Submitted:
03 March 2026
Posted:
04 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
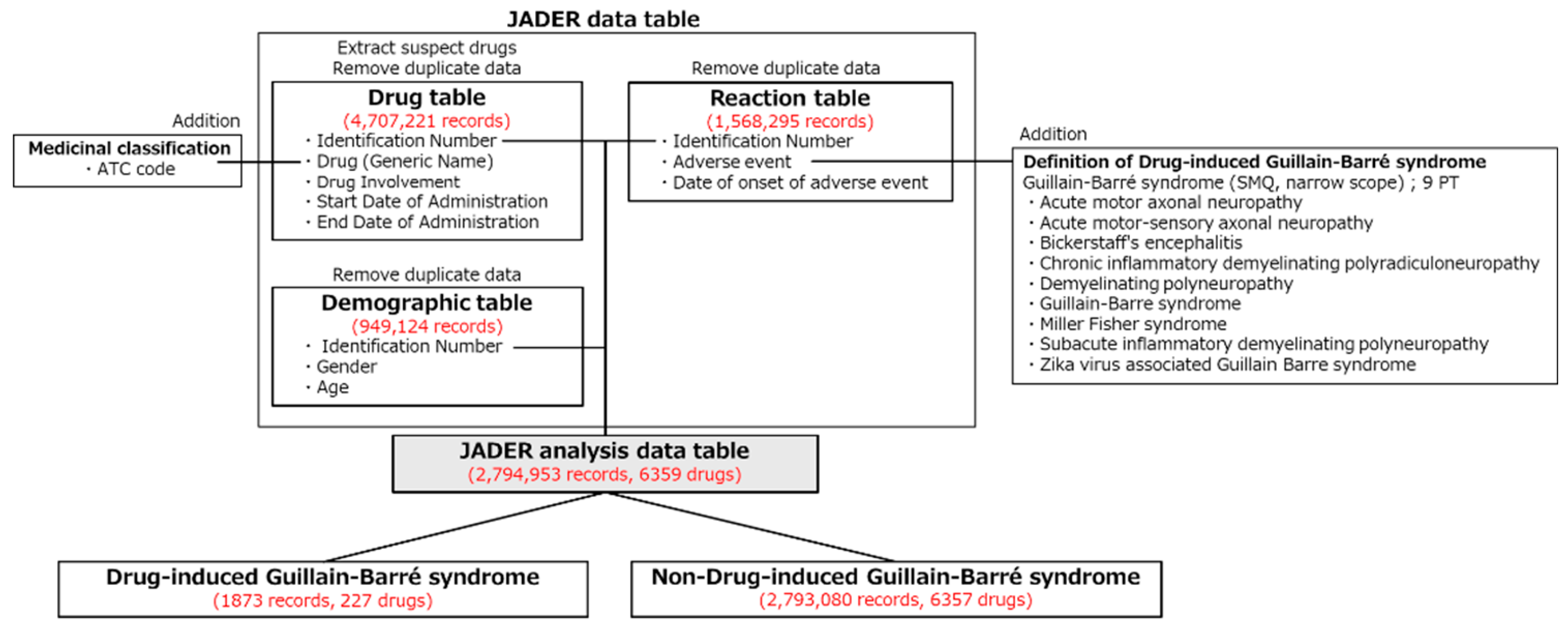
2.1. JADER Analysis Dataset
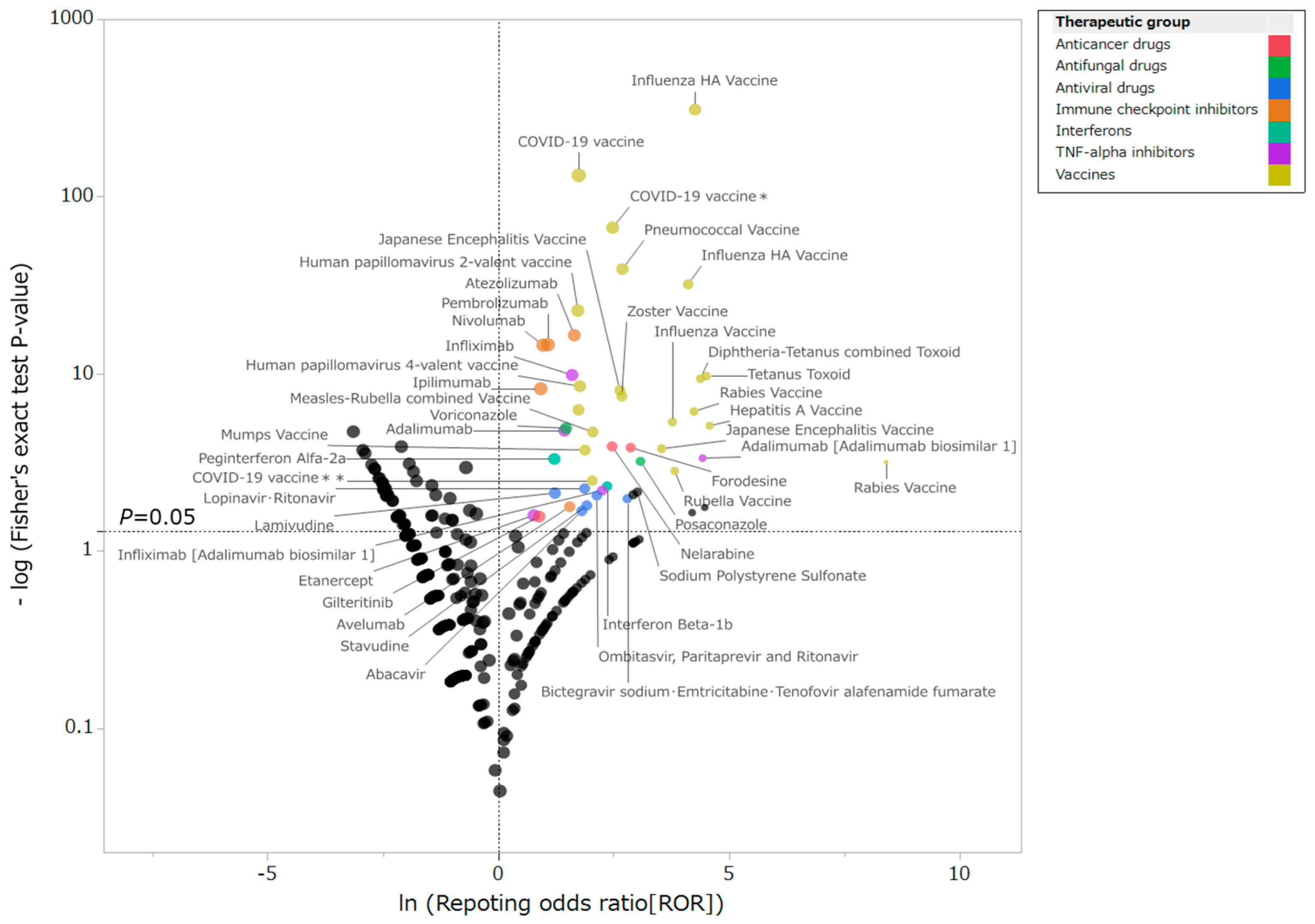
2.2. Drugs Associated with GBS and Patient Characteristics
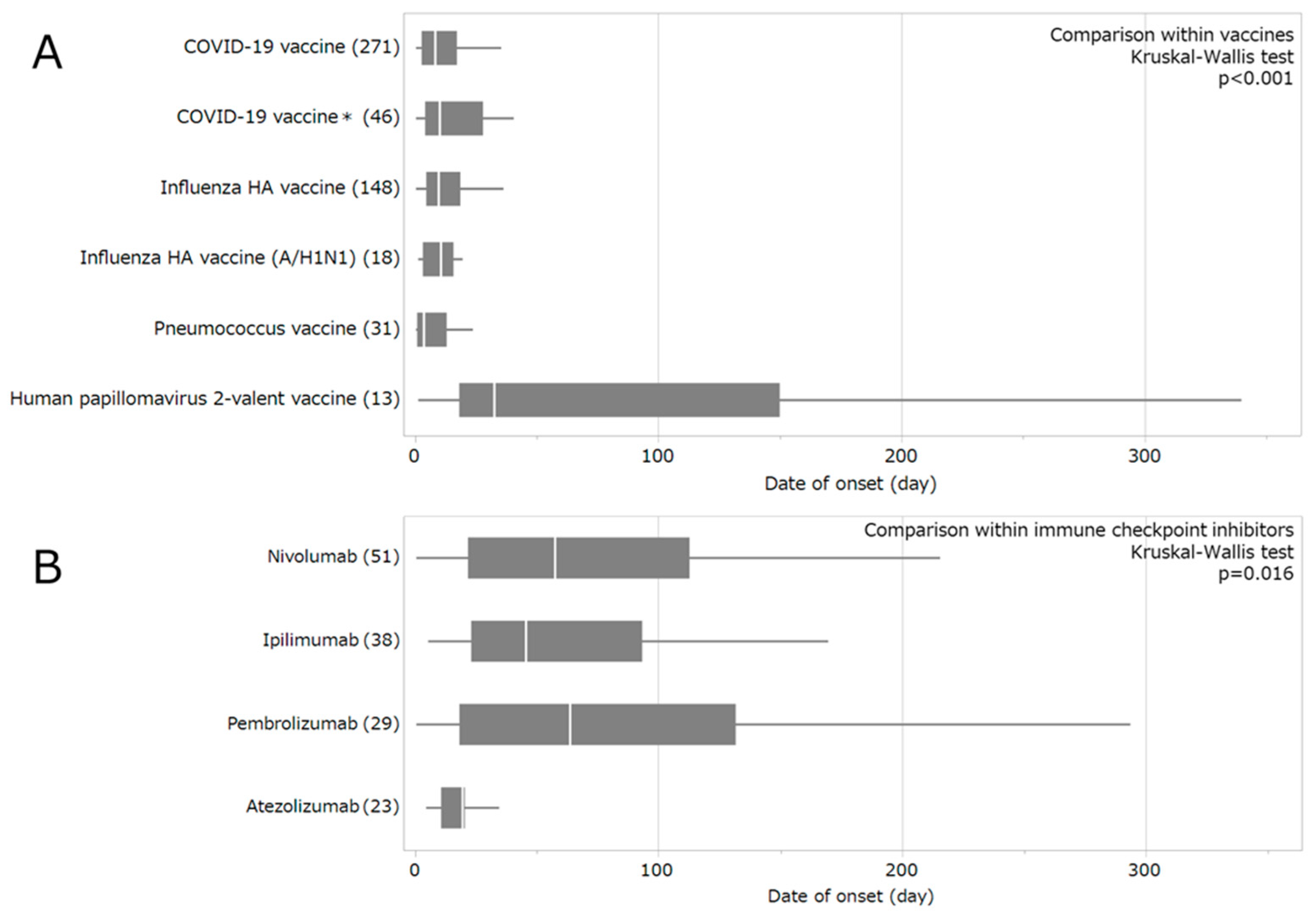
2.3. Time-to-Event Analysis and Weibull Distribution
3. Discussion
3.1. Drugs Associated with GBS
3.2. Timing of Vaccine- and Immune Checkpoint Inhibitor-Associated GBS Onset
3.3. Limitations
4. Materials and Methods
4.1. Identification of Drugs Associated with Drug-Induced GBS
4.1.1. Construction of the JADER Analysis Data Table
4.1.2. Drugs Associated with Drug-Induced GBS and Patient Characteristics
4.2. Onset Timing of Drug-Induced GBS
4.2.1. Construction of a Data Table for Time-to-Onset Analysis
4.2.2. Evaluation of Adverse Event Onset Profiles
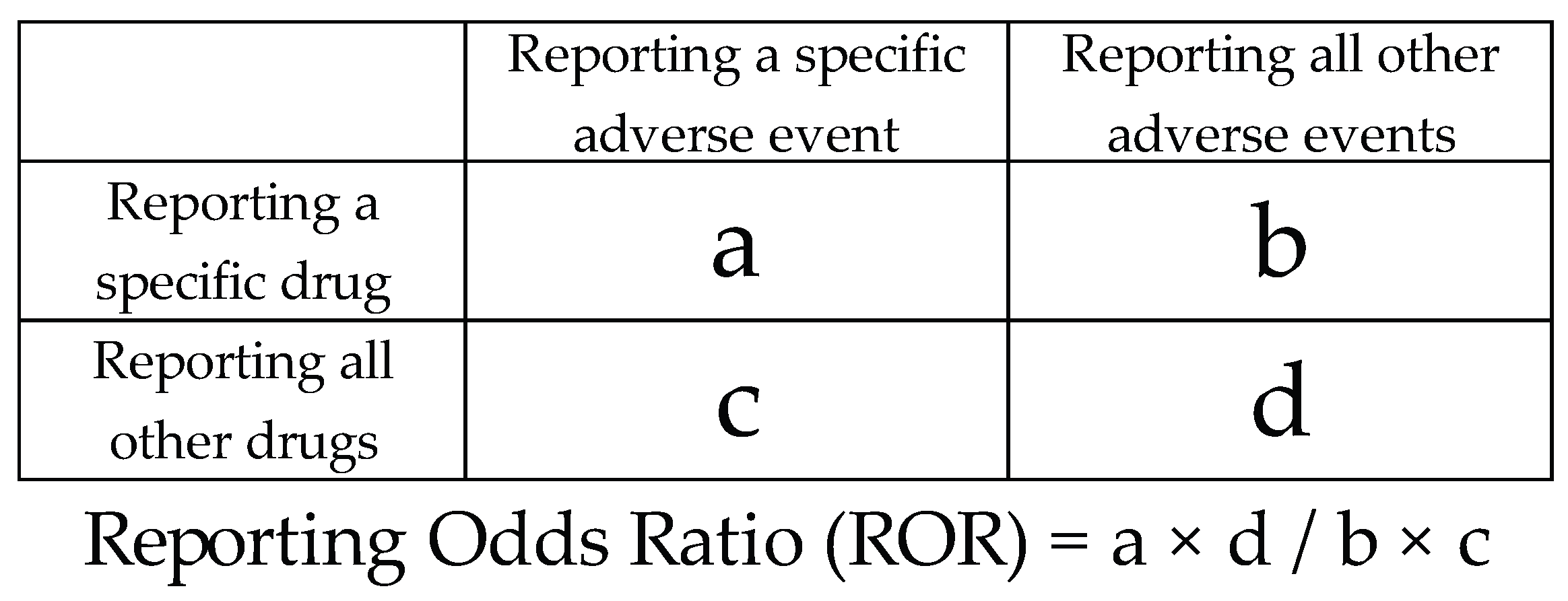
4.3. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ATC | Anatomical Therapeutic Chemical Classification System |
| COVID-19 vaccine | Coronavirus disease 2019 vaccine. |
| CTLA-4 | Cytotoxic T lymphocyte-associated protein 4 |
| GBS | Guillain-Barré syndrome |
| ID | Identification number |
| IrAEs | Immune-mediated adverse events |
| JADER | Japan adverse event reporting database |
| MedDRA/J | Medical Dictionary for Regulatory Activities / Japanese version |
| PD-1 | Programmed death-1 |
| PD-L1 | Programmed death-Ligand 1 |
| PMDA | Pharmaceuticals and Medical Devices Agency |
| ROR | Reporting odds ratio |
| SMQ | Standard MedDRA query |
| TNF-α | Tumor necrosis factor alpha |
References
- van Doorn, P.A.; Van den Bergh, P.Y.K.; Hadden, R.D.M.; Avau, B.; Vankrunkelsven, P.; Attarian, S.; Blomkwist-Markens, P.H.; Cornblath, D.R.; Goedee, H.S.; Harbo, T.; et al. European Academy of Neurology/Peripheral Nerve Society Guideline on diagnosis and treatment of Guillain-Barré syndrome. Eur J Neurol. 2023, 30, 3646–3674. [Google Scholar] [CrossRef] [PubMed]
- Shahrizaila, N.; Lehmann, H.C.; Kuwabara, S. Guillain-Barré syndrome. Lancet. 2021, 397, 1214–1228. [Google Scholar] [CrossRef]
- Yuki, N.; Hartung, H.P. Guillain-Barré syndrome. N Engl J Med. 2012, 366, 2294–2304. [Google Scholar] [CrossRef]
- Yuki, N.; Taki, T.; Inagaki, F.; Kasama, T.; Takahashi, M.; Saito, K.; Handa, S.; Miyatake, T. A bacterium lipopolysaccharide that elicits Guillain-Barré syndrome has a GM1 ganglioside-like structure. J Exp Med. 1993, 178, 1771–1775. [Google Scholar] [CrossRef] [PubMed]
- Hahn, A.F. Guillain-Barré syndrome. Lancet 1998, 352, 635–641. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Zhao, C.; Bao, Y.; Liu, Y.; Liang, Y.; Wei, J.; Liu, G.; Wang, J.; Zhan, S.; Wang, S.; et al. Variation in worldwide incidence of Guillain-Barré syndrome: a population-based study in urban China and existing global evidence. Front Immunol. 2024, 15, 1415986. [Google Scholar] [CrossRef]
- van den Berg, B.; Walgaard, C.; Drenthen, J.; Fokke, C.; Jacobs, B.C.; van Doorn, P.A. Guillain-Barré syndrome: pathogenesis, diagnosis, treatment and prognosis. Nat Rev Neurol. 2014, 10, 469–482. [Google Scholar] [CrossRef] [PubMed]
- Haber, P.; Sejvar, J.; Mikaeloff, Y.; DeStefano, F. Vaccines and Guillain-Barré syndrome. Drug Saf. 2009, 32, 309–323. [Google Scholar] [CrossRef]
- Schneiderbauer, R.; Schneiderbauer, M.; Wick, W.; Enk, A.H.; Haenssle, H.A.; Hassel, J.C. PD-1 Antibody-induced Guillain-Barré Syndrome in a Patient with Metastatic Melanoma. Acta Derm Venereol. 2017, 97, 395–396. [Google Scholar] [CrossRef]
- Fusaroli, M.; Salvo, F.; Begaud, B.; AlShammari, T.M.; Bate, A.; Battini, V.; Brueckner, A.; Candore, G.; Carnovale, C.; Crisafulli, S.; et al. The Reporting of a Disproportionality Analysis for Drug Safety Signal Detection Using Individual Case Safety Reports in PharmacoVigilance (READUS-PV): Development and Statement. Drug Saf. 2024, 47, 575–584. [Google Scholar] [CrossRef]
- Fusaroli, M.; Salvo, F.; Begaud, B.; AlShammari, T.M.; Bate, A.; Battini, V.; Brueckner, A.; Candore, G.; Carnovale, C.; Crisafulli, S.; et al. The Reporting of A Disproportionality Analysis for DrUg Safety Signal Detection Using Individual Case Safety Reports in PharmacoVigilance (READUS-PV): Explanation and Elaboration. Drug Saf. 2024, 47, 585–599. [Google Scholar] [CrossRef] [PubMed]
- Nomura, K.; Takahashi, K.; Hinomura, Y.; Kawaguchi, G.; Matsushita, Y.; Marui, H.; Anzai, T.; Hashiguchi, M.; Mochizuki, M. Effect of database profile variation on drug safety assessment: an analysis of spontaneous adverse event reports of Japanese cases. Drug Des Devel Ther. 2015, 9, 3031–3041. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, A.; Uesawa, Y. Steroid-Induced Thrombosis: A Comprehensive Analysis Using the FAERS Database. Pharmaceuticals (Basel). 2025, 18, 1463. [Google Scholar] [CrossRef]
- Yamaoka, K.; Masago, S.; Uchida, M.; Uesawa, Y.; Muroi, N.; Shimizu, T. Adverse events of antibody-drug conjugates: comparative analysis of agents with a common payload using the adverse event spontaneous reporting database. Oncologist. 2025, 30, oyaf298. [Google Scholar] [CrossRef]
- Toriumi, S.; Kobayashi, A.; Sueki, H.; Yamamoto, M.; Uesawa, Y. Exploring the Mechanisms Underlying Drug-Induced Fractures Using the Japanese Adverse Drug Event Reporting Database. Pharmaceuticals (Basel). 2021, 14, 1299. [Google Scholar] [CrossRef]
- Pharmaceutical and Medical Devices Agency. Available online: https://www.pmda.go.jp/safety/info-services/drugs/adr-info/suspected-adr/0005.html (accessed on 16 March 2025).
- Toriumi, S.; Mimori, R.; Sakamoto, H.; Sueki, H.; Yamamoto, M.; Uesawa, Y. Examination of Risk Factors and Expression Patterns of Atypical Femoral Fractures Using the Japanese Adverse Drug Event Report Database: A Retrospective Pharmacovigilance Study. Pharmaceuticals (Basel). 2023, 16, 626. [Google Scholar] [CrossRef]
- Pulendran, B.; Ahmed, R. Immunological mechanisms of vaccination. Nat Immunol. 2011, 12, 509–517. [Google Scholar] [CrossRef]
- Censi, S.; Bisaccia, G.; Gallina, S.; Tomassini, V.; Uncini, A. Guillain-Barré syndrome and COVID-19 vaccination: a systematic review and meta-analysis. J Neurol. 2024, 271, 1063–1071. [Google Scholar] [CrossRef]
- Patone, M.; Handunnetthi, L.; Saatci, D.; Pan, J.; Katikireddi, S.V.; Razvi, S.; Hunt, D.; Mei, X.W.; Dixon, S.; Zaccardi, F.; et al. Neurological complications after first dose of COVID-19 vaccines and SARS-CoV-2 infection. Nat Med. 2021, 12, 2144–2153. [Google Scholar] [CrossRef]
- Martín Arias, L.H.; Sanz, R.; Sáinz, M.; Treceño, C.; Carvajal, A. Guillain-Barré syndrome and influenza vaccines: A meta-analysis. Vaccine. 2015, 33, 3773–3778. [Google Scholar] [CrossRef] [PubMed]
- Nachamkin, I.; Shadomy, S.V.; Moran, A.P.; Cox, N.; Fitzgerald, C.; Ung, H.; Corcoran, A.T.; Iskander, J.K.; Schonberger, L.B.; Chen, R.T. Anti-ganglioside antibody induction by swine (A/NJ/1976/H1N1) and other influenza vaccines: insights into vaccine-associated Guillain-Barré syndrome. J Infect Dis. 2008, 198, 226–233. [Google Scholar] [CrossRef] [PubMed]
- Sato, S.; Katsuta, T.; Kawazoe, Y.; Takahashi, M.; Murata, F.; Maeda, M.; Fukuda, H.; Kamidani, S. Immune thrombocytopenic purpura and Guillain-Barré syndrome after 23-valent pneumococcal polysaccharide vaccination in Japan: The vaccine effectiveness, networking, and universal safety (VENUS) study. Vaccine. 2024, 42, 4–7. [Google Scholar] [CrossRef]
- Mbata, O.E.; Atiku, P.M. Rapid Onset of Guillain Barré Syndrome following Quadrivalent Human Papilloma Virus Vaccination in a Young Female: A Case Report. Ann Vaccines Immunization 2025, 9, 1026. [Google Scholar]
- Hegde, N.R.; Gore, M.M. Japanese encephalitis vaccines: Immunogenicity, protective efficacy, effectiveness, and impact on the burden of disease. Hum Vaccin Immunother. 2017, 13, 1–18. [Google Scholar] [CrossRef]
- Nelson, J.C.; Ulloa-Pérez, E.; Yu, O.; Cook, A.J.; Jackson, M.L.; Belongia, E.A.; Daley, M.F.; Harpaz, R.; Kharbanda, E.O.; Klein, N.P.; et al. Active Postlicensure Safety Surveillance for Recombinant Zoster Vaccine Using Electronic Health Record Data. Am J Epidemiol. 2023, 192, 205–216. [Google Scholar] [CrossRef]
- Di Pietrantonj, C.; Rivetti, A.; Marchione, P.; Debalini, M.G.; Demicheli, V. Vaccines for measles, mumps, rubella, and varicella in children. Cochrane Database Syst Rev. 2021, 11, CD004407. [Google Scholar] [CrossRef] [PubMed]
- Pan, M.; Sun, T.; Zhu, W.; Liu, H.; Dong, H. Guillain Barré syndrome after combined diphtheria, tetanus, and acellular pertussis (DTaP) vaccine: A rare pediatric case report and review of literature. Hum Vaccin Immunother. 2023, 19, 2261199. [Google Scholar] [CrossRef]
- Souayah, N.; Nasar, A.; Suri, M.F.; Qureshi, AI. Guillain-Barré syndrome after vaccination in United States: data from the Centers for Disease Control and Prevention/Food and Drug Administration Vaccine Adverse Event Reporting System (1990-2005). J Clin Neuromuscul Dis. 2009, 11, 1–6. [Google Scholar] [CrossRef]
- Principi, N.; Esposito, S. Vaccine-preventable diseases, vaccines and Guillain-Barre' syndrome. Vaccine. 2019, 37, 5544–5550. [Google Scholar] [CrossRef] [PubMed]
- Leach, D.R.; Krummel, M.F.; Allison, J.P. Enhancement of antitumor immunity by CTLA-4 blockade. Science 1996, 271, 1734–1736. [Google Scholar] [CrossRef]
- Ishida, Y.; Agata, Y.; Shibahara, K.; Honjo, T. Induced expression of PD-1, a novel member of the immunoglobulin gene superfamily, upon programmed cell death. EMBO J. 1992, 11, 3887–3895. [Google Scholar] [CrossRef]
- Sonpavde, G.P.; Grivas, P.; Lin, Y.; Hennessy, D.; Hunt, J.D. Immune-related adverse events with PD-1 versus PD-L1 inhibitors: a meta-analysis of 8730 patients from clinical trials. Future Oncol. 2021, 17, 2545–2558. [Google Scholar] [CrossRef]
- Salim, A.; Tapia Rico, G.; Shaikh, A.; Brown, M.P. A systematic review of immune checkpoint inhibitor-related neurological adverse events and association with anti-neuronal autoantibodies. Expert Opin Biol Ther. 2021, 21, 1237–1251. [Google Scholar] [CrossRef] [PubMed]
- Möhn, N.; Beutel, G.; Gutzmer, R.; Ivanyi, P.; Satzger, I.; Skripuletz, T. Neurological Immune Related Adverse Events Associated with Nivolumab, Ipilimumab, and Pembrolizumab Therapy-Review of the Literature and Future Outlook. J Clin Med. 2019, 8, 1777. [Google Scholar] [CrossRef]
- Cuzzubbo, S.; Javeri, F.; Tissier, M.; Roumi, A.; Barlog, C.; Doridam, J.; Lebbe, C.; Belin, C.; Ursu, R.; Carpentier, A.F. Neurological adverse events associated with immune checkpoint inhibitors: Review of the literature. Eur J Cancer. 2017, 73, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Kyriazoglou, A.; Liontos, M.; Papadopoulos, C.; Bilali, A.; Kostouros, E.; Pagoni, S.; Doumas, K.; Dimopoulos, M.A.; Bamias, A. Guillain-Barré Syndrome Related to Nivolumab: Case Report of a Patient With Urothelial Cancer and Review of the Literature. Clin Genitourin Cancer. 2019, 17, e360–e364. [Google Scholar] [CrossRef] [PubMed]
- Oguri, T.; Sasada, S.; Shimizu, S.; Shigematsu, R.; Tsuchiya, Y.; Ishioka, K.; Takahashi, S.; Oki, K.; Kimura, Y.; Seki, R.; et al. A Case of Guillain-Barré Syndrome and Stevens-Johnson Syndrome/Toxic Epidermal Necrosis Overlap After Pembrolizumab Treatment. J Investig Med High Impact Case Rep. 2021, 9, 23247096211037462. [Google Scholar] [CrossRef]
- Kelly Wu, W.; Broman, K.K.; Brownie, E.R.; Kauffmann, R.M. Ipilimumab-induced Guillain-Barré Syndrome Presenting as Dysautonomia: An Unusual Presentation of a Rare Complication of Immunotherapy. J Immunother. 2017, 40, 196–199. [Google Scholar] [CrossRef]
- Yamanaka, N.; Oishi, M.; Shimizu, F.; Koga, M.; Kanda, T. Atezolizumab-induced Guillain-Barré syndrome-like acute demyelinating polyneuropathy responsive to steroid therapy: a case report. Rinsho Shinkeigaku. 2021, 61, 653–657. [Google Scholar] [CrossRef]
- Abrahao, A.; Tenório, P.H.M.; Rodrigues, M.; Mello, M.; Nascimento, OJM. Guillain-Barré syndrome and checkpoint inhibitor therapy: insights from pharmacovigilance data. BMJ Neurol Open. 2024, 6, e000544. [Google Scholar] [CrossRef]
- Johnson, D.B.; Manouchehri, A.; Haugh, A.M.; Quach, HT.; Balko, J.M.; Lebrun-Vignes, B.; Mammen, A.; Moslehi, J.J.; Salem, J.E. Neurologic toxicity associated with immune checkpoint inhibitors: a pharmacovigilance study. J Immunother Cancer. 2019, 7, 134. [Google Scholar] [CrossRef]
- Freeman, G.J.; Long, A.J.; Iwai, Y.; Bourque, K.; Chernova, T.; Nishimura, H.; Fitz, L.J.; Malenkovich, N.; Okazaki, T.; Byrne, M.C.; et al. Engagement of the PD-1 immunoinhibitory receptor by a novel B7 family member leads to negative regulation of lymphocyte activation. J Exp Med. 2000, 192, 1027–1034. [Google Scholar] [CrossRef] [PubMed]
- Buchbinder, E.I.; Desai, A. CTLA-4 and PD-1 Pathways: Similarities, Differences, and Implications of Their Inhibition. Am J Clin Oncol. 2016, 39, 98–106. [Google Scholar] [CrossRef]
- Zhang, H.; Shi, N.; Diao, Z.; Chen, Y.; Zhang, Y. Therapeutic potential of TNFα inhibitors in chronic inflammatory disorders: Past and future. Genes Dis. 2020, 8, 38–47. [Google Scholar] [CrossRef]
- Mohan, N.; Edwards, E.T.; Cupps, T.R.; Oliverio, P.J.; Sandberg, G.; Crayton, H.; Richert, J.R.; Siegel, J.N. Demyelination occurring during anti-tumor necrosis factor alpha therapy for inflammatory arthritides. Arthritis Rheum. 2001, 44, 2862–2869. [Google Scholar] [CrossRef]
- Silburn, S.; McIvor, E.; McEntegart, A.; Wilson, H. Guillain-Barré syndrome in a patient receiving anti-tumour necrosis factor alpha for rheumatoid arthritis: a case report and discussion of literature. Ann Rheum Dis. 2008, 67, 575–576. [Google Scholar] [CrossRef]
- Natividade, N.B.; Felix, P.A.O.; Lerer, C. Guillain-Barré syndrome in a patient on adalimumab for the treatment of psoriasis. An Bras Dermatol. 2017, 92, 85–87. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, S.D.C.; Vasconcelos, J.R.O.; Rezende, R.E.F.; Aprile, L.R.O.; Troncon, L.E.A. Guillain-Barré Syndrome in a Patient Receiving Anti-Tumor Necrosis Factor for Crohn Disease: Coincidence or Consequence? Am J Case Rep. 2024, 25, e943709. [Google Scholar] [CrossRef]
- Doden, M.H.; Manasra, M.R.; AbuIrayyeh, B.M.; Al-Ihribat, A.R.; Albandak, M. Guillain-Barrè syndrome after treatment with anti-tumour necrosis factor α (etanercept) in a rheumatoid arthritis patient: Case report and literature review. Sci Prog. 2024, 107, 368504241304203. [Google Scholar] [CrossRef]
- Billmeier, U.; Dieterich, W.; Neurath, M.F.; Atreya, R. Molecular mechanism of action of anti-tumor necrosis factor antibodies in inflammatory bowel diseases. World J Gastroenterol. 2016, 22, 9300–9313. [Google Scholar] [CrossRef] [PubMed]
- Beigel, F.; Schnitzler, F.; Paul Laubender, R.; Pfennig, S.; Weidinger, M.; Göke, B.; Seiderer, J.; Ochsenkühn, T.; Brand, S. Formation of antinuclear and double-strand DNA antibodies and frequency of lupus-like syndrome in anti-TNF-α antibody-treated patients with inflammatory bowel disease. Inflamm Bowel Dis. 2011, 17, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Kunchok, A.; Aksamit, A.J, Jr.; Davis, J.M., 3rd; Kantarci, O.H.; Keegan, BM.; Pittock, S.J.; Weinshenker, B.G.; McKeon, A. Association Between Tumor Necrosis Factor Inhibitor Exposure and Inflammatory Central Nervous System Events. JAMA Neurol. 2020, 77, 937–946. [Google Scholar] [CrossRef]
- Girgin, N.K.; İşçimen, R.; Yılmaz, E.; Kahveci, Ş.F.; Kutlay, O. Guillain-Barré Syndrome and Human Immunodeficiency Virus. Turk J Anaesthesiol Reanim. 2014, 42, 100–102. [Google Scholar] [CrossRef]
- Piliero, P.J.; Fish, D.G.; Preston, S.; Cunningham, D.; Kinchelow, T.; Salgo, M.; Qian, J.; Drusano, G.L. Guillain-Barré syndrome associated with immune reconstitution. Clin Infect Dis. 2003, 36, e111–e114. [Google Scholar] [CrossRef]
- Braish, J.S.; Kugler, E.; Jabbour, E.; Woodman, K.; Ravandi, F.; Nicholas, S.; Jain, N.; Kantarjian, H.; Sasaki, K. Incidence and Clinical Presentation of Severe Neurotoxicity from Nelarabine in Patients with T-Cell Acute Lymphoblastic Leukemia. Clin Lymphoma Myeloma Leuk. 2024, 24, 783–788. [Google Scholar] [CrossRef]
- Tsiodras, S.; Zafiropoulou, R.; Kanta, E.; Demponeras, C.; Karandreas, N.; Manesis, E.K. Painful peripheral neuropathy associated with voriconazole use. Arch Neurol. 2005, 62, 144–146. [Google Scholar] [CrossRef]
- Benitez, LL; Carver, PL. Adverse Effects Associated with Long-Term Administration of Azole Antifungal Agents. Drugs 2019, 79, 833–853. [Google Scholar] [CrossRef] [PubMed]
- Niazi, M.A.; Azhar, A.; Tufail, K.; Feyssa, E.L.; Penny, S.F.; McGregory, M.; Araya, V.; Ortiz, J.A. Guillain-Barre syndrome associated with peginterferon alfa-2a for chronic hepatitis C: A case report. World J Hepatol. 2010, 2, 162–166. [Google Scholar] [CrossRef] [PubMed]
- Ogunjimi, O.B.; Tsalamandris, G.; Paladini, A.; Varrassi, G.; Zis, P. Guillain-Barré Syndrome Induced by Vaccination Against COVID-19: A Systematic Review and Meta-Analysis. Cureus. 2023, 15, e37578. [Google Scholar] [CrossRef] [PubMed]
- Chalela, J.A.; Andrews, C.; Bashmakov, A.; Kapoor, N.; Snelgrove, D. Reports of Guillain-Barre Syndrome Following COVID-19 Vaccination in the USA: An Analysis of the VAERS Database. J Clin Neurol. 2023, 19, 179–185. [Google Scholar] [CrossRef]
- Schonberger, L.B.; Bregman, D.J.; Sullivan-Bolyai, J.Z.; Keenlyside, R.A.; Ziegler, D.W.; Retailliau, H.F.; Eddins, D.L.; Bryan, J.A. Guillain-Barre syndrome following vaccination in the National Influenza Immunization Program, United States, 1976--1977. Am J Epidemiol. 1979, 110, 105–123. [Google Scholar] [CrossRef] [PubMed]
- Lasky, T.; Terracciano, G.J.; Magder, L.; Koski, C.L.; Ballesteros, M.; Nash, D.; Clark, S.; Haber, P.; Stolley, P.D.; Schonberger, L.B.; et al. The Guillain-Barré syndrome and the 1992-1993 and 1993-1994 influenza vaccines. N Engl J Med. 1998, 339, 1797–1802. [Google Scholar] [CrossRef]
- Kwong, JC.; Vasa, PP.; Campitelli, MA.; Hawken, S.; Wilson, K.; Rosella, LC.; Stukel, TA.; Crowcroft, NS.; McGeer, AJ.; Zinman, L.; et al. Risk of Guillain-Barré syndrome after seasonal influenza vaccination and influenza health-care encounters: a self-controlled study. Lancet Infect Dis. 2013, 13, 769–776. [Google Scholar] [CrossRef]
- Kaneda, Y.; Hashimoto, T.; Kaneda, U.; Higuchi, Y.; Murakami, J.; Inada, M.; Senoo, Y.; Fujieda, T.; Murata, Y.; Tanimoto, T. Guillain-Barre Syndrome After COVID-19 Vaccination: A Secondary Analysis of Domestic Safety Data by the Japanese Government. Cureus. 2022, 14, e30905. [Google Scholar] [CrossRef] [PubMed]
- Ravishankar, N. Guillain-Barre Syndrome Following PCV Vaccine. Clin Surg. 2017, 2, 1413. [Google Scholar]
- Oliveira, M.; Marquez, P.; Ennulat, C.; Blanc, P.; Welsh, K.; Nair, N.; Taminato, M.; Moro, P.L. Post-licensure Safety Surveillance of 20-Valent Pneumococcal Conjugate Vaccine (PCV20) Among US Adults in the Vaccine Adverse Event Reporting System (VAERS). Drug Saf. 2025, 48, 279–286. [Google Scholar] [CrossRef]
- Boender, T.S.; Bartmeyer, B.; Coole, L.; Wichmann, O.; Harder, T. Risk of Guillain-Barré syndrome after vaccination against human papillomavirus: a systematic review and meta-analysis, 1 January 2000 to 4 April 2020. Euro Surveill. 2022, 27, 2001619. [Google Scholar] [CrossRef]
- Roodbol, J.; de Wit, M.C.; Walgaard, C.; de Hoog, M.; Catsman-Berrevoets, C.E.; Jacobs, B.C. Recognizing Guillain-Barre syndrome in preschool children. Neurology. 2011, 76, 807–810. [Google Scholar] [CrossRef]
- Hagen, K.M.; Ousman, S.S. The Neuroimmunology of Guillain-Barré Syndrome and the Potential Role of an Aging Immune System. Front Aging Neurosci. 2021, 12, 613628. [Google Scholar] [CrossRef] [PubMed]
- Sejvar, J.J.; Baughman, A.L.; Wise, M.; Morgan, O.W. Population incidence of Guillain-Barré syndrome: a systematic review and meta-analysis. Neuroepidemiology. 2011, 36, 123–133. [Google Scholar] [CrossRef]
- O'Hare, M.; Guidon, A.C. Peripheral nervous system immune-related adverse events due to checkpoint inhibition. Nat Rev Neurol. 2024, 20, 509–525. [Google Scholar] [CrossRef] [PubMed]
- Albarrán, V.; Chamorro, J.; Rosero, D.I.; Saavedra, C.; Soria, A.; Carrato, A.; Gajate, P. Neurologic Toxicity of Immune Checkpoint Inhibitors: A Review of Literature. Front Pharmacol. 2022, 13, 774170. [Google Scholar] [CrossRef]
- Fan, Q.; Hu, Y.; Wang, X.; Zhao, B. Guillain-Barré syndrome in patients treated with immune checkpoint inhibitors. J Neurol. 2021, 268, 2169–2174. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, X.; Zhao, C. Guillain-Barré Syndrome-Like Polyneuropathy Associated with Immune Checkpoint Inhibitors: A Systematic Review of 33 Cases. Biomed Res Int. 2021, 2021, 9800488. [Google Scholar] [CrossRef]
- Sun, X.; Roudi, R.; Dai, T.; Chen, S.; Fan, B.; Li, H.; Zhou, Y.; Zhou, M.; Zhu, B.; Yin, C.; et al. Immune-related adverse events associated with programmed cell death protein-1 and programmed cell death ligand 1 inhibitors for non-small cell lung cancer: a PRISMA systematic review and meta-analysis. BMC Cancer. 2019, 19, 558. [Google Scholar] [CrossRef] [PubMed]
- Kanaoka, K.; Matsumoto, K.; Shiroyama, T.; Tsukaguchi, A.; Shoshihara, N.; Moritomo, K.; Kinehara, Y.; Mihashi, Y.; Kuge, T.; Yoneda, M.; et al. A Multicenter, Retrospective, Real-World Study of Atezolizumab Plus Chemotherapy and Pembrolizumab Plus Chemotherapy for Older Patients With NSCLC. JTO Clin Res Rep. 2025, 6, 100891. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.S.; Irving, B.A.; Hodi, F.S. Molecular pathways: next-generation immunotherapy--inhibiting programmed death-ligand 1 and programmed death-1. Clin Cancer Res. 2012, 18, 6580–6587. [Google Scholar] [CrossRef] [PubMed]
- Paterson, A.M.; Brown, K.E.; Keir, M.E.; Vanguri, V.K.; Riella, L.V.; Chandraker, A.; Sayegh, M.H.; Blazar, B.R.; Freeman, G.J.; Sharpe, A.H. The programmed death-1 ligand 1: B7-1 pathway restrains diabetogenic effector T cells in vivo. J Immunol. 2011, 187, 1097–1105. [Google Scholar] [CrossRef]
- Yang, J.; Riella, L.V.; Chock, S.; Liu, T.; Zhao, X.; Yuan, X.; Paterson, A.M.; Watanabe, T.; Vanguri, V.; Yagita, H.; et al. The novel costimulatory programmed death ligand 1/B7.1 pathway is functional in inhibiting alloimmune responses in vivo. J Immunol. 2011, 187, 1113–1119. [Google Scholar] [CrossRef]
- Alemayehu, D. Evaluation of Reporting Bias in Postmarketing Risk Assessment Based on Spontaneous Reporting Systems. Pharm Med. 2009, 23, 195–200. [Google Scholar] [CrossRef]
- Caster, O.; Aoki, Y.; Gattepaille, L.M.; Grundmark, B. Disproportionality Analysis for Pharmacovigilance Signal Detection in Small Databases or Subsets: Recommendations for Limiting False-Positive Associations. Drug Saf. 2020, 43, 479–487. [Google Scholar] [CrossRef]
- Matsuda, S.; Aoki, K.; Kawamata, T.; Kimotsuki, T.; Kobayashi, T.; Kuriki, H.; Nakayama, T.; Okugawa, S.; Sugimura, Y.; Tomita, M.; et al. Bias in spontaneous reporting of adverse drug reactions in Japan. PLoS One. 2015, 10, e0126413. [Google Scholar] [CrossRef] [PubMed]
- Pariente, A.; Avillach, P.; Salvo, F.; Thiessard, F.; Miremont-Salamé, G.; Fourrier-Reglat, A.; Haramburu, F.; Bégaud, B.; Moore, N. Effect of competition bias in safety signal generation: analysis of a research database of spontaneous reports in France. Drug Saf. 2012, 35, 855–864. [Google Scholar] [CrossRef]
- García-Abeijon, P.; Costa, C.; Taracido, M.; Herdeiro, M.T.; Torre, C.; Figueiras, A. Factors Associated with Underreporting of Adverse Drug Reactions by Health Care Professionals: A Systematic Review Update. Drug Saf. 2023, 46, 625–636. [Google Scholar] [CrossRef]
- ATC/DDD Index. Available online: https://atcddd.fhi.no/atc_ddd_index/ (accessed on 3 March 2025).
- MedDRA Japanese Maintenance Organization. Available online: https://www.pmrj.jp/jmo/php/indexj.php (accessed on 16 February 2025).
- Medical Dictionary for Regulatory Activities. Available online: https://www.meddra.org/standardised-meddra-queries (accessed on 10 February 2025).
- Hirooka, T.; Yamada, M. Evaluation of AEs Risk Using the “Japanese Adverse Drug Event Report Database” of PMDA. In Proceedings of the SAS User General Assembly, Tokyo, Japan, 1–3 December 2012; pp. 263–270. [Google Scholar]
- Hosoya, R.; Ishii-Nozawa, R.; Kurosaki, K.; Uesawa, Y. Analysis of Factors Associated with Hiccups Using the FAERS Database. Pharmaceuticals (Basel). 2021, 15, 27. [Google Scholar] [CrossRef]
- Ruxton, G.D.; Neuhäuser, M. Review of alternative approaches to calculation of a confidence interval for the odds ratio of a 2 × 2 contingency table. Methods Ecol Evol. 2013, 4, 9–13. [Google Scholar] [CrossRef]
- Yajima, A.; Uesawa, Y. A Comprehensive Analysis of Adverse Events Associated with HER2 Inhibitors Approved for Breast Cancer Using the FDA Adverse Event Report System (FAERS). Pharmaceuticals (Basel). 2025, 18, 1510. [Google Scholar] [CrossRef] [PubMed]
- van Puijenbroek, E.P.; Bate, A.; Leufkens, HG.; Lindquist, M.; Orre, R.; Egberts, AC. A comparison of measures of disproportionality for signal detection in spontaneous reporting systems for adverse drug reactions. Pharmacoepidemiol Drug Saf. 2002, 11, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Rothman, K.J.; Lanes, S.; Sacks, S.T. The reporting odds ratio and its advantages over the proportional reporting ratio. Pharmacoepidemiol Drug Saf. 2004, 13, 519–523. [Google Scholar] [CrossRef]
- Harpaz, R.; DuMouchel, W.; LePendu, P.; Bauer-Mehren, A.; Ryan, P.; Shah, N.H. Performance of pharmacovigilance signal-detection algorithms for the FDA adverse event reporting system. Clin Pharmacol Ther. 2013, 93, 539–546. [Google Scholar] [CrossRef]
- European Medicines Agency. “Guideline on the Use of Statistical Signal Detection Methods in the Eudravigilance Data Analysis System”. Available online: https://www.ema.europa.eu/en/documents/regulatory-procedural-guideline/draft-guideline use-statistical-signal-detection-methods-eudravigilance-data-analysis-system_en.pdf (accessed on 5 July 2022).
- Kurosaki, K.; Uesawa, Y. Molecular Initiating Events Associated with Drug-Induced Liver Malignant Tumors: An Integrated Study of the FDA Adverse Event Reporting System and Toxicity Predictions. Biomolecules 2021, 11, 944. [Google Scholar] [CrossRef] [PubMed]
- Toriumi, S.; Shimokawa, K.; Yamamoto, M.; Uesawa, Y. Development of a Medication-Related Osteonecrosis of the Jaw Prediction Model Using the FDA Adverse Event Reporting System Database and Machine Learning. Pharmaceuticals (Basel). 2025, 18, 423. [Google Scholar] [CrossRef] [PubMed]
- Toriumi, S.; Kobayashi, A.; Uesawa, Y. Comprehensive Study of the Risk Factors for Medication-Related Osteonecrosis of the Jaw Based on the Japanese Adverse Drug Event Report Database. Pharmaceuticals (Basel). 2020, 13, 467. [Google Scholar] [CrossRef]
- Kan, Y.; Doi, M.; Uesawa, Y. Investigation of the total anticholinergic load of reported anticholinergic drug-related adverse events using the Japanese adverse drug event report database: a retrospective pharmacovigilance study. J Pharm Health Care Sci. 2025, 11, 8. [Google Scholar] [CrossRef]
- Kan, Y.; Nagai, J.; Uesawa, Y. Evaluation of antibiotic-induced taste and smell disorders using the FDA adverse event reporting system database. Sci Rep. 2021, 11, 9625. [Google Scholar] [CrossRef] [PubMed]
- Nakao, S.; Hasegawa, S.; Umetsu, R.; Shimada, K.; Mukai, R.; Tanaka, M.; Matsumoto, K.; Yoshida, Y.; Inoue, M.; Satake, R.; et al. Pharmacovigilance study of anti-infective-related acute kidney injury using the Japanese adverse drug event report database. BMC Pharmacol Toxicol. 2021, 22, 47. [Google Scholar] [CrossRef]
- Hasegawa, S.; Ikesue, H.; Satake, R.; Inoue, M.; Yoshida, Y.; Tanaka, M.; Matsumoto, K.; Wakabayashi, W.; Oura, K.; Muroi, N.; et al. Osteonecrosis of the Jaw Caused by Denosumab in Treatment-Naïve and Pre-Treatment with Zoledronic Acid Groups: A Time-to-Onset Study Using the Japanese Adverse Drug Event Report (JADER) Database. Drugs Real World Outcomes. 2022, 9, 659–665. [Google Scholar] [CrossRef]
- Sauzet, O.; Carvajal, A.; Escudero, A.; Molokhia, M.; Cornelius, V.R. Illustration of the weibull shape parameter signal detection tool using electronic healthcare record data. Drug Saf. 2013, 36, 995–1006. [Google Scholar] [CrossRef]
| Drug | N | Gender male/female/Unknown and not reported |
Age median (min–max) |
|---|---|---|---|
| Vaccines | |||
| COVID-19 vaccine | 354 | 165/185/4 | 55 (5–95) |
| COVID-19 vaccine* | 98 | 56/42/0 | 45 (15–85) |
| Influenza HA vaccine | 250 | 131/115/4 | 55 (5–85) |
| Influenza HA vaccine (A/H1N1) | 23 | 13/10/0 | 65 (5–75) |
| Human papillomavirus 2-valent vaccine | 56 | 0/56/0 | 15 (15–35) |
| Human papillomavirus 4-valent vaccine | 19 | 0/19/0 | 15 (15–45) |
| Pneumococcal vaccine | 50 | 28/15/7 | 75 (45–85) |
| Hepatitis B vaccine | 14 | 7/7/0 | 25 (5–35) |
| Japanese encephalitis vaccine | 10 | 5/5/0 | 10 (5–15) |
| Varicella zoster vaccine | 9 | 4/5/0 | 65 (55–75) |
| Immune checkpoint inhibitors | |||
| Nivolumab | 91 | 62/26/3 | 65 (35–85) |
| Pembrolizumab | 76 | 44/27/5 | 75 (25–85) |
| Ipilimumab | 54 | 37/17/0 | 65 (45–85) |
| Atezolizumab | 43 | 26/14/3 | 75 (45–95) |
| Avelumab | 4 | 3/0/1 | 75 (75–75) |
| Others | |||
| Infliximab | 26 | 22/4/0 | 65 (15–75) |
| Drug | Onset days | Weibull distribution | N | |||
|---|---|---|---|---|---|---|
| Median (min–max) |
Scale Parameter α (95%CI) |
Shape Parameter β (95%CI) |
Shape Pattern | |||
| Vaccines | ||||||
| COVID-19 vaccine | 8.5 (0.5 –225.5) |
13.41 (11.21–16.00) |
0.71 (0.65–0.77) |
Early failure type | 271 | |
| COVID-19 vaccine* | 10.5 (0.5–347.5) |
27.43 (16.27–45.28) |
0.61 (0.49–0.75) |
Early failure type | 46 | |
| Influenza HA vaccine | 10.5 (0.5–212.5) |
15.44 (12.66–18.76) |
0.87 (0.77–0.98) |
Early failure type | 148 | |
| Influenza HA vaccine (A/H1N1) | 10.5 (1.5–51.5) |
13.36 (8.23–21.12) |
1.09 (0.74–1.51) |
Random failure type | 18 | |
| Pneumococcal vaccine | 4.5 (0.5–212.5) |
10.83 (5.44–20.94) |
0.57 (0.43–0.72) |
Early failure type | 31 | |
| Human papillomavirus 2-valent vaccine | 32.5 (1.5–352.5) |
81.92 (35.55–179.17) |
0.77 (0.48–1.13) |
Random failure type | 13 | |
| Immune checkpoint inhibitors | ||||||
| Nivolumab | 57.5 (0.5–359.5) |
83.75 (62.50–110.90) |
1.03 (0.82 –1.27) |
Random failure type | 51 | |
| Ipilimumab | 45.5 (5.5–169.5) |
65.52 (50.42–84.10) |
1.35 (1.03 –1.71) |
Wear-out failure type | 38 | |
| Pembrolizumab | 63.5 (0.5–314.5) |
92.13 (57.51–144.19) |
0.86 (0.63–1.14) |
Random failure type | 29 | |
| Atezolizumab | 19.5 (4.5–147.5) |
33.68 (21.36–51.90) |
1.02 (0.74–1.33) |
Random failure type | 23 | |
| Drug | Gender | Onset days | Weibull distribution | N | P value# | |||
|---|---|---|---|---|---|---|---|---|
| Median (Min -Max) |
Scale Parameter α (95%CI) |
Shape Parameter β (95%CI) |
||||||
| Vaccine | ||||||||
| COVID-19 vaccine | 0.432 | |||||||
| Male | 8.5 (0.5–175.5) | 13.64 (10.77–17.18) | 0.81 (0.71–0.92) | 122 | ||||
| Female | 8 (0.5–242.5) | 13.44 (10.28–17.48) | 0.66 (0.58–0.74) | 144 | ||||
| COVID-19 vaccine* | 0.005 | |||||||
| Male | 16.5 (1.5–287.5) | 40.01 (22.62–68.82) | 0.70 (0.53–0.90) | 30 | ||||
| Female | 4.5 (0.5–347.5) | 11.49 (4.04–31.15) | 0.53 (0.37–0.72) | 16 | ||||
| Influenza HA vaccine | 0.416 | |||||||
| Male | 9.5 (0.5–71.5) | 13.71 (10.74–17.36) | 0.98 (0.83–1.15) | 79 | ||||
| Female | 10 (0.5–108.5) | 16.20 (12.11– 21.46) | 0.89 (0.74–1.06) | 68 | ||||
| Influenza HA vaccine (A/H1N1) | 0.032 | |||||||
| Male | 14.5 (2.5–51.5) | 19.65 (11.20–33.32) | 1.33 (0.77–2.03) | 10 | ||||
| Female | 5.5 (1.5–16.5) | 7.03 (3.57–13.24) | 1.28 (0.68–2.11) | 8 | ||||
| Human papillomavirus 2-valent vaccine | - | |||||||
| Male | - | - | - | - | ||||
| Female | 32.5 (1.5–352.5) | 81.92 (35.55–179.17) | 0.77 (0.48–1.13) | 13 | ||||
| Pneumococcal vaccine | 0.496 | |||||||
| Male | 3 (0.5–23.5) | 6.07 (3.09 - 11.39) | 0.80 (0.53–1.12) | 18 | ||||
| Female | 4.5 (0.5–31.5) | 8.23 (3.98 - 16.23) | 0.98 (0.58–1.47) | 11 | ||||
| Immune checkpoint inhibitors | ||||||||
| Nivolumab | 0.422 | |||||||
| Male | 57.5 (0.5–359.5) | 76.83 (53.49–108.62) | 1.01 (0.77–1.29) | 35 | ||||
| Female | 67 (7.5–215.5) | 99.97 (59.12–163.25) | 1.09 (0.71–1.59) | 16 | ||||
| Ipilimumab | 0.649 | |||||||
| Male | 46.5 (5.5–112.5) | 59.91 (45.95–76.97) | 1.64 (1.17–2.20) | 26 | ||||
| Female | 44.5 (7.5–169.5) | 77.04 (42.21–135.01) | 1.13 (0.68–1.71) | 12 | ||||
| Pembrolizumab | 0.853 | |||||||
| Male | 63.5 (1.5–314.5) | 104.55(55.05–191.02) | 0.89 (0.57–1.28) | 16 | ||||
| Female | 97 (0.5–293.5) | 81.32 (35.13–179.83) | 0.85 (0.49–1.33) | 11 | ||||
| Atezolizumab | 0.203 | |||||||
| Male | 20.5 (4.5–147.5) | 46.27 (23.87–85.81) | 0.97 (0.61–1.43) | 13 | ||||
| Female | 15.5 (8.5–20.5) | 16.97 (14.36–19.85) | 4.85 (2.63–7.91) | 9 | ||||
| Drug | Age | Onset days | Weibull distribution | N | P value# | |||
|---|---|---|---|---|---|---|---|---|
| Median (Min -Max) |
Scale Parameter α (95%CI) |
Shape Parameter β (95%CI) |
||||||
| Vaccine | ||||||||
| COVID-19 vaccine | 0.610 | |||||||
| <20y | 9.5 (0.5–153.5) | 14.96 (7.54–28.65) | 0.75 (0.52–1.00) | 19 | ||||
| ≧20y | 7.5 (0.5–242.5) | 13.41 (11.12–16.12) | 0.71 (0.65–0.77) | 250 | ||||
| COVID-19 vaccine* | 0.524 | |||||||
| <20y | 347.5 (347.5–347.5) | - | - | 1 | ||||
| ≧20y | 9.5 (0.5–287.5) | 24.57 (14.81–39.98) | 0.64 (0.51–0.78) | 45 | ||||
| Influenza HA vaccine | 0.311 | |||||||
| <20y | 12.5 (1.5–108.5) | 18.52 (12.42–27.18) | 0.98 (0.75–1.23) | 31 | ||||
| ≧20y | 9.5 (0.5–90.5) | 13.94 (11.27–17.15) | 0.92 (0.80–1.06) | 116 | ||||
| Influenza HA vaccine (A/H1N1) | 0.333 | |||||||
| <20y | 16.5 (16.5–16.5) | - | - | 1 | ||||
| ≧20y | 10.5 (1.5–51.5) | 12.98 (7.73–21.16) | 1.06 (0.71–1.47) | 17 | ||||
| Human papillomavirus 2-valent vaccine | 0.894 | |||||||
| <20y | 32.5 (1.5–352.5) | 82.62 (33.25–193.62) | 0.74 (0.45–1.11) | 12 | ||||
| ≧20y | 52.5 (52.5–52.5) | - | - | 1 | ||||
| Pneumococcal vaccine | - | |||||||
| <20y | - | - | - | 0 | ||||
| ≧20y | 3.5 (0.5–31.5) | 6.86 (4.25–10.79) | 0.86 (0.63–1.12) | 29 | ||||
| Immune checkpoint inhibitors | ||||||||
| Nivolumab | - | |||||||
| <20y | - | - | - | 0 | ||||
| ≧20y | 57.5 (0.5–359.5) | 83.75 (62.50–110.90) | 1.03 (0.82–1.27) | 51 | ||||
| Ipilimumab | - | |||||||
| <20y | - | - | - | 0 | ||||
| ≧20y | 45.5 (5.5–169.5) | 65.52 (50.42–84.10) | 1.35 (1.03–1.71) | 38 | ||||
| Pembrolizumab | - | |||||||
| <20y | - | - | - | 0 | ||||
| ≧20y | 63.5 (0.5–314.5) | 90.05 (55.02–143.74) | 0.84 (0.61–1.12) | 28 | ||||
| Atezolizumab | - | |||||||
| <20y | - | - | - | 0 | ||||
| ≧20y | 19.5 (4.5–147.5) | 33.68 (21.36–51.90) | 1.02 (0.74–1.33) | 23 | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




