Submitted:
02 March 2026
Posted:
03 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
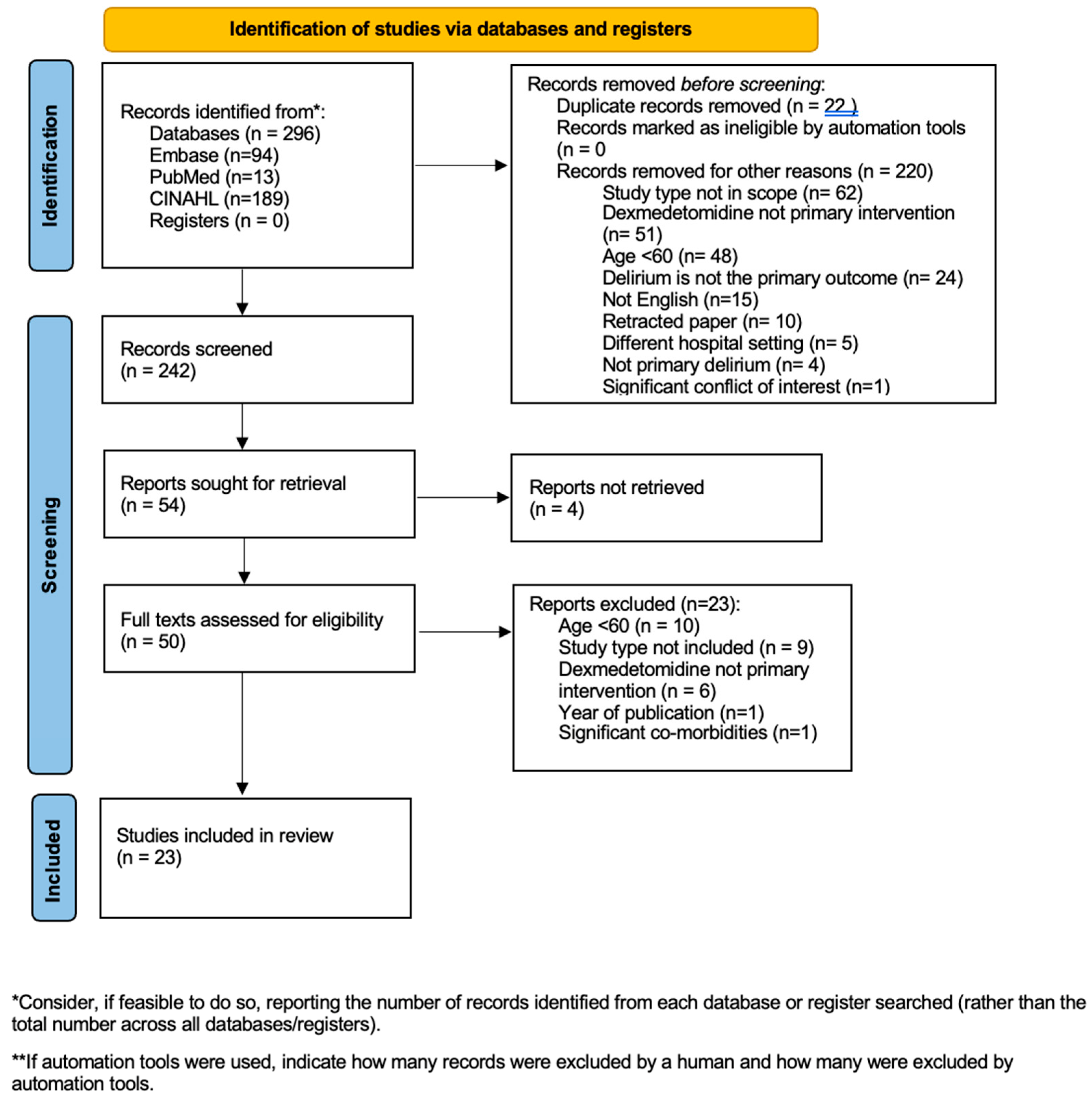
2.1. Data Sources and Search Strategy
2.2. Study Selection
2.3. Eligibility Criteria
2.4. Data Extraction
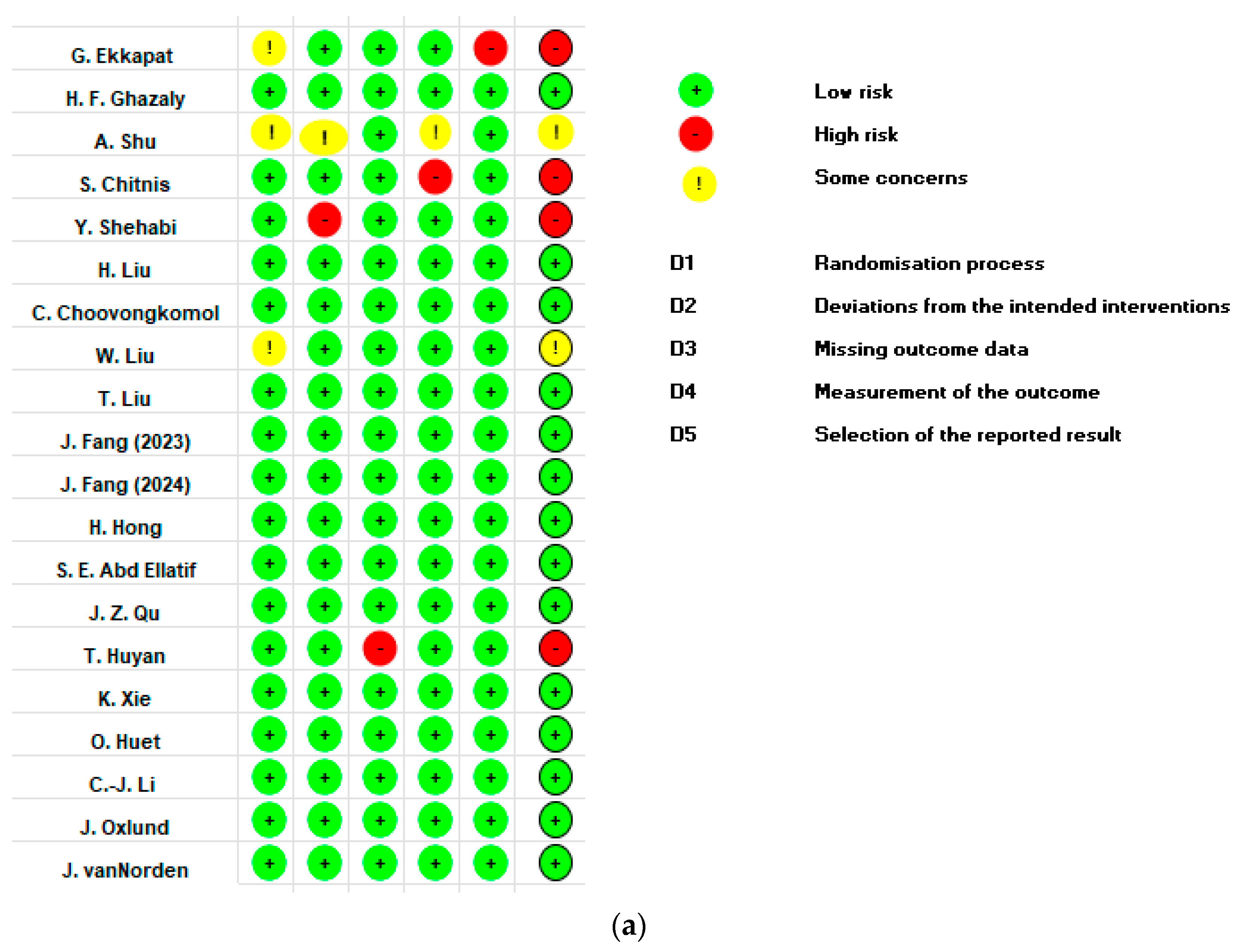
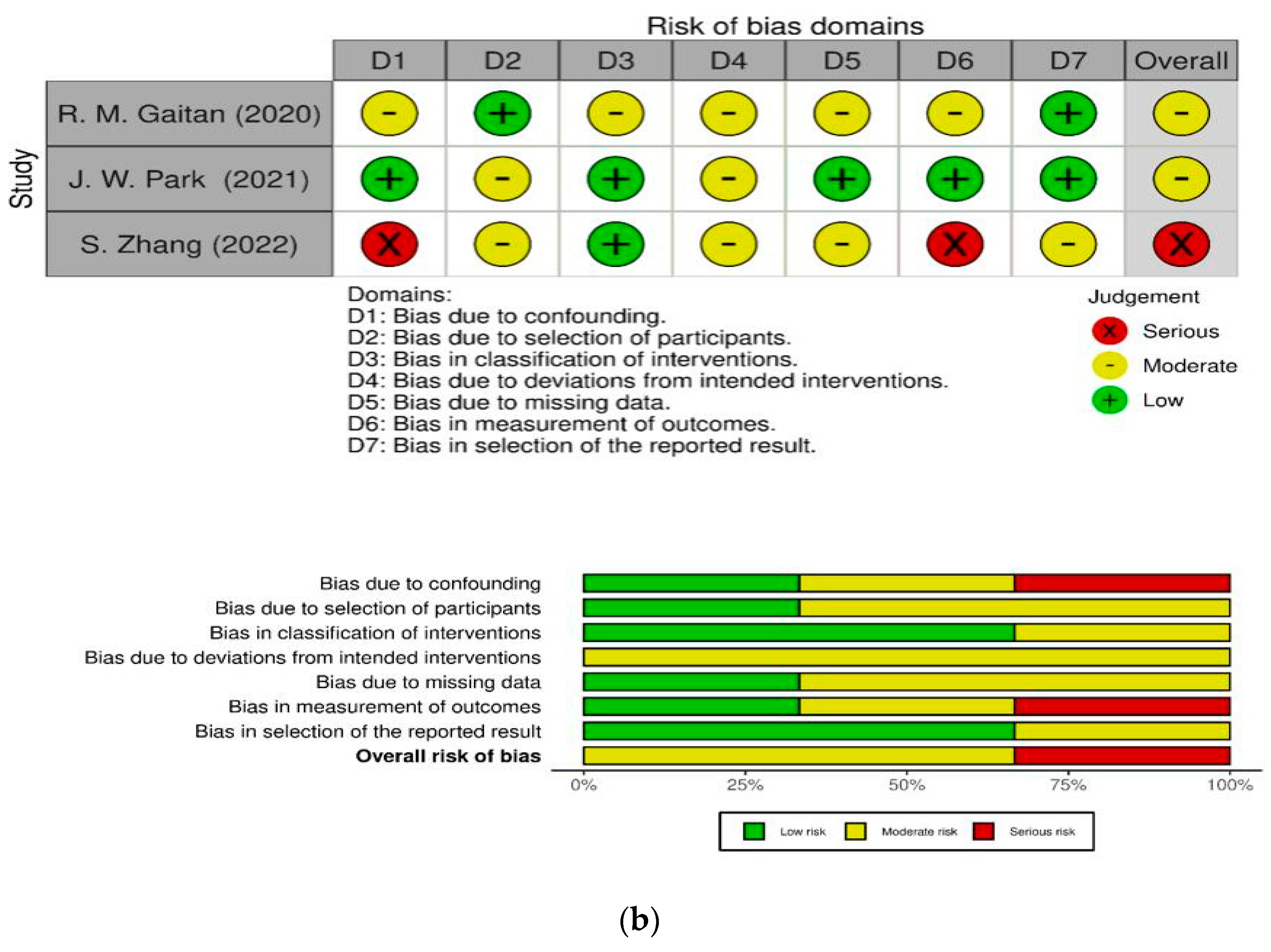
2.5. Quality Assessment
3. Results
3.1. Study Characteristics
3.2. Risk of Bias in Individual Studies
3.3. Primary Outcomes
3.3.1. Incidence of Delirium (IOD)
3.3.2. Timing of Dexmedetomidine Administration
3.4. Secondary Outcomes
3.4.1. Length of stay in ICU
3.4.2. Adverse effects of Dexmedetomidine
3.4.3. Mortality
4. Discussion
5. Strengths and Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Abbreviations
| α2-AR | α2-adrenergic receptors |
| 5-HT | 5-Hydroxytryptamine |
| CAM-ICU | Confusion Assessment Method for ICU |
| CRP | C-Reactive Protein |
| GABA | Gamma-Aminobutyric Acid |
| IOD | Incidence of delirium |
| ICDSC | Intensive Care Delirium Screening Checklist |
| ICU | Intensive Care Unit |
| RCTs | Randomized Controlled Trials |
| RASS | Richmond Agitation-Sedation Scale |
| RoB | Risk of Bias |
| ROBINS-I | Risk Of Bias In Non-randomized Studies - of Interventions |
References
- Thom, RP; Levy-Carrick, NC; Bui, M; Silbersweig, D. Delirium. American Journal of Psychiatry 2019, 176(10), 785–93. [Google Scholar] [CrossRef]
- Lawlor, PG; Bush, SH. Delirium diagnosis, screening and management. Current opinion in supportive and palliative care 2014, 8(3), 286–95. [Google Scholar] [PubMed]
- Delirium is prevalent in older hospital inpatients and associated with adverse outcomes: results of a prospective multi-centre study on World Delirium Awareness Day. BMC medicine 2019, 17, 1–11.
- Senderovich, H; Gardner, S; Berall, A; Ganion, M; Zhang, D; Vinoraj, D; et al. Benzodiazepine Use and Morbidity-Mortality outcomes in a geriatric Palliative Care Unit: a retrospective review. Dementia and Geriatric Cognitive Disorders 2021, 50(6), 559–67. [Google Scholar] [CrossRef]
- Wu, TT; Ko, S; Kooken, R; van den Boogaard, M; Devlin, JW. Exploring ketamine analgosedation use and its effect on incident delirium in critically ill adults. Critical Care Explorations 2021, 3(10), e0544. [Google Scholar] [CrossRef]
- Brown, KE; Mirrakhimov, AE; Yeddula, K; Kwatra, MM. Propofol and the risk of delirium: exploring the anticholinergic properties of propofol. Medical Hypotheses 2013, 81(4), 536–9. [Google Scholar] [CrossRef] [PubMed]
- Gertler R, Brown HC, Mitchell DH, Silvius EN, editors. Dexmedetomidine: a novel sedative-analgesic agent. Baylor University Medical Center Proceedings 2001; 14(1): 13-21.
- Kaur, M; Singh, P. Current role of dexmedetomidine in clinical anesthesia and intensive care. Anesthesia Essays and Researches 2011, 5(2), 128–33. [Google Scholar] [CrossRef] [PubMed]
- Carollo, DS; Nossaman, BD; Ramadhyani, U. Dexmedetomidine: a review of clinical applications. Current Opinion in Anesthesiology 2008, 21(4), 457–61. [Google Scholar] [CrossRef]
- Lewis, K; Alshamsi, F; Carayannopoulos, KL; Granholm, A; Piticaru, J; Al Duhailib, Z; et al. Dexmedetomidine vs other sedatives in critically ill mechanically ventilated adults: a systematic review and meta-analysis of randomized trials. Intensive care medicine 2022, 48(7), 811–40. [Google Scholar] [CrossRef]
- Ungarian, J; Rankin, JA; Then, KL. Delirium in the Intensive Care Unit: Is Dexmedetomidine Effective? Critical care nurse 2019, 39(4), e8–e21. [Google Scholar]
- Chitnis, S; Mullane, D; Brohan, J; Noronha, A; Paje, H; Grey, R; et al. Dexmedetomidine use in intensive care unit sedation and post-operative recovery in elderly patients post-cardiac surgery (DIRECT). Journal of Cardiothoracic and Vascular Anesthesia 2022, 36(3), 880–92. [Google Scholar] [CrossRef]
- Gaitan, RM; Vicent, L; Rodriguez-Queralto, O; Lopez-de-Sa, E; Elorriaga, A; Pastor, G; et al. Dexmedetomidine in medical cardiac intensive care units. Data from a multicenter prospective registry. International Journal of Cardiology 2020, 310, 162–6. [Google Scholar] [CrossRef]
- Ekkapat, G; Kampitak, W; Theerasuwipakorn, N; Kittipongpattana, J; Engsusophon, P; Phannajit, J; et al. A comparison of efficacy between low-dose dexmedetomidine and propofol for prophylaxis of post-operative delirium in elderly patients undergoing hip fracture surgery: A randomized controlled trial. Indian Journal of Critical Care Medicine: Peer-reviewed, Official Publication of Indian Society of Critical Care Medicine 2024, 28(5), 467. [Google Scholar] [CrossRef]
- Baumgartner, K; Joseph, M; Lothet, E; Fuller, BM. Dexmedetomidine in the emergency department: A prospective observational cohort study. Academic Emergency Medicine 2024, 31(3), 263–72. [Google Scholar] [CrossRef]
- Gibb, K; Seeley, A; Quinn, T; Siddiqi, N; Shenkin, S; Rockwood, K; et al. The consistent burden in published estimates of delirium occurrence in medical inpatients over four decades: a systematic review and meta-analysis study. Age and ageing 2020, 49(3), 352–60. [Google Scholar] [CrossRef]
- Sterne, JA; Savović, J; Page, MJ; Elbers, RG; Blencowe, NS; Boutron, I; et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ 2019, 366. [Google Scholar]
- Sterne, JA; Hernán, MA; Reeves, BC; Savović, J; Berkman, ND; Viswanathan, M; et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ 2016, 355. [Google Scholar] [CrossRef]
- Ghazaly, HF; Hemaida, TS; Zaher, ZZ; Elkhodary, OM; Hammad, SS. A pre-anesthetic bolus of ketamine versus Dexmedetomidine for prevention of post-operative delirium in elderly patients undergoing emergency surgery: a randomized, double-blinded, placebo-controlled study. BMC anesthesiology 2023, 23(1), 407. [Google Scholar]
- Shu, A; Fu, Y; Luo, Y; Cao, J; Jian, W; Sun, B. An investigation on delirium and hemodynamics influenced by Dexmedetomidine for sedating elderly patients in mechanical ventilation. Int J Clin Exp Med 2019, 12(2), 1942–6. [Google Scholar]
- Shehabi, Y; Howe, BD; Bellomo, R; Arabi, YM; Bailey, M; Bass, FE; et al. Early sedation with dexmedetomidine in critically ill patients. New England Journal of Medicine 2019, 380(26), 2506–17. [Google Scholar] [CrossRef] [PubMed]
- Liu, H; Gao, M; Zheng, Y; Sun, C; Lu, Q; Shao, D. Effects of Dexmedetomidine at different dosages on peri-operative haemodynamics and post-operative recovery quality in elderly patients undergoing hip replacement surgery under general anaesthesia: a randomized controlled trial. Trials 2023, 24(1), 386. [Google Scholar] [PubMed]
- Choovongkomol, C; Sinchai, S; Choovongkomol, K. Effect of a Single-dose Dexmedetomidine on Post-operative Delirium and Intraoperative Hemodynamic Outcomes in Elderly Hip Surgery; A Randomized Controlled Trial Dexmedetomidine for Post-operative Delirium: Dexmedetomidine for Post-operative Delirium. Siriraj Medical Journal 2024, 76(2), 80–9. [Google Scholar] [CrossRef]
- Liu, W; Wang, Y; Chen, K; Ye, M; Lu, W; Chen, K; et al. Effect of intraoperative dexmedetomidine use on postoperative delirium in the elderly after laryngectomy: a randomized controlled clinical trial. Drug Design, Development and Therapy 2023, 2933–41. [Google Scholar] [CrossRef]
- Liu, T; Tuo, J; Wei, Q; Sun, X; Zhao, H; Zhao, X; et al. Effect of perioperative dexmedetomidine infusion on postoperative delirium in elderly patients undergoing oral and maxillofacial surgery: a randomized controlled clinical trial. International Journal of General Medicine 2022, 6105–13. [Google Scholar] [CrossRef]
- Fang, J; Yang, J; Zhai, M; Zhang, Q; Zhang, M; Xie, Y. Effects of dexmedetomidine dosage on the short-term cognitive function of elderly patients undergoing cardiac surgery. BMC anesthesiology 2023, 23(1), 380. [Google Scholar] [CrossRef]
- Fang, J; Yang, J; Zhai, M; Zhang, Q; Zhang, M; Xie, Y. Effects of short-term preoperative intranasal dexmedetomidine plus conventional treatment on delirium following cardiac surgery in patients with sleep disorders. Perioperative Medicine 2024, 13(1), 17. [Google Scholar] [CrossRef]
- Hong, H; Zhang, D-Z; Li, M; Wang, G; Zhu, S-N; Zhang, Y; et al. Impact of dexmedetomidine supplemented analgesia on delirium in patients recovering from orthopedic surgery: a randomized controlled trial. BMC anesthesiology 2021, 21, 1–13. [Google Scholar] [CrossRef]
- Abd Ellatif, SE; Mowafy, SM; Shahin, MA. Ketofol versus Dexmedetomidine for preventing postoperative delirium in elderly patients undergoing intestinal obstruction surgeries: a randomized controlled study. BMC anesthesiology 2024, 24(1), 1. [Google Scholar] [CrossRef]
- Qu, JZ; Mueller, A; McKay, TB; Westover, MB; Shelton, KT; Shaefi, S; et al. Nighttime dexmedetomidine for delirium prevention in non-mechanically ventilated patients after cardiac surgery (MINDDS): a single-centre, parallel-arm, randomised, placebo-controlled superiority trial. EClinicalMedicine 2023, 56. [Google Scholar] [CrossRef] [PubMed]
- Huyan, T; Hu, X; Peng, H; Zhu, Z; Li, Q; Zhang, W. Perioperative dexmedetomidine reduces delirium in elderly patients after lung cancer surgery. Psychiatria Danubina 2019, 31(1), 95–101. [Google Scholar] [CrossRef] [PubMed]
- Xie, K; Chen, J; Tian, L; Gu, F; Pan, Y; Huang, Z; et al. Postoperative infusion of dexmedetomidine via intravenous patient-controlled analgesia for prevention of post-operative delirium in elderly patients undergoing surgery. Aging Clinical and Experimental Research 2023, 35(10), 2137–44. [Google Scholar] [CrossRef] [PubMed]
- Huet, O; Gargadennec, T; Oilleau, J-F; Rozec, B; Nesseler, N; Bouglé, A; et al. Prevention of post-operative delirium using an overnight infusion of dexmedetomidine in patients undergoing cardiac surgery: a pragmatic, randomized, double-blind, placebo-controlled trial. Critical Care 2024, 28(1), 64. [Google Scholar]
- Li, C-J; Wang, B-J; Mu, D-L; Hu, J; Guo, C; Li, X; et al. Randomized clinical trial of intraoperative dexmedetomidine to prevent delirium in the elderly undergoing major non-cardiac surgery. Journal of British Surgery 2020, 107(2), e123–e32. [Google Scholar]
- Oxlund, J; Knudsen, T; Sörberg, M; Strøm, T; Toft, P; Jennum, PJ. Sleep quality and quantity determined by polysomnography in mechanically ventilated critically ill patients randomized to dexmedetomidine or placebo. Acta Anaesthesiologica Scandinavica 2023, 67(1), 66–75. [Google Scholar]
- Van Norden, J; Spies, C; Borchers, F; Mertens, M; Kurth, J; Heidgen, J; et al. The effect of peri-operative dexmedetomidine on the incidence of postoperative delirium in cardiac and non-cardiac surgical patients: a randomised, double-blind placebo-controlled trial. Anaesthesia 2021, 76(10), 1342–1351. [Google Scholar] [PubMed]
- Park, J-W; Kim, E-K; Lee, H-T; Park, S; Do, S-H. The effects of propofol or dexmedetomidine sedation on postoperative recovery in elderly patients receiving lower limb surgery under spinal anesthesia: a retrospective propensity score-matched analysis. J Clin Med 2021, 10(1), 135. [Google Scholar]
- Zhang S, Yin Q, Wang Y, Yu K, Shen P, editors. The Preventive Effect of Dexmedetomidine Against Delirium in Patients with Aortic Dissection: A Retrospective Cohort Study. The Heart Surgery Forum 2022; 25(4): e489-e493.
- Rahimi-Bashar, F; Abolhasani, G; Manouchehrian, N; Jiryaee, N; Vahedian-Azimi, A; Sahebkar, A. Incidence and risk factors of delirium in the intensive care unit: a prospective cohort. BioMed research international 2021, 2021(1), 6219678. [Google Scholar]
- Maldonado, JR. Pathoetiological model of delirium: a comprehensive understanding of the neurobiology of delirium and an evidence-based approach to prevention and treatment. Critical care clinics 2008, 24(4), 789–856. [Google Scholar]
- Wang, S; Hong, Y; Li, S; Kuriyama, A; Zhao, Y; Hu, J; et al. Effect of dexmedetomidine on delirium during sedation in adult patients in intensive care units: A systematic review and meta-analysis. Journal of Clinical Anesthesia 2021, 69, 110157. [Google Scholar] [CrossRef]
- Heybati, K; Zhou, F; Ali, S; Deng, J; Mohananey, D; Villablanca, P; et al. Outcomes of dexmedetomidine versus propofol sedation in critically ill adults requiring mechanical ventilation: a systematic review and meta-analysis of randomised controlled trials. British journal of anaesthesia 2022, 129(4), 515–26. [Google Scholar] [CrossRef]
- Fondeur, J; Mendez, LE; Srinivasan, M; Hamouda, RK; Ambedkar, B; Arzoun, H; et al. Dexmedetomidine in prevention of post-operative delirium: a systematic review. Cureus 2022, 14(6). [Google Scholar]
- Sanders, RD; Maze, M. Contribution of sedative-hypnotic agents to delirium via modulation of the sleep pathway. Canadian journal of anaesthesia 2010, 58(2), 149. [Google Scholar] [CrossRef]
- Maddocks, I; Somogyi, A; Abbott, F; Hayball, P; Parker, D. Attenuation of morphine-induced delirium in palliative care by substitution with infusion of oxycodone. Journal of pain and symptom management 1996, 12(3), 182–9. [Google Scholar] [CrossRef]
- Azimaraghi, O; Wongtangman, K; Wachtendorf, LJ; Santer, P; Rumyantsev, S; Ahn, C; et al. Differential effects of gamma-aminobutyric acidergic sedatives on risk of post-extubation delirium in the ICU: A retrospective cohort study from a New England health care network. Critical care medicine 2022, 50(5), e434–e44. [Google Scholar] [CrossRef]
- Wang, C; Chen, Q; Wang, P; Jin, W; Zhong, C; Ge, Z; et al. The effect of dexmedetomidine as a sedative agent for mechanically ventilated patients with sepsis: a systematic review and meta-analysis. Frontiers in Medicine 2021, 8, 776882. [Google Scholar] [CrossRef]
- Zhang, T; Mei, Q; Dai, S; Liu, Y; Zhu, H. Use of dexmedetomidine in patients with sepsis: a systematic review and meta-analysis of randomized-controlled trials. Annals of Intensive Care 2022, 12(1), 81. [Google Scholar] [PubMed]
- Tilouche, N; Hassen, MF; Ali, HBS; Jaoued, O; Gharbi, R; El Atrous, SS. Delirium in the intensive care unit: incidence, risk factors, and impact on outcome. Indian journal of critical care medicine: peer-reviewed, official publication of Indian Society of Critical Care Medicine 2018, 22(3), 144. [Google Scholar] [CrossRef] [PubMed]
- Wozniak, H; Beckmann, TS; Dos Santos Rocha, A; Pugin, J; Heidegger, C-P; Cereghetti, S. Long-stay ICU patients with frailty: mortality and recovery outcomes at 6 months. Annals of Intensive Care 2024, 14(1), 31. [Google Scholar] [CrossRef]
- Huang, J-X; Zhang, S-S; Wang, S-X; Xi, D-S; Luo, F-R; Liu, C-J; et al. The role of peri-operative sedative anesthetics in preventing postoperative delirium: a systematic review and network-meta analysis including 6679 patients. BMC Cardiovascular Disorders 2024, 24(1), 147. [Google Scholar] [CrossRef] [PubMed]
- Sscarpace, PJ; Abrass, IB. Alpha-and beta-adrenergic receptor function in the brain during senescence. Neurobiology of Aging 1988, 9, 53–8. [Google Scholar]
- Zhao, W; Li, J; Wang, N; Wang, Z; Zhang, M; Zhang, H; et al. Effect of dexmedetomidine on postoperative nausea and vomiting in patients under general anaesthesia: an updated meta-analysis of randomised controlled trials. BMJ open 2023, 13(8), e067102. [Google Scholar] [CrossRef]
- Jin, S; Liang, DD; Chen, C; Zhang, M; Wang, J. Dexmedetomidine prevent post-operative nausea and vomiting on patients during general anesthesia: A PRISMA-compliant meta analysis of randomized controlled trials. Medicine 2017, 96(1), e5770. [Google Scholar] [CrossRef] [PubMed]
- Tang, J; Zhong, Z-G; Wu, C-D. Exploring the impact of dexmedetomidine on short-term outcomes in critically ill sepsis-associated encephalopathy patients. European Review for Medical & Pharmacological Sciences 2024, 28(7). [Google Scholar]
- Peng, K; Ji, F-h; Liu, H-y; Zhang, J; Chen, Q-c; Jiang, Y-h. Effects of peri-operative dexmedetomidine on post-operative mortality and morbidity: a systematic review and meta-analysis. Clinical Therapeutics 2019, 41(1), 138–54. [Google Scholar] [CrossRef] [PubMed]
- Li, R; Lai, IK; Pan, JZ; Zhang, P; Maze, M. Dexmedetomidine exerts an anti-inflammatory effect via α2 adrenoceptors to prevent lipopolysaccharide-induced cognitive decline in mice. Anesthesiology 2020, 133(2), 393–407. [Google Scholar] [CrossRef]
- Wang, L; Dai, X; Yu, L; Li, H; Zhang, X; Yu, Q; et al. Dexmedetomidine therapy promotes cardiac dysfunction and increases mortality in sepsis: A translational study. International Immunopharmacology 2025, 146, 113924. [Google Scholar] [CrossRef]
| First author | Year | Study type | Country | Compara-tor(s) | Total | DEX | Comparator | Route | Timing | Assessment tool |
|---|---|---|---|---|---|---|---|---|---|---|
| G. Ekkapat (14) | 2024 | RCT | Thailand | Propofol Saline |
108 | 36 | 72 | IV | Post-op | CAM-ICU |
| H. F. Ghazaly (19) | 2023 | RCT | Egypt | Saline | 2023 | RCT | Egypt | |||
| A. Shu (20) | 2019 | RCT | China | Midazolam | 80 | 40 | 40 | IV | ICU | Not stated |
| R. M. Gaitan (13) | 2020 | Prospective / Registries | Spain | No comparator | 410 | 410 | 0 | IV | ICU | RASS |
| S. Chitnis (12) | 2022 | RCT | Canada | Propofol | 67 | 34 | 33 | IV | Post-op | ICDSC |
| Y. Shehabi (21) | 2019 | RCT | Ireland Australia Italy, Malaysia New Zealand Saudi Arabia Switzerland |
Usual care | 3918 | 1954 | 1964 | IV | ICU | CAM-ICU |
| H. Liu (22) | 2023 | RCT | China | Saline Midazolam |
150 | 76 | 74 | IV | Intra-op | RASS |
| C. Choovongkomol (23) | 2024 | RCT | Thailand | Saline | 200 | 100 | 100 | IV | Intra-op | CAM |
| W. Liu (24) | 2023 | RCT | China | Saline | 299 | 149 | 150 | IV | Intra-op | CAM |
| T. Liu(25) | 2022 | RCT | China | Saline | 120 | 60 | 60 | IV | Pre-op | 3D-CAM |
| J. Fang (26) | 2023 | RCT | China | No comparator | 88 | 88 | 0 | IV | Pre-op | CAM-ICU |
| J. Fang (27) | 2024 | RCT | China | Saline | 100 | 50 | 50 | Intra-nasal | Pre-op | CAM-ICU |
| H. Hong (28) | 2021 | RCT | China | Saline | 712 | 356 | 356 | IV | Post-op | CAM / CAM-ICU |
| S. E. Abd Ellatif (29) | 2024 | RCT | Egypt | Ketofol | 2024 | RCT | Egypt | Intra-op Post-op |
CAM-ICU | |
| J. Z. Qu (30) | 2022 | RCT | USA | Ketofol, Saline | 394 | 188 | 206 | IV | Post-op | CAM |
| T. Huyan (31) | 2018 | RCT | China | Saline | 346 | 173 | 173 | IV | Pre-op Intra-op |
ICDSC |
| K. Xie (32) | 2023 | RCT | China | Saline | 236 | 117 | 119 | PCIA / pump | Post-op | RASS |
| O. Huet (33) | 2024 | RCT | France | Saline | 331 | 165 | 166 | IV | Pre-op Intra-op Post-op |
CAM-ICU |
| C.-J. Li (34) | 2020 | RCT | China | Saline | 619 | 309 | 310 | IV | Intra-op | CAM / CAM-ICU |
| J. Oxlund (35) | 2023 | RCT | Denmark | Saline | 30 | 20 | 10 | IV | ICU | CAM-ICU |
| J. vanNorden (36) | 2021 | RCT | Germany | Saline | 63 | 30 | 33 | IV | Pre-op Intra-op Post-op |
CAM-ICU |
| J.-W. Park (37) | 2021 | Observational | Korea | Propofol | 714 | 357 | 357 | IV | Intra-op | CAM-ICU |
| S. Zhang (38) | 2022 | Observational | China | Saline | 60 | 28 | 32 | IV | N/A | CAM-ICU |
| Number of studies reporting | |
|---|---|
| Incidence of delirium in DEX group | 20 |
| Significant decrease in the incidence of delirium in DEX group | 9 |
| Significant increase in the incidence of delirium in DEX group | 0 |
| No significant difference in the incidence of delirium in DEX group | 8 |
| No p-value reported | 3 |
| Adverse effect | Number of studies reporting |
|---|---|
| Bradycardia | 17 |
| Hypotension | 15 |
| Nausea and vomiting | 4 |
| Tachycardia | 3 |
| Hypertension | 3 |
| Pneumonia | 1 |
| Prolonged sinus arrest | 1 |
| Sedation | 1 |
| Nightmares | 1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





