Submitted:
27 February 2026
Posted:
02 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
3. Results
3.1. Emerging Evidence of Cholesterol Dysregulation in ASD and COVID-19
3.1.1. Lipid Rafts and Membrane Function
3.1.2. Lipid Profile Abnormalities
3.1.3. Inflammation and Oxidative Stress
3.2. Glucose Dysregulation: Possible Mechanistic Links Between ASD and COVID-19
3.2.1. Systemic Inflammation and Insulin Resistance
3.2.2. Oxidative Stress and Glucose Dysregulation
3.2.3. Developmental Impacts and Long-Term Risks
3.2.4. Glucose Transport and Therapeutic Targets
3.3. White Blood Cell (WBC) Differentials: Immune Dysregulation in ASD and COVID-19
3.3.1. Elevated NLR in ASD and COVID-19
3.3.2. Cytokine Profiles and Immune Activation
3.3.3. Implications for Clinical Management
3.4. Ferritin and Iron Homeostasis: Divergent Roles in ASD and COVID-19
3.4.1. Low Ferritin in ASD: Neurodevelopmental and Behavioral Implications
3.4.2. Hyperferritinemia in COVID-19: Inflammatory Marker and Prognostic Tool
3.4.3. Iron Dysregulation in ASD–COVID-19 Intersection
4. Discussion: Summary, Future Treatments/Directions
4.1. Cholesterol Dysregulation
4.2. Glucose Dysregulation
4.3. White Blood Cell and Neutrophil Dysregulation
4.4. Ferritin
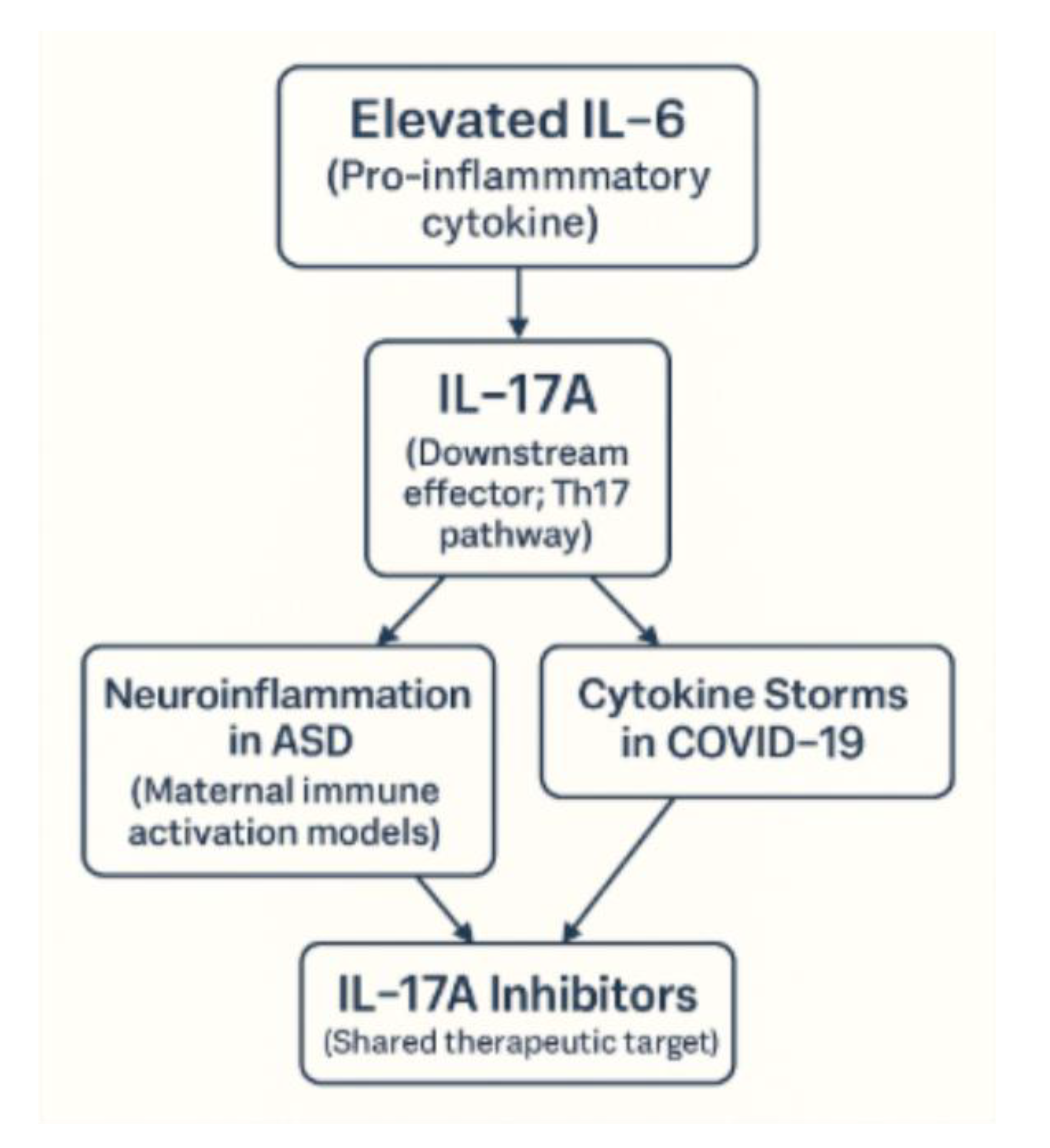
4.5. Cytokines and IL-17A as a Bridge
4.6. IL-6/IL-17A Axis: Impact on Microglial and Mitochondrial Dysfunction
4.7. Il-6/Il-17A Axis: Oxidative Stress Pathways
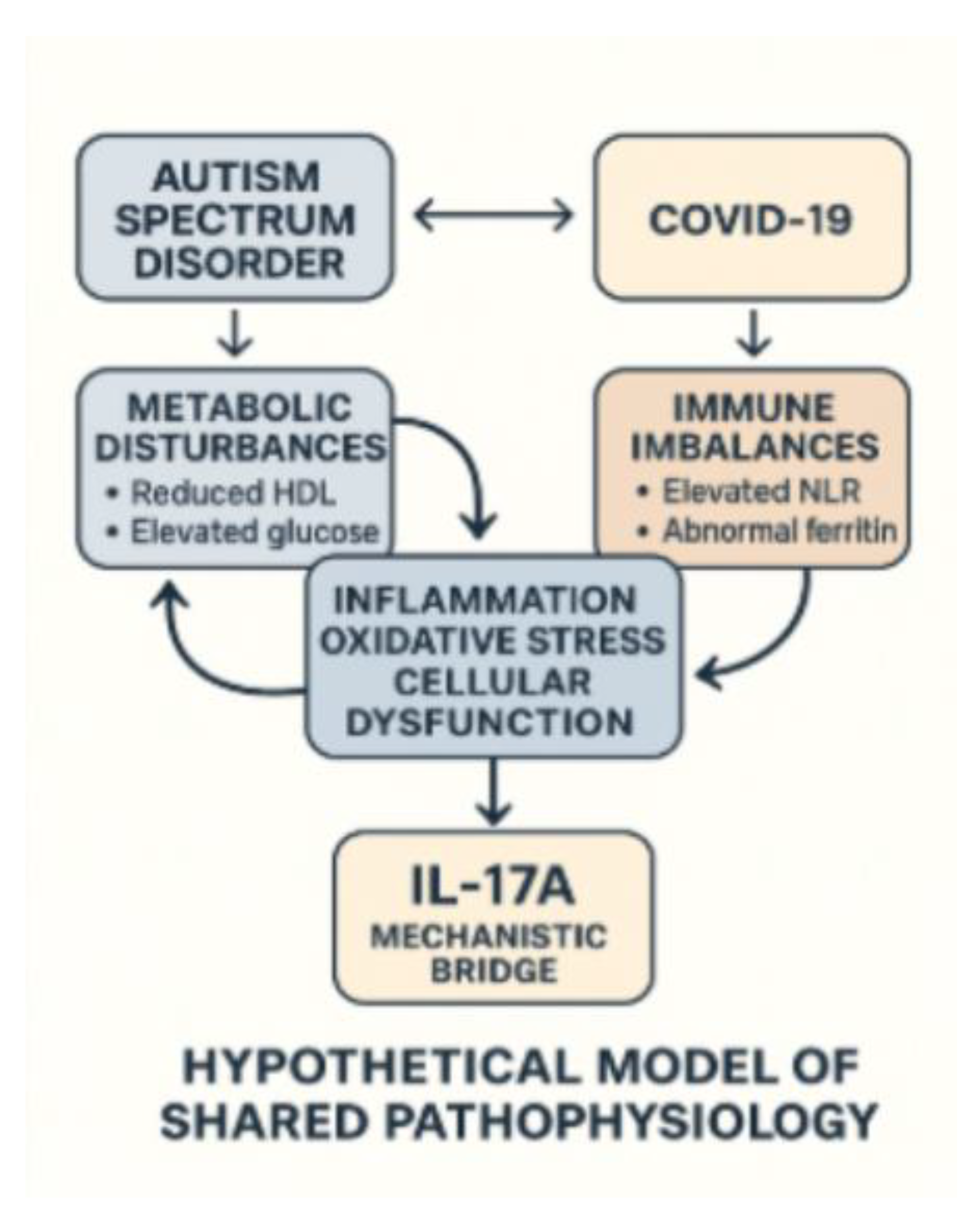
4.9. Hypothetical Model of Overlapping Metabolic and Immune Pathways in ASD and COVID-19
4.10. Clinical Implications
4.11. Limitations and Future Directions
- Age- and sex-stratified biomarker analyses
- Longitudinal biomarker monitoring in ASD-COVID cohorts
- Nutritional and behavioral interventions to enhance metabolic resilience
- Immune-modulating therapies (e.g. IL-6, IL-17A inhibitors, 2-DG)
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
Appendix A
| Cholesterol Function | ASD | COVID-19 |
| Typical Trend | Low HDL (Tierney et al., 2021; Benachenhou et al., 2019), variable LDL/total cholesterol (Gaspar et al., 2024; Kim et al., 2010) | Low HDL, LDL, total cholesterol (especially in severe cases) (Fan et al., 2020; Wei et al., 2020; Ding et al., 2020) |
| Clinical Role | Neurodevelopment, synaptic plasticity, membrane signaling (Wang 2014; Kalinowska et al., 2015) | Viral entry, disease progression, immune modulation (Kluck et al., 2021; Masana et al., 2021) |
| Key Mechanism | Lipid raft disruption, oxidative stress (Lingampelly et al., 2024; Kalinowska et al., 2015) | Lipid raft-dependent ACE2 entry, inflammation-induced dyslipidemia (Sorice et al., 2021; Palacios-Rápalo et al., 2021; Wang et al., 2020; Kluck et al., 2021; Al-Kuraishy et al., 2023) |
| Prognostic Value | Emerging (adaptive function, oxidative stress markers) (Goicoechea et al., 2023) | Moderate to strong (severity, ICU admission, prolonged shedding) (Tanaka et al., 2020; Ding et al., 2020) |
| Glucose Function | ASD | COVID-19 |
| Typical Trend | Insulin resistance, variable fasting glucose (Manco et al., 2021; Zhang et al., 2019) | Hyperglycemia, insulin resistance (Logette et al., 2021; Michaels et al., 2024; Shestakova et al., 2022; Soto et al., 2022) |
| Clinical Role | Neurodevelopment, energy metabolism, cognitive function (Manco et al., 2021; Liu M. et al., 2023) | Predictor of severity, β-cell infection, cytokine storm trigger (Michaels et al., 2024; Soto et al., 2022) |
| Key Mechanism | Impaired insulin signaling, oxidative stress, β-cell dysfunction (Qin et al., 2025; Cheng, J. et al., 2021; Liu X. et al., 2022, Zhang et al., 2019) | Glycosylation of ACE2, cytokine-induced insulin resistance (Liu S. et al., 2020; Codo et al., 2020; Huang Z. et al., 2022) |
| Prognostic Value | Moderate (diabetes risk, developmental impact) (Hoirisch-Clapauch & Nardi 2019; Liu M. et al., 2023) | Strong (severity, ICU, new-onset diabetes post-infection) (Shestakova et al., 2022; Logette et al., 2021; Soto et al., 2022) |
| WBC Parameters | ASD | COVID-19 |
| Typical Trend | Elevated neutrophils, reduced lymphocytes, high NLR (Kutlu et al., 2018; Kulaksizoglu et al., 2019; Siniscalco et al., 2018) | Elevated neutrophils, lymphopenia, high NLR (Toori et al., 2021; La Torre et al., 2022; Ergenç et al., 2021) |
| Clinical Role | Marker of immune dysfunction and chronic inflammation (Kutlu et al., 2018; Hesapcioglu et al., 2017) | Severity indicator, inflammation monitor (Rathod et al., 2022; La Torre et al., 2022 ) |
| Key Mechanism | Chronic low-grade inflammation, innate-adaptive imbalance (Kutlu et al., 2018; Goines & Ashwood 2013) | Acute inflammation, cytokine storm, immune exhaustion (Paranga et al., 2024; Toori et al., 2021) |
| Prognostic Value | Limited to emerging (Kutlu et al., 2018; Kulaksizoglu et al., 2019; Siniscalco et al., 2018; Hesapcioglu et al., 2017) | Strong (severity, hospitalization, mortality risk) (Rathod et al., 2022) |
| Ferritin Function | ASD | COVID-19 |
| Typical Trend | Iron deficiency, low ferritin (Reynolds et al., 2012; Giacomo et al., 2022; Samy et al., 2024; Sidrak et al., 2013; Chen et al., 2013) | Elevated ferritin (Rio et al., 2020; Girelli et al., 2021; Kaushal et al., 2022; Cheng L. et al., 2020; Kappart et al., 2020) |
| Clinical Role | Iron deficiency, sleep disturbances, neuroinflammation (Youssef et al., (2013); Zhou et al., 2024; DelRosso et al., 2022) | Prognostic biomarker, cytokine storm indicator (Rio et al., 2020; Girelli et al., 2021; Cheng L. et al., 2020; Mahat et al., 2020) |
| Key Mechanism | Dysregulated ferritinophagy, chronic inflammation (Zhou et al., 2024; Cheng R. et al., 2021; McCarthy et al., 2022) | IL-6-mediated synthesis, acute hyperinflammation (Hippchen et al., 2020; Peng et al., 2022; Hirschhorn et al., 2019; Chaubey et al., 2023) |
| Prognostic Value | Limited | Strong (severity, mortality, brain fog) (Rio et al., 2020; Girelli et al., 2021; Kappert et al., 2020) |
| Biomarker | ASD Trend | COVID-19 Trend | Clinical Implication |
| Cholesterol | ↓HDL, mixed LDL (Tierney et al., 2021; Benachenhou et al., 2019; Gaspar et al., 2024; Kim et al., 2010) | ↓ HDL & LDL (acute phase) (Fan et al., 2020; Wei et al., 2020; Ding et al., 2020) | Neuroinflammation, synaptic dysfunction (Kluck et al., 2021; Lingampelly et al., 2024; Goicoechea et al., 2023; Masana et al., 2021) |
| Glucose | ↑ Insulin resistance, sometimes ↓ fasting BG (Manco et al., 2021; Zhang et al., 2019) | ↑ Hyperglycemia (Logette et al., 2021) |
Neuronal energy deficits, metabolic stress (Manco et al., 2021; Michaels et al., 2024) |
| Neutrophil-to-Lymphocyte Ratio (NLR) | ↑ (Chronic low-grade inflammation) (Kulaksizoglu et al., 2019; Siniscalco et al., 2018) |
↑ (acute inflammation) (Rathod et al., 2022; Paranga et al., 2024; Toori et al., 2021) | Immune dysregulation, possible risk stratification marker (Paranga et al., 2024; Rathod et al., 2022; Kutlu et al., 2018) |
| Ferritin | ↓ (due to iron deficiency) (Giacomo et al., 2022; Samy et al 2024) | ↑ (acute-phase reactant) (Kaushal et al., 2022; Cheng L. et al., 2020; Kappert et al., 2020) | Iron metabolism imbalance, oxidative stress (Youssef et al., 2013; DelRosso et al., 2022; Gomez-Pastor et al., 2020; Mahat et al., 2020; Kernan et al., 2017) |
| Biomarker | Findings | Clinical Application |
| Cholesterol | Dysregulated levels seen in ASD and COVID-19 (Tierney et al., 2021; Gaspar et al. 2024; Fan et al. 2020; Wei et al. 2020) | May help identify metabolic subtypes of ASD and flag individuals at higher risk for infection-related inflammation. Could guide dietary or pharmacologic interventions. |
| Glucose | Altered regulation linked to both ASD and COVID-19 (Manco et al., 2021; Zhang et al., 2019; Michaels et al., 2024; Shestakova et al., 2022) | Supports monitoring for insulin resistance, metabolic stress, or risk of complications. May inform lifestyle or pharmacologic interventions targeting glucose homeostasis. |
| Neutrophil-to-Lymphocyte Ratio (NLR) | Increased in ASD and in severe COVID-19 (Kulaksizoglu et al., 2019; Siniscalo et al., 2018; Toori et al., 2021; La Torre et al., 2022; Ergenç et al., 2021) | Low-cost marker of systemic inflammation; could aid risk stratification for severe infection or neuroinflammatory complications. Useful for monitoring treatment response. |
| Ferritin | Dysregulated in ASD and COVID-19; elevated post-COVID may reflect chronic immune activation (Giacomo et al., 2022; Samy et al., 2024; Kaushal et al., 2022; Cheng et al., 2020; Daru et al., 2017) | Marker of hyperinflammation or immune activation, especially in COVID-19; may guide monitoring or interventions in select cases. In ASD, utility is less established and may require stratification by iron status. |
| IL-6 / IL-17A | Elevated in neuroinflammation and systemic immune dysregulation (Akintunde et al., 2015; Wong & Hoeffer, 2018; Aksakal et al., 2024; Zou et al., 2021) | Potential targets for immunomodulatory therapy. Biomarker-guided interventions could be tailored to ASD individuals with elevated cytokines, reducing immune-mediated morbidity. Linked to microglial activation, oxidative stress, and acute-phase responses. Existing FDA-approved inhibitors make these actionable for precision medicine. |
References
- Aguilar-Ballester, M. Herrero-Cervera, A. Vinué, Á. Martínez-Hervás, S. & González-Navarro, H. (2020). Impact of cholesterol metabolism in immune cell function and atherosclerosis. Nutrients, 12(7), 2021. [CrossRef]
- Akintunde, M. E. Rose, M. Krakowiak, P. Heuer, L. Ashwood, P. Hansen, R. Hertz-Picciotto, I. & Van De Water, J. (2015). Increased production of IL-17 in children with autism spectrum disorders and co-morbid asthma. Journal of Neuroimmunology, 286, 33–41. [CrossRef]
- Aksakal, S. & Gorgun, S. (2024). The protective role of IL-17 and IL-22 in COVID-19 infection. PubMed, 21(3). [CrossRef]
- Al-Beltagi, M. Saeed, N. K. Bediwy, A. S. Alhawamdeh, R. & Qaraghuli, S. (2022). Effects of COVID-19 on children with autism. World Journal of Virology, 11(6), 411–425. [CrossRef]
- Al-kuraishy, H. M. Hussien, N. R. Al-Niemi, M. S. Fahad, E. H. Al-Buhadily, A. K. Al-Gareeb, A. I. Al-Hamash, S. M. Tsagkaris, C. Papadakis, M. Alexiou, A. & Batiha, G. E. (2023). SARS-CoV-2 induced HDL dysfunction may affect the host’s response to and recovery from COVID-19. Immunity Inflammation and Disease, 11(5). [CrossRef]
- Alyaseen, F. F. (2024). Serum cholesterol in children with ASD. Romanian Journal of Pediatrics, 73(3), 133 – 139. [CrossRef]
- Ashwood, P. Krakowiak, P. Hertz-Picciotto, I. Hansen, R. Pessah, I. N. & Van De Water, J. (2010). Associations of impaired behaviors with elevated plasma chemokines in autism spectrum disorders. Journal of Neuroimmunology, 232(1–2), 196–199. [CrossRef]
- Benachenhou, S. Etcheverry, A. Galarneau, L. Dubé, J. & Çaku, A. (2019). Implication of hypocholesterolemia in autism spectrum disorder and its associated comorbidities: A retrospective case–control study. Autism Research, 12(12), 1860–1869. [CrossRef]
- Buonacera, A. Stancanelli, B. Colaci, M. & Malatino, L. (2022). Neutrophil to Lymphocyte Ratio: An Emerging Marker of the Relationships between the Immune System and Diseases. International Journal of Molecular Sciences, 23(7), 3636. [CrossRef]
- Carter, M., Casey, S., O'Keeffe, G. W., Gibson, L., Gallagher, L., & Murray, D. M. (2022). Maternal Immune Activation and Interleukin 17A in the Pathogenesis of Autistic Spectrum Disorder and Why It Matters in the COVID-19 Era. Frontiers in psychiatry, 13, 823096. [CrossRef]
- Chaubey, G. K. Dilawari, R. Modanwal, R. Talukdar, S. Dhiman, A. Raje, C. I. & Raje, M. (2023). Excess iron aggravates the severity of COVID-19 infection. Free radical biology & medicine, 208, 186–193. [CrossRef]
- Chen, M. H. Su, T. P. Chen, Y. S. Hsu, J. W. Huang, K. L. Chang, W. H. Chen, T. J. & Bai, Y. M. (2013). Association between psychiatric disorders and iron deficiency anemia among children and adolescents: a nationwide population-based study. BMC psychiatry, 13, 161. [CrossRef]
- Cheng, J. Zhang, R. Xu, Z. Ke, Y. Sun, R. Yang, H. Zhang, X. Zhen, X. & Zheng, L. (2021). Early glycolytic reprogramming controls microglial inflammatory activation. Journal of Neuroinflammation, 18(1). [CrossRef]
- Cheng, L. Li, H. Li, L. Liu, C. Yan, S. Chen, H. & Li, Y. (2020). Ferritin in the coronavirus disease 2019 (COVID-19): A systematic review and meta-analysis. Journal of Clinical Laboratory Analysis, 34(10). [CrossRef]
- Cheng, R. Dhorajia, V. V. Kim, J. & Kim, Y. (2021). Mitochondrial iron metabolism and neurodegenerative diseases. NeuroToxicology, 88, 88–101. [CrossRef]
- Choi, G. B. Yim, Y. S. Wong, H. Kim, S. Kim, H. Kim, S. V. Hoeffer, C. A. Littman, D. R. & Huh, J. R. (2016). The maternal interleukin-17a pathway in mice promotes autism-like phenotypes in offspring. Science, 351(6276), 933–939. [CrossRef]
- Codo, A. C. Davanzo, G. G. De Brito Monteiro, L. De Souza, G. F. Muraro, S. P. Virgilio-Da-Silva, J. V. Prodonoff, J. S. Carregari, V. C. De Biagi, C. a. O. Junior, Crunfli, F. Restrepo, J. L. J. Vendramini, P. H. Reis-De-Oliveira, G. Santos, K. B. D. Toledo-Teixeira, D. A. Parise, P. L. Martini, M. C. Marques, R. E. Carmo, H. R. . . . Moraes-Vieira, P. M. (2020). Elevated Glucose Levels Favor SARS-CoV-2 Infection and Monocyte Response through a HIF-1α/Glycolysis-Dependent Axis. Cell Metabolism, 32(3), 437-446.e5. [CrossRef]
- Daru, J. Colman, K. Stanworth, S. J. De La Salle, B. Wood, E. M. & Pasricha, S. (2017). Serum ferritin as an indicator of iron status: what do we need to know? American Journal of Clinical Nutrition, 106, 1634S-1639S. [CrossRef]
- Davinelli, S. Medoro, A. Siracusano, M. Savino, R. Saso, L. Scapagnini, G. & Mazzone, L. (2025). Oxidative stress response and NRF2 signaling pathway in autism spectrum disorder. Redox biology, 83, 103661. [CrossRef]
- de Farias, E. C. F., do Nascimento, L. M. P. P., Pavão Junior, M. J. C., Pavão, D. C. A., Pinheiro, A. P. S., Pinheiro, A. H. O., Alves, M. C. B., Ferraro, K. M. M. M., Aires, L. F. Q., Dias, L. G., Machado, M. M. M., Serrão, M. J. D., Gomes, R. R., de Moraes, S. M. P., Pontes, G. C. L., Carvalho, R. D. F. P., Silva, C. T. C., Neves, C. M. A. D., Dos Santos, J. C. L., de Sousa, A. M. B., … Terreri, M. T. (2025). Plasma IL-17A is increased in patients with critical MIS-C and associated to in-hospital mortality. Frontiers in immunology, 15, 1485009. [CrossRef]
- De Giacomo, A. Medicamento, S. Pedaci, C. Giambersio, D. Giannico, O. V. Petruzzelli, M. G. Simone, M. Corsalini, M. Marzulli, L. & Matera, E. (2022). Peripheral Iron Levels in Autism Spectrum Disorders vs. Other Neurodevelopmental Disorders: Preliminary Data. International Journal of Environmental Research and Public Health, 19(7), 4006. [CrossRef]
- DelRosso, L. M. Reuter-Yuill, L. M. Cho, Y. Ferri, R. Mogavero, M. P. & Picchietti, D. L. (2022). Clinical efficacy and safety of intravenous ferric carboxymaltose treatment for restless legs symptoms and low serum ferritin in children with autism spectrum disorder. Sleep Medicine, 100, 488–493. [CrossRef]
- Ding, X. Zhang, J. Liu, L. Yuan, X. Zang, X. Lu, F. He, P. Wang, Q. Zhang, X. Xu, Y. Li, X. Liu, Y. Li, Q. Tan, X. Zheng, Y. Lin, X. & Liu, Y. (2020). High-density lipoprotein cholesterol as a factor affecting virus clearance in covid-19 patients. Respiratory Medicine, 175, 106218. [CrossRef]
- Durkin, M. S., Maenner, M. J., Meaney, F. J., Levy, S. E., DiGuiseppi, C., Nicholas, J. S., Kirby, R. S., Pinto-Martin, J. A., & Schieve, L. A. (2010). Socioeconomic inequality in the prevalence of autism spectrum disorder: evidence from a U.S. cross-sectional study. PloS one, 5(7), e11551. [CrossRef]
- Eloseily, E. M. A. Minoia, F. Crayne, C. B. Beukelman, T. Ravelli, A. & Cron, R. Q. (2019). Ferritin to Erythrocyte Sedimentation Rate Ratio: Simple Measure to Identify Macrophage Activation Syndrome in Systemic Juvenile Idiopathic Arthritis. ACR open rheumatology, 1(6), 345–349. [CrossRef]
- Ergenç, H. Ergenç, Z. Dog˘an, M. Usanmaz, M. & Gozdas, H. T. (2021). C-reactive protein and neutrophil–lymphocyte ratio as predictors of mortality in coronavirus disease 2019. Revista da Associação Médica Brasileira, 67(10), 1498–1502. [CrossRef]
- Eshraghi, A. A. Li, C. Alessandri, M. Messinger, D. S. Eshraghi, R. S. Mittal, R. & Armstrong, F. D. (2020b). COVID-19: overcoming the challenges faced by individuals with autism and their families. The Lancet Psychiatry, 7(6), 481–483. [CrossRef]
- Esposito, M., Mirizzi, P., Fadda, R., Pirollo, C., Ricciardi, O., Mazza, M., & Valenti, M. (2023). Food Selectivity in Children with Autism: Guidelines for Assessment and Clinical Interventions. International journal of environmental research and public health, 20(6), 5092. [CrossRef]
- Fan, J. Wang, H. Ye, G. Cao, X. Xu, X. Tan, W. & Zhang, Y. (2020). Letter to the Editor: Low-density lipoprotein is a potential predictor of poor prognosis in patients with coronavirus disease 2019. Metabolism, 107, 154243. [CrossRef]
- Fekete, R., Simats, A., Bíró, E., Pósfai, B., Cserép, C., Schwarcz, A. D., Szabadits, E., Környei, Z., Tóth, K., Fichó, E., Szalma, J., Vida, S., Kellermayer, A., Dávid, C., Acsády, L., Kontra, L., Silvestre-Roig, C., Moldvay, J., Fillinger, J., Csikász-Nagy, A., … Dénes, Á. (2025). Microglia dysfunction, neurovascular inflammation and focal neuropathologies are linked to IL-1- and IL-6-related systemic inflammation in COVID-19. Nature neuroscience, 28(3), 558–576. [CrossRef]
- Gaspar, J. M. Ferreira, J. P. Carvalho, H. M. & Toscano, C. V. A. (2024). Metabolic and Inflammatory Profiles in Children and Adolescents with Autism Spectrum Disorder: A Cross-Sectional Study. Brain Sciences, 14(11), 1052. [CrossRef]
- Goicoechea, L. Conde, L. Torres, S. García-Ruiz, C. & Fernández-Checa, J. C. (2023). Mitochondrial cholesterol: Metabolism and impact on redox biology and disease. Redox Biology, 61, 102643–102643. [CrossRef]
- Goines, P. E. & Ashwood, P. (2012). Cytokine dysregulation in autism spectrum disorders (ASD): Possible role of the environment. Neurotoxicology and Teratology, 36, 67–81. [CrossRef]
- Guo, Z. Zhang, Z. Prajapati, M. & Li, Y. (2021). Lymphopenia caused by virus infections and the mechanisms beyond. Viruses, 13(9), 1876. [CrossRef]
- Hesapcioglu, S. T. Kasak, M. Kurt, A. N. C. & Ceylan, M. F. (2017). High monocyte level and low lymphocyte to monocyte ratio in autism spectrum disorders. International Journal of Developmental Disabilities, 65(2), 73–81. [CrossRef]
- Hoirisch-Clapauch, S. & Nardi, A. E. (2019). Autism spectrum disorders: let’s talk about glucose? Translational Psychiatry, 9(1). [CrossRef]
- Howard, R., Scheiner, A., Kanetsky, P. A., & Egan, K. M. (2019). Sociodemographic and lifestyle factors associated with the neutrophil-to-lymphocyte ratio. Annals of epidemiology, 38, 11–21.e6. [CrossRef]
- Hu, X. Chen, D. Wu, L. He, G. & Ye, W. (2020). Low Serum Cholesterol Level Among Patients with COVID-19 Infection in Wenzhou, China. SSRN Electronic Journal. [CrossRef]
- Huang, C. Wang, Y. Li, X. Ren, L. Zhao, J. Hu, Y. Zhang, L. Fan, G. Xu, J. Gu, X. Cheng, Z. Yu, T. Xia, J. Wei, Y. Wu, W. Xie, X. Yin, W. Li, H. Liu, M. . . . Cao, B. (2020). Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. The Lancet, 395(10223), 497–506. [CrossRef]
- Huang, Z. Chavda, V. P. Vora, L. K. Gajjar, N. Apostolopoulos, V. Shah, N. & Chen, Z.-S. (2022). 2-Deoxy-D-Glucose and its Derivatives for the COVID-19 Treatment: An Update. Frontiers in Pharmacology, 13. [CrossRef]
- Kalinowska, M. Castillo, C. & Francesconi, A. (2015). Quantitative Profiling of Brain Lipid Raft Proteome in a Mouse Model of Fragile X Syndrome. PLOS ONE, 10(4), e0121464. [CrossRef]
- Kappert, K. Jahić, A. & Tauber, R. (2020). Assessment of serum ferritin as a biomarker in COVID-19: bystander or participant? Insights by comparison with other infectious and non-infectious diseases. Biomarkers, 25(8), 616–625. [CrossRef]
- Kartasheva-Ebertz, D. M., Pol, S., & Lagaye, S. (2021). Retinoic Acid: A New Old Friend of IL-17A in the Immune Pathogeny of Liver Fibrosis. Frontiers in immunology, 12, 691073. [CrossRef]
- Kaushal, K. Kaur, H. Sarma, P. Bhattacharyya, A. Sharma, D. J. Prajapat, M. Pathak, M. Kothari, A. Kumar, S. Rana, S. Kaur, M. Prakash, A. Mirza, A. A. Panda, P. K. Vivekanandan, S. Omar, B. J. Medhi, B. & Naithani, M. (2021). Serum ferritin as a predictive biomarker in COVID-19. A systematic review, meta-analysis and meta-regression analysis. Journal of Critical Care, 67, 172–181. [CrossRef]
- Kernan, K. F. & Carcillo, J. A. (2017). Hyperferritinemia and inflammation. International immunology, 29(9), 401–409. [CrossRef]
- Khaliulin, I., Hamoudi, W., & Amal, H. (2025). The multifaceted role of mitochondria in autism spectrum disorder. Molecular psychiatry, 30(2), 629–650. [CrossRef]
- Kim, E. Neggers, Y. H. Shin, C. Kim, E. & Kim, E. M. (2010). Alterations in lipid profile of autistic boys: a case control study. Nutrition Research, 30(4), 255–260. [CrossRef]
- Kluck, G. E. G. Yoo, J.-A. Sakarya, E. H. & Trigatti, B. L. (2021). Good Cholesterol Gone Bad? HDL and COVID-19. International Journal of Molecular Sciences, 22(19), 10182. [CrossRef]
- Kotla, N. K. Dutta, P. Parimi, S. & Das, N. K. (2022). The role of Ferritin in Health and Disease: Recent advances and understandings. Metabolites, 12(7), 609. [CrossRef]
- Kutlu, Ayşe & Bınıcı, N.. (2018). Does increased neutrophil-lymphocyte ratio predict autism spectrum disorder?. Anatolian Journal of Psychiatry. 19. 607-614. [CrossRef]
- Kulaksizoglu S. Koparan C. (2019). High neutrophil to lymphocyte ratio and low mean platelet volume level in autism spectrum disorders. Annals of Medical Research. 26(10), 2382-5. https://annalsmedres.org/index.php/aomr/article/view/1774.
- La Torre, G. Marte, M. Massetti, A. P. Carli, S. M. Romano, F. Mastroianni, C. M. Minorenti, M. Alessandri, F. Ajassa, C. Fusconi, M. De Vincentiis, M. De Meo, D. Villani, C. Cardi, M. Pugliese, F. & Author_Id, N. (2022). The neutrophil/lymphocyte ratio as a prognostic factor in COVID-19 patients: a case-control study. DOAJ (DOAJ: Directory of Open Access Journals), 26(3), 1056–1064. [CrossRef]
- Lee, E., Ozigbo, A. A., Varon, J., Halma, M., Laezzo, M., Ang, S. P., & Iglesias, J. (2025). Mitochondrial Reactive Oxygen Species: A Unifying Mechanism in Long COVID and Spike Protein-Associated Injury: A Narrative Review. Biomolecules, 15(9), 1339. [CrossRef]
- Lingampelly, S. S. Naviaux, J. C. Heuer, L. S. Monk, J. M. Li, K. Wang, L. Haapanen, L. Kelland, C. A. Van De Water, J. & Naviaux, R. K. (2024). Metabolic network analysis of pre-ASD newborns and 5-year-old children with autism spectrum disorder. Communications Biology, 7(1). [CrossRef]
- Lingwood, D. & Simons, K. (2009). Lipid Rafts as a Membrane-Organizing Principle. Science, 327(5961), 46–50. [CrossRef]
- Liu, M. Chen, Y. Sun, M. Du, Y. Bai, Y. Lei, G. Zhang, C. Zhang, M. Zhang, Y. Xi, C. Ma, Y. & Wang, G. (2023). Auts2 regulated autism-like behavior, glucose metabolism and oxidative stress in mice. Experimental Neurology, 361, 114298. [CrossRef]
- Liu, S. Zhang, Q. Wang, W. Zhang, M. Liu, C. Xiao, X. Liu, Z. Hu, W. & Jin, P. (2020). Hyperglycemia is a strong predictor of poor prognosis in COVID-19. Diabetes Research and Clinical Practice, 167, 108338. [CrossRef]
- Liu, X. Lin, J. Zhang, H. Khan, N. U. Zhang, J. Tang, X. Cao, X. & Shen, L. (2022). Oxidative Stress in Autism Spectrum Disorder-Current Progress of Mechanisms and Biomarkers. Frontiers in psychiatry, 13, 813304. [CrossRef]
- Logette, E. Lorin, C. Favreau, C. Oshurko, E. Coggan, J. S. Casalegno, F. Sy, M. F. Monney, C. Bertschy, M. Delattre, E. Fonta, P. Krepl, J. Schmidt, S. Keller, D. Kerrien, S. Scantamburlo, E. Kaufmann, A. & Markram, H. (2021). A Machine-Generated View of the role of blood glucose levels in the severity of COVID-19. Frontiers in Public Health, 9. [CrossRef]
- Lou, Q. (2024). Neutrophil-to-Lymphocyte ratio: a promising predictor of mortality in patients with acute respiratory distress syndrome. A retrospective analysis of a single hospital center. Journal of Cardiothoracic and Vascular Anesthesia, 38(8), 1716–1726. [CrossRef]
- Malech, H. L. DeLeo, F. R. & Quinn, M. T. (2014). The role of neutrophils in the immune system: an overview. Methods in Molecular Biology, 3–10. [CrossRef]
- Manco, M. Guerrera, S. Ravà, L. Ciofi degli Atti, M. Di Vara, S. Valeri, G. & Vicari, S. (2021, April 20). Cross-sectional investigation of insulin resistance in youths with autism spectrum disorder. Any role for reduced brain glucose metabolism?. Nature News. https://www.nature.com/articles/s41398-021-01345-3.
- Masana, L. Correig, E. Ibarretxe, D. Anoro, E. Arroyo, J. A. Jericó, C. Guerrero, C. Miret, M. Näf, S. Pardo, A. Perea, V. Pérez-Bernalte, R. Plana, N. Ramírez-Montesinos, R. Royuela, M. Soler, C. Urquizu-Padilla, M. Zamora, A. & Pedro-Botet, J. (2021). Low HDL and high triglycerides predict COVID-19 severity. Scientific Reports, 11(1), 7217. [CrossRef]
- McCarthy, E. K. Murray, D. M. & Kiely, M. E. (2021). Iron deficiency during the first 1000 days of life: are we doing enough to protect the developing brain? Proceedings of the Nutrition Society, 81(1), 108–118. [CrossRef]
- Micai, M., Fatta, L. M., Gila, L., Caruso, A., Salvitti, T., Fulceri, F., Ciaramella, A., D'Amico, R., Del Giovane, C., Bertelli, M., Romano, G., Schünemann, H. J., & Scattoni, M. L. (2023). Prevalence of co-occurring conditions in children and adults with autism spectrum disorder: A systematic review and meta-analysis. Neuroscience and biobehavioral reviews, 155, 105436. [CrossRef]
- Michaels, T. M. M. Faadiel Essop, & Joseph, D. E. (2024). Potential Effects of Hyperglycemia on SARS-CoV-2 Entry Mechanisms in Pancreatic Beta Cells. Viruses, 16(8), 1243–1243. [CrossRef]
- Nadeem, A., Ahmad, S. F., Attia, S. M., Bakheet, S. A., Al-Harbi, N. O., & Al-Ayadhi, L. Y. (2018a). Activation of IL-17 receptor leads to increased oxidative inflammation in peripheral monocytes of autistic children. Brain, behavior, and immunity, 67, 335–344. [CrossRef]
- Nadeem, A., Al-Harbi, N. O., Alfardan, A. S., Ahmad, S. F., AlAsmari, A. F., & Al-Harbi, M. M. (2018b). IL-17A-induced neutrophilic airway inflammation is mediated by oxidant-antioxidant imbalance and inflammatory cytokines in mice. Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie, 107, 1196–1204. [CrossRef]
- Orth, M. & Bellosta, S. (2012). Cholesterol: Its regulation and role in central nervous system disorders. Cholesterol, 2012, 1–19. [CrossRef]
- Palacios-Rápalo, S. N. De Jesús-González, L. A. Cordero-Rivera, C. D. Farfan-Morales, C. N. Osuna-Ramos, J. F. Martínez-Mier, G. Quistián-Galván, J. Muñoz-Pérez, A. Bernal-Dolores, V. Del Ángel, R. M. & Reyes-Ruiz, J. M. (2021). Cholesterol-Rich lipid rafts as platforms for SARS-COV-2 entry. Frontiers in Immunology, 12. [CrossRef]
- Pandrangi, S. L. Chittineedi, P. Chalumuri, S. S. Meena, A. S. Mosquera, J. a. N. Llaguno, S. N. S. Pamuru, R. R. Mohiddin, G. J. & Mohammad, A. (2022). Role of intracellular iron in switching apoptosis to ferroptosis to target Therapy-Resistant cancer stem cells. Molecules, 27(9), 3011. [CrossRef]
- Paranga, T. G. Mitu, I. Pavel-Tanasa, M. Rosu, M. F. Miftode, I. Constantinescu, D. Obreja, M. Plesca, C. E. & Miftode, E. (2024). Cytokine Storm in COVID-19: Exploring IL-6 signaling and Cytokine-Microbiome interactions as Emerging therapeutic Approaches. International Journal of Molecular Sciences, 25(21), 11411. [CrossRef]
- Rosário, C. Zandman-Goddard, G. Meyron-Holtz, E. G. D’Cruz, D. P. & Shoenfeld, Y. (2013). The Hyperferritinemic Syndrome: macrophage activation syndrome, Still’s disease, septic shock and catastrophic antiphospholipid syndrome. BMC Medicine, 11(1). [CrossRef]
- Sasaki, T., Tome, S. & Takei, Y. Intraventricular IL-17A administration activates microglia and alters their localization in the mouse embryo cerebral cortex. Mol Brain 13, 93 (2020). [CrossRef]
- Scahill, L., Jeon, S., Boorin, S. J., McDougle, C. J., Aman, M. G., Dziura, J., McCracken, J. T., Caprio, S., Arnold, L. E., Nicol, G., Deng, Y., Challa, S. A., & Vitiello, B. (2016). Weight Gain and Metabolic Consequences of Risperidone in Young Children With Autism Spectrum Disorder. Journal of the American Academy of Child and Adolescent Psychiatry, 55(5), 415–423. [CrossRef]
- Shibabaw T. (2020). Inflammatory Cytokine: IL-17A Signaling Pathway in Patients Present with COVID-19 and Current Treatment Strategy. Journal of inflammation research, 13, 673–680. [CrossRef]
- Qin, Q. Fan, L. Zeng, X. Zheng, D. Wang, H. Li, M. Jiang, Y. Wang, H. Liu, H. Liang, S. Wu, L. & Liang, S. (2025). Mesenchymal stem cell-derived extracellular vesicles alleviate autism by regulating microglial glucose metabolism reprogramming and neuroinflammation through PD-1/PD-L1 interaction. Journal of Nanobiotechnology, 23(1). [CrossRef]
- Reynolds, A. Krebs, N. F. Stewart, P. A. Austin, H. Johnson, S. L. Withrow, N. et al., (2012). Iron Status in Children With Autism Spectrum Disorder. Pediatrics, 130(Supplement 2), S154–S159. [CrossRef]
- Rodríguez-Rodríguez, E., Salas-González, M. D., Ortega, R. M., & López-Sobaler, A. M. (2022). Leukocytes and Neutrophil-Lymphocyte Ratio as Indicators of Insulin Resistance in Overweight/Obese School-Children. Frontiers in nutrition, 8, 811081. [CrossRef]
- Rosário, C. Zandman-Goddard, G. Meyron-Holtz, E. G. D'Cruz, D. P. & Shoenfeld, Y. (2013). The hyperferritinemic syndrome: macrophage activation syndrome, Still's disease, septic shock and catastrophic antiphospholipid syndrome. BMC medicine, 11, 185. [CrossRef]
- Sammels, O. Karjalainen, L. Dahlgren, J. & Wentz, E. (2022). Autism spectrum disorder and obesity in children: A systematic review and meta-analysis. Obesity Facts, 15(3). [CrossRef]
- Samy, A. ElRouby, I. Eldin, E. S. & El-Ghaffar, E. S. A. (2024). Evaluation of iron, zinc and vitamin D deficiencies in children with autism spectrum disorder at the Hearing and Speech Institute, in Egypt. Journal of Medicine in Scientific Research. https://jmisr.researchcommons.org/home/vol8/iss2/11/.
- Segatto, M. Tonini, C. Pfrieger, F. W. Trezza, V. & Pallottini, V. (2019). Loss of Mevalonate/Cholesterol Homeostasis in the Brain: A Focus on Autism Spectrum Disorder and Rett Syndrome. International journal of molecular sciences, 20(13), 3317. [CrossRef]
- Shestakova, M. Kononenko, I. Zilya Kalmykovа, Markova, T. Kaplun, E. Lysenko, M. & Mokrysheva, N. (2022). Glycated hemoglobin level dynamics in COVID-19 survivors: 12 months follow-up study after discharge from hospital. PloS One, 17(11), e0275381–e0275381. [CrossRef]
- Sidrak, S. Yoong, T. & Woolfenden, S. (2013). Iron deficiency in children with global developmental delay and autism spectrum disorder. Journal of Paediatrics and Child Health, 50(5), 356–361. [CrossRef]
- Simons, K. & Sampaio, J. L. (2011). Membrane Organization and Lipid Rafts. Cold Spring Harbor Perspectives in Biology, 3(10), a004697–a004697. [CrossRef]
- Siniscalco, D. Schultz, S. Brigida, A. L. & Antonucci, N. (2018). Inflammation and Neuro-Immune dysregulations in autism spectrum disorders. Pharmaceuticals, 11(2), 56. [CrossRef]
- Sorice, M. Misasi, R. Riitano, G. Manganelli, V. Martellucci, S. Longo, A. Garofalo, T. & Mattei, V. (2021). Targeting lipid rafts as a strategy against coronavirus. Frontiers in Cell and Developmental Biology, 8. [CrossRef]
- Soto, M. E. Guarner-Lans, V. Díaz-Díaz, E. Manzano-Pech, L. Palacios-Chavarría, A. Valdez-Vázquez, R. R. Aisa-Álvarez, A. Saucedo-Orozco, H. & Pérez-Torres, I. (2022). Hyperglycemia and loss of redox homeostasis in COVID-19 patients. Cells, 11(6), 932. [CrossRef]
- Sural-Fehr, T. Singh, H. Cantuti-Catelvetri, L. Zhu, H. Marshall, M. S. Rebiai, R. Jastrzebski, M. J. Givogri, M. I. Rasenick, M. M. & Bongarzone, E. R. (2019). Inhibition of IGF-1-PI3K-Akt-mTORC2 in lipid rafts increases neuronal vulnerability in a genetic lysosomal glycosphingolipidosis. Disease Models & Mechanisms. [CrossRef]
- Tanaka, S. Christian de Tymowski, Assadi, M. Zappella, N. Sylvain Jean-Baptiste, Robert, T. Katell Peoc'h, Lortat-Jacob, B. Fontaine, L. Donia Bouzid, Alexy Tran-Dinh, Parvine Tashk, Olivier Meilhac, & Philippe Montravers. (2020). Lipoprotein concentrations over time in the intensive care unit COVID-19 patients: Results from the ApoCOVID study. 15(9), e0239573–e0239573. [CrossRef]
- Thawley, A. J. Veneziani, L. P. Rabelo-da-Ponte, F. D. Riederer, I. Mendes-da-Cruz, D. A. & Bambini-Junior, V. (2022). Aberrant IL-17 Levels in Rodent Models of Autism Spectrum Disorder: A Systematic Review. Frontiers in Immunology, 13. [CrossRef]
- Tierney, E. Remaley, A. T. Thurm, A. Jager, L. R. Wassif, C. A. Kratz, L. E. Bailey-Wilson, J. E. Bukelis, I. Geeta Sarphare, Jung, E. S. Brand, B. Noah, K. K. & Porter, F. D. (2021). Sterol and lipid analyses identifies hypolipidemia and apolipoprotein disorders in autism associated with adaptive functioning deficits. Translational Psychiatry, 11(1). [CrossRef]
- Toma C. (2020). Genetic Variation across Phenotypic Severity of Autism. Trends in genetics : TIG, 36(4), 228–231. [CrossRef]
- Toori, K. U. Qureshi, M. A. Chaudhry, A. & Safdar, M. F. (2021). Neutrophil to lymphocyte ratio (NLR) in COVID-19: A cheap prognostic marker in a resource constraint setting. Pakistan Journal of Medical Sciences, 37(5). [CrossRef]
- Tural Hesapcioglu, S. Kasak, M. Cıtak Kurt, A. N. & Ceylan, M. F. (2017). High monocyte level and low lymphocyte to monocyte ratio in autism spectrum disorders. International journal of developmental disabilities, 65(2), 73–81. [CrossRef]
- Viljetić, B. Blažetić, S. Labak, I. Ivić, V. Zjalić, M. Heffer, M. & Balog, M. (2024). Lipid rafts: the maestros of normal brain development. Biomolecules, 14(3), 362. [CrossRef]
- Voss, K. Sewell, A. E. Krystofiak, E. S. Gibson-Corley, K. N. Young, A. C. Basham, J. H. Sugiura, A. Arner, E. N. Beavers, W. N. Kunkle, D. E. Dickson, M. E. Needle, G. A. Skaar, E. P. Rathmell, W. K. Ormseth, M. J. Major, A. S. & Rathmell, J. C. (2023). Elevated transferrin receptor impairs T cell metabolism and function in systemic lupus erythematosus. Science immunology, 8(79), eabq0178. [CrossRef]
- Wang, H. (2014). Lipid rafts: a signaling platform linking cholesterol metabolism to synaptic deficits in autism spectrum disorders. Frontiers in Behavioral Neuroscience, 8. [CrossRef]
- Wang, H. Yuan, Z. Pavel, M. A. Jablonski, S. M. Jablonski, J. Hobson, R. Valente, S. Reddy, C. B. & Hansen, S. B. (2023). The role of high cholesterol in SARS-CoV-2 infectivity. Journal of Biological Chemistry, 104763. [CrossRef]
- Wei, X. Zeng, W. Su, J. Wan, H. Yu, X. Cao, X. Tan, W. & Wang, H. (2020). Hypolipidemia is associated with the severity of COVID-19. Journal of Clinical Lipidology, 14(3), 297–304. [CrossRef]
- Wong, H. & Hoeffer, C. (2018). Maternal IL-17A in autism. Experimental Neurology, 299, 228–240. [CrossRef]
- Yap, C. X. Henders, A. K. Alvares, G. A. Giles, C. Huynh, K. Nguyen, A. Wallace, L. McLaren, T. Yang, Y. Hernandez, L. M. Gandal, M. J. Hansell, N. K. Cleary, D. Grove, R. Hafekost, C. Harun, A. Holdsworth, H. Jellett, R. Khan, F. . . . Gratten, J. (2023). Interactions between the lipidome and genetic and environmental factors in autism. Nature Medicine, 29(4), 936–949. [CrossRef]
- Youssef, J. Singh, K. Huntington, N. Becker, R. & Kothare, S. V. (2013). Relationship of serum ferritin levels to sleep fragmentation and periodic limb movements of sleep on polysomnography in autism spectrum disorders. Pediatric Neurology, 49(4), 274–278. [CrossRef]
- Zhang, M. Ding, R. Wang, Z. Zhang, J. Liu, J. & Wang, J. (2019). Altered Peak C-peptide and Fasting Blood Glucose in Children with Autism Spectrum Disorder. Journal of Diabetes and Clinical Research, 1(2). [CrossRef]
- Zheng, M. Y., & Luo, L. Z. (2025). The Role of IL-17A in Mediating Inflammatory Responses and Progression of Neurodegenerative Diseases. International journal of molecular sciences, 26(6), 2505. [CrossRef]
- Zhou, P. Yang, X. Wang, X. Hu, B. Zhang, L. Zhang, W. Si, H. Zhu, Y. Li, B. Huang, C. Chen, H. Chen, J. Luo, Y. Guo, H. Jiang, R. Liu, M. Chen, Y. Shen, X. Wang, X. . . . Shi, Z. (2020). Addendum: A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature, 588(7836), E6. [CrossRef]
- Zou, Y. & Meng, Z. (2021). Literature Overview of the IL-17 Inhibition from Psoriasis to COVID-19. Journal of Inflammation Research, 14, 5611–5618. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




