Introduction
Device-aided therapies (DATs) in Parkinson’s disease (PD) are advanced treatment modalities used when oral or transdermal medications no longer provide adequate control of motor fluctuations or dyskinesias [
1]. For many years, 3 DATs have been used in many countries: deep brain stimulation (DBS), levodopa-carbidopa intestinal gel (LCIG) infusion, and continuous subcutaneous apomorphine infusion (CSAI) [
2]. However, new formulations such as foslevodopa-foscarbidopa (fLD/fCD), a prodrug-based subcutaneous infusion, and levodopa-carbidopa-entacapone intestinal gel (LECIG) have emerged with the aim to provide continuous dopaminergic stimulation and reduce device-related complications [
3]. In particular, fLD/fCD offers a non-surgical, continuous, and adjustable alternative to existing DATs such as levodopa-carbidopa intestinal gel and DBS for patients with advanced PD and motor fluctuations not adequately controlled by oral medication. Clinical trials have demonstrated that continuous subcutaneous fLD/fCD significantly increases “On” time without troublesome dyskinesia and reduces “Off” time compared to oral immediate-release levodopa/carbidopa, with benefits observed as early as the first post-baseline assessment and sustained over 12 weeks [
4]. Given its ease of implementation and the good profile of levodopa in terms of safety and effectiveness, fLD/fCD is a good DAT option in many patients with PD, being the most frequently used option currently in Spain [
5,
6]. However, adverse events such as infusion site reactions (erythema, pain, cellulitis, edema), which are generally mild to moderate and manageable with site rotation and aseptic technique, or others related to the drug (visual hallucinations, psychosis, etc.) may lead to discontinuation of the treatment in some cases [
4,
7]. LECIG can be an alternative option for people with PD (PwP) who failed fLD/fCD, and in fact, a prospective, international, non-interventional phase IV study (SWITCH-ON) designed to assess the effectiveness of LECIG on the reduction in “Off” time from baseline at 12 months in PwP who failed fLD/fCD, is ongoing.
To date, no data has been published on the outcome of patients with PD treated with fLD/fCD who switch to LECIG. Here, we report our experience in Spain with PwPD treated with LECIG who had previously failed subcutaneous fLD/fCD.
Material and Methods
PwP treated with LECIG in Spain until November 30, 2025, who previously failed fLD/fCD, were included in this multicenter, longitudinal, retrospective, observational study. An in-person visit (post-LECIG) was conducted in different centers of Spain from December 1, 2025, to 10, January, 2026, to assess patients already receiving LECIG and to collect data on their outcome. During this visit, the patient signed the informed consent form. Information on sociodemographic aspects, comorbidity, factors related to PD, and treatment including levodopa equivalent daily dose (LEDD) [
8], fLD/fCD failure, and LECIG indication, was retrospectively collected.
The change from pre-LECIG (Vpre) to post-LECIG (Vpost) in the mean daily OFF time, health-related quality of life (PDQ-39 [39-item Parkinson’s Disease Questionnaire] total score), motor symptoms burden, and non-motor symptoms (NMS) burden was assessed. To assess motor symptoms burden, a motor symptoms score (MSs) with a range from 0 to 38 was calculated, being the result of the sum of the score of “daily OFF time” (from 0 to 4 according to the UPDRS [Unified Parkinson’s Disease Rated Scale] part IV - item 42) + “daily dyskinesia time” (from 0 to 4 according to the UPDRS part IV - item 35) + other motor symptoms (dyskinesia severity; painful dyskinesia; morning dystonia; freezing of gait during the “Off” state; freezing of gait during the “On” state; falls; posture; tremor; hypomimia; speech problems) that were scored as 0 (none), 1 (slight), 2 (moderate) or 3 (severe). Regarding NMS burden, the same methodology was applied with a NMS score (NMSs) from 0 to 54 that was the result of the sum of the score of different NMS, from 0 (none) to 3 (severe) (non-motor fluctuations; cognitive impairment; visual hallucinations; psychosis; impulse control disorder; depression; anxiety; apathy; sleep behavior disorder; diurnal somnolence; urinary symptoms; gastrointestinal symptoms; symptomatic orthostatic hypotension; constipation; sialorrhea; dysphagia; fatigue; pain). Data about the impression of change with fLD/fCD and with LECIG (post-LECIG) was collected according to the Clinical Global Impression of Change (CGI-C) scale. Adverse events were also collected.
Data were processed using SPSS 20.0 for Windows. Different variables were expressed as quantitative and/or qualitative variables. Distribution for variables was verified by one-sample Kolmogorov–Smirnov test. The change from pre-LECIG to post-LECIG in the mean daily OFF time, PDQ-39 total score, MSs, and NMSs was assessed using the Wilcoxon rank-sum test. The marginal homogeneity test was used to compare the CGI-C with f/LD/fCD vs. with LECIG. Values of p < 0.05 were considered significant.
Results
Data about 14 patients (57.1% males; 66.6 ± 8.6 years old) from 12 hospitals in Spain out of a total of 15 who were treated with LECIG were included. All patients received a 24-hour/day continuous subcutaneous infusion of fLD/fCD with a mean exposure time of 98.6 ± 92.3 days, with a range from 20 to 301. The frequency of use of other dopaminergic drugs associated with fLD/fCD was 28.6% for MAO-B inhibitors, 21.4% for amantadine, 14.3% for COMT inhibitors, and 7.1% for dopamine agonists. Regarding the cause of fLD/fCD failure, up to 92.9% and 57.1% of the patients experienced side effects and lack of response, respectively. Specifically, significant subcutaneous nodules with/without other skin problems were reported in up to 64.3% of patients, whereas visual hallucinations with psychotic symptoms were in 28.6% (in 2 patients it was the reason to switch to LECIG). Five patients (35.7%) received fLD/fCD for more than 3 months, with nodules and other skin problems being the major reason to switch to LECIG in 4 of them.
LECIG was a direct switch from fLD/fCD in 35.7% of the patients. At the time LECIG was indicated by the neurologist, the mean time from diagnosis of PD was 13.4 ± 8.2 years, and 100% of the patients had motor fluctuations and non-motor fluctuations with a mean daily time during the “Off” state of 5.2 ± 3.0 hours. Patients had a good response to levodopa from the “Off” to the “On” state (Hoeh&Yahr stage and UPDRS part III), with 50% of them presenting cognitive impairment and 28.6% psychosis (
Table 1). In addition to fLD/fCD, 1 patient had previously been treated with CSAI and 2 with LCIG. LECIG was initiated with hospitalization in more than half of the cases (57.1%), with the average optimization time being 14.9 ± 24.1 days (
Table 1). At LECIG initiation, the mean cartridge volume (mL) used was 56.6 ± 15.9, and LEDD was significantly lower than at LECIG indication (1664.6 ± 449.0 vs. 1992.1 ± 689.9; p=0.019) and at fLD/fCD withdrawal (1664.6 ± 449.0 vs. 2084.9 ± 726.3; p=0.030).
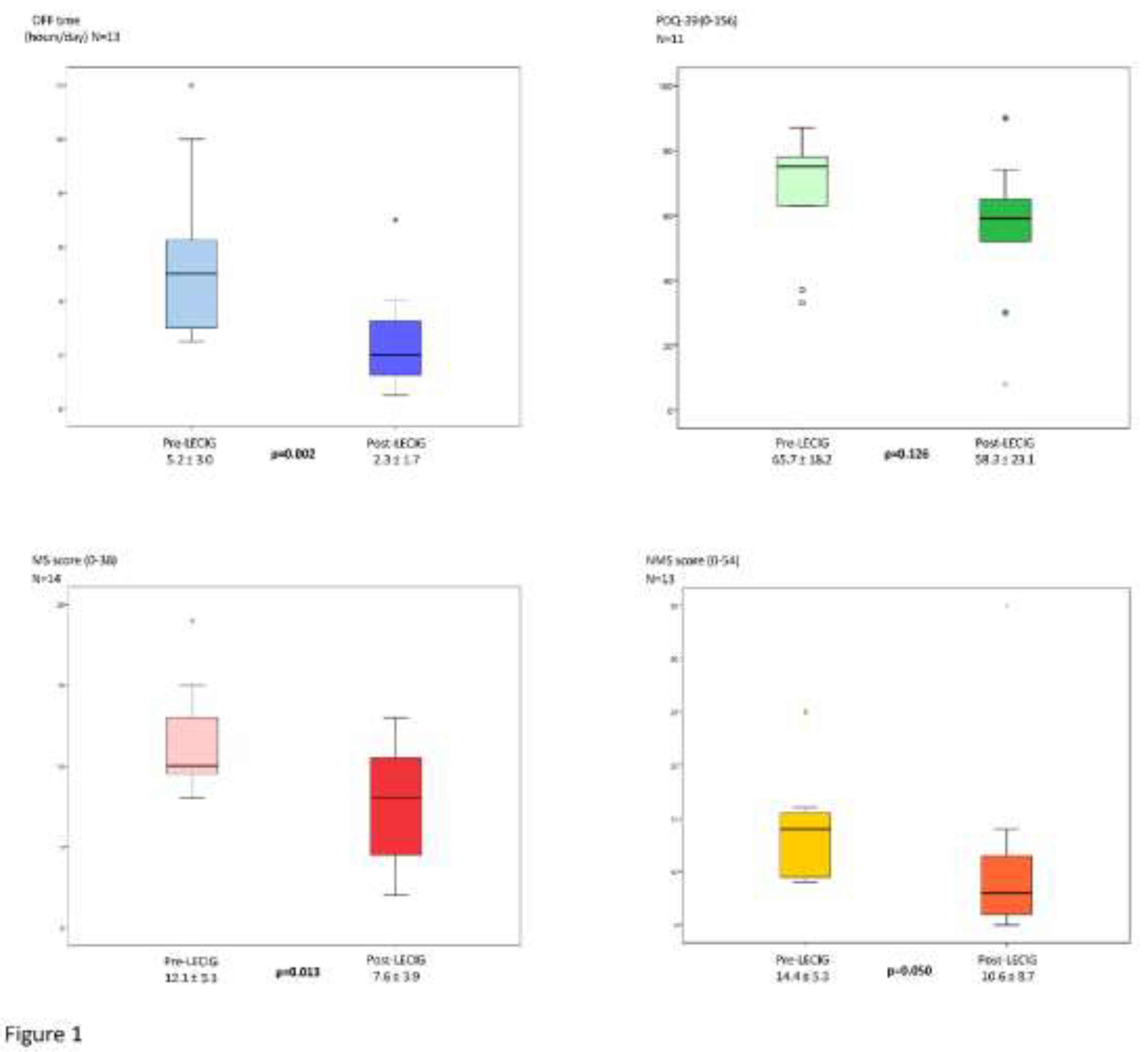
The mean exposure time to LECIG (from Vpre to Vpost) was 233.7 ± 157.4 days. Daily OFF time was significantly reduced by 2.9 ± 1.9 hours (p=0.002), from 5.2 ± 3 at Vpre to 2.3 ± 1.7 at Vpost (
Figure 1). PwP improved significantly from Vpre to Vpost in motor symptoms, with a decrease in the MSs from 12.1 ± 3.3 to 7.6 ± 3.9 points (-37.2%; p=0.013). Regarding NMS burden, a trend of significance was detected in their improvement with a decrease in the NMSs from 14.4 ± 5.3 points at Vpre to 10.6 ± 8.7 at Vpost (-35.8%; p=0.050). In terms of quality of life, the total score in the PDQ-39 from Vpre to Vpost was reduced in 7.4 points, but it was not statistically significant (
Figure 1). No significant change was detected in the mean LEDD (mg) from Vpre (1664.6 ± 449.0) to Vpost (1718.0 ± 468.6) (p=0.508). No differences were detected either (p=0.726) between the UPDRS-III in the “On” state at Vpre (19.2 ± 16.8) and at Vpost (18.3 ± 16.0). Weight remained stable between both visits (70.5 ± 15.3 at Vpre vs. 69.8 ± 16.4 at Vpost; p=0.505). The clinical perception of improvement experienced by patients based to the CGI-C was significantly better in the case of LECIG compared to fLD/fCD according to the neurologist’s (p=0.017) and the patient’s own opinion (p=0.012), but not in the case of the caregiver (p=0.072) (
Figure 1- Supplementary Material).
LECIG was well tolerated, with only 1 dropout (7.1%) due to complications related to dementia (the Vpost data was also collected in this case). Adverse events were reported in 28.6% and 35.7% of the patients in the optimization and final follow-up evaluation phases, respectively. Specifically, in the LECIG optimization phase, the following adverse events were recorded: stoma infection (N=2), stoma erythema (N=2), dyskinesia impairment (N=2), tube migration (N=1) and problems with gastrostomy (N=1). In global terms, a total of 20 adverse events were reported, with complications in the stoma (N=11) being the most frequent (
Table 2). Fifty percent of the patients suffered from at least one possibly related (LECIG and/or device) adverse event.
Discussion
The present study observed a good outcome in the medium term (near 8 months on average) of PwP treated with LECIG who had previously failed fLD/fCD. In particular, a reduction in “Off” time and improvement in motor symptoms were found, with a trend towards improvement in quality of life and NMS burden. The tolerability profile of LECIG was good, in line with previous studies [
9,
10,
11]. Despite the small sample size and pending data from the SWITCH-ON study, this is the first report on the outcome of patients treated with LECIG who previously failed fLD/fCD.
Most patients from our cohort who dropped fLD/fCD did so because of side effects, including more than 60% with nodules that in many cases were associated with a suboptimal response due to probable problems with drug absorption. The frequency of skin problems in patients treated with fLD/fCD is high, with 62% of patients experiencing infusion/catheter site reactions and 28% experiencing infusion/catheter site infections in phase 3 clinical trials [
12]. The most common skin reactions include erythema (27%), pain (26%), cellulitis (19%), edema (12%), bruising (3%), and hemorrhage (3%). These reactions are generally mild to moderate but can lead to treatment discontinuation in a subset of patients (8% for site reactions, 5% for site infections) [
4,
7,
12,
13]. The development of hallucinations or psychotic events can also be a reason for therapeutic failure in 15% of the patients [
4], as in 14.3% of our patients. The global frequency of fLD/fCD withdrawal in clinical practice is at least 20% during the initial 12 weeks of therapy [
4,
7], with adverse events—primarily skin reactions—occurring in up to 87% of patients. When fLD/fCD fails, DATs are the main alternatives for advanced disease refractory to oral and adjunctive pharmacotherapy. CSAI would not be recommended in a patient with psychosis or pre-existing skin problems [
14]. Alternatively, enteral levodopa infusion could be considered in these cases, with LECIG providing an equivalent efficacy to LCIG with a lower levodopa dose, similar safety and tolerability, and practical device advantages due to the addition of entacapone [
15]. Data from previous studies on the use of DATs in Spain have shown that LECIG is indeed a frequently used therapeutic option when other DATs have previously failed [
5,
6]. But even in these cases, many patients show a good response, with improvement in motor and NMS and maintenance of therapy [
9,
10,
11]. In line with the findings observed in the ELEGANCE and LECIPARK studies, patients from this cohort reduced “Off” time by nearly 3 hours and improved motor symptoms, with a trend towards improvement in NMS and their quality of life. In fact, the reduction of 7.4 points on the PDQ-39 total score is equivalent to 4.7 points on the PDQ-39SI, which is exactly what has been defined in the literature as the minimal clinically important difference [
16]. It is noteworthy that these are patients with an advanced stage of the disease who did not improve satisfactorily with fLD/fCD. In this regard, there is data suggesting that patients who switch from LCIG to LECIG and are not optimally controlled may also improve [
9]. The addition of entacapone increases levodopa bioavailability, allowing for a 20–35% reduction in levodopa maintenance dose while achieving similar systemic exposure and motor response as LCIG, regardless of COMT genotype [
17]. The reduction in LEDD observed with LECIG in this cohort supports this aspect. In this context, and although treatment should always be individualized, in general, for patients eligible for a DAT other than deep brain stimulation, and based on costs, invasiveness and ease of implementation, the subcutaneous route would be preferred over gastrostomy, with LECIG being a good treatment option in patients with previous failure [
5,
6,
18,
19,
20].
The present study has important limitations. First, the sample size is small. Second, the methodology is that of an open and retrospective study. Third, in the follow-up evaluation (Vpost) with LECIG, the data for the variables in which the change was measured were not present in all cases. Fourth, we used non-validated scores (MSs and NMSs) to calculate the change in motor and NMS. Fifth, underreporting of adverse events cannot be ruled out. Finally, the average follow-up time is almost 8 months, making it important to know the longer-term evolution. Despite these limitations, the findings are novel since, pending the results of the ongoing SWITCH-ON study, no data have been previously reported on PwP treated with LECIG who failed fLD/fCD.
In conclusion, based on this first Spanish experience report, LECIG could be a good therapeutic option in PwP who failed fLD/fCD. Data from prospective multicenter studies with a larger number of patients are needed to corroborate this initial observation.
Legend of figures
Figure 2. Clinical Impression Global of Change according to the opinion of the neurologist, patient, and principal caregiver with fLD/fCD (A) and with LECIG (B) (N=14). P was significant for neurologist’s (p=0.017) and the patient’s own opinion (p=0.012), but not in the case of the caregiver (p=0.072). The marginal homogeneity test was used. CGI-C, Clinical Global Impression of Change; fLD/fCD, foslevodopa/foscarbidopa; LECIG, levodopa-carbidopa-entacapone intestinal gel.
Supplementary Materials
1 figure.
Role of the authors of this manuscript
Diego Santos García: conception, organization, and execution of the project; statistical analysis; writing of the first draft of the manuscript; recruitment and/or evaluation of participants. Inés Legarda: recruitment and/or evaluation of participants, data collection, and review of the manuscript. Tamara M. González Fernández: recruitment and/or evaluation of participants, data collection, and review of the manuscript. Ana Rodríguez Sanz: recruitment and/or evaluation of participants, data collection, and review of the manuscript. Maria Isabel Morales-Casado: recruitment and/or evaluation of participants, data collection, and review of the manuscript. Alejandro Peral: recruitment and/or evaluation of participants, data collection, and review of the manuscript. Nuria Caballol: recruitment and/or evaluation of participants, data collection, and review of the manuscript. María Álvarez Sauco: recruitment and/or evaluation of participants, data collection, and review of the manuscript. Iria Campos Rodríguez: recruitment and/or evaluation of participants, data collection, and review of the manuscript. Déborah Alonso Modino: recruitment and/or evaluation of participants, data collection, and review of the manuscript. Lydia López Manzanares: recruitment and/or evaluation of participants, data collection, and review of the manuscript. Jesús Olivares Romero: recruitment and/or evaluation of participants, data collection, and review of the manuscript. Alberto Blanco Ollero: recruitment and/or evaluation of participants, data collection, and review of the manuscript.
Statement of Ethics
The project was conducted in accordance with the ICH Good Clinical Practice version 6 Revision 2 standard, the fundamental ethical principles established in the Declaration of Helsinki and the Oviedo Convention, as well as the Spanish legal requirements for biomedical research (Biomedical Research Law 14/2007). For this study, we received approval from the Comité de Ética de Investigación de medicamentos de Galicia of Spain (2025/445; 31 October 2025). Written informed consent from all participants (patients and controls) in this study was obtained.
Data Availability Statement
The protocol, statistical analysis plan and unidentified participant data will be available on request.
Acknowledgments
We would like to thank all patients and their caregivers who collaborated in this study.
Financial Disclosures/Conflicts of interest
Diego Santos-García has received honoraria for educational presentations and advice service by Abbvie, UCB Pharma, Lundbeck, KRKA, Zambon, Bial, Italfarmaco, Teva, Archímedes, Esteve, Stada, Orion Pharma, financial compensation for INIBIC / Fundación Professor Novoa Santos in relation to activity as PI in phase 2 to phase 4 clinical trials for Parkinson’s disease and other movement disorders, compensation for Fundación Degen in relation to different projects, and grants from the “Fundación Professor Novoa Santos” as a result of the “CONVOCATORIA DE AYUDAS PARA LA REALIZACIÓN DE PROYECTOS DE INVESTIGACIÓN PARA GRUPOS EMERGENTES Y ASOCIADOS DEL INIBIC (2023/2024)”. Inés Legarda has received honoraria for educational presentations and advice service by Abbvie, UCB Pharma, Zambon, Bial, and Teva. Tamara M. González Fernández has received honoraria for educational presentations from Merck, Bial and Stada. Ana Rodríguez Sanz has received honoraria for educational presentations and advice service by Stada, KRKA, Bial, Zambon and Esteve. María Isabel Morales-Casado has received honoraria for educational presentations by Abbvie, UCB Pharma, Zambon, Bial, Lundbeck, Stada and Steve. Alejandro Peral has received honoraria from Stada and Esteve and sponsorship from Bial, Zambon, Stada, Esteve, Italfármaco, Nuria Caballol has has received honoraria for advisory services and educational presentations, and support for conference attendance, from Italfármaco, Orion Pharma, Abbvie, Stada, KRKA, and Zambon. María Álvarez Sauco has received honoraria for educational presentations and advice service by Abbvie, UCB Pharma, Zambon, Bial, Teva, Stada and Orphalan. Iria Campos Rodríguez: None. Déborah Alonso Modino has received honoraria for educational presentations and advice service by Abbvie, UCB Pharma, Italfarmaco, Stada, Esteve and Medtronic. Lydia López Manzanares compensated advisory services, consulting, research grant support, or speaker honoraria: AbbVie, Acorda, Bial, Intec Pharma, Italfarmaco, Pfizer, Roche, Teva, UCB, and Zambon. Jesús Olivares Romero has received honoraria from Bial, Italfármaco, Zambon, Abbvie, Esteve and Stada for attending medical conferences. Alberto Blanco Ollero has received honoraria from Esteve, Italfármaco, and Bial.
Abbreviations
CGI–C, Clinical Global Impression of Change; CSAI, continuous subcutaneous apomorphine infusion; DATs, device-aided therapies; DBS, deep brain stimulation; fLD/fCD, foslevodopa/foscarbidopa; LCIG, levodopa-carbidopa intestinal gel infusion; LECIG, levodopa-carbidopa-entacapone intestinal gel; LEDD, levodopa equivalent daily dose; MSs, motor symptoms score; NMS; non-motor symptoms; NMSs, non-motor symptoms score; PDQ-39, 39-item Parkinson’s Disease Questionnaire; Parkinson’s disease; PwP, people with PD; UPDRS, Unified Parkinson’s Disease Rated Scale.
References
- Tanner, CM; Ostrem, JL. Parkinson’s Disease. N Engl J Med 2024, 391, 442–52. [Google Scholar] [CrossRef] [PubMed]
- Marsili, L; Bologna, M; Miyasaki, JM; Colosimo, C. Parkinson’s disease advanced therapies - A systematic review: More unanswered questions than guidance. Parkinsonism Relat Disord 2021, 83, 132–9. [Google Scholar]
- Schröter, N; Sajonz, BEA; Jost, WH; Rijntjes, M; Coenen, VA; Groppa, S. Advanced therapies in Parkinson’s disease: an individualized approach to their indication. J Neural Transm (Vienna) 2024, 131, 1285–93. [Google Scholar] [CrossRef] [PubMed]
- Soileau, MJ; Aldred, J; Budur, K; et al. Safety and efficacy of continuous subcutaneous foslevodopa-foscarbidopa in patients with advanced Parkinson’s disease: a randomised, double-blind, active-controlled, phase 3 trial. Lancet Neurol 2022, 21, 1099–109. [Google Scholar] [CrossRef]
- Santos-García, D; Solleiro, Á; González-Ortega, G. DATs-PD GETM Spanish Registry Group. Profile of People With Parkinson’s Disease Treated With a Device-Aided Therapy in Spain. A Comparative Multicenter Observational Study. Mov Disord Clin Pract 2026, 13, 154–72. [Google Scholar] [CrossRef]
- Santos-García, D; Solleiro, Á; González-Ortega, G. DATs-PD GETM Spanish Registry Group. Impact of device-aided therapies on quality of life in patients with Parkinson’s disease. A comparative multicenter observational study. J Neural Transm (Vienna) 2025. [Google Scholar] [CrossRef] [PubMed]
- Blair, HA. Foslevodopa/Foscarbidopa: A Review in Advanced Parkinson’s Disease. CNS Drugs 2025, 39, 621–632. [Google Scholar] [CrossRef] [PubMed]
- Jost, ST; Kaldenbach, MA; Antonini, A. International Parkinson and Movement Disorders Society Non-Motor Parkinson Disease Study Group. Levodopa Dose Equivalency in Parkinson’s Disease: Updated Systematic Review and Proposals. Mov Disord 2023, 38, 1236–52. [Google Scholar] [CrossRef] [PubMed]
- Santos-García, D; López-Manzanares, L; Muro, I; et al. Effectiveness and safety of levodopa-entacapone-carbidopa infusion in Parkinson disease: A real-world data study. Eur J Neurol 2025, 32, e16535. [Google Scholar] [CrossRef] [PubMed]
- Warnecke, T; Odin, P; García-Ramos, R; et al. Effects of levodopa-entacapone-carbidopa intrajejunal infusion on non-motor symptoms of advanced Parkinson’s disease-interim analysis of the ELEGANCE study. J Neural Transm (Vienna) 2025. [Google Scholar] [CrossRef] [PubMed]
- Weiss, D; Jost, WH; Szász, JA; et al. Levodopa-Entacapone-Carbidopa Intrajejunal Infusion in Advanced Parkinson’s Disease - Interim Analysis of the ELEGANCE Study. Mov Disord Clin Pract 2025, 12, 1075–85. [Google Scholar] [CrossRef] [PubMed]
- Katzenschlager, R; Bergquist, F. Continuous subcutaneous infusion therapies in Parkinson’s disease: evidence of efficacy and safety. Parkinsonism Relat Disord 2025, 139 (Suppl 1), 107905. [Google Scholar] [CrossRef] [PubMed]
- Aldred, J; Freire-Alvarez, E; Amelin, AV; et al. Continuous Subcutaneous Foslevodopa/Foscarbidopa in Parkinson’s Disease: Safety and Efficacy Results From a 12-Month, Single-Arm, Open-Label, Phase 3 Study. Neurol Ther 2023, 12, 1937–58. [Google Scholar] [CrossRef] [PubMed]
- Henriksen, T; Katzenschlager, R; Bhidayasiri, R; Staines, H; Lockhart, D; Lees, A. Practical use of apomorphine infusion in Parkinson’s disease: lessons from the TOLEDO study and clinical experience. J Neural Transm (Vienna) 2023, 130, 1475–84. [Google Scholar] [CrossRef] [PubMed]
- Jost, WH. Apprends-moi l’art des petits pas: Levodopa, Carbidopa Intestinal Gel plus Entacapone. J Neural Transm (Vienna) 2023, 130, 1379–82. [Google Scholar] [CrossRef] [PubMed]
- Horváth, K; Aschermann, Z; Kovács, M; et al. Changes in Quality of Life in Parkinson’s Disease: How Large Must They Be to Be Relevant? Neuroepidemiology 2017, 48, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Nyholm, D; Jost, WH. Levodopa-entacapone-carbidopa intestinal gel infusion in advanced Parkinson’s disease: real-world experience and practical guidance. Ther Adv Neurol Disord 2022, 15, 17562864221108018. [Google Scholar] [CrossRef]
- Poplawska-Domaszewicz, K; Batzu, L; Falup-Pecurariu, C; Chaudhuri, KR. Subcutaneous Levodopa: A New Engine for the Vintage Molecule. Neurol Ther 2024, 13, 1055–68. [Google Scholar] [CrossRef]
- Jenner, P; Nyholm, D. COMT inhibition with entacapone for patients with Parkinson’s disease and motor complications: the novelty of continuous infusion. J Neural Transm (Vienna) 2026, 133, 361–75. [Google Scholar] [CrossRef]
- Szatmári, S; Szász, JA; Orbán-Kis, K; et al. Levodopa-Entacapone-Carbidopa Intestinal Gel in the Treatment of Advanced Parkinson’s Disease: A Single Center Real-World Experience. Pharmaceutics 2024, 16, 453. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).