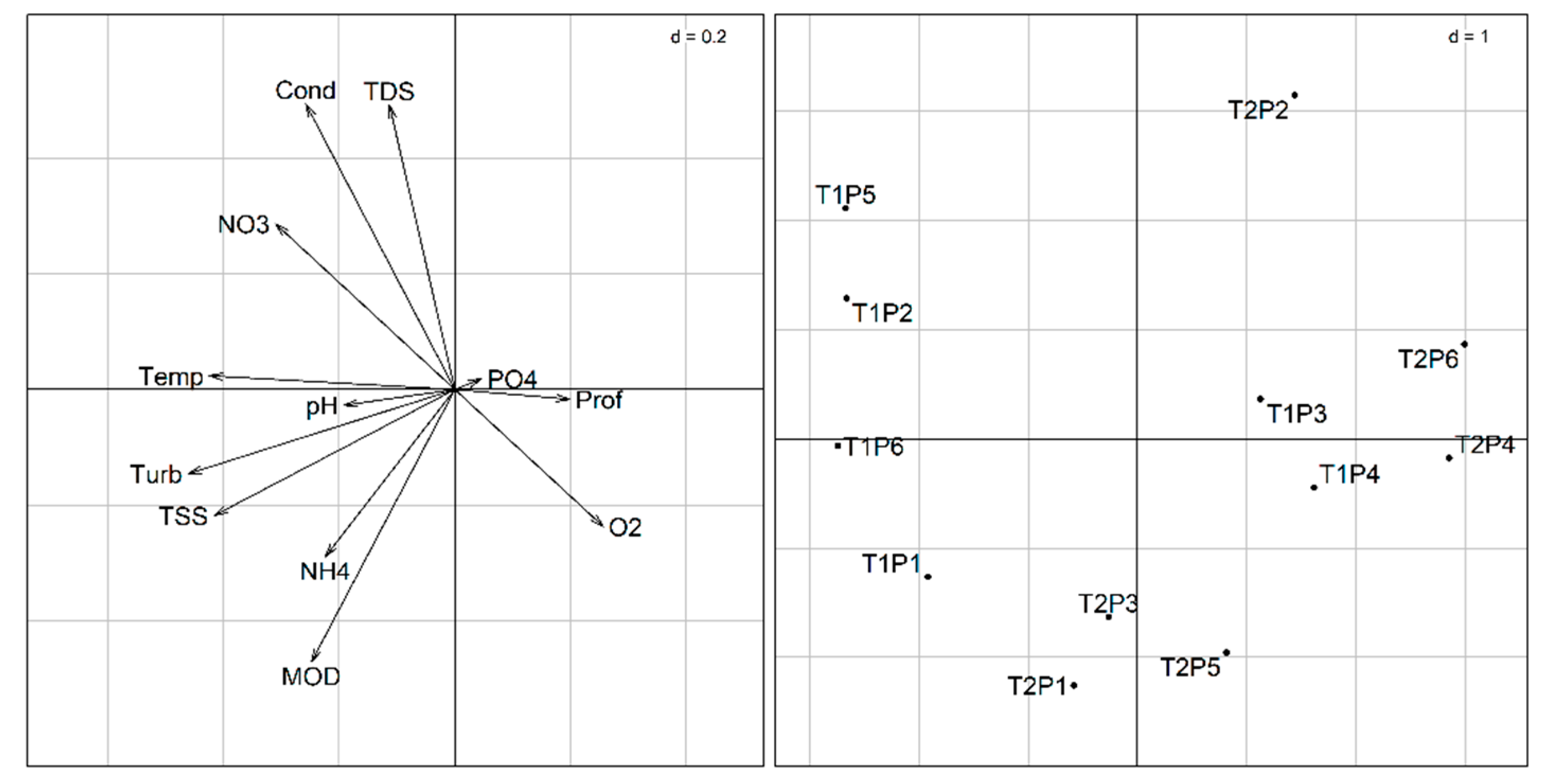
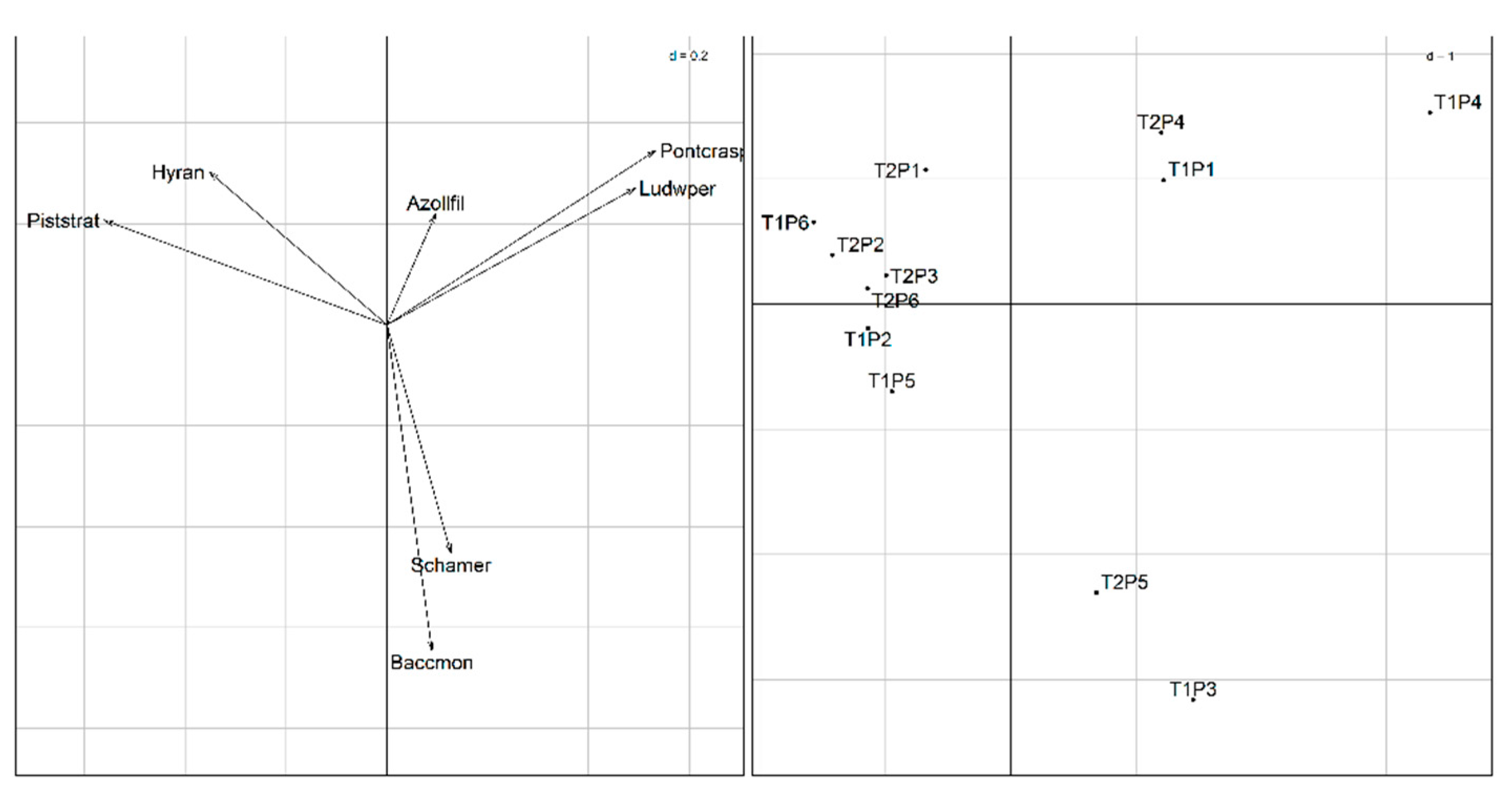
3.3. Relationship Between Biotic and Abiotic Matrices
3.3.1. Principal Component Analysis (PCA)
3.3.2. Co-Inertia
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| TDS | Total dissolved solids |
| TSS | Total suspended solids |
| DO | Dissolved oxygen |
| Cond | Conductivity |
| Turb | Turbidity |
| DOM | Dissolved organic matter |
| DOC | Drivers of change |
| PCA | Principal Component Analysis |
| PCoA | Principal Coordinate Analysis |
| MRPP | Multiple Response Permutation Procedure |
| CUD | Uncontrolled urban growth |
| DGA | Agricultural degradation |
| DGP | Livestock degradation |
| IEE | Introduction of exotic species |
| COQ | Chemical contamination |
| CORG | Organic contamination |
| RSS | Solid waste |
| RSC | Construction waste |
| SDE | Burial and drying |
| PGR | Presence of farms |
| COM | Microbiological contamination |
| GMI | Governance failures associated with poor management and inadequate policies |
| EFL | Effluents |
| OTP | Other disturbances (alteration of residence time due to drainage excavation) |
References
- Grizzetti, B.; Lanzanova, D.; Liquete, C.; Reynaud, A.; Cardoso, A.C. Assessing Water Ecosystem Services for Water Resource Management. Environmental Science & Policy 2016, 61, 194–203. [CrossRef]
- Moomaw, W.R.; Chmura, G.L.; Davies, G.T.; Finlayson, C.M.; Middleton, B.A.; Natali, S.M.; Perry, J.E.; Roulet, N.; Sutton-Grier, A.E. Wetlands In a Changing Climate: Science, Policy and Management. Wetlands 2018, 38, 183–205. [CrossRef]
- Mitsch, W.J.; Gosselink, J.G. Wetlands: Human Use and Science. In Wetlands, 5th Edition; Wiley, 2015; pp. 3–26.
- Davidson, N.C.; Finlayson, C.M. Updating Global Coastal Wetland Areas Presented in Davidson and Finlayson (2018). Mar. Freshwater Res. 2019, 70, 1195. [CrossRef]
- Davidson, N.C. How Much Wetland Has the World Lost? Long-Term and Recent Trends in Global Wetland Area. Mar. Freshwater Res. 2014, 65, 934–941. [CrossRef]
- Fluet-Chouinard, E.; Stocker, B.D.; Zhang, Z.; Malhotra, A.; Melton, J.R.; Poulter, B.; Kaplan, J.O.; Goldewijk, K.K.; Siebert, S.; Minayeva, T.; et al. Extensive Global Wetland Loss over the Past Three Centuries. Nature 2023, 614, 281–286. [CrossRef]
- IPBES Global Assessment Report on Biodiversity and Ecosystem Services of the Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Services; Zenodo, 2019;
- Xu, T.; Weng, B.; Yan, D.; Wang, K.; Li, X.; Bi, W.; Li, M.; Cheng, X.; Liu, Y. Wetlands of International Importance: Status, Threats, and Future Protection. International Journal of Environmental Research and Public Health 2019, 16, 1818. [CrossRef]
- An, S.; Yin, S.; Verhoeven, J.T.A.; Jeelani, N. Chapter 29 - Wetlands. In Wetzel’s Limnology (Fourth Edition); Jones, I.D., Smol, J.P., Eds.; Academic Press: San Diego, 2024; pp. 999–1013 ISBN 978-0-12-822701-5.
- Mitsch, W.J.; Gosselink, J.G. Wetland Biogeochemistry. In Wetlands, 5th Edition; Wiley, 2015; pp. 179–214.
- Rasmussen, T.C.; Deemy, J.B.; Long, S.L. Wetland Hydrology. In The Wetland Book: I: Structure and Function, Management, and Methods; Finlayson, C.M., Everard, M., Irvine, K., McInnes, R.J., Middleton, B.A., van Dam, A.A., Davidson, N.C., Eds.; Springer Netherlands: Dordrecht, 2018; pp. 201–216 ISBN 978-90-481-9659-3.
- Tiner, R.W. Wetland Indicators: A Guide to Wetland Identification, Delineation, Classification, and Mapping; Second edition.; Taylor & Francis: Boca Raton, 2017; ISBN 978-1-4398-5369-6.
- Mitsch, W.J.; Gosselink, J.G. Wetland Hydrology. In Wetlands, 5th Edition; Wiley, 2015; pp. 111–160.
- Reddy, K.R.; DeLaune, R.D. Biogeochemistry of Wetlands: Science and Applications; CRC Press: Boca Raton, 2008; ISBN 978-0-429-09576-4.
- Davidson, N.C.; Middleton, B.A.; McInnes, R.J.; Everard, M.; Irvine, K.; Van Dam, A.A.; Finlayson, C.M. Introduction to the Wetland Book 1: Wetland Structure and Function, Management, and Methods. The Wetland Book: I: Structure and Function, Management, and Methods 2018, 3–14.
- Ramsar Convention on Wetlands Global wetland outlook: state of the World’s wetlands and their services to people; Ramsar Convention Secretariat: Gland, Switzerland, 2018;
- Chambers, P.A.; Maberly, S.C. Chapter 24 - Freshwater Plants. In Wetzel’s Limnology (Fourth Edition); Jones, I.D., Smol, J.P., Eds.; Academic Press: San Diego, 2024; pp. 759–816 ISBN 978-0-12-822701-5.
- Bornette, G.; Puijalon, S. Response of Aquatic Plants to Abiotic Factors: A Review. Aquatic Sciences 2011, 73. [CrossRef]
- Mitsch, W.J.; Gosselink, J.G. Wetland Vegetation and Succession. In Wetlands, 5th Edition; Wiley, 2015; pp. 215–255.
- Siddiqui, A.J.; Jahan, S.; Adnan, M.; Ashraf, S.A.; Singh, R. Macrophytes and Their Role in Wetland Ecosystems. In Aquatic Macrophytes: Ecology, Functions and Services; Kumar, S., Bauddh, K., Singh, R., Kumar, N., Kumar, R., Eds.; Springer Nature Singapore: Singapore, 2023; pp. 119–138 ISBN 978-981-99-3821-6.
- Maltchik, L.; Rolon, A.S.; Schott, P. Effects of Hydrological Variation on the Aquatic Plant Community in a Floodplain Palustrine Wetland of Southern Brazil. Limnology 2007, 8, 23–28. [CrossRef]
- Freedman, B.; Lacoul, P. Environmental Influences on Aquatic Plants in Freshwater Ecosystems. Environmental Reviews 2006, 14, 89–136. [CrossRef]
- Dar, N.A.; Pandit, A.K.; Ganai, B.A. Factors Affecting the Distribution Patterns of Aquatic Macrophytes. Limnological Review 2014, 14, 75–81. [CrossRef]
- Rameshkumar, S.; Radhakrishnan, K.; Aanand, S.; Rajaram, R. Influence of Physicochemical Water Quality on Aquatic Macrophyte Diversity in Seasonal Wetlands. Appl Water Sci 2019, 9, 12. [CrossRef]
- Dubey, D.; Kumar, S.; Dutta, V. Impact of Nutrient Enrichment on Habitat Heterogeneity and Species Richness of Aquatic Macrophytes: Evidence from Freshwater Tropical Lakes of Central Ganga Plain, India. Int. J. Environ. Sci. Technol. 2022, 19, 5529–5546. [CrossRef]
- Florescu, L.I.; Catana, R.D.; Mihai, R.A.; Dumitrache, A.C.; Moldoveanu, M.M. Macrophyte Community Distribution in Relation to Anthropization Influences and Phytoplankton Development in an Urban Lake Chain. Water 2024, 16, 3467. [CrossRef]
- Madzivanzira, T.C.; Coetzee, J.A.; Dalu, T. Factors Structuring Aquatic Macrophytes. In Aquatic Macrophytes: Ecology, Functions and Services; Kumar, S., Bauddh, K., Singh, R., Kumar, N., Kumar, R., Eds.; Springer Nature Singapore: Singapore, 2023; pp. 21–52 ISBN 978-981-99-3821-6.
- Cerna-Arrue, A.; Aponte, H. El origen de los humedales peruanos y su relación con los procesos geológico-ambientales de Sudamérica: Una revisión. Caldasia 2024, 46, 314–325. [CrossRef]
- Díaz, A.; Sáenz-Zúñiga, L.; Zutta, B. Formación de un humedal en la costa norte del Perú: Estabilidad biofísica y diversidad biológica. Rev peru biol 2021, 28, e21132. [CrossRef]
- INGEMMET, I.G.M. y M.D. de G.A. y R. Estudio hidrogeológico del Refugio de Vida Silvestre Los Pantanos de Villa, Chorrillos - Lima. Instituto Geológico, Minero y Metalúrgico – INGEMMET 2019.
- Verones, F.; Bartl, K.; Pfister, S.; Jiménez Vílchez, R.; Hellweg, S. Modeling the Local Biodiversity Impacts of Agricultural Water Use: Case Study of a Wetland in the Coastal Arid Area of Peru. Environ. Sci. Technol. 2012, 46, 4966–4974. [CrossRef]
- Aponte, H.; Gonzales, S.; Gomez, A. Impulsores de cambio en los humedales de América Latina: El caso de los humedales costeros de Lima. South Sustainability 2020, 1, e023–e023. [CrossRef]
- Aponte, H.; Cano, A. Estudio florístico comparativo de seis humedales de la costa de lima (Perú): Actualización y nuevos retos para su conservación. 2013, 3, 13.
- Gonzales, S.; Aponte, H.; Cano, A. Update of the Vascular Flora of the Santa Rosa Wetland - Chancay (Lima, Peru). Arnaldoa 2019, 26, 867–882. [CrossRef]
- Castillo Zavala, J.M.; Mostacero León, J.; De La Cruz-Castillo, A.J.; Castillo- Zavala, J.L.; Anhuamán-Castañeda, V.A. Inventario florístico de los humedales de la costa de la provincia de Trujillo, La Libertad, Perú. HYW 2022, 1, 23–39. [CrossRef]
- Pulido, V. Ciento quince años de registros de aves en Pantanos de Villa. Rev peru biol 2018, 25, 291–306. [CrossRef]
- Aguirre-Soto, L.; Nieto-Juárez, J.I. Evaluación de La Calidad Del Agua En El Humedal Costero Pantanos de Villa, Chorrillos, Perú. Tecnia 2024, 34, 11–25. [CrossRef]
- Benites-Alfaro, E.; Mendivil, V.T.; Farfán, E.E.; Rodriguez, C. Trophic Status Index of the Waters of the Pantanos de Villa Protected Natural Reserve, Chorrillos, Perú. 2020, 7.
- Huaman Vilca, S.; Lucen, M.; Paredes Vite, M.; Changanaqui Alfaro, D. Evaluación de la calidad del agua de la Laguna Marvilla en los Pantanos de Villa (Lima – Perú). S. Sust. 2020, e019. [CrossRef]
- Canales Cuadros, H.; Cabrera Carranza, C.; Arana Maestre, J. Macroinvertebrados acuáticos y calidad de agua en el Área de Conservación Regional Humedales de Ventanilla, Callao. Rev. Inst. investig. Fac. minas metal cienc. geogr 2022, 25, 295–301. [CrossRef]
- Castillo-Velásquez, R.-M.; Huamantinco-Araujo, A.-A. Variación espacial de la comunidad de macroinvertebrados acuáticos en la zona litoral del humedal costero Santa Rosa, Lima, Perú. Revista de Biología Tropical 2020, 68, 50–68. [CrossRef]
- Córdova-Tello, C.A.; Huamantinco-Araujo, A.A. Efecto de las variables fisicoquímicas en la comunidad de coleópteros acuáticos en un humedal costero, Lima, Perú. Acta biol. Colomb. 2023, 28, 204–219. [CrossRef]
- Bopp, G.; Peláez, F. Evaluación de La Flora Vascular de Los Humedales Costeros de La Libertad, Perú. Manglar 2019, 16, 151–156. [CrossRef]
- Jindo, K.; Morikawa, M. Innovative Feasibility Study for the Reclamation of the Cascajo Wetlands in Peru Utilizing Sustainable Technologies. Water 2020, 12. [CrossRef]
- Aldana-Domínguez, J.; Rojas Quezada, C.; Munizaga, J.; Moschella, P.; Stamm, C.; Martínez, C. Urban Wetland Losses and Land-Use Conservation Challenges in Three Latin American Cities. In Urban Wetlands in Latin America: Protection, Conservation, Innovation, Restoration, and Community for Sustainable and Water Sensitive Cities; Rojas Quezada, C., Ed.; Springer Nature Switzerland: Cham, 2024; pp. 3–18 ISBN 978-3-031-69590-2.
- Aponte, H.; Coello-Sarmiento, M.-P.; Montes-Iturrizaga, D. New Insights About the Drivers of Change in the Coastal Wetlands of Peru: Results of a Rapid Field Survey. Water 2025, 17, 1473. [CrossRef]
- Romero-Mariscal, G.; Garcia-Chevesich, P.A.; Morales-Paredes, L.; Arenazas-Rodriguez, A.; Ticona-Quea, J.; Vanzin, G.; Sharp, J.O. Peruvian Wetlands: National Survey, Diagnosis, and Further Steps toward Their Protection. Sustainability 2023, 15, 8255. [CrossRef]
- Urbina, D.; Rivera-Cáceda, F.; Aponte, H. ¿Se Están Reduciendo Los Humedales de La Costa Del Pacífico Suramericano? El Caso de Los Humedales de Lima. Rev. Acad. Colomb. Cienc. Ex. Fis. Nat. 2022, 46, 985–998. [CrossRef]
- Ramirez, D.W.; Aponte, H.; Cano, A. Flora vascular y vegetación del humedal de Santa Rosa (Chancay, Lima). Revista Peruana de Biología 2011, 17. [CrossRef]
- Zamora-Aranda, A.; Aponte, H. Evaluation of Two Ecosystem Services Provided by a Pistia Stratiotes Population on the Pacific Coast of South America. Biology 2024, 13, 573. [CrossRef]
- Ochoa-Balbacea, D.O.; Gonzales, S.; Apeño, A.; Aponte, H. Hábitats prioritarios en un humedal costero del Pacífico: propuesta de un índice basado en servicios ecosistémicos. Revista de la Academia Colombiana de Ciencias Exactas, Físicas y Naturales 2022, 46, 182–191. [CrossRef]
- ANA Protocolo Nacional Para El Monitoreo de La Calidad de Los Recursos Hídricos Superficiales (R.J. N· 010-2016-ANA) 2016.
- Baird, R.; Rice, E.W.; Eaton, A.D.; Bridgewater, L.; Federation, W.E. Standard Methods for the Examination of Water and Wastewater; American Public Health Association, 2017; ISBN 978-0-87553-287-5.
- Methods in Stream Ecology; Hauer, F.R., Lamberti, G.A., Eds.; 2nd ed.; Academic Press/Elsevier: San Diego, Calif, 2006; ISBN 978-0-12-332908-0.
- Dengler, J.; Dembicz, I. Should We Estimate Plant Cover in Percent or on Ordinal Scales? VCS 2023, 4, 131–138. [CrossRef]
- Cirujano, S.; Meco Molina, A.; García Murillo, P. Flora acuática española. Hidrófitos vasculares Available online: https://bibdigital.rjb.csic.es/records/item/16128-flora-acuatica-espanola-hidrofitos-vasculares?offset=1 (accessed on 28 March 2025).
- Sarkar, P.; Salami, M.; Githiora, Y.; Vieira, R.; Navarro, A.; Clavijo, D.; Padgurschi, M. A Conceptual Model to Understand the Drivers of Change in Tropical Wetlands: A Comparative Assessment in India and Brazil. Biota Neotrop. 2020, 20, e20190913. [CrossRef]
- Oksanen, J.; Simpson, G.L.; Blanchet, F.G.; Kindt, R.; Legendre, P.; Minchin, P.R.; O’Hara, R.B.; Solymos, P.; Stevens, M.H.H.; Szoecs, E.; et al. Vegan: Community Ecology Package 2025.
- Borcard, D.; Gillet, F.; Legendre, P. Numerical Ecology with R; Use R!; Springer International Publishing: Cham, 2018; ISBN 978-3-319-71403-5.
- Legendre, P.; Legendre, L. Numerical Ecology; Developments in environmental modelling; 3rd English ed.; Elsevier: Amsterdam, 2012; ISBN 978-0-444-53868-0.
- Mielke, P.W.; Berry, K.J. Description of MRPP. In Permutation Methods: A Distance Function Approach; Mielke, P.W., Berry, K.J., Eds.; Springer: New York, NY, 2001; pp. 9–65 ISBN 978-1-4757-3449-2.
- Doledec, S.; Chessel, D. Co-Inertia Analysis: An Alternative Method for Studying Species-Environment Relationships. Freshwater Biol 1994, 31, 277–294. [CrossRef]
- Dray, S.; Dufour, A.-B.; Thioulouse, J.; Chessel, D.; Jombart, T.; Pavoine, S.; Lobry, J.R.; Ollier, S.; Borcard, D.; Legendre, P.; et al. Ade4: Analysis of Ecological Data: Exploratory and Euclidean Methods in Environmental Sciences 2025.
- R Core Team R: A Language and Environment for Statistical Computing. 2020.
- RStudio Team RStudio: Integrated Development for R. RStudio 2020.
- Agidie, A.; Wondie, A.; Beneberu, G. Wetland Disturbance Level and Macrophyte Composition in Natural Wetlands and Paddy Fields in the Eastern Part of Lake Tana, Ethiopia. Heliyon 2024, 10, e40701. [CrossRef]
- Galatowitsch, S.M. Natural and Anthropogenic Drivers of Wetland Change. In The Wetland Book; Finlayson, C.M., Milton, G.R., Prentice, R.C., Davidson, N.C., Eds.; Springer Netherlands: Dordrecht, 2018; pp. 359–367 ISBN 978-94-007-4000-6.
- Gomez Vilca, A.J.; Aponte, H.; Gonzales, S. ¿Cómo Proteger Los Humedales Costeros Peruanos? Una Respuesta a Partir de Un Modelo Conceptual de Sus Impulsores de Cambio. Bol. Investig. Mar. Costeras 2023, 52, 125–142. [CrossRef]
- Pacioglu, O.; Tușa, I.M.; Sidoroff, M.E.; Ițcuș, C. The Best Management Practices in Agriculture for Protection of Inland Water Ecosystems. In Encyclopedia of Inland Waters (Second Edition); Mehner, T., Tockner, K., Eds.; Elsevier: Oxford, 2022; pp. 58–67 ISBN 978-0-12-822041-2.
- Meerhoff, M.; Beklioğlu, M. Chapter 26 - Shallow Lakes and Ponds. In Wetzel’s Limnology (Fourth Edition); Jones, I.D., Smol, J.P., Eds.; Academic Press: San Diego, 2024; pp. 859–892 ISBN 978-0-12-822701-5.
- Hadad, H.R.; Mufarrege, M.M.; Di Luca, G.A.; Maine, M.A. Salinity and pH Effects on Floating and Emergent Macrophytes in a Constructed Wetland. Water Sci Technol 2018, 2017, 270–275. [CrossRef]
- Haller, W.T.; Sutton, D.L.; Barlowe, W.C. Effects of Salinity on Growth of Several Aquatic Macrophytes. Ecology 1974, 55, 891–894. [CrossRef]
- Pereto, S.C.A. da S.; Padial, A.A. Macrophyte Functional Composition Is Stable across a Strong Environmental Gradient of a Neotropical Floodplain. Acta Bot. Bras. 2021, 35, 62–69. [CrossRef]
- Rolon, A.; Homem, H.; Maltchik, L. Aquatic Macrophytes in Natural and Managed Wetlands of Rio Grande Do Sul State, Southern Brazil. Acta Limnologica Brasiliensia 2010, 22, 133–146. [CrossRef]
- Muñoz, M.; Tinoco, K.; Arenas, J.; Aponte, H. Dinámica de La Planta Acuática Pistia Stratiotes y Su Incorporación a Un Sistema de Producción de Compost. Tecnología y ciencias del agua 17. [CrossRef]
- Rejmankova, E. The Role of Macrophytes in Wetland Ecosystems. Journal of Ecology and Field Biology 2011, 34, 333–345. [CrossRef]
- Saluja, R.; Garg, J.K. Macrophyte Species Composition and Structure along Littoral Region in Relation to Limnological Variables of a Tropical Wetland Ecosystem. Chemistry and Ecology 2017, 33, 499–515. [CrossRef]
- Aponte, H.; Jiménez, R.; Alcántara, B. Challenges for Management and Conservation of Santa Rosa Wetland (Lima-Peru). Cientifica 2012, 9, 257–264.
- Loayza-Basan, K.; Castillejos Paniagua, K.; Mestas Valero, R.; Quiliche Duran, J.P.J. Estudio de la Teledetección y Caracterización Fisicoquímica del Humedal “El Cascajo”, Santa Rosa, Chancay- Lima, Perú. Infinitum... 2017, 7. [CrossRef]
- Leiva, J.V.Y.; Ibarra, J.A. Evaluación de la calidad del agua en el canal Surco de la Zona de Reglamentación Especial del Humedal Los Pantanos de Villa (Lima, Perú). South Sustainability 2024, 5, e090–e090. [CrossRef]
- Mendoza-Caballero, W.; Quiroz-Santos, R.L. EVALUACIÓN DE LOS ESTADOS TRÓFICOS DE LA LAGUNA PRINCIPAL DEL ÁREA DE CONSERVACIÓN REGIONAL ALBUFERA DE MEDIO MUNDO, HUAURA-LIMA, PERÚ. Biotempo 2022, 19, 149–164. [CrossRef]
- Jyothi, P.V.; Sureshkumar, S. Patterns of Vegetation Dynamics across Mild Disturbance Gradient in a Freshwater Wetland System in Southern India. Wetlands 2018, 38, 807–817. [CrossRef]
- Domínguez-Machín, M.E.; Silva-López, G. Consumo de plantas por el ganado vacuno en una zona de Manglar-Espartal de Alvarado, Veracruz, México. Foresta Veracruzana 2020, 22, 7–11.
- Marca-Zevallos, M.J.; Galiano, W.; Chevarría, R.; Sebastián, G.; Huamani, K.; Ochoa, J.A.; Mendoza, J. Influencia de factores ambientales en la composición de macrofitas acuáticas en lagos (cochas) de la Amazonía sur del Perú. Darwiniana, nueva serie 2024, 12, 170–191. [CrossRef]
- Schneider, B.; Cunha, E.R.; Marchese, M.; Thomaz, S.M. Explanatory Variables Associated with Diversity and Composition of Aquatic Macrophytes in a Large Subtropical River Floodplain. Aquatic Botany 2015, 121, 67–75. [CrossRef]
- Aoki, C.; Teixeira-Gamarra, M.C.; Gamarra, R.M.; De Medeiros, S.C.H.; Pott, V.J.; Damasceno-Junior, G.A.; Pott, A.; Scremin-Dias, E. Abiotic Factors Drive the Structure of Aquatic Plant Assemblages in Riverine Habitats of the Brazilian “Pantanal.” Braz. J. Bot 2017, 40, 405–415. [CrossRef]
- Rolon, A.S.; Maltchik, L. Environmental Factors as Predictors of Aquatic Macrophyte Richness and Composition in Wetlands of Southern Brazil. Hydrobiologia 2006, 556, 221–231. [CrossRef]
- Adams, D.C. Evaluating Modularity in Morphometric Data: Challenges with the RV Coefficient and a New Test Measure. Methods Ecol Evol 2016, 7, 565–572. [CrossRef]
- Mitsch, W.J.; Gosselink, J.G. Wetland Definitions. In Wetlands, 5th Edition; Wiley, 2015; pp. 27–44.
- Gómez-Sánchez, R.; Cuba, D.; Aponte, H. Sobre la necesidad de descentralización y diversificación de la investigación en humedales costeros peruanos. TB 2022, 20, 121–150. [CrossRef]
- Rivera, G.; Gonzales, S.; Aponte, H. Wetlands of the South American Pacific Coast: A Bibliometric Analysis. Wetlands Ecol Manage 2021. [CrossRef]
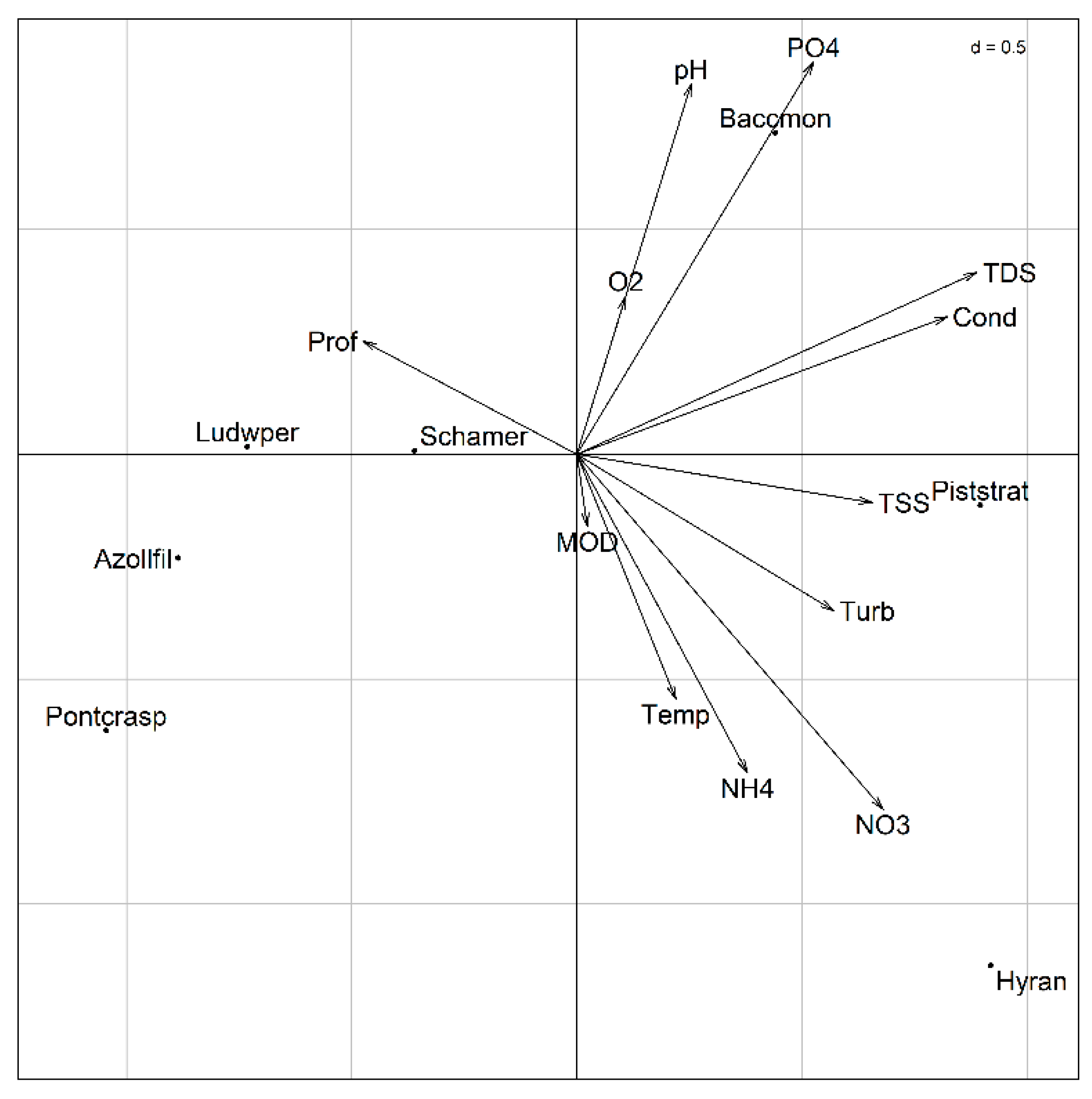
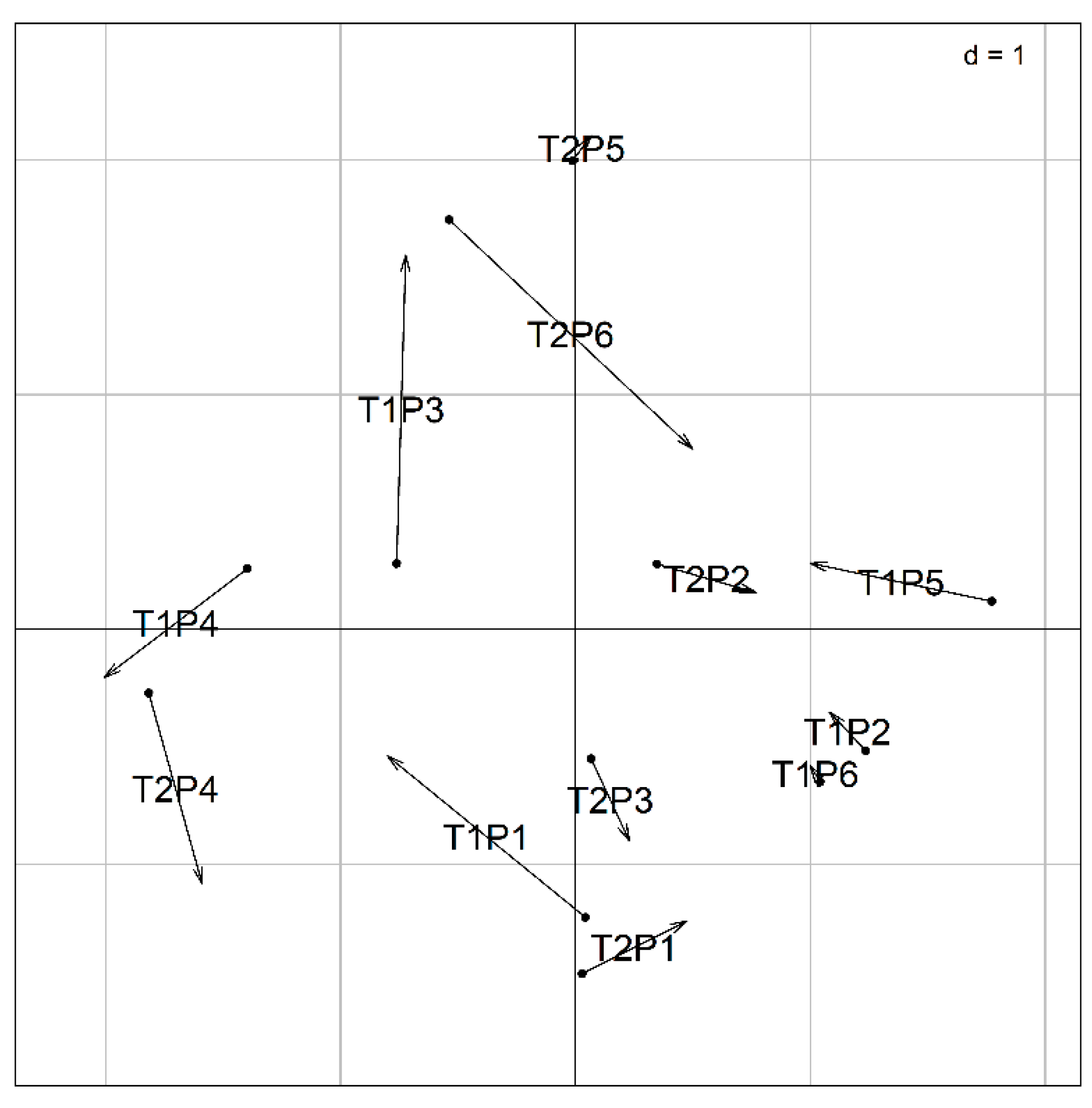
| Sample Points | pH | Conductivity (µS/cm) |
TDS (mg/L) |
DO (mg/L) |
Temperature (°C) |
Depth (m) |
TSS (mg/L) |
Turbidity (NTU) |
PO43- (mg/L) |
NH4 (mg/L) |
NO3 (mg/L) |
||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| T1 | T2 | T1 | T2 | T1 | T2 | T1 | T2 | T1 | T2 | T1 | T2 | T1 | T2 | T1 | T2 | T1 | T2 | T1 | T2 | T1 | T2 | ||
| P1 | 8 | 7.45 | 1610 | 1166 | 505 | 788 | 0.8 | 6 | 25 | 21.8 | 0.12 | 0.12 | 52 | 50 | 57 | 40.77 | 0.605 | 0.66 | 1.54 | 3.43 | 7 | 6 | |
| P2 | 8.3 | 7.13 | 3000 | 2740 | 1260 | 1940 | 0.4 | 3.2 | 27 | 20.5 | 0.08 | 0.19 | 50 | 8 | 50 | 9.47 | 0.9 | 1.06 | 1.75 | 1.26 | 7.5 | 7 | |
| P3 | 7.9 | 7.43 | 1770 | 1401 | 907 | 940 | 0.5 | 3.4 | 22 | 22.1 | 0.13 | 0.20 | 19 | 45 | 25 | 45.92 | 0.785 | 1.28 | 1.49 | 3.03 | 0.05 | 3 | |
| P4 | 7.9 | 7.42 | 1360 | 992 | 610 | 670 | 0.3 | 4.18 | 23 | 19.9 | 0.75 | 0.39 | 19 | 4 | 18 | 10.51 | 0.97 | 0.29 | 1.55 | 0.81 | 2 | 4 | |
| P5 | 8.5 | 8.72 | 3280 | 1632 | 1968 | 1095 | 0.5 | 8.2 | 23.8 | 19.5 | 0.40 | 0.84 | 56 | 56 | 56 | 34.87 | 1.03 | 1.65 | 1.81 | 1.22 | 9 | 0.05 | |
| P6 | 8.1 | 8.06 | 2045 | 1735 | 1227 | 1155 | 0.3 | 6.5 | 25.9 | 16.7 | 0.40 | 0.50 | 74 | 20 | 72 | 12.8 | 1.05 | 1.54 | 1.9 | 0.97 | 7 | 4 | |
| DOC | CUD | DGA | DGP | IEE | COQ | CORG | RSS | RSC | SDE | PGR | COM | GMI | EFL | OTP |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| T2P1 | 0 | 0 | 1 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 1 | 1 | 0 | 0 |
| T2P2 | 0 | 0 | 1 | 1 | 0 | 1 | 0 | 0 | 0 | 1 | 1 | 1 | 0 | 0 |
| T2P3 | 0 | 1 | 1 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 1 | 1 | 0 | 0 |
| T2P4 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 1 |
| T2P5 | 0 | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 0 |
| T2P6 | 0 | 0 | 1 | 1 | 1 | 1 | 1 | 0 | 0 | 0 | 1 | 1 | 0 | 0 |
| T1P1 | 0 | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 1 | 1 | 1 | 0 |
| T1P2 | 0 | 0 | 1 | 1 | 0 | 1 | 0 | 0 | 0 | 1 | 1 | 1 | 0 | 0 |
| T1P3 | 0 | 1 | 1 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 1 | 1 | 0 | 0 |
| T1P4 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 0 |
| T1P5 | 0 | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 0 |
| T1P6 | 0 | 0 | 1 | 1 | 1 | 1 | 1 | 0 | 0 | 0 | 1 | 1 | 0 | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




