Submitted:
27 February 2026
Posted:
02 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. MRD and Metastasis
3. MRD in Hematologic and Solid Tumors
3.1. In Hematologic Malignancies
3.1.1. In Acute Myeloid Leukemia
3.1.2. In BCR/ABL1 and Philadelphia Chromosome-Positive Leukemia
3.1.3. In Multiple Myeloma
3.2. In Solid Tumors
3.2.1. Colorectal Cancer
3.2.2. Non-Small Cell Lung Cancer
3.2.3. Breast Cancer
4. Techniques for MRD Detection
4.1. Conventional Techniques
4.2. Emerging Methods and Multi-Omics Integration
5. Eradicating MRD Positivity
5.1. Immunotherapy
5.2. Targeted Therapies
6. Limitations
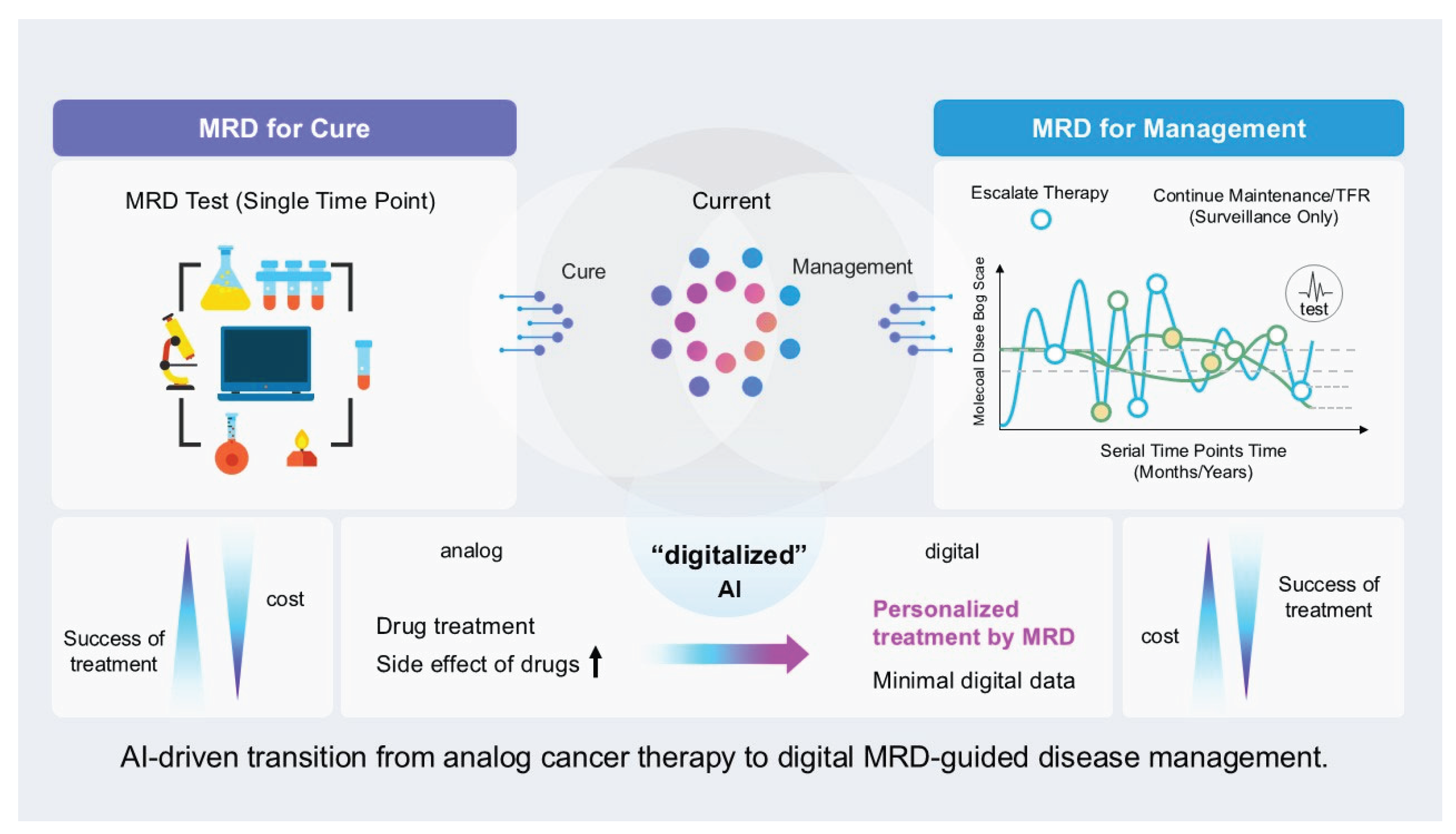
7. Conclusions and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ABL1 | Abelson tyrosine-protein kinase 1 |
| ADHFE1 | alcohol dehydrogenase iron containing 1 |
| AI | Artificial intelligence |
| ALL | acute lymphoblastic leukemia |
| alloSCT | allogeneic stem cell transplantation |
| AML | acute myeloid leukemia |
| ASCT | autologous stem cell transplantation |
| BCL2 | B cell lymphoma2 |
| BM | bone marrow |
| cfDNA | cell-free DNA |
| CLL | chronic lymphocytic leukemia |
| CML | chronic myeloid leukemia |
| CRC | colorectal cancer |
| CTC | circulating tumor cell |
| ctDNA | circulating tumor DNA |
| ddPCR | droplet-based dPCR |
| dPCR | digital PCR |
| DTC | disseminated tumor cells |
| EDGE | Enrichment and detection using Genomic Enrichment |
| EGFR | epidermal growth factor receptor |
| ELN | European LeukemiaNet |
| EMA | European Medicines Agency |
| EMT | epithelial-mesenchymal transition |
| ER | estrogen receptor |
| EV | tumor-derived extracellular vesicles |
| FLT3 | fms-like tyrosine kinase 3 |
| GC | gastric cancer |
| GMM | Gaussian mixture model |
| GPRC5D | G protein-coupled receptor, class C, group 5, member D |
| HER2 | human epidermal growth factor receptor |
| IDH | isocitrate dehydrogenase |
| ITD | internal tandem duplication |
| MFC | multiparameter flow cytometry |
| MM | multiple myeloma |
| MRD | minimal residual disease |
| NGS | next-generation sequencing |
| NPM1 | nucleophosmin |
| NSCLC | non-small cell lung cancer |
| OS | overall survival |
| PCR | polymerase chain reaction |
| PFS | progression-free survival |
| Ph | Philadelphia |
| Ph-positive ALL | Philadelphia chromosome-positive Acute Lymphoblastic Leukemia |
| PIK3CA | phosphatidylinositol 3-kinase catalytic subunit alpha |
| PML | promyelocytic leukemia |
| PPP2R5C | protein phosphatase 2 regulatory subunit B gamma |
| qPCR | quantitative real-time PCR |
| RFS | recurrence-free survival |
| RUNX1T1 | RUNX family transcription factor 1 partner transcriptional co-repressor 1 |
| scMRD | single-cell MRD |
| scRNA-seq | Single-cell RNA sequencing |
| SDC2 | syndecan2 |
| TFR | treatment-free remission |
| TP53 | tumor protein p53 |
| TSG | tumor suppressor gene |
| UDS | ultra-deep sequencing |
| WGS | whole-genome sequencing |
References
- Brown, J.S.; Amend, S.R.; Austin, R.H.; Gatenby, R.A.; Hammarlund, E.U.; Pienta, K.J. Updating the Definition of Cancer. Mol Cancer Res 2023, 21, 1142–1147. [Google Scholar] [CrossRef]
- Sonnenschein, C.; Davis, B.; Soto, A.M. A novel pathogenic classification of cancers. Cancer Cell Int 2014, 14, 113. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Zhou, H.; Tan, L.; Siu, K.T.H.; Guan, X.Y. Exploring treatment options in cancer: Tumor treatment strategies. Signal Transduct Target Ther 2024, 9, 175. [Google Scholar] [CrossRef]
- Lone, S.N.; Nisar, S.; Masoodi, T.; Singh, M.; Rizwan, A.; Hashem, S.; El-Rifai, W.; Bedognetti, D.; Batra, S.K.; Haris, M.; et al. Liquid biopsy: a step closer to transform diagnosis, prognosis and future of cancer treatments. Mol Cancer 2022, 21, 79. [Google Scholar] [CrossRef]
- Nikanjam, M.; Kato, S.; Kurzrock, R. Liquid biopsy: current technology and clinical applications. J Hematol Oncol 2022, 15, 131. [Google Scholar] [CrossRef]
- Connal, S.; Cameron, J.M.; Sala, A.; Brennan, P.M.; Palmer, D.S.; Palmer, J.D.; Perlow, H.; Baker, M.J. Liquid biopsies: the future of cancer early detection. J Transl Med 2023, 21, 118. [Google Scholar] [CrossRef] [PubMed]
- Kurtz, D.M.; Soo, J.; Co Ting Keh, L.; Alig, S.; Chabon, J.J.; Sworder, B.J.; Schultz, A.; Jin, M.C.; Scherer, F.; Garofalo, A.; et al. Enhanced detection of minimal residual disease by targeted sequencing of phased variants in circulating tumor DNA. Nat Biotechnol 2021, 39, 1537–1547. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Gong, Y.; Lam, V.K.; Shi, Y.; Guan, Y.; Zhang, Y.; Ji, L.; Chen, Y.; Zhao, Y.; Qian, F.; et al. Deep sequencing of circulating tumor DNA detects molecular residual disease and predicts recurrence in gastric cancer. Cell Death Dis 2020, 11, 346. [Google Scholar] [CrossRef]
- Nong, J.; Gong, Y.; Guan, Y.; Yi, X.; Yi, Y.; Chang, L.; Yang, L.; Lv, J.; Guo, Z.; Jia, H.; et al. Circulating tumor DNA analysis depicts subclonal architecture and genomic evolution of small cell lung cancer. Nat Commun 2018, 9, 3114. [Google Scholar] [CrossRef]
- Chatterjee, T.; Mallhi, R.S.; Venkatesan, S. Minimal residual disease detection using flow cytometry: Applications in acute leukemia. Med J Armed Forces India 2016, 72, 152–156. [Google Scholar] [CrossRef]
- Dohner, H.; Wei, A.H.; Appelbaum, F.R.; Craddock, C.; DiNardo, C.D.; Dombret, H.; Ebert, B.L.; Fenaux, P.; Godley, L.A.; Hasserjian, R.P.; et al. Diagnosis and management of AML in adults: 2022 recommendations from an international expert panel on behalf of the ELN. Blood 2022, 140, 1345–1377. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Shields, M.D.; Chauhan, P.S.; Ramirez, R.J.; Harris, P.K.; Reimers, M.A.; Zevallos, J.P.; Davis, A.A.; Pellini, B.; Chaudhuri, A.A. Commercial ctDNA Assays for Minimal Residual Disease Detection of Solid Tumors. Mol Diagn Ther 2021, 25, 757–774. [Google Scholar] [CrossRef]
- Steeg, P.S. Tumor metastasis: mechanistic insights and clinical challenges. Nat Med 2006, 12, 895–904. [Google Scholar] [CrossRef]
- Jongen-Lavrencic, M.; Grob, T.; Hanekamp, D.; Kavelaars, F.G.; Al Hinai, A.; Zeilemaker, A.; Erpelinck-Verschueren, C.A.J.; Gradowska, P.L.; Meijer, R.; Cloos, J.; et al. Molecular Minimal Residual Disease in Acute Myeloid Leukemia. N Engl J Med 2018, 378, 1189–1199. [Google Scholar] [CrossRef] [PubMed]
- Jamroziak, K.; Pula, B. Measurable Residual Disease in Hematological Cancers. Cancers (Basel) 2024, 16. [Google Scholar] [CrossRef]
- Honore, N.; Galot, R.; van Marcke, C.; Limaye, N.; Machiels, J.P. Liquid Biopsy to Detect Minimal Residual Disease: Methodology and Impact. Cancers (Basel) 2021, 13. [Google Scholar] [CrossRef]
- Fares, J.; Fares, M.Y.; Khachfe, H.H.; Salhab, H.A.; Fares, Y. Molecular principles of metastasis: a hallmark of cancer revisited. Signal Transduct Target Ther 2020, 5, 28. [Google Scholar] [CrossRef]
- Nagrath, S.; Sequist, L.V.; Maheswaran, S.; Bell, D.W.; Irimia, D.; Ulkus, L.; Smith, M.R.; Kwak, E.L.; Digumarthy, S.; Muzikansky, A.; et al. Isolation of rare circulating tumour cells in cancer patients by microchip technology. Nature 2007, 450, 1235–1239. [Google Scholar] [CrossRef]
- Kahn, H.J.; Presta, A.; Yang, L.Y.; Blondal, J.; Trudeau, M.; Lickley, L.; Holloway, C.; McCready, D.R.; Maclean, D.; Marks, A. Enumeration of circulating tumor cells in the blood of breast cancer patients after filtration enrichment: correlation with disease stage. Breast Cancer Res Treat 2004, 86, 237–247. [Google Scholar] [CrossRef]
- Badia-Ramentol, J.; Linares, J.; Gomez-Llonin, A.; Calon, A. Minimal Residual Disease, Metastasis and Immunity. Biomolecules 2021, 11. [Google Scholar] [CrossRef] [PubMed]
- Massague, J.; Obenauf, A.C. Metastatic colonization by circulating tumour cells. Nature 2016, 529, 298–306. [Google Scholar] [CrossRef]
- Dongre, A.; Weinberg, R.A. New insights into the mechanisms of epithelial-mesenchymal transition and implications for cancer. Nat Rev Mol Cell Biol 2019, 20, 69–84. [Google Scholar] [CrossRef]
- Janin, M.; Davalos, V.; Esteller, M. Cancer metastasis under the magnifying glass of epigenetics and epitranscriptomics. Cancer Metastasis Rev 2023, 42, 1071–1112. [Google Scholar] [CrossRef]
- Marusiak, A.A.; Prelowska, M.K.; Mehlich, D.; Lazniewski, M.; Kaminska, K.; Gorczynski, A.; Korwat, A.; Sokolowska, O.; Kedzierska, H.; Golab, J.; et al. Upregulation of MLK4 promotes migratory and invasive potential of breast cancer cells. Oncogene 2019, 38, 2860–2875. [Google Scholar] [CrossRef] [PubMed]
- Ilango, S.; Paital, B.; Jayachandran, P.; Padma, P.R.; Nirmaladevi, R. Epigenetic alterations in cancer. Front Biosci (Landmark Ed) 2020, 25, 1058–1109. [Google Scholar] [CrossRef]
- Szyf, M.; Pakneshan, P.; Rabbani, S.A. DNA methylation and breast cancer. Biochem Pharmacol 2004, 68, 1187–1197. [Google Scholar] [CrossRef]
- Mian, H.; Wildes, T.M.; Venner, C.P.; Fonseca, R. MRD negativity: considerations for older adults with multiple myeloma. Blood Cancer J 2023, 13, 166. [Google Scholar] [CrossRef] [PubMed]
- Boyiadzis, M.; Wei, A.H.; Paiva, B.; Freeman, S.D.; Kaspers, G.; Chyla, B.; Hersey, S.; Patel, R.; Maloney, B.; Blanchet Zumofen, M.H.; et al. Measurable residual disease (MRD) as a surrogate end point for clinical drug approval in acute myeloid leukemia (AML): Perspectives from the MRD Partnership and Alliance in AML Clinical Treatment Consortium. Cancer 2025, 131, e35960. [Google Scholar] [CrossRef] [PubMed]
- Shimony, S.; Stahl, M.; Stone, R.M. Acute Myeloid Leukemia: 2025 Update on Diagnosis, Risk-Stratification, and Management. Am J Hematol 2025, 100, 860–891. [Google Scholar] [CrossRef]
- Ivey, A.; Hills, R.K.; Simpson, M.A.; Jovanovic, J.V.; Gilkes, A.; Grech, A.; Patel, Y.; Bhudia, N.; Farah, H.; Mason, J.; et al. Assessment of Minimal Residual Disease in Standard-Risk AML. N Engl J Med 2016, 374, 422–433. [Google Scholar] [CrossRef]
- Short, N.J.; Zhou, S.; Fu, C.; Berry, D.A.; Walter, R.B.; Freeman, S.D.; Hourigan, C.S.; Huang, X.; Nogueras Gonzalez, G.; Hwang, H.; et al. Association of Measurable Residual Disease With Survival Outcomes in Patients With Acute Myeloid Leukemia: A Systematic Review and Meta-analysis. JAMA Oncol 2020, 6, 1890–1899. [Google Scholar] [CrossRef]
- Pratz, K.W.; Jonas, B.A.; Pullarkat, V.; Recher, C.; Schuh, A.C.; Thirman, M.J.; Garcia, J.S.; DiNardo, C.D.; Vorobyev, V.; Fracchiolla, N.S.; et al. Measurable Residual Disease Response and Prognosis in Treatment-Naive Acute Myeloid Leukemia With Venetoclax and Azacitidine. J Clin Oncol 2022, 40, 855–865. [Google Scholar] [CrossRef] [PubMed]
- Othman, J.; Tiong, I.S.; O'Nions, J.; Dennis, M.; Mokretar, K.; Ivey, A.; Austin, M.; Latif, A.L.; Amer, M.; Chan, W.Y.; et al. Molecular MRD is strongly prognostic in patients with NPM1-mutated AML receiving venetoclax-based nonintensive therapy. Blood 2024, 143, 336–341. [Google Scholar] [CrossRef]
- Pasquini, M.C.; Wallace, P.K.; Logan, B.; Kaur, M.; Tario, J.D.; Howard, A.; Zhang, Y.; Brunstein, C.; Efebera, Y.; Geller, N.; et al. Minimal Residual Disease Status in Multiple Myeloma 1 Year After Autologous Hematopoietic Cell Transplantation and Lenalidomide Maintenance Are Associated With Long-Term Overall Survival. J Clin Oncol 2024, 42, 2757–2768. [Google Scholar] [CrossRef]
- Tiong, I.S.; Loo, S. Targeting Measurable Residual Disease (MRD) in Acute Myeloid Leukemia (AML): Moving beyond Prognostication. Int J Mol Sci 2023, 24. [Google Scholar] [CrossRef] [PubMed]
- Nachmias, B.; Krichevsky, S.; Gatt, M.E.; Gross Even-Zohar, N.; Shaulov, A.; Haran, A.; Aumann, S.; Vainstein, V. Standardization of Molecular MRD Levels in AML Using an Integral Vector Bearing ABL and the Mutation of Interest. Cancers (Basel) 2023, 15. [Google Scholar] [CrossRef] [PubMed]
- Zuna, J.; Hovorkova, L.; Krotka, J.; Koehrmann, A.; Bardini, M.; Winkowska, L.; Fronkova, E.; Alten, J.; Koehler, R.; Eckert, C.; et al. Minimal residual disease in BCR::ABL1-positive acute lymphoblastic leukemia: different significance in typical ALL and in CML-like disease. Leukemia 2022, 36, 2793–2801. [Google Scholar] [CrossRef]
- Chandhok, N.S.; Sekeres, M.A. Measurable residual disease in hematologic malignancies: a biomarker in search of a standard. EClinicalMedicine 2025, 86, 103348. [Google Scholar] [CrossRef]
- Chen, B.; Pan, Q.; Dong, Y. Advancing MRD Detection in Multiple Myeloma: Technologies, Applications, and Future Perspectives. Am J Clin Oncol 2025, 48, 376–380. [Google Scholar] [CrossRef]
- Munshi, N.C.; Avet-Loiseau, H.; Anderson, K.C.; Neri, P.; Paiva, B.; Samur, M.; Dimopoulos, M.; Kulakova, M.; Lam, A.; Hashim, M.; et al. A large meta-analysis establishes the role of MRD negativity in long-term survival outcomes in patients with multiple myeloma. Blood Adv 2020, 4, 5988–5999. [Google Scholar] [CrossRef]
- Anderson, K.C.; Auclair, D.; Adam, S.J.; Agarwal, A.; Anderson, M.; Avet-Loiseau, H.; Bustoros, M.; Chapman, J.; Connors, D.E.; Dash, A.; et al. Minimal Residual Disease in Myeloma: Application for Clinical Care and New Drug Registration. Clin Cancer Res 2021, 27, 5195–5212. [Google Scholar] [CrossRef] [PubMed]
- Szalat, R.; Anderson, K.; Munshi, N. Role of minimal residual disease assessment in multiple myeloma. Haematologica 2024, 109, 2049–2059. [Google Scholar] [CrossRef]
- Jimenez-Vicente, C.; Cardus, A.; Castano-Diez, S.; Ramil, G.; Garcia-Avila, S.; Gomez-Perez, L.; Esteban, D.; Arribas, I.; Sturla, A.L.; Lopez-Guerra, M.; et al. Prognostic value of measurable residual disease (MRD) for venetoclax in combination with hypomethylating agents in patients diagnosed with acute myeloid leukemia: validation of the ELN-2021 MRD recommendations. Blood Cancer J 2025, 15, 94. [Google Scholar] [CrossRef]
- Reinert, T.; Henriksen, T.V.; Christensen, E.; Sharma, S.; Salari, R.; Sethi, H.; Knudsen, M.; Nordentoft, I.; Wu, H.T.; Tin, A.S.; et al. Analysis of Plasma Cell-Free DNA by Ultradeep Sequencing in Patients With Stages I to III Colorectal Cancer. JAMA Oncol 2019, 5, 1124–1131. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Murillas, I.; Schiavon, G.; Weigelt, B.; Ng, C.; Hrebien, S.; Cutts, R.J.; Cheang, M.; Osin, P.; Nerurkar, A.; Kozarewa, I.; et al. Mutation tracking in circulating tumor DNA predicts relapse in early breast cancer. Sci Transl Med 2015, 7, 302ra133. [Google Scholar] [CrossRef]
- Zheng, J.; Qin, C.; Wang, Q.; Tian, D.; Chen, Z. Circulating tumour DNA-Based molecular residual disease detection in resectable cancers: a systematic review and meta-analysis. EBioMedicine 2024, 103, 105109. [Google Scholar] [CrossRef] [PubMed]
- Mannucci, A.; Goel, A. Stool and blood biomarkers for colorectal cancer management: an update on screening and disease monitoring. Mol Cancer 2024, 23, 259. [Google Scholar] [CrossRef]
- Niu, F.; Wen, J.; Fu, X.; Li, C.; Zhao, R.; Wu, S.; Yu, H.; Liu, X.; Zhao, X.; Liu, S.; et al. Stool DNA Test of Methylated Syndecan-2 for the Early Detection of Colorectal Neoplasia. Cancer Epidemiol Biomarkers Prev 2017, 26, 1411–1419. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Park, S.C. Syndecan-2 Methylation as a New Biomarker for Early Detection of Colorectal Neoplasm. Gut Liver 2018, 12, 479–480. [Google Scholar] [CrossRef]
- Ma, D.; Gao, X.; Wang, L.; Yin, H.; Feng, L.; Zhu, Y. Circulating tumor DNA for MRD detection in colorectal cancer: recent advances and clinical implications. Biomark Res 2025, 13, 89. [Google Scholar] [CrossRef]
- Peng, Y.; Mei, W.; Ma, K.; Zeng, C. Circulating Tumor DNA and Minimal Residual Disease (MRD) in Solid Tumors: Current Horizons and Future Perspectives. Front Oncol 2021, 11, 763790. [Google Scholar] [CrossRef]
- Chin, R.I.; Chen, K.; Usmani, A.; Chua, C.; Harris, P.K.; Binkley, M.S.; Azad, T.D.; Dudley, J.C.; Chaudhuri, A.A. Detection of Solid Tumor Molecular Residual Disease (MRD) Using Circulating Tumor DNA (ctDNA). Mol Diagn Ther 2019, 23, 311–331. [Google Scholar] [CrossRef]
- Hoang, V.-A.N.; Nguyen, N.; Le, D.N.; Nguyen, V.S.; Thi, H.G.N.; Vu, H.T.; Ho, H.H.; Le, H.M.; Nguyen, T.D.; Vo, H.N.; et al. Real-World Utilization and Performance of Circulating Tumor DNA Monitoring to Predict Recurrence in Solid Tumors. JCO Oncology Advances 2025, e2400084. [Google Scholar] [CrossRef]
- Duineveld, L.A.; van Asselt, K.M.; Bemelman, W.A.; Smits, A.B.; Tanis, P.J.; van Weert, H.C.; Wind, J. Symptomatic and Asymptomatic Colon Cancer Recurrence: A Multicenter Cohort Study. Ann Fam Med 2016, 14, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Tie, J.; Wang, Y.; Tomasetti, C.; Li, L.; Springer, S.; Kinde, I.; Silliman, N.; Tacey, M.; Wong, H.L.; Christie, M.; et al. Circulating tumor DNA analysis detects minimal residual disease and predicts recurrence in patients with stage II colon cancer. Sci Transl Med 2016, 8, 346ra392. [Google Scholar] [CrossRef]
- Tie, J.; Wang, Y.; Cohen, J.; Li, L.; Hong, W.; Christie, M.; Wong, H.L.; Kosmider, S.; Wong, R.; Thomson, B.; et al. Circulating tumor DNA dynamics and recurrence risk in patients undergoing curative intent resection of colorectal cancer liver metastases: A prospective cohort study. PLoS Med 2021, 18, e1003620. [Google Scholar] [CrossRef]
- Parikh, A.R.; Van Seventer, E.E.; Siravegna, G.; Hartwig, A.V.; Jaimovich, A.; He, Y.; Kanter, K.; Fish, M.G.; Fosbenner, K.D.; Miao, B.; et al. Minimal Residual Disease Detection using a Plasma-only Circulating Tumor DNA Assay in Patients with Colorectal Cancer. Clin Cancer Res 2021, 27, 5586–5594. [Google Scholar] [CrossRef]
- Abbosh, C.; Birkbak, N.J.; Wilson, G.A.; Jamal-Hanjani, M.; Constantin, T.; Salari, R.; Le Quesne, J.; Moore, D.A.; Veeriah, S.; Rosenthal, R.; et al. Phylogenetic ctDNA analysis depicts early-stage lung cancer evolution. Nature 2017, 545, 446–451. [Google Scholar] [CrossRef] [PubMed]
- Lu, D.; Lin, N.; Li, S.; Jing, Q.; Yin, J.C.; Shi, L.; Zhang, Z.; Chen, Z.; Wang, Z.; Tong, Y.; et al. Predictive Effectiveness of Circulating Tumor DNA in Recurrent Early-Stage Non-Small Cell Lung Cancer: An Updated Meta-Analysis. JCO Precis Oncol 2025, 9, e2500489. [Google Scholar] [CrossRef] [PubMed]
- Bartolomucci, A.; Nobrega, M.; Ferrier, T.; Dickinson, K.; Kaorey, N.; Nadeau, A.; Castillo, A.; Burnier, J.V. Circulating tumor DNA to monitor treatment response in solid tumors and advance precision oncology. NPJ Precis Oncol 2025, 9, 84. [Google Scholar] [CrossRef]
- Elliott, M.J.; Echelard, P.; Pipinikas, C.; Main, S.; Fuentes Antras, J.; Dou, A.; Veitch, Z.; Amir, E.; Nadler, M.B.; Meti, N.; et al. Longitudinal evaluation of circulating tumor DNA in patients undergoing neoadjuvant therapy for early breast cancer using a tumor-informed assay. Nat Commun 2025, 16, 1837. [Google Scholar] [CrossRef]
- Cheng, X. A Comprehensive Review of HER2 in Cancer Biology and Therapeutics. Genes (Basel) 2024, 15. [Google Scholar] [CrossRef]
- Chen, H.; Gu, M.; Liang, J.; Song, H.; Zhang, J.; Xu, W.; Zhao, F.; Shen, D.; Shen, H.; Liao, C.; et al. Minimal residual disease detection by next-generation sequencing of different immunoglobulin gene rearrangements in pediatric B-ALL. Nat Commun 2023, 14, 7468. [Google Scholar] [CrossRef] [PubMed]
- Pulsipher, M.A.; Han, X.; Maude, S.L.; Laetsch, T.W.; Qayed, M.; Rives, S.; Boyer, M.W.; Hiramatsu, H.; Yanik, G.A.; Driscoll, T.; et al. Next-Generation Sequencing of Minimal Residual Disease for Predicting Relapse after Tisagenlecleucel in Children and Young Adults with Acute Lymphoblastic Leukemia. Blood Cancer Discov 2022, 3, 66–81. [Google Scholar] [CrossRef] [PubMed]
- Corne, J.; Le Du, F.; Quillien, V.; Godey, F.; Robert, L.; Bourien, H.; Brunot, A.; Crouzet, L.; Perrin, C.; Lefeuvre-Plesse, C.; et al. Development of multiplex digital PCR assays for the detection of PIK3CA mutations in the plasma of metastatic breast cancer patients. Sci Rep 2021, 11, 17316. [Google Scholar] [CrossRef] [PubMed]
- Taly, V.; Pekin, D.; Benhaim, L.; Kotsopoulos, S.K.; Le Corre, D.; Li, X.; Atochin, I.; Link, D.R.; Griffiths, A.D.; Pallier, K.; et al. Multiplex picodroplet digital PCR to detect KRAS mutations in circulating DNA from the plasma of colorectal cancer patients. Clin Chem 2013, 59, 1722–1731. [Google Scholar] [CrossRef]
- Shong, L.Y.; Deng, J.Y.; Kwok, H.H.; Lee, N.C.; Tseng, S.C.; Ng, L.Y.; Yee, W.K.; Lam, D.C. Detection of EGFR mutations in patients with suspected lung cancer using paired tissue-plasma testing: a prospective comparative study with plasma ddPCR assay. Sci Rep 2024, 14, 25701. [Google Scholar] [CrossRef]
- Pilcher, W.; Thomas, B.E.; Bhasin, S.S.; Jayasinghe, R.G.; Yao, L.; Gonzalez-Kozlova, E.; Dasari, S.; Kim-Schulze, S.; Rahman, A.; Patton, J.; et al. Cross center single-cell RNA sequencing study of the immune microenvironment in rapid progressing multiple myeloma. NPJ Genom Med 2023, 8, 3. [Google Scholar] [CrossRef]
- Matera, A.; Marella, A.; Maeda, A.; Da Via, M.C.; Lazzaroni, F.; Fabris, S.; Pioggia, S.; Porretti, L.; Colombo, F.; Torricelli, F.; et al. Single-Cell RNA Sequencing for the Detection of Clonotypic V(D)J Rearrangements in Multiple Myeloma. Int J Mol Sci 2022, 23. [Google Scholar] [CrossRef]
- Soh, K.T.; Tario, J.D., Jr.; Hahn, T.E.; Hillengass, J.; McCarthy, P.L.; Wallace, P.K. Methodological considerations for the high sensitivity detection of multiple myeloma measurable residual disease. Cytometry B Clin Cytom 2020, 98, 161–173. [Google Scholar] [CrossRef]
- Engelmann, R.; Flores-Montero, J.; Schilperoord-Vermeulen, J.; Ritgen, M.; Hengeveld, P.J.; Kohlscheen, S.; Grigore, G.; Rodriguez, R.F.; Lecrevisse, Q.; Philippe, J.; et al. Novel Flow Cytometric Antibody Panel and Dedicated Analysis Algorithm for Automated Fully Standardized Minimal Residual Disease Detection in Chronic Lymphocytic Leukemia. Am J Hematol 2025, 100, 724–728. [Google Scholar] [CrossRef]
- Furstenau, M.; Weiss, J.; Giza, A.; Franzen, F.; Robrecht, S.; Fink, A.M.; Fischer, K.; Schneider, C.; Tausch, E.; Stilgenbauer, S.; et al. Circulating Tumor DNA-Based MRD Assessment in Patients with CLL Treated with Obinutuzumab, Acalabrutinib, and Venetoclax. Clin Cancer Res 2022, 28, 4203–4211. [Google Scholar] [CrossRef]
- Lim, J.; Park, C.; Kim, M.; Kim, H.; Kim, J.; Lee, D.S. Advances in single-cell omics and multiomics for high-resolution molecular profiling. Exp Mol Med 2024, 56, 515–526. [Google Scholar] [CrossRef]
- Li, Y.; Jiang, G.; Wu, W.; Yang, H.; Jin, Y.; Wu, M.; Liu, W.; Yang, A.; Chervova, O.; Zhang, S.; et al. Multi-omics integrated circulating cell-free DNA genomic signatures enhanced the diagnostic performance of early-stage lung cancer and postoperative minimal residual disease. EBioMedicine 2023, 91, 104553. [Google Scholar] [CrossRef] [PubMed]
- Tivey, A.; Lee, R.J.; Clipson, A.; Hill, S.M.; Lorigan, P.; Rothwell, D.G.; Dive, C.; Mouliere, F. Mining nucleic acid "omics" to boost liquid biopsy in cancer. Cell Rep Med 2024, 5, 101736. [Google Scholar] [CrossRef]
- Saeed, B.R.R.; Saboli, A.; Rehammar, A.; Molnar, A.; Delsing Malmberg, E.; Depreter, B.; Shah Barkhordar, G.; De Moerloose, B.; Lammens, T.; Palmqvist, L.; et al. Ultra-Deep Sequencing and Single Cell Multi-Omics Characterize Measurable Residual Disease and Identify Therapy-Survived Clone in Pediatric Acute Myeloid Leukemia. Blood 2024, 144, 4994. [Google Scholar] [CrossRef]
- Thompson, K.; Geller, B.; Nousheen, L.; Krishnan, I.; Wang, S.; Mendoza, D.; Druley, T.E.; Sciambi, A. A Multiomic, Single-Cell Measurable Residual Disease (scMRD) Assay for Simultaneous Assessment of DNA Mutations and Surface Immunophenotypes in Acute Myeloid Leukemia. Blood 2024, 144, 6168–6168. [Google Scholar] [CrossRef]
- Sanches, P.H.G.; de Melo, N.C.; Porcari, A.M.; de Carvalho, L.M. Integrating Molecular Perspectives: Strategies for Comprehensive Multi-Omics Integrative Data Analysis and Machine Learning Applications in Transcriptomics, Proteomics, and Metabolomics. Biology (Basel) 2024, 13. [Google Scholar] [CrossRef] [PubMed]
- Widman, A.J.; Shah, M.; Frydendahl, A.; Halmos, D.; Khamnei, C.C.; Ogaard, N.; Rajagopalan, S.; Arora, A.; Deshpande, A.; Hooper, W.F.; et al. Ultrasensitive plasma-based monitoring of tumor burden using machine-learning-guided signal enrichment. Nat Med 2024, 30, 1655–1666. [Google Scholar] [CrossRef]
- Mocking, T.R.; Kelder, A.; Reuvekamp, T.; Ngai, L.L.; Rutten, P.; Gradowska, P.; van de Loosdrecht, A.A.; Cloos, J.; Bachas, C. Computational assessment of measurable residual disease in acute myeloid leukemia using mixture models. Commun Med (Lond) 2024, 4, 271. [Google Scholar] [CrossRef]
- Pawlyn, C.; Davies, F.E. Predicting the Future: Machine-Based Learning for MRD Prognostication. Clin Cancer Res 2022, 28, 2482–2484. [Google Scholar] [CrossRef]
- Moritz, J.; Schwab, A.; Reinisch, A.; Zebisch, A.; Sill, H.; Wolfler, A. Measurable Residual Disease Detection in Acute Myeloid Leukemia: Current Challenges and Future Directions. Biomedicines 2024, 12. [Google Scholar] [CrossRef]
- Chan, H.T.; Nagayama, S.; Otaki, M.; Chin, Y.M.; Fukunaga, Y.; Ueno, M.; Nakamura, Y.; Low, S.K. Tumor-informed or tumor-agnostic circulating tumor DNA as a biomarker for risk of recurrence in resected colorectal cancer patients. Front Oncol 2022, 12, 1055968. [Google Scholar] [CrossRef]
- Ruiz-Torres, D.A.; Merkin, R.D.; Bryan, M.E.; Mendel, J.; Efthymiou, V.; Roberts, T.; Patel, M.J.; Park, J.C.; Chevalier, A.; Murray, C.; et al. Personalized circulating tumor DNA dynamics inform survival and response to immune checkpoint blockade in recurrent/metastatic head and neck cancer. NPJ Precis Oncol 2025, 9, 298. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Nawaz, M.A.; Zuo, Y.; Zeng, P. Next-generation immunotherapy in relapsed/refractory multiple myeloma: Strategies to achieve sustained MRD negativity. Crit Rev Oncol Hematol 2025, 214, 104913. [Google Scholar] [CrossRef]
- Gökbuget, N.; Dombret, H.; Bonifacio, M.; Reichle, A.; Graux, C.; Faul, C.; Diedrich, H.; Topp, M.S.; Brüggemann, M.; Horst, H.-A.; et al. Blinatumomab for minimal residual disease in adults with B-cell precursor acute lymphoblastic leukemia. Blood 2018, 131, 1522–1531. [Google Scholar] [CrossRef] [PubMed]
- FDA; U.S.F.a.D.A. FDA granted accelerated approval to blinatumomab (Blincyto, Amgen Inc.) for the treatment of adult and pediatric patients with B-cell precursor acute lymphoblastic leukemia. Available online: https://www.fda.gov/drugs/resources-information-approved-drugs/fda-granted-accelerated-approval-blinatumomab-blincyto-amgen-inc-treatment-adult-and-pediatric (accessed on).
- Curran, E.; Stock, W. Taking a "BiTE out of ALL": blinatumomab approval for MRD-positive ALL. Blood 2019, 133, 1715–1719. [Google Scholar] [CrossRef]
- Boissel, N.; Chiaretti, S.; Papayannidis, C.; Ribera, J.M.; Bassan, R.; Sokolov, A.N.; Alam, N.; Brescianini, A.; Pezzani, I.; Kreuzbauer, G.; et al. Real-world use of blinatumomab in adult patients with B-cell acute lymphoblastic leukemia in clinical practice: results from the NEUF study. Blood Cancer J 2023, 13, 2. [Google Scholar] [CrossRef] [PubMed]
- Short, N.J.; Jabbour, E.; Jamison, T.; Paul, S.; Cuglievan, B.; McCall, D.; Gibson, A.; Jain, N.; Haddad, F.G.; Nasr, L.F.; et al. Dose-Dense Mini-Hyper-CVD, Inotuzumab Ozogamicin and Blinatumomab Achieves Rapid MRD-Negativity in Philadelphia Chromosome-Negative B-cell Acute Lymphoblastic Leukemia. Clin Lymphoma Myeloma Leuk 2024, 24, e168–e173. [Google Scholar] [CrossRef]
- Ghorashian, S.; Lucchini, G.; Richardson, R.; Nguyen, K.; Terris, C.; Guvenel, A.; Oporto-Espuelas, M.; Yeung, J.; Pinner, D.; Chu, J.; et al. CD19/CD22 targeting with cotransduced CAR T cells to prevent antigen-negative relapse after CAR T-cell therapy for B-cell ALL. Blood 2024, 143, 118–123. [Google Scholar] [CrossRef]
- Shim, K.G.; Fonseca, R. Measurable Residual Disease Testing in Multiple Myeloma Following T-Cell Redirecting Therapies. Cancers 2024, 16, 3288. [Google Scholar] [CrossRef]
- Chari, A.; Minnema, M.C.; Berdeja, J.G.; Oriol, A.; van de Donk, N.; Rodriguez-Otero, P.; Askari, E.; Mateos, M.V.; Costa, L.J.; Caers, J.; et al. Talquetamab, a T-Cell-Redirecting GPRC5D Bispecific Antibody for Multiple Myeloma. N Engl J Med 2022, 387, 2232–2244. [Google Scholar] [CrossRef]
- Lesokhin, A.M.; Tomasson, M.H.; Arnulf, B.; Bahlis, N.J.; Miles Prince, H.; Niesvizky, R.; Rodriotaguez-Otero, P.; Martinez-Lopez, J.; Koehne, G.; Touzeau, C.; et al. Elranatamab in relapsed or refractory multiple myeloma: phase 2 MagnetisMM-3 trial results. Nat Med 2023, 29, 2259–2267. [Google Scholar] [CrossRef]
- Garfall, A.L.; Nooka, A.K.; van de Donk, N.W.; Moreau, P.; Bhutani, M.; Oriol, A.; Martin, T.G.; Rosiñol, L.; Mateos, M.-V.; Bahlis, N.J. Long-term follow-up from the phase 1/2 MajesTEC-1 trial of teclistamab in patients with relapsed/refractory multiple myeloma. 2024. [Google Scholar]
- Rasche, L.; Schinke, C.; Touzeau, C.; Minnema, M.C.; van de Donk, N.W.C.J.; Rodríguez-Otero, P.; Mateos, M.-V.; Christine Ye, J.; Vishwamitra, D.; Singh, I.; et al. MM-492 Long-term Efficacy and Safety Results From the Phase 1/2 MonumenTAL-1 Study of Talquetamab, a GPRC5D×CD3 Bispecific Antibody, in Patients With Relapsed/Refractory Multiple Myeloma (RRMM). Clinical Lymphoma Myeloma and Leukemia 2024, 24, S561–S562. [Google Scholar] [CrossRef]
- Dohner, H.; Wei, A.H.; Roboz, G.J.; Montesinos, P.; Thol, F.R.; Ravandi, F.; Dombret, H.; Porkka, K.; Sandhu, I.; Skikne, B.; et al. Prognostic impact of NPM1 and FLT3 mutations in patients with AML in first remission treated with oral azacitidine. Blood 2022, 140, 1674–1685. [Google Scholar] [CrossRef]
- Levis, M.J.; Erba, H.P.; Montesinos, P.; Vrhovac, R.; Patkowska, E.; Kim, H.; Zak, P.; Wang, P.-N.; Rohrbach, J.E.C.; Chang, K.C.; et al. Quantum-First Trial: FLT3-ITD-Specific MRD Clearance Is Associated with Improved Overall Survival. Blood 2022, 140, 546–548. [Google Scholar] [CrossRef]
- Othman, J.; Potter, N.; Mokretar, K.; Taussig, D.; Khan, A.; Krishnamurthy, P.; Latif, A.-L.; Cahalin, P.; Aries, J.; Amer, M.; et al. High Molecular Response Rate and Overall Survival with FLT3 Inhibitors As MRD-Guided Salvage Treatment for Molecular Failure in AML. Blood 2022, 140, 2002–2004. [Google Scholar] [CrossRef]
- Wood, H.; Bourlon, C.; Kulasekararaj, A.; Borg, A.; Pavlu, J.; Elder, P.; Taussig, D.C.; Veitch, S.; Knapper, S.; Othman, J.; et al. Venetoclax-Based Non-Intensive Combinations Successfully Salvage Molecular Relapse of Acute Myeloid Leukemia and Are an Important Bridge to Cellular Therapy in Relapsed/Refractory Disease - Real-World Data from a UK-Wide Programme. Blood 2022, 140, 9016–9018. [Google Scholar] [CrossRef]
- Bhutani, M.; Robinson, M.; Foureau, D.; Atrash, S.; Paul, B.; Guo, F.; Grayson, J.M.; Ivanina-Foureau, A.; Pineda-Roman, M.; Varga, C.; et al. MRD-driven phase 2 study of daratumumab, carfilzomib, lenalidomide, and dexamethasone in newly diagnosed multiple myeloma. Blood Adv 2025, 9, 507–519. [Google Scholar] [CrossRef]
- Chen, J.; Gale, R.P.; Hu, Y.; Yan, W.; Wang, T.; Zhang, W. Measurable residual disease (MRD)-testing in haematological and solid cancers. Leukemia 2024, 38, 1202–1212. [Google Scholar] [CrossRef] [PubMed]
- Song, M.; Pan, W.; Yu, X.; Ren, J.; Tang, C.; Chen, Z.; Wang, Z.; Deng, Y.; He, N.; Liu, H.; et al. Minimal Residual Disease Detection: Implications for Clinical Diagnosis and Cancer Patient Treatment. MedComm (2020) 2025, 6, e70193. [Google Scholar] [CrossRef] [PubMed]
- Li, W. Measurable Residual Disease Testing in Acute Leukemia: Technology and Clinical Significance. In Leukemia; Li, W., Ed.; Brisbane (AU), 2022. [Google Scholar]
- Moding, E.J.; Nabet, B.Y.; Alizadeh, A.A.; Diehn, M. Detecting Liquid Remnants of Solid Tumors: Circulating Tumor DNA Minimal Residual Disease. Cancer Discov 2021, 11, 2968–2986. [Google Scholar] [CrossRef] [PubMed]
| MRD Method | Sample Type | Typical Sensitivity | Primary Clinical Use | Strengths | Limitations | Current Clinical Actionability | References |
|---|---|---|---|---|---|---|---|
| Multiparameter Flow Cytometry (MFC) | Bone marrow, blood | 10⁻⁴–10⁻⁵ | Hematologic malignancies (ALL, AML, CLL, MM) | Broad applicability; rapid turnaround | Lower sensitivity than molecular methods; inter-lab variability | High in hematologic malignancies (risk stratification, transplant decisions) | [70] |
| RT-qPCR | Bone marrow, blood | 10⁻⁵–10⁻⁶ | Fusion genes / recurrent mutations (e.g. BCR-ABL1, NPM1) | High sensitivity; standardized for selected targets | Limited to known targets; relative quantification | High for selected molecular subtypes | [82] |
| Next-Generation Sequencing (NGS) | Bone marrow, blood, plasma | 10⁻⁵–10⁻⁶ | Hematologic and solid tumors | Detects clonal heterogeneity; tumor-informed assays | Cost; bioinformatics complexity; clonal hematopoiesis confounding | High (hematologic); Moderate (solid tumors) | [63] |
| Digital PCR (ddPCR / cdPCR) | Blood, plasma | 10⁻⁴–10⁻⁵ | Targeted mutation tracking | Absolute quantification; high precision | Limited multiplexing; target-restricted | Moderate–High for known mutations | [65,66,67] |
| ctDNA (tumor-informed) | Plasma | 10⁻⁵–10⁻⁶ | Solid tumor MRD detection | High specificity; early relapse detection | Requires tumor tissue; cost | Moderate (mainly within trials) | [83] |
| ctDNA (tumor-agnostic) | Plasma | 10⁻³–10⁻⁵ | Broad surveillance, variant discovery | No tumor tissue required; broader mutation capture | Lower specificity for MRD | Low–Moderate | [83] |
| Single-cell sequencing (scRNA-seq / scMRD) | Bone marrow, tissue | <10⁻⁶ (theoretical) | Clonal architecture, resistant subpopulations | Ultra-high resolution; biological insight | Cost; scalability; limited clinical validation | Exploratory | [69] |
| Multi-omics / AI-integrated platforms | Plasma, tissue | Variable | Advanced MRD detection and prediction | Signal enhancement; low tumor fraction detection | Regulatory and interpretability challenges | Exploratory / Early clinical | [80,81] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




