Submitted:
28 February 2026
Posted:
04 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
Materials
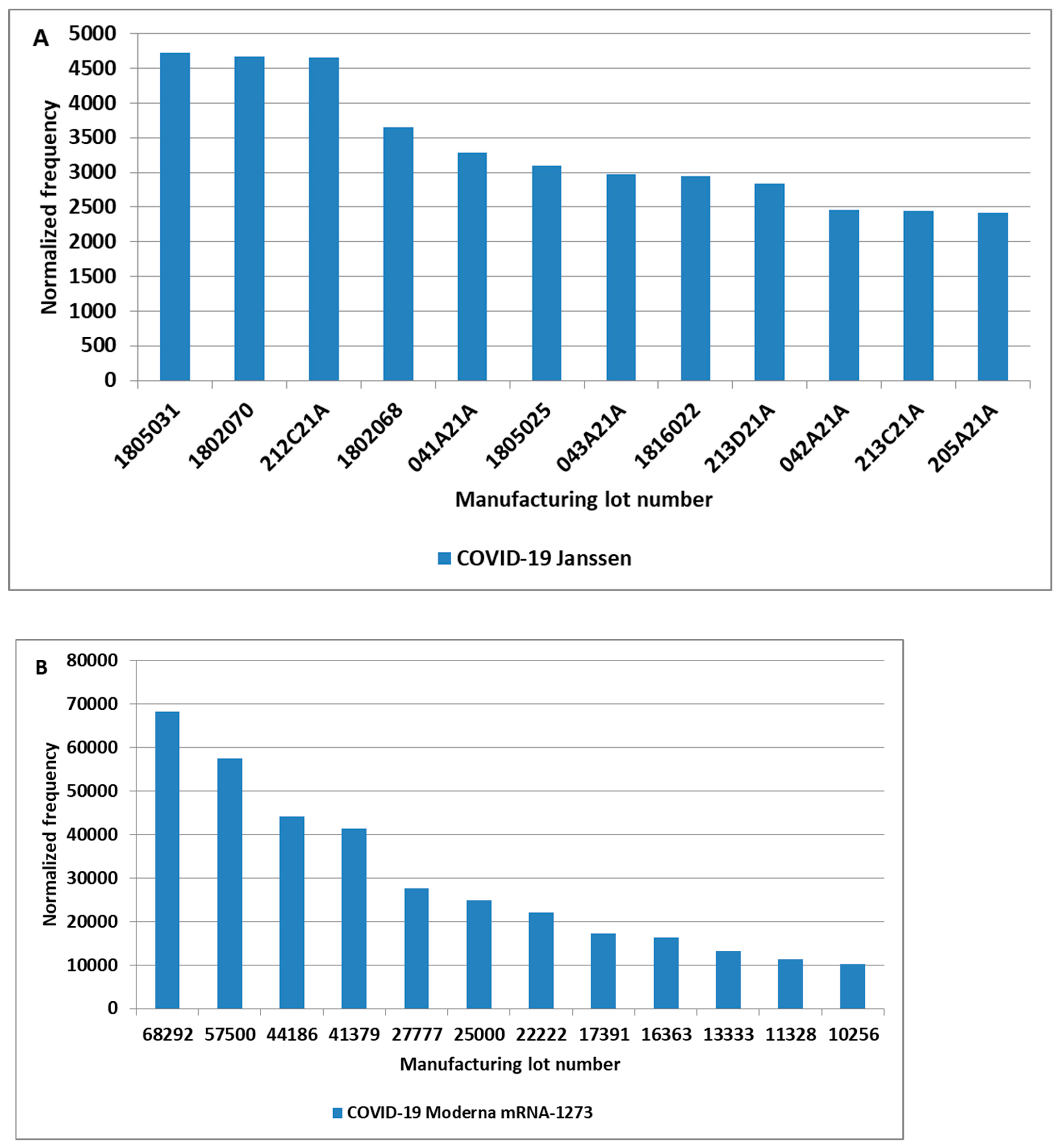
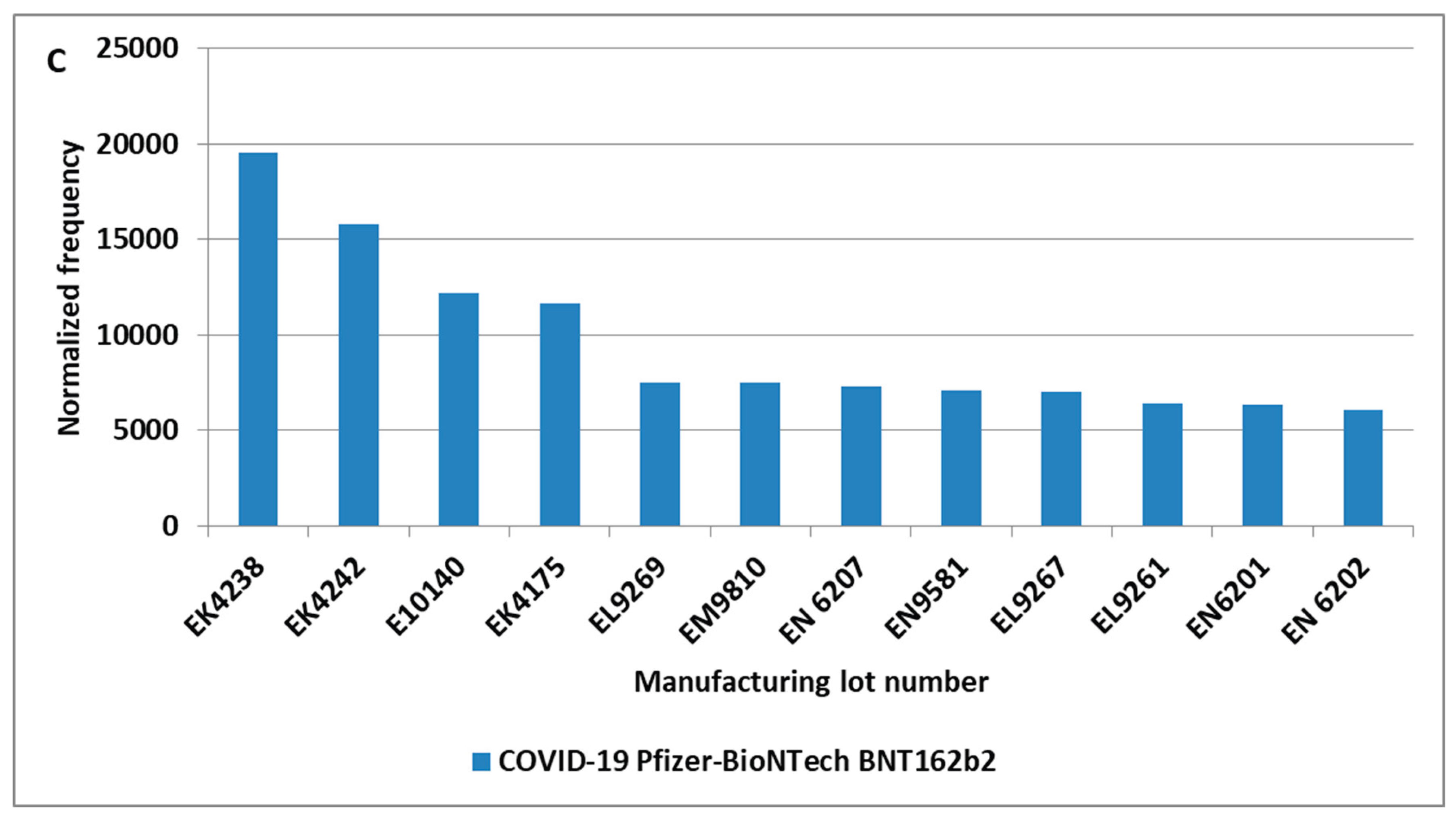
3. Results
4. Discussion
4.1. Frequent Adverse Events in the Age Group 80 to 89
4.2. COVID-19 Breakthrough Infection
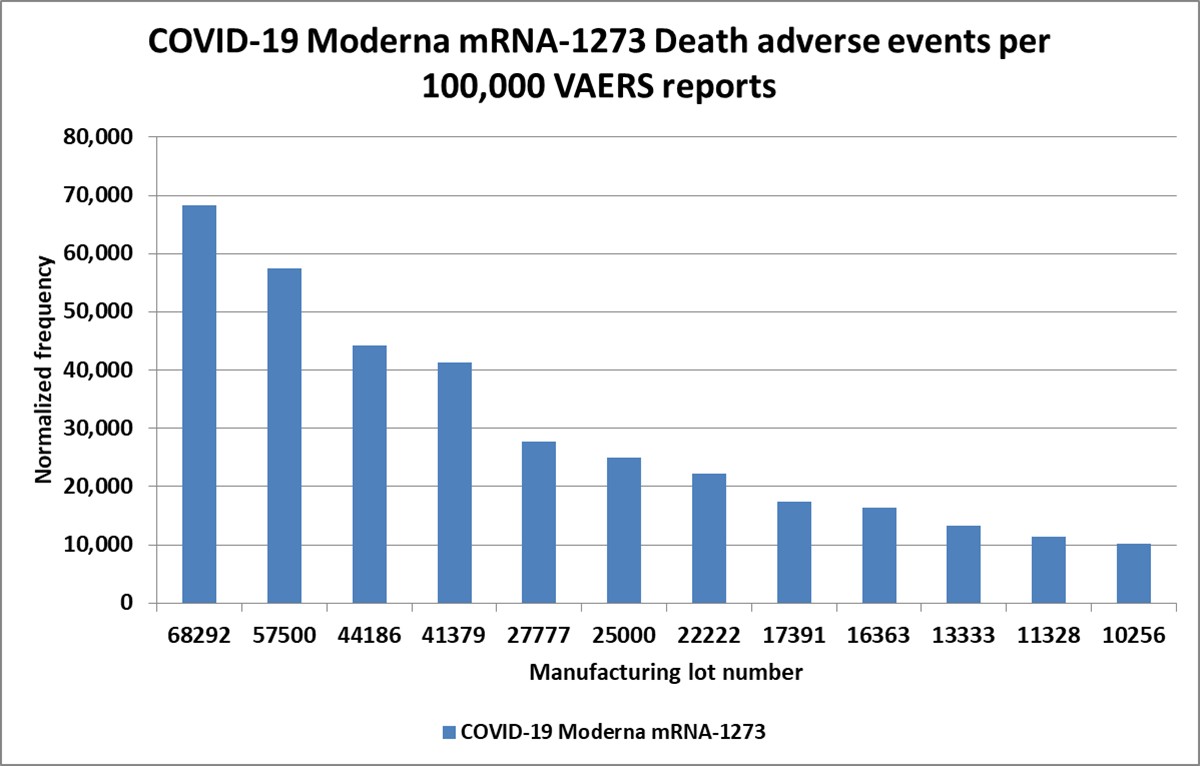
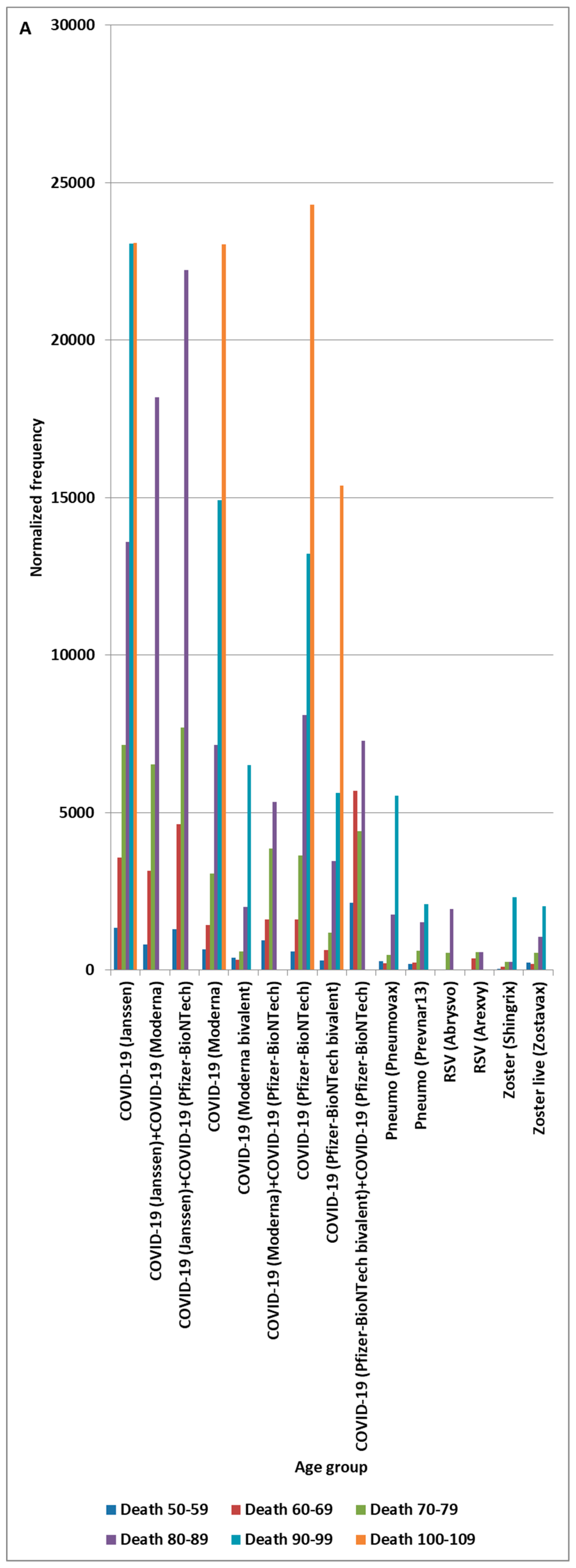
4.3. Death Adverse Event
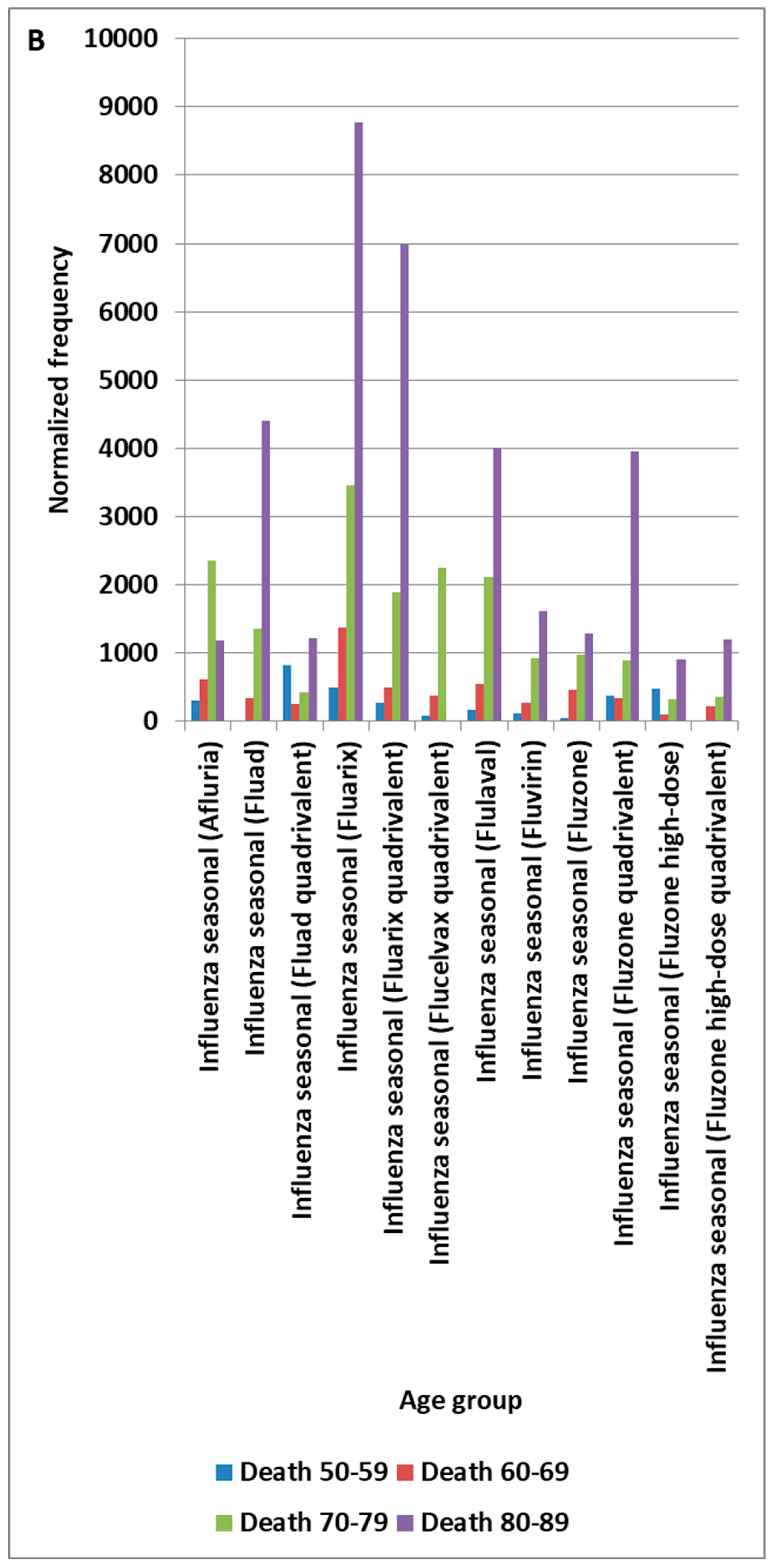
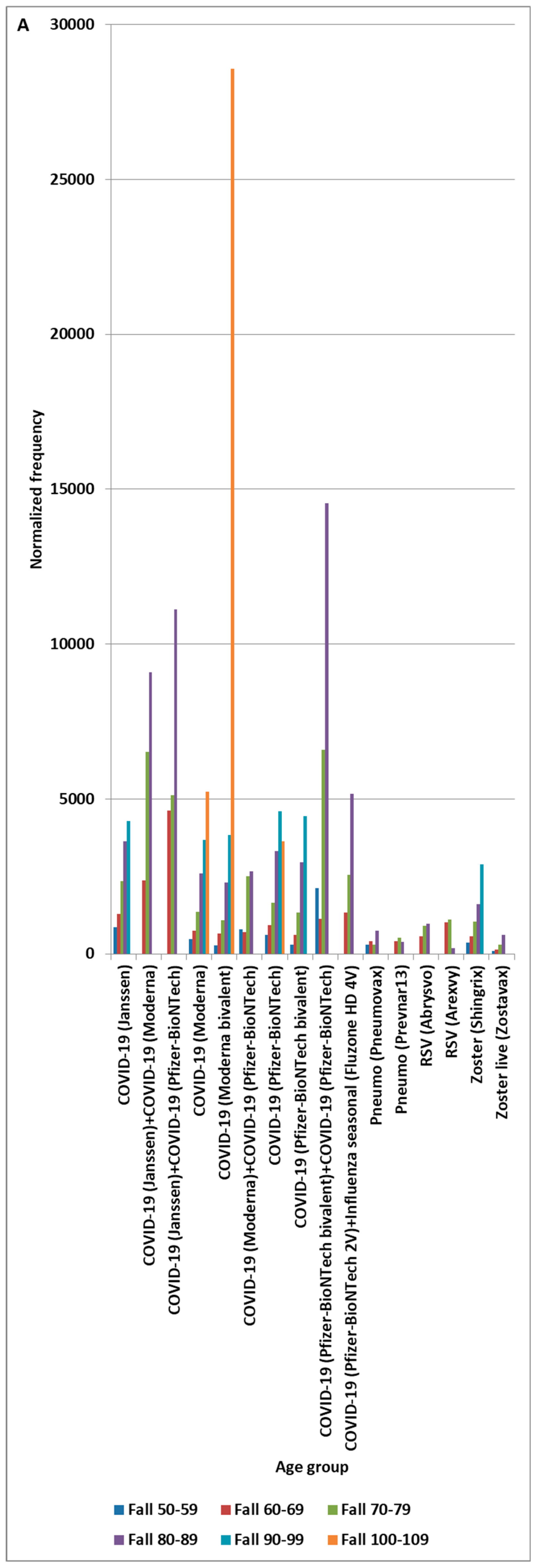
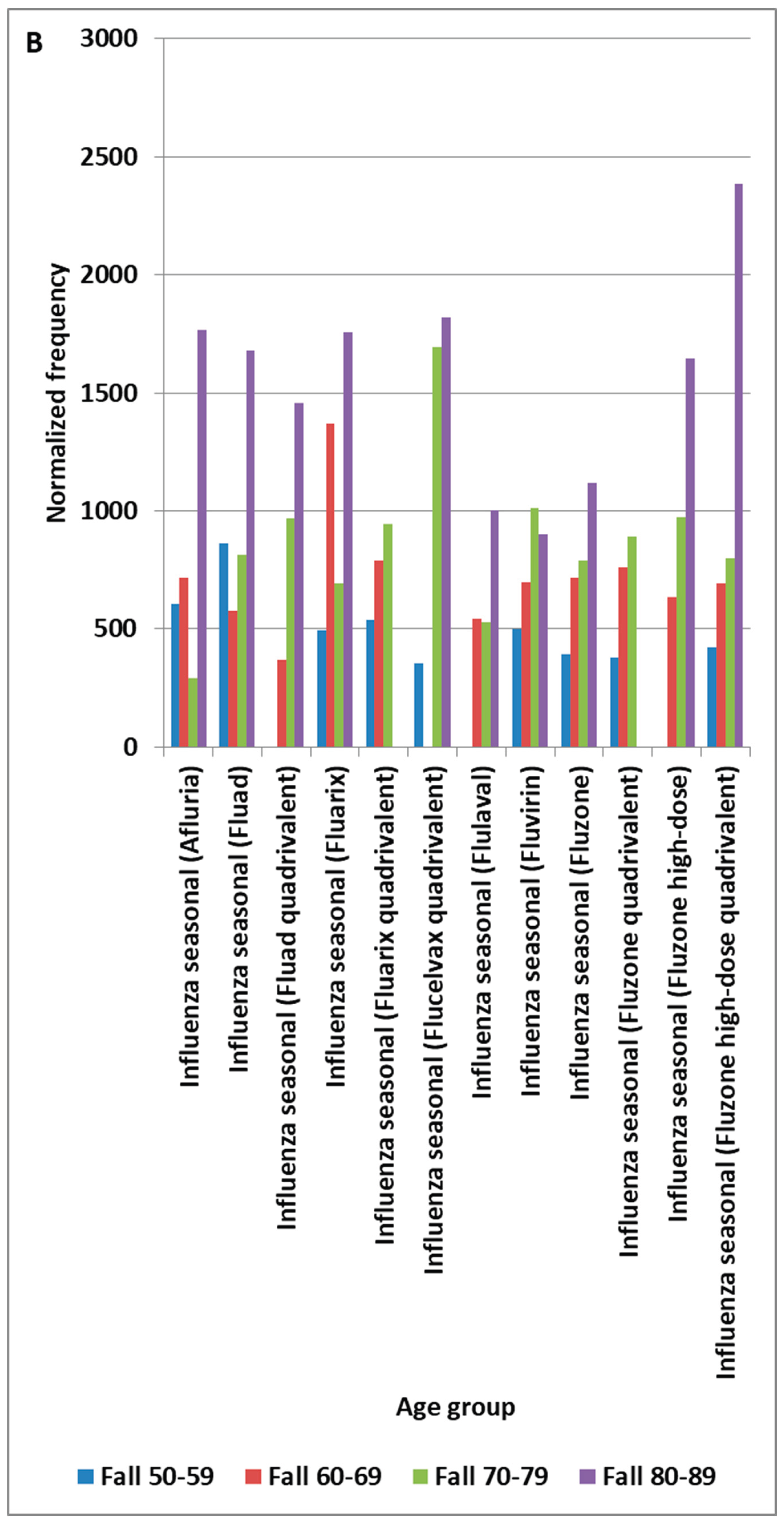
4.4. Fall Adverse Event
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Ethical approval
References
- Collier, DA; Ferreira, IATM; Kotagiri, P; et al. Age-related immune response heterogeneity to SARS-CoV-2 vaccine BNT162b2. Nature 2021, 596, 417–422. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, T; Tober-Lau, P; Hillus, D; et al. Delayed Antibody and T-Cell Response to BNT162b2 Vaccination in the Elderly, Germany. Emerg Infect Dis J. 2021, 27, 2174. [Google Scholar] [CrossRef]
- Canaday, DH; Carias, L; Oyebanji, OA; et al. Reduced BNT162b2 Messenger RNA Vaccine Response in Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2)–Naive Nursing Home Residents. Clin Infect Dis. 2021, 73, 2112–2115. [Google Scholar] [CrossRef] [PubMed]
- Demaret, J; Corroyer-Simovic, B; Alidjinou, EK; et al. Impaired Functional T-Cell Response to SARS-CoV-2 After Two Doses of BNT162b2 mRNA Vaccine in Older People. Front Immunol. 2021, 12-2021. Available online: https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2021.778679. [CrossRef]
- Breznik, JA; Zhang, A; Huynh, A; et al. Antibody Responses 3-5 Months Post-Vaccination with mRNA-1273 or BNT163b2 in Nursing Home Residents. J Am Med Dir Assoc. 2021, 22, 2512–2514. [Google Scholar] [CrossRef]
- Björk, J; Dietler, D; Bonander, C; et al. Vaccine protection against COVID-19 mortality in relation to time since last booster dose among nursing home residents in Sweden – A case-control study over 35 months. Vaccine 2026, 71, 128043. [Google Scholar] [CrossRef]
- Yang, XH; Bao, WJ; Zhang, H; Fu, SK; Jin, HM. The Efficacy of SARS-CoV-2 Vaccination in the Elderly: A Systemic Review and Meta-analysis. J Gen Intern Med. 2023. [Google Scholar] [CrossRef]
- VAERS. Vaccine Adverse Event Reporting System; U.S. Department of Health & Human Services, 2025. Available online: https://vaers.hhs.gov/data/datasets.html (accessed on 10 November 2025).
- Ricke, DO. VAERS-Tools. 2022. Available online: https://github.com/doricke/VAERS-Tools (accessed on 10 November 2025).
- Ricke, DO. Cardiac adverse events post-vaccination. Brain Heart 2025, 3, 1–15. [Google Scholar] [CrossRef]
- Ricke, DO. Menstrual adverse events post-COVID-19 and human papillomavirus immunization. Microbes Immun. 2025. [Google Scholar] [CrossRef]
- Geier, M R; Stanbro, H; Merril, C R. Endotoxins in commercial vaccines. Appl Environ Microbiol. 1978, 36, 445–449. [Google Scholar] [CrossRef] [PubMed]
- Brito, LA; Singh, M. COMMENTARY: Acceptable Levels of Endotoxin in Vaccine Formulations During Preclinical Research. J Pharm Sci. 2011, 100, 34–37. [Google Scholar] [CrossRef] [PubMed]
- Costa, JP; Jesus, S; Colaço, M; Duarte, A; Soares, E; Borges, O. Endotoxin contamination of nanoparticle formulations: A concern in vaccine adjuvant mechanistic studies. Vaccine 2023, 41, 3481–3485. [Google Scholar] [CrossRef] [PubMed]
- Jaume, M.; Yip, M.S.; Cheung, C.Y.; et al. Anti-Severe Acute Respiratory Syndrome Coronavirus Spike Antibodies Trigger Infection of Human Immune Cells via a pH- and Cysteine Protease-Independent FcγR Pathway. J Virol. 2011, 85, 10582–10597. [Google Scholar] [CrossRef]
- Ricke, DO. Two Different Antibody-Dependent Enhancement (ADE) Risks for SARS-CoV-2 Antibodies. Front Immunol. 2021, 12, 443. [Google Scholar] [CrossRef]
- Ricke, DO. Antibodies and infected monocytes and macrophages in COVID-19 patients. AIMS Allergy Immunol. 2022, 6, 64–70. [Google Scholar] [CrossRef]
- Wan, Y; Shang, J; Sun, S; et al. Molecular Mechanism for Antibody-Dependent Enhancement of Coronavirus Entry. J Virol. 2022, 94, e02015-19. [Google Scholar] [CrossRef]
- Liu, JY; Chen, TJ; Hou, MC. Does COVID-19 vaccination cause excess deaths? J Chin Med Assoc. 2021, 84. Available online: https://journals.lww.com/jcma/Fulltext/2021/09000/Does_COVID_19_vaccination_cause_excess_deaths_.2.aspx. [CrossRef]
- Torjesen, I. Covid-19: Pfizer-BioNTech vaccine is “likely” responsible for deaths of some elderly patients, Norwegian review finds. BMJ. 2021, 373, n1372. [Google Scholar] [CrossRef]
- Kakeya, H; Nitta, T; Kamijima, Y; Miyazawa, T. Significant Increase in Excess Deaths after Repeated COVID-19 Vaccination in Japan. JMA J. 2025, 8, 584–586. [Google Scholar] [CrossRef]
- Levi, R; Mansuri, F; Jordan, MM; Ladapo, JA. Twelve-Month All-Cause Mortality after Initial COVID-19 Vaccination with Pfizer-BioNTech or mRNA-1273 among Adults Living in Florida. medRxiv 2025. [Google Scholar] [CrossRef]
- Speicher, DJ; Rose, J; McKernan, K. Quantification of residual plasmid DNA and SV40 promoter-enhancer sequences in Pfizer/BioNTech and Moderna modRNA COVID-19 vaccines from Ontario, Canada. Autoimmunity 2025, 58, 2551517. [Google Scholar] [CrossRef] [PubMed]
- Ricke, DO. Rare dizziness, syncope, loss of consciousness, seizure, and risk of falling after vaccination. AIMS Allergy Immunol. 2023, 7, 164–175. [Google Scholar] [CrossRef]
| Adverse event | COVID-19 (Janssen) | COVID-19 (Moderna) | COVID-19 (Moderna bivalent) | COVID-19 (Pfizer-BioNTech) | COVID-19 (Pfizer-BioNTech bivalent) | Top 50 |
|---|---|---|---|---|---|---|
| COVID-19 | 30,540 | 15,462 | 15,502 | 20,500 | 18495 | JmMpP |
| SARS-CoV-2 test positive | 25,263 | 12,054 | 12,816 | 15,586 | 17651 | JmMpP |
| Dyspnoea | 13,588 | 6752 | 4834 | 9652 | 6680 | JmMpP |
| Fatigue | 8707 | 9157 | 8902 | 8036 | 4571 | JmMpP |
| Asthenia | 9234 | 7476 | 5679 | 7848 | 8509 | JmMpP |
| Pyrexia | 7387 | 8229 | 6983 | 8085 | 5555 | JmMpP |
| Death | 13,588 | 7147 | 1995 | 8089 | 3445 | JmMpP |
| Cough | 9036 | 4045 | 6753 | 5746 | 7383 | JmMpP |
| Expired product administered | 791 | 5410 | 11,588 | 1603 | 9845 | mM P |
| Product storage error | 1451 | 3903 | 4297 | 2342 | 13994 | mMpP |
| Headache | 4815 | 6305 | 5602 | 5334 | 2812 | JmMpP |
| Pain | 4617 | 4903 | 5909 | 4134 | 3445 | JmMpP |
| Pain in extremity | 4881 | 6088 | 5141 | 4615 | 2039 | JmMpP |
| Acute respiratory failure | 4089 | 2061 | 3146 | 2873 | 9704 | JmMpP |
| Dizziness | 4485 | 4638 | 3607 | 5142 | 2672 | JmMpP |
| Chills | 3891 | 5895 | 3146 | 4215 | 3305 | JmMpP |
| Nausea | 4485 | 4839 | 3530 | 4538 | 3023 | JmMpP |
| Malaise | 3759 | 2889 | 4758 | 4522 | 3305 | JmMpP |
| Hypoxia | 5343 | 2173 | 2686 | 2836 | 5977 | JmMpP |
| Condition aggravated | 4815 | 2647 | 2609 | 3224 | 4992 | JmMpP |
| Vaccine breakthrough infection | 5277 | 2539 | 2609 | 3048 | 2742 | JmMpP |
| Arthralgia | 2572 | 3549 | 3914 | 3273 | 1828 | JmMpP |
| Fall | 3627 | 2606 | 2302 | 3309 | 2953 | JmMpP |
| COVID-19 pneumonia | 5540 | 1961 | 1074 | 3485 | 2672 | Jm pP |
| Vomiting | 3166 | 3009 | 2455 | 3416 | 2531 | JmMpP |
| Diarrhoea | 3562 | 3180 | 2302 | 3428 | 1969 | JmMpP |
| Chest X-ray abnormal | 4485 | 1771 | 1688 | 2424 | 3375 | JmMpP |
| Extra dose administered | 1121 | 1334 | 4451 | 1285 | 4219 | M P |
| Myalgia | 1781 | 3419 | 2686 | 2489 | 1195 | mMp |
| Rash | 1385 | 3501 | 1611 | 2461 | 1969 | mMpP |
| Pneumonia | 3166 | 1879 | 1381 | 2718 | 1687 | Jm pP |
| Confusional state | 2770 | 1886 | 1534 | 2167 | 2461 | JmMpP |
| Decreased appetite | 2968 | 2382 | 1611 | 2767 | 984 | Jm pP |
| Atrial fibrillation | 2506 | 1659 | 1458 | 2424 | 2390 | JmMpP |
| Injection site pain | 1978 | 2457 | 1841 | 906 | 2109 | JmM P |
| Gait disturbance | 2044 | 1588 | 1841 | 1881 | 1687 | JmMpP |
| Peripheral swelling | 2440 | 2158 | 1995 | 1424 | 984 | JmM |
| Feeling abnormal | 1583 | 2102 | 2302 | 1816 | 1125 | mM |
| Pulmonary embolism | 2836 | 1107 | 997 | 1787 | 1898 | J P |
| Pruritus | 857 | 3818 | 1074 | 2028 | 843 | m p |
| Chest pain | 1715 | 1487 | 1841 | 1954 | 1336 | mMp |
| Cerebrovascular accident | 2440 | 1510 | 690 | 1848 | 1687 | Jm pP |
| Acute kidney injury | 2506 | 1017 | 920 | 1477 | 1969 | J P |
| Erythema | 1187 | 3143 | 1458 | 1461 | 632 | mM |
| Anticoagulant therapy | 2638 | 913 | 1151 | 1073 | 2039 | J P |
| Unevaluable event | 2968 | 1856 | 383 | 1885 | 703 | Jm p |
| Inappropriate schedule of product administration | 395 | 1935 | 1688 | 2301 | 1476 | mMp |
| Oropharyngeal pain | 1319 | 820 | 1918 | 1020 | 2672 | M P |
| Sepsis | 2308 | 816 | 1074 | 1048 | 2109 | J P |
| Injection site erythema | 263 | 3161 | 1841 | 624 | 914 | mM |
| Respiratory tract congestion | 791 | 678 | 1611 | 722 | 2039 | M |
| Injection site swelling | 329 | 1979 | 1688 | 453 | 1195 | mM |
| Adverse event | Influenza seasonal (Fluzone high-dose) | Pneumo (Pneumovax) | Pneumo (Prevnar13) | Zoster (Shingrix) | Zoster live (Zostavax) | Top 50 |
|---|---|---|---|---|---|---|
| Injection site erythema | 9978 | 25,530 | 28,231 | 13,499 | 12,782 | IOPSZ |
| Erythema | 7749 | 21,378 | 19,391 | 9769 | 8134 | IOPSZ |
| Injection site pain | 11,093 | 18,074 | 16,444 | 14,068 | 4770 | IOPSZ |
| Injection site swelling | 8439 | 21,472 | 17,395 | 9010 | 5932 | IOPSZ |
| Pain in extremity | 12,951 | 12,364 | 12,452 | 15,333 | 3363 | IOPSZ |
| Pain | 10,350 | 10,382 | 10,076 | 12,867 | 11,192 | IOPSZ |
| Pyrexia | 11,677 | 11,845 | 9410 | 9895 | 2568 | IOPSZ |
| Rash | 4299 | 3445 | 7889 | 10,907 | 15,902 | IOPSZ |
| Herpes zoster | 106 | 235 | 95 | 7556 | 32,782 | SZ |
| Pruritus | 3980 | 4907 | 9885 | 7714 | 10,275 | IOPSZ |
| Injection site warmth | 4989 | 10,335 | 11,501 | 5153 | 4036 | IOPSZ |
| Chills | 11,411 | 2642 | 5893 | 9326 | 1039 | IOPSZ |
| Peripheral swelling | 3237 | 9202 | 8935 | 5026 | 366 | IOPS |
| Headache | 6157 | 1793 | 4372 | 8409 | 3792 | IOPSZ |
| Fatigue | 5414 | 1840 | 4847 | 10,116 | 1834 | IOPSZ |
| Skin warm | 4299 | 6606 | 7414 | 3446 | 1896 | IOPSZ |
| Injection site pruritus | 2388 | 2501 | 8935 | 4331 | 4403 | IOPSZ |
| Asthenia | 5732 | 2737 | 3992 | 5501 | 2568 | IOPSZ |
| Swelling | 3078 | 6984 | 5513 | 3035 | 1651 | IOPSZ |
| Nausea | 5891 | 2548 | 3707 | 6165 | 1773 | IOPSZ |
| Dizziness | 4670 | 2595 | 3231 | 4552 | 3302 | IOPSZ |
| Malaise | 3397 | 3303 | 3326 | 4204 | 2385 | IOPSZ |
| Myalgia | 3343 | 2312 | 3897 | 5279 | 1590 | IOPSZ |
| Arthralgia | 2600 | 1604 | 3422 | 4805 | 1773 | IOPSZ |
| Mobility decreased | 3556 | 2878 | 2756 | 3793 | 733 | IOPS |
| Cellulitis | 636 | 7126 | 4182 | 885 | 550 | OP |
| Dyspnoea | 3927 | 2548 | 3802 | 1549 | 1345 | IOPSZ |
| Injection site rash | 1433 | 1179 | 4277 | 2402 | 3608 | I PSZ |
| Rash erythematous | 1804 | 1226 | 2756 | 2971 | 4036 | I PSZ |
| Vomiting | 5520 | 1321 | 1996 | 2307 | 856 | I PS |
| Urticaria | 2388 | 1038 | 2091 | 2655 | 2507 | I PSZ |
| Diarrhoea | 3450 | 566 | 2281 | 2845 | 978 | I PS |
| Tremor | 4989 | 943 | 855 | 2655 | 672 | I S |
| Injection site reaction | 1645 | 2925 | 1615 | 1517 | 2140 | IOP Z |
| Musculoskeletal pain | 3237 | 1793 | 1901 | 1738 | 1039 | IOPSZ |
| Injected limb mobility decreased | 2282 | 2831 | 2091 | 1896 | 122 | IOPS |
| Oedema peripheral | 796 | 6134 | 665 | 126 | 1406 | O Z |
| Extra dose administered | 3450 | 519 | 1235 | 2877 | 672 | I S |
| Tenderness | 1008 | 3445 | 1996 | 1201 | 978 | OP |
| Blister | 265 | 330 | 380 | 2023 | 5382 | SZ |
| Back pain | 1698 | 1274 | 1045 | 2023 | 2140 | I SZ |
| Rash pruritic | 1008 | 188 | 1330 | 2529 | 3058 | PSZ |
| Cough | 2813 | 1415 | 2376 | 758 | 550 | IOP |
| Pneumonia | 583 | 3916 | 2661 | 316 | 428 | OP |
| Decreased appetite | 1910 | 1179 | 2376 | 1802 | 550 | I PS |
| Muscular weakness | 1804 | 991 | 2091 | 1960 | 795 | I PS |
| Injection site induration | 690 | 1746 | 2756 | 695 | 1590 | OP Z |
| Neck pain | 1645 | 991 | 1996 | 1738 | 795 | I PS |
| Feeling abnormal | 1380 | 802 | 1235 | 2307 | 795 | I PS |
| Paraesthesia | 1433 | 519 | 570 | 1580 | 2262 | I SZ |
| Condition aggravated | 955 | 991 | 475 | 1580 | 1896 | SZ |
| Hypoaesthesia | 1857 | 849 | 570 | 1738 | 856 | I S |
| Gait disturbance | 1273 | 471 | 1520 | 1612 | 856 | PS |
| Death | 902 | 1746 | 1520 | 252 | 1039 | OP Z |
| Injection site inflammation | 424 | 1604 | 2186 | 663 | 489 | OP |
| Fall | 1645 | 755 | 380 | 1612 | 611 | I S |
| Adverse event | Age group | COVID-19 (Janssen) | COVID-19 (Moderna) | COVID-19 (Moderna bivalent) | COVID-19 (Pfizer-BioNTech) | COVID-19 (Pfizer-BioNTech bivalent) |
|---|---|---|---|---|---|---|
| Acute kidney injury |
50-59 | 428 | 154 | 108 | 212 | 247 |
| 60-69 | 829 | 326 | 201 | 465 | 344 | |
| 70-79 | 1430 | 629 | 425 | 1045 | 789 | |
| 80-89 | 2506 | 1017 | 920 | 1477 | 1969 | |
| Acute respiratory failure |
50-59 | 714 | 200 | 325 | 379 | 949 |
| 60-69 | 1469 | 463 | 717 | 955 | 1895 | |
| 70-79 | 2861 | 957 | 1567 | 1862 | 3996 | |
| 80-89 | 4089 | 2061 | 3146 | 2873 | 9704 | |
| Atrial fibrillation |
50-59 | 436 | 407 | 595 | 508 | 289 |
| 60-69 | 965 | 746 | 1091 | 1017 | 1033 | |
| 70-79 | 1379 | 1223 | 1461 | 1726 | 1934 | |
| 80-89 | 2506 | 1659 | 1458 | 2424 | 2390 | |
| Cerebrovascular accident |
50-59 | 1045 | 463 | 325 | 601 | 371 |
| 60-69 | 1280 | 630 | 229 | 832 | 590 | |
| 70-79 | 1788 | 900 | 478 | 1224 | 1272 | |
| 80-89 | 2440 | 1510 | 690 | 1848 | 1687 | |
| Chest pain |
50-59 | 3683 | 2669 | 2329 | 3379 | 2105 |
| 60-69 | 3222 | 1880 | 1608 | 2680 | 1624 | |
| 70-79 | 3193 | 1554 | 1567 | 2254 | 1374 | |
| 80-89 | 1715 | 1487 | 1841 | 1954 | 1336 | |
| Chest X-ray abnormal |
50-59 | 729 | 257 | 270 | 393 | 660 |
| 60-69 | 1605 | 524 | 660 | 735 | 812 | |
| 70-79 | 2989 | 926 | 1036 | 1362 | 1960 | |
| 80-89 | 4485 | 1771 | 1688 | 2424 | 3375 | |
| Confusional state | 50-59 | 766 | 547 | 433 | 632 | 578 |
| 60-69 | 923 | 669 | 717 | 738 | 541 | |
| 70-79 | 1379 | 1085 | 744 | 1130 | 1069 | |
| 80-89 | 2770 | 1886 | 1534 | 2167 | 2461 | |
| COVID-19 |
50-59 | 7352 | 6030 | 12,026 | 7911 | 12,262 |
| 60-69 | 12,248 | 7494 | 20,017 | 11,670 | 19,374 | |
| 70-79 | 20,975 | 10,033 | 18,841 | 15,991 | 22,683 | |
| 80-89 | 30,540 | 15,462 | 15,502 | 20,500 | 18,495 | |
| COVID-19 pneumonia |
50-59 | 1075 | 232 | 108 | 432 | 165 |
| 60-69 | 1994 | 557 | 201 | 936 | 492 | |
| 70-79 | 3960 | 1045 | 451 | 2129 | 1145 | |
| 80-89 | 5540 | 1961 | 1074 | 3485 | 2672 | |
| Hypoxia |
50-59 | 781 | 214 | 54 | 405 | 743 |
| 60-69 | 1458 | 545 | 459 | 801 | 1452 | |
| 70-79 | 2938 | 970 | 876 | 1662 | 2316 | |
| 80-89 | 5343 | 2173 | 2686 | 2836 | 5977 | |
| Oropharyngeal pain |
50-59 | 1714 | 1673 | 4767 | 2216 | 4830 |
| 60-69 | 1700 | 1719 | 7495 | 2099 | 6794 | |
| 70-79 | 1303 | 1452 | 5793 | 1938 | 5804 | |
| 80-89 | 1319 | 820 | 1918 | 1020 | 2672 | |
| Pulmonary embolism |
50-59 | 1278 | 730 | 325 | 746 | 495 |
| 60-69 | 1731 | 859 | 373 | 977 | 861 | |
| 70-79 | 2120 | 916 | 558 | 1408 | 763 | |
| 80-89 | 2836 | 1107 | 997 | 1787 | 1898 | |
| Sepsis |
50-59 | 293 | 121 | 270 | 198 | 289 |
| 60-69 | 472 | 253 | 258 | 322 | 295 | |
| 70-79 | 919 | 456 | 558 | 733 | 992 | |
| 80-89 | 2308 | 816 | 1074 | 1048 | 2109 | |
| Unevaluable event |
50-59 | 699 | 740 | 812 | 731 | 1321 |
| 60-69 | 1143 | 946 | 459 | 1019 | 713 | |
| 70-79 | 1328 | 1259 | 558 | 1498 | 636 | |
| 80-89 | 2968 | 1856 | 383 | 1885 | 703 | |
| Vaccine breakthrough infection |
50-59 | 714 | 447 | 1083 | 559 | 660 |
| 60-69 | 1721 | 866 | 1062 | 1077 | 960 | |
| 70-79 | 2887 | 1228 | 1222 | 1851 | 2163 | |
| 80-89 | 5277 | 2539 | 2609 | 3048 | 2742 |
| Vaccine name | Day 0 | Day 1 | Day 2 | Day 3 | Day 4 | Day 5 | Day 6 | Day 7 |
|---|---|---|---|---|---|---|---|---|
| COVID-19 (Janssen) | 62 | 73 | 32 | 34 | 20 | 16 | 20 | 23 |
| COVID-19 (Moderna) | 567 | 649 | 289 | 170 | 124 | 129 | 84 | 124 |
| COVID-19 (Moderna bivalent) | 11 | 19 | 9 | 2 | 2 | 4 | 3 | |
| COVID-19 (Pfizer-BioNTech) | 1022 | 795 | 404 | 287 | 216 | 201 | 138 | 131 |
| COVID-19 (Pfizer-BioNTech bivalent) | 15 | 30 | 11 | 7 | 3 | 4 | 5 | 2 |
| Pneumo (Pneumovax) | 56 | 19 | 15 | 11 | 5 | 4 | 3 | 2 |
| Pneumo (Prevnar13) | 138 | 189 | 67 | 49 | 34 | 9 | 13 | 11 |
| RSV (Abrysvo) | 4 | 2 | 3 | 1 | 1 | |||
| RSV (Arexvy) | 7 | 7 | 3 | 3 | 1 | 1 | ||
| Zoster (Shingrix) | 40 | 36 | 7 | 5 | 3 | 6 | 1 | 4 |
| Zoster live (Zostavax) | 13 | 7 | 10 | 2 | 1 | 2 | 4 | 3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




