Submitted:
02 March 2026
Posted:
02 March 2026
You are already at the latest version
Abstract
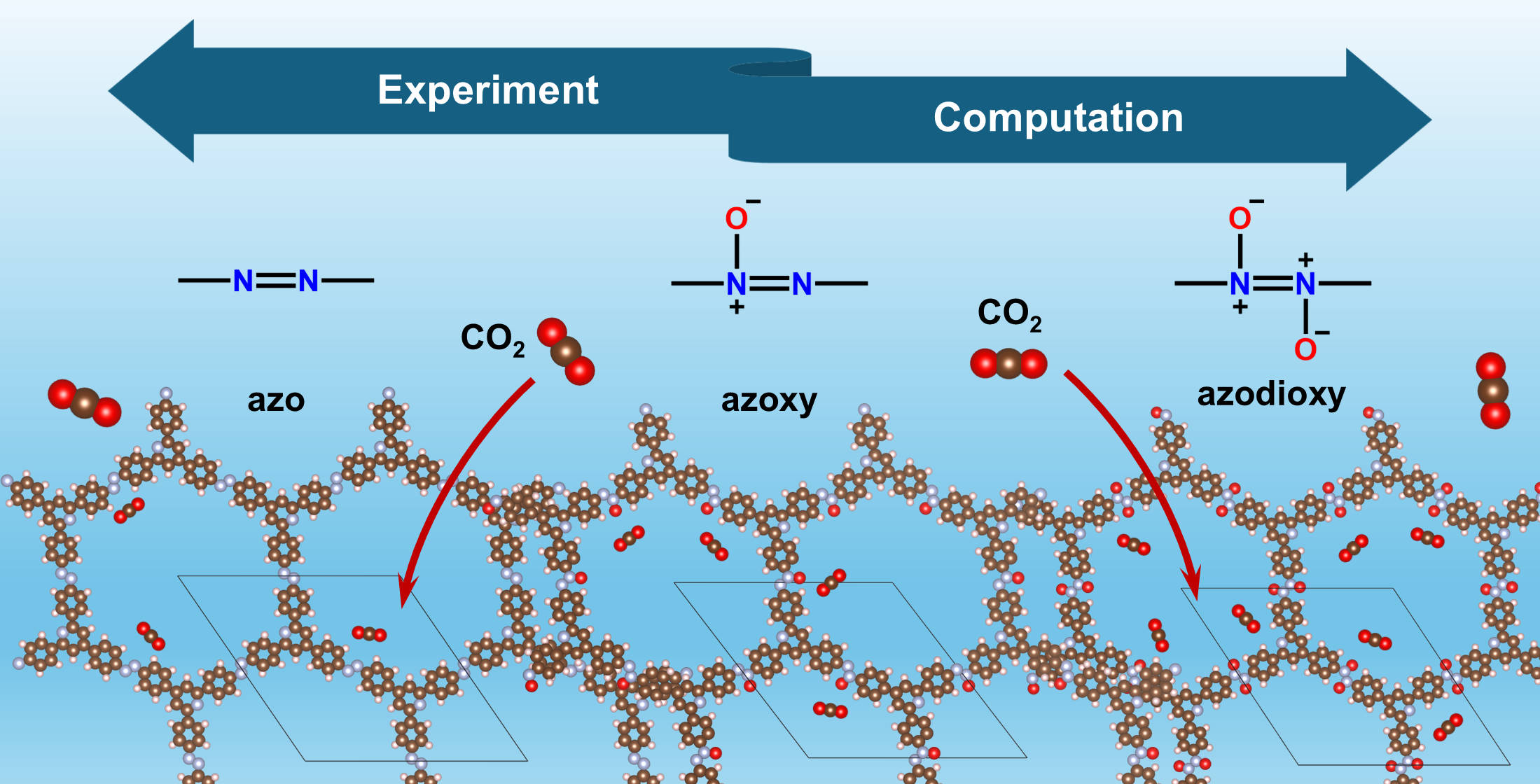
Keywords:
1. Introduction
2. Synthesis and CO2 Capture Performance of Azo-Linked POPs
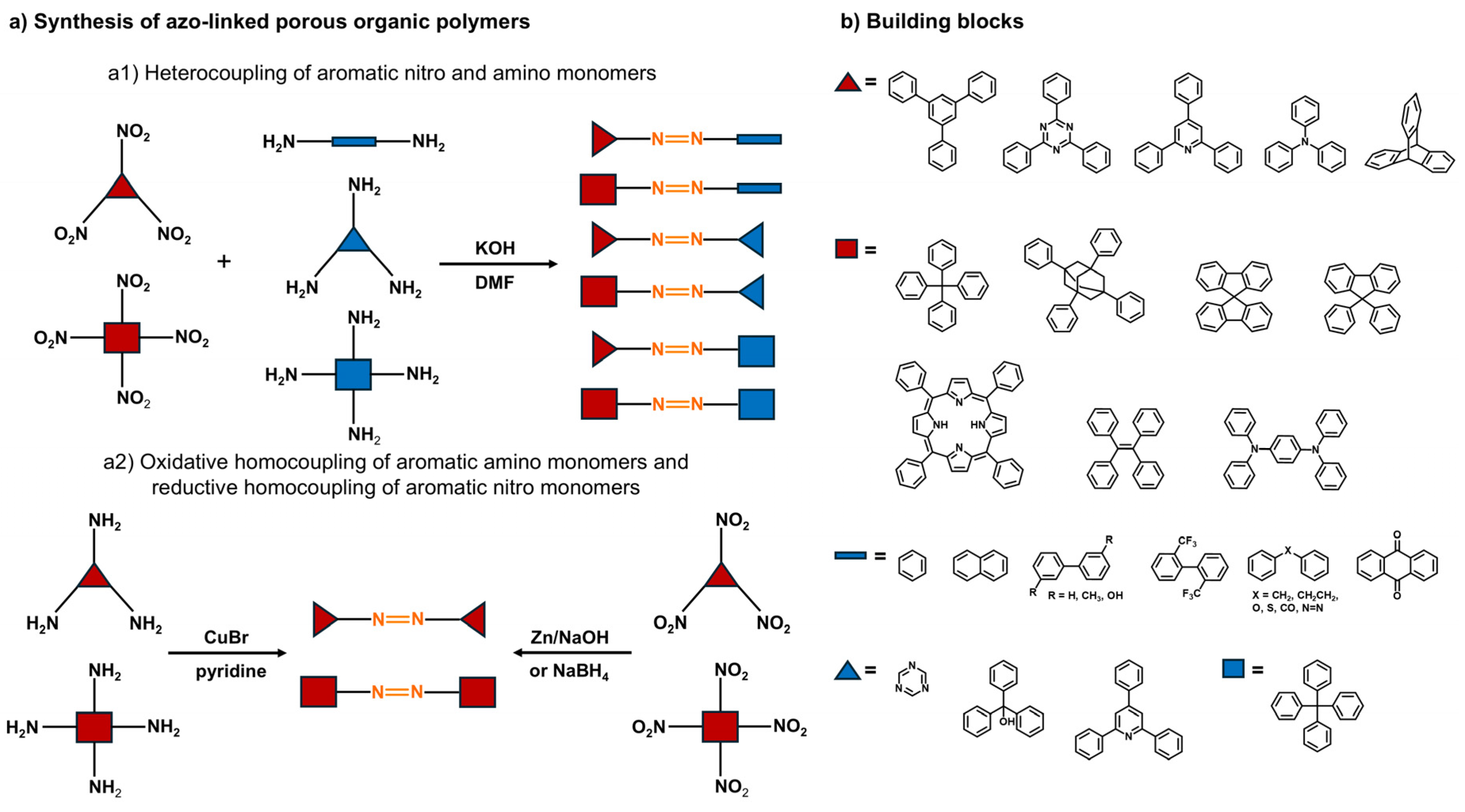
2.1. Synthesis of Azo-Linked POPs
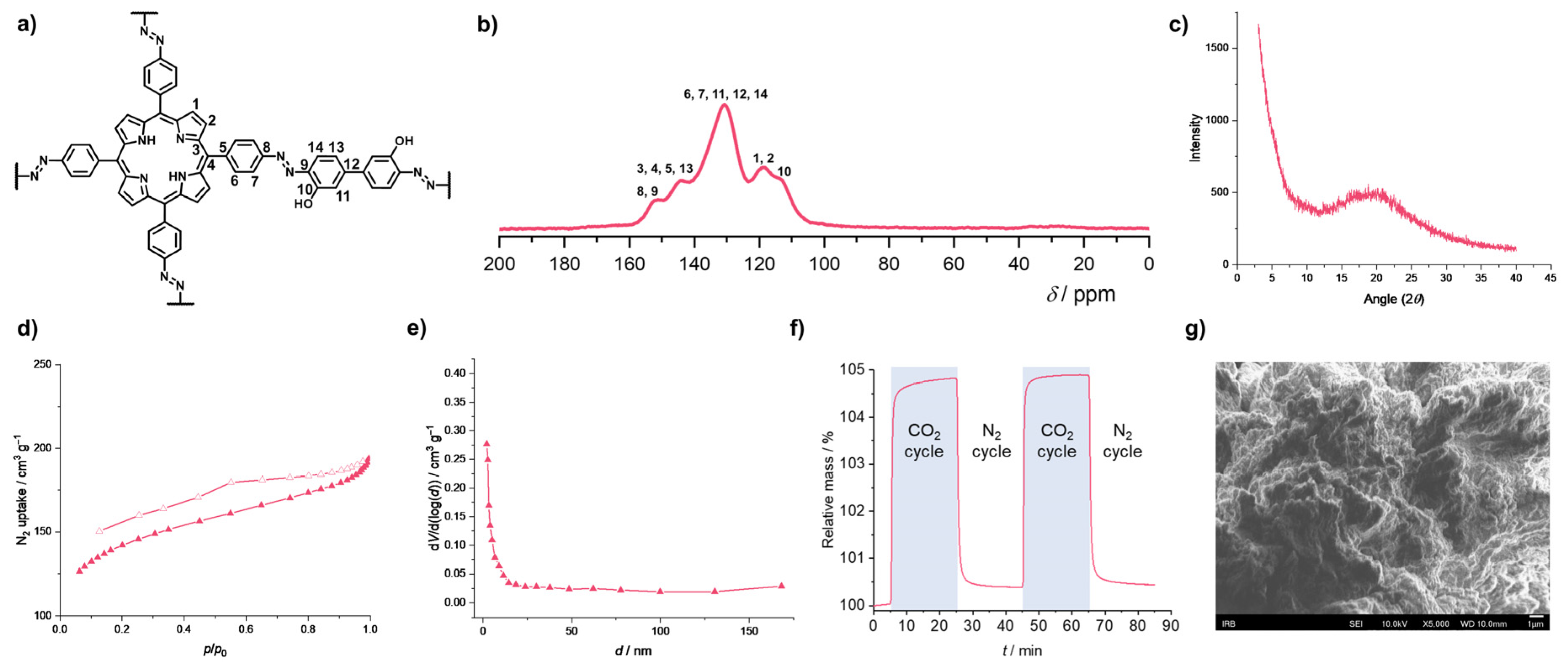
2.2. Azo-Linked POPs for CO2 Capture
3. Synthesis and CO2 Capture Performance of Azodioxy and Azoxy-Linked POPs
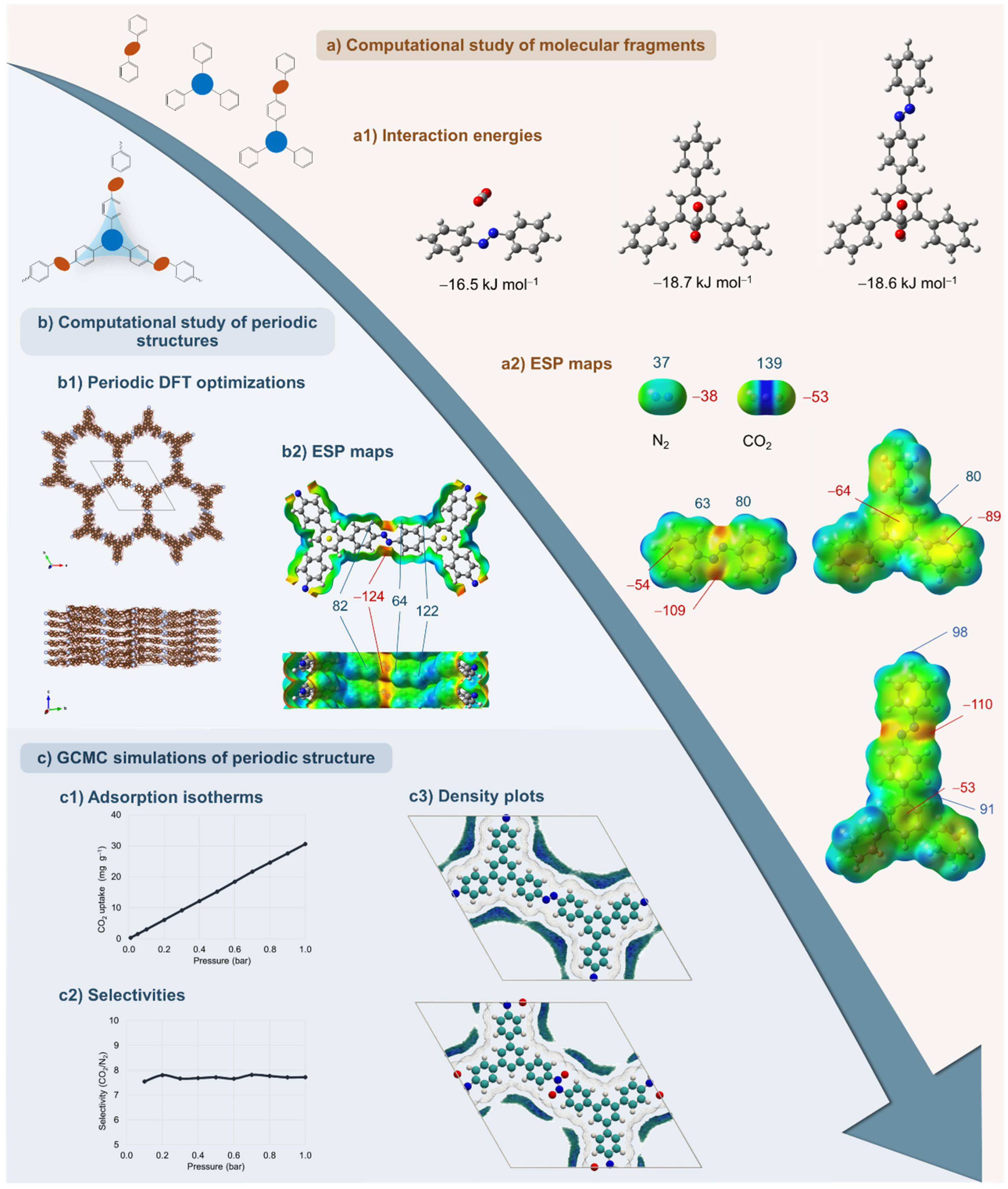
4. Computational Studies of N−N-Linked POPs
4.1. Computational Methodology for Studying Gas Adsorption in N−N-Linked POPs
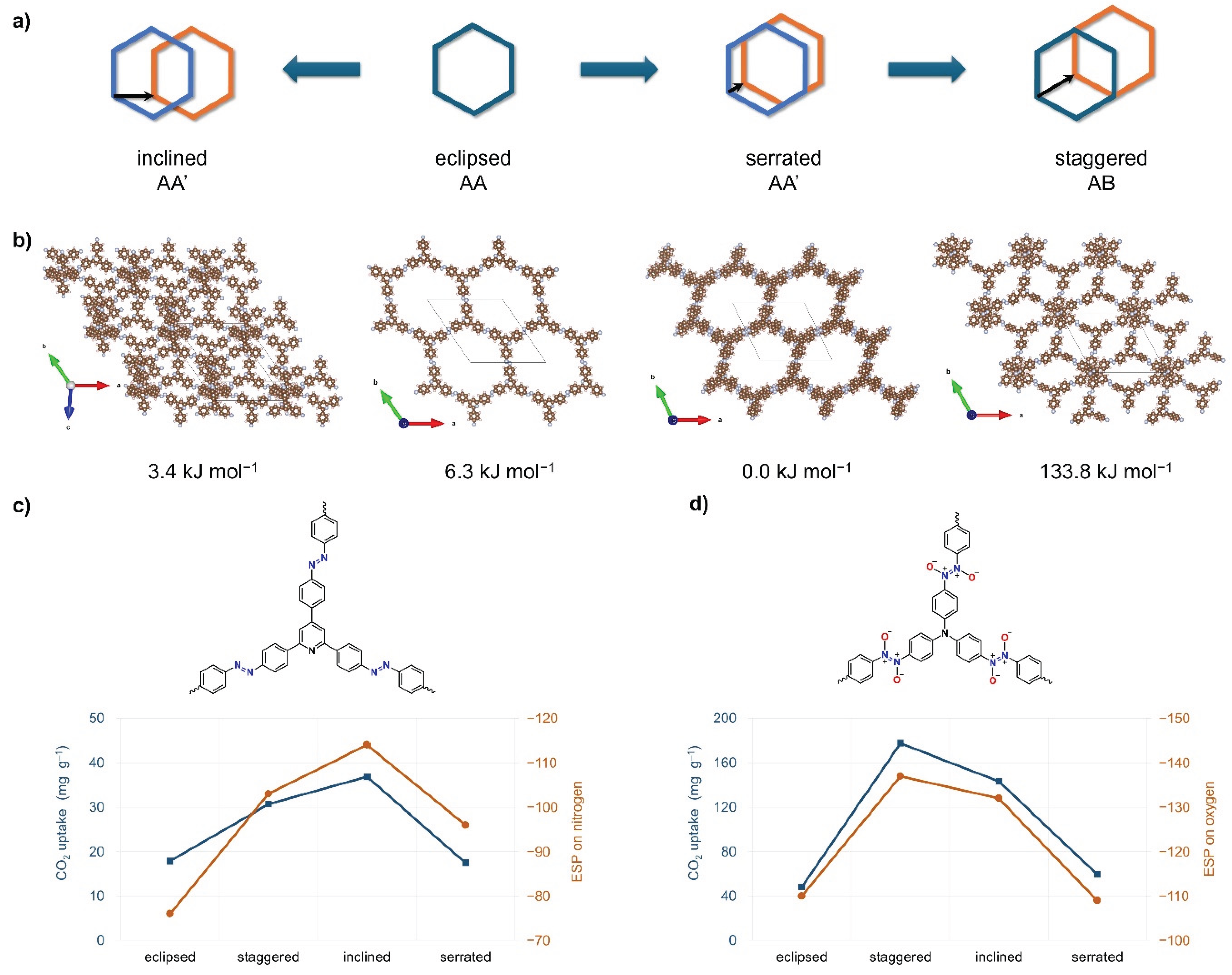
4.2. Computational Studies of Azo-Linked Frameworks for CO2 Adsorption
4.3. Computational Studies of Azo-Linked POPs Beyond CO2 Adsorption
4.4. Computational Study of Azodioxy and Azoxy-Linked Frameworks for CO2 Adsorption
4. Conclusion and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ma, H.; Fu, H.; Tong, Y.; Umar, A.; Hung, Y.M.; Wang, X. Advances in CO2 Capture and Separation Materials: Emerging Trends, Challenges, and Prospects for Sustainable Applications. Carbon Capture Sci. Technol. 2025, 15, 100441. [Google Scholar] [CrossRef]
- Zhong, Z.; Wang, X.; Tan, B. Porous Organic Polymers for CO2 Capture and Catalytic Conversion. Chem. Eur. J. 2025, 31. [Google Scholar] [CrossRef]
- Yoro, K.O.; Daramola, M.O. CO2 Emission Sources, Greenhouse Gases, and the Global Warming Effect. In Advances in Carbon Capture; Elsevier, 2020; pp. 3–28. [Google Scholar]
- Nunes, L.J.R. The Rising Threat of Atmospheric CO2: A Review on the Causes, Impacts, and Mitigation Strategies. Environments 2023, 10, 66. [Google Scholar] [CrossRef]
- Carbon Dioxide Now More than 50% Higher than Pre-Industrial Levels. Available online: https://www.noaa.gov/news-release/carbon-dioxide-now-more-than-50-higher-than-pre-industrial-levels (accessed on 23 February 2026).
- Carbon Dioxide - Earth Indicator. Available online: https://science.nasa.gov/earth/explore/earth-indicators/carbon-dioxide/ (accessed on 23 February 2026).
- Zentou, H.; Hoque, B.; Abdalla, M.A.; Saber, A.F.; Abdelaziz, O.Y.; Aliyu, M.; Alkhedhair, A.M.; Alabduly, A.J.; Abdelnaby, M.M. Recent Advances and Challenges in Solid Sorbents for CO2 Capture. Carbon Capture Sci. Technol. 2025, 15, 100386. [Google Scholar] [CrossRef]
- Singh, G.; Lee, J.; Karakoti, A.; Bahadur, R.; Yi, J.; Zhao, D.; AlBahily, K.; Vinu, A. Emerging Trends in Porous Materials for CO2 Capture and Conversion. Chem. Soc. Rev. 2020, 49, 4360–4404. [Google Scholar] [CrossRef] [PubMed]
- Song, K.S.; Fritz, P.W.; Coskun, A. Porous Organic Polymers for CO2 Capture, Separation and Conversion. Chem. Soc. Rev. 2022, 51, 9831–9852. [Google Scholar] [CrossRef] [PubMed]
- Sumida, K.; Rogow, D.L.; Mason, J.A.; McDonald, T.M.; Bloch, E.D.; Herm, Z.R.; Bae, T.-H.; Long, J.R. Carbon Dioxide Capture in Metal–Organic Frameworks. Chem. Rev. 2012, 112, 724–781. [Google Scholar] [CrossRef]
- Sai Bhargava Reddy, M.; Ponnamma, D.; Sadasivuni, K.K.; Kumar, B.; Abdullah, A.M. Carbon Dioxide Adsorption Based on Porous Materials. RSC Adv. 2021, 11, 12658–12681. [Google Scholar] [CrossRef]
- Kumar, S.; Srivastava, R.; Koh, J. Utilization of Zeolites as CO2 Capturing Agents: Advances and Future Perspectives. J. CO2 Util. 2020, 41, 101251. [Google Scholar] [CrossRef]
- Boer, D.G.; Langerak, J.; Pescarmona, P.P. Zeolites as Selective Adsorbents for CO2 Separation. ACS Appl. Energy Mater. 2023, 6, 2634–2656. [Google Scholar] [CrossRef]
- Li, Z.; Liu, P.; Ou, C.; Dong, X. Porous Metal–Organic Frameworks for Carbon Dioxide Adsorption and Separation at Low Pressure. ACS Sustain. Chem. Eng. 2020, 8, 15378–15404. [Google Scholar] [CrossRef]
- Ghanbari, T.; Abnisa, F.; Wan Daud, W.M.A. A Review on Production of Metal Organic Frameworks (MOF) for CO2 Adsorption. Sci. Total Environ. 2020, 707, 135090. [Google Scholar] [CrossRef] [PubMed]
- Trickett, C.A.; Helal, A.; Al-Maythalony, B.A.; Yamani, Z.H.; Cordova, K.E.; Yaghi, O.M. The Chemistry of Metal–Organic Frameworks for CO2 Capture, Regeneration and Conversion. Nat. Rev. Mater. 2017, 2, 17045. [Google Scholar] [CrossRef]
- Ding, M.; Flaig, R.W.; Jiang, H.-L.; Yaghi, O.M. Carbon Capture and Conversion Using Metal–Organic Frameworks and MOF-Based Materials. Chem. Soc. Rev. 2019, 48, 2783–2828. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Xie, L.-H.; Li, J.-R.; Ma, Y.; Seminario, J.M.; Balbuena, P.B. CO2 Capture and Separations Using MOFs: Computational and Experimental Studies. Chem. Rev. 2017, 117, 9674–9754. [Google Scholar] [CrossRef]
- Bhanja, P.; Modak, A.; Bhaumik, A. Porous Organic Polymers for CO2 Storage and Conversion Reactions. ChemCatChem 2019, 11, 244–257. [Google Scholar] [CrossRef]
- Mohamed, M.G.; EL-Mahdy, Ahmed.F.M.; Kotp, M.G.; Kuo, S.-W. Advances in Porous Organic Polymers: Syntheses, Structures, and Diverse Applications. Mater. Adv. 2022, 3, 707–733. [Google Scholar] [CrossRef]
- Acevedo, S.; Giraldo, L.; Moreno-Piraján, J.C. Adsorption of CO2 on Activated Carbons Prepared by Chemical Activation with Cupric Nitrate. ACS Omega 2020, 5, 10423–10432. [Google Scholar] [CrossRef]
- Zhang, Z.; Liu, Z.; Xue, C.; Chen, H.; Han, X.; Ren, Y. Amorphous Porous Organic Polymers Containing Main Group Elements. Commun. Chem. 2023, 6, 271. [Google Scholar] [CrossRef]
- Zhu, Y.; Long, H.; Zhang, W. Imine-Linked Porous Polymer Frameworks with High Small Gas (H2, CO2, CH4, C2H2) Uptake and CO2/N2 Selectivity. Chem. Mater. 2013, 25, 1630–1635. [Google Scholar] [CrossRef]
- Gu, C.; Liu, D.; Huang, W.; Liu, J.; Yang, R. Synthesis of Covalent Triazine-Based Frameworks with High CO2 Adsorption and Selectivity. Polym. Chem. 2015, 6, 7410–7417. [Google Scholar] [CrossRef]
- Wang, W.; Zhou, M.; Yuan, D. Carbon Dioxide Capture in Amorphous Porous Organic Polymers. J. Mater. Chem. A 2017, 5, 1334–1347. [Google Scholar] [CrossRef]
- Nandi, S.; Werner-Zwanziger, U.; Vaidhyanathan, R. A Triazine–Resorcinol Based Porous Polymer with Polar Pores and Exceptional Surface Hydrophobicity Showing CO2 Uptake under Humid Conditions. J. Mater. Chem. A 2015, 3, 21116–21122. [Google Scholar] [CrossRef]
- Patel, H.A.; Hyun Je, S.; Park, J.; Chen, D.P.; Jung, Y.; Yavuz, C.T.; Coskun, A. Unprecedented High-Temperature CO2 Selectivity in N2-Phobic Nanoporous Covalent Organic Polymers. Nat. Commun. 2013, 4, 1357. [Google Scholar] [CrossRef]
- Patel, H.A.; Je, S.H.; Park, J.; Jung, Y.; Coskun, A.; Yavuz, C.T. Directing the Structural Features of N2 -Phobic Nanoporous Covalent Organic Polymers for CO2 Capture and Separation. Chem. Eur. J. 2014, 20, 772–780. [Google Scholar] [CrossRef] [PubMed]
- Gu, S.; Guo, J.; Huang, Q.; He, J.; Fu, Y.; Kuang, G.; Pan, C.; Yu, G. 1,3,5-Triazine-Based Microporous Polymers with Tunable Porosities for CO2 Capture and Fluorescent Sensing. Macromolecules 2017, 50, 8512–8520. [Google Scholar] [CrossRef]
- Zhu, X.; Mahurin, S.M.; An, S.-H.; Do-Thanh, C.-L.; Tian, C.; Li, Y.; Gill, L.W.; Hagaman, E.W.; Bian, Z.; Zhou, J.-H.; et al. Efficient CO2 Capture by a Task-Specific Porous Organic Polymer Bifunctionalized with Carbazole and Triazine Groups. Chem. Commun. 2014, 50, 7933. [Google Scholar] [CrossRef]
- Li, G.; Qin, L.; Yao, C.; Xu, Y. Controlled Synthesis of Conjugated Polycarbazole Polymers via Structure Tuning for Gas Storage and Separation Applications. Sci. Rep. 2017, 7, 15394. [Google Scholar] [CrossRef] [PubMed]
- Car, Ž.; Borovina, M.; Panić, B.; Biljan, I. Microwave-Assisted Reductive Homocoupling of Aromatic Nitro Monomers: Synthesis of Azo-Linked Porous Organic Polymers for CO2 Capture. RSC Adv. 2025, 15, 7332–7339. [Google Scholar] [CrossRef] [PubMed]
- Panić, B.; Frey, T.; Borovina, M.; Ištoković, P.; Kodrin, I.; Biljan, I. Synergistic Experimental and Computational Investigation of Azo-Linked Porphyrin-Based Porous Organic Polymers for CO2 Capture. RSC Adv. 2025, 15, 13774–13785. [Google Scholar] [CrossRef]
- Popović, M.; Frey, T.; Borovina, M.; Kodrin, I.; Biljan, I. Tuning Linkers in Azo-Linked Porphyrin-Based Porous Organic Polymers for Enhanced CO2 Capture. Langmuir 2026, 42, 4641–4652. [Google Scholar] [CrossRef]
- Rabbani, M.G.; El-Kaderi, H.M. Synthesis and Characterization of Porous Benzimidazole-Linked Polymers and Their Performance in Small Gas Storage and Selective Uptake. Chem. Mater. 2012, 24, 1511–1517. [Google Scholar] [CrossRef]
- Arab, P.; Rabbani, M.G.; Sekizkardes, A.K.; İslamoğlu, T.; El-Kaderi, H.M. Copper(I)-Catalyzed Synthesis of Nanoporous Azo-Linked Polymers: Impact of Textural Properties on Gas Storage and Selective Carbon Dioxide Capture. Chem. Mater. 2014, 26, 1385–1392. [Google Scholar] [CrossRef]
- Arab, P.; Parrish, E.; İslamoğlu, T.; El-Kaderi, H.M. Synthesis and Evaluation of Porous Azo-Linked Polymers for Carbon Dioxide Capture and Separation. J. Mater. Chem. A 2015, 3, 20586–20594. [Google Scholar] [CrossRef]
- Lu, J.; Zhang, J. Facile Synthesis of Azo-Linked Porous Organic Frameworks via Reductive Homocoupling for Selective CO2 Capture. J. Mater. Chem. A 2014, 2, 13831–13834. [Google Scholar] [CrossRef]
- Zhou, J.-X.; Luo, X.-S.; Liu, X.; Qiao, Y.; Wang, P.; Mecerreyes, D.; Bogliotti, N.; Chen, S.-L.; Huang, M.-H. Azo-Linked Porous Organic Polymers: Robust and Time-Efficient Synthesis via NaBH4-Mediated Reductive Homocoupling on Polynitro Monomers and Adsorption Capacity towards Aniline in Water. J. Mater. Chem. A 2018, 6, 5608–5612. [Google Scholar] [CrossRef]
- Modak, A.; Nandi, M.; Mondal, J.; Bhaumik, A. Porphyrin Based Porous Organic Polymers: Novel Synthetic Strategy and Exceptionally High CO2 Adsorption Capacity. Chem. Commun. 2012, 48, 248–250. [Google Scholar] [CrossRef]
- Feng, L.; Wang, K.-Y.; Joseph, E.; Zhou, H.-C. Catalytic Porphyrin Framework Compounds. Trends Chem. 2020, 2, 555–568. [Google Scholar] [CrossRef]
- Ji, W.; Wang, T.-X.; Ding, X.; Lei, S.; Han, B.-H. Porphyrin- and Phthalocyanine-Based Porous Organic Polymers: From Synthesis to Application. Coord. Chem. Rev. 2021, 439, 213875. [Google Scholar] [CrossRef]
- Jin, L.; Lv, S.; Miao, Y.; Liu, D.; Song, F. Recent Development of Porous Porphyrin-based Nanomaterials for Photocatalysis. ChemCatChem 2021, 13, 140–152. [Google Scholar] [CrossRef]
- Zhu, Y.; Zhu, D.; Chen, Y.; Yan, Q.; Liu, C.-Y.; Ling, K.; Liu, Y.; Lee, D.; Wu, X.; Senftle, T.P.; et al. Porphyrin-Based Donor–Acceptor COFs as Efficient and Reusable Photocatalysts for PET-RAFT Polymerization under Broad Spectrum Excitation. Chem. Sci. 2021, 12, 16092–16099. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Li, Z.; Zhang, F.; Zhuang, X.; Zeng, Z.; Wei, J. New Nitrogen-Rich Azo-Bridged Porphyrin-Conjugated Microporous Networks for High Performance of Gas Capture and Storage. RSC Adv. 2016, 6, 30048–30055. [Google Scholar] [CrossRef]
- Bera, R.; Ansari, M.; Alam, A.; Das, N. Nanoporous Azo Polymers (NAPs) for Selective CO2 Uptake. J. CO2 Util. 2018, 28, 385–392. [Google Scholar] [CrossRef]
- Jiang, X.; Liu, Y.; Liu, J.; Luo, Y.; Lyu, Y. Facile Synthesis of Porous Organic Polymers Bifunctionalized with Azo and Porphyrin Groups. RSC Adv. 2015, 5, 98508–98513. [Google Scholar] [CrossRef]
- Tao, L.; Niu, F.; Zhang, D.; Liu, J.; Wang, T.; Wang, Q. Azo-Bridged Covalent Porphyrinic Polymers (Azo-CPPs): Synthesis and CO2 Capture Properties. RSC Adv. 2015, 5, 96871–96878. [Google Scholar] [CrossRef]
- Youm, K.; Choi, Y.; Byun, H.; Kumar, S.; Cho, Y.; Hsan, N.; Koh, J. A Highly Efficient Porphyrin-Based Azo-Porous Organic Polymer for Selective CO2 Capture and Conversion. J. CO2 Util. 2024, 84, 102854. [Google Scholar] [CrossRef]
- Šutalo, P.; Pisačić, M.; Biljan, I.; Kodrin, I. Benzene and Triazine-Based Porous Organic Polymers with Azo, Azoxy and Azodioxy Linkages: A Computational Study. CrystEngComm 2022, 24, 4748–4763. [Google Scholar] [CrossRef]
- Frey, T.; Panić, B.; Šutalo, P.; Borovina, M.; Biljan, I.; Kodrin, I. Prediction of CO2 Adsorption Properties of Azo, Azoxy and Azodioxy-Linked Porous Organic Polymers Guided by Electrostatic Potential. CrystEngComm 2023, 25, 3870–3884. [Google Scholar] [CrossRef]
- Beaudoin, D.; Wuest, J.D. Dimerization of Aromatic C-Nitroso Compounds. Chem. Rev. 2016, 116, 258–286. [Google Scholar] [CrossRef]
- Vančik, H. Aromatic C-Nitroso Compounds; Springer Netherlands: Dordrecht, 2013; ISBN 978-94-007-6336-4. [Google Scholar]
- Rončević, I.; Bibulić, P.; Vančik, H.; Biljan, I. Solution Equilibria of Aromatic Dinitroso Compounds: A Combined NMR and DFT Study. Struct. Chem. 2018, 29, 1489–1497. [Google Scholar] [CrossRef]
- Bibulić, P.; Rončević, I.; Špadina, M.; Biljan, I.; Vančik, H. Isothermal and Isoconversional Modeling of Solid-State Nitroso Polymerization. J. Phys. Chem. A 2020, 124, 10726–10735. [Google Scholar] [CrossRef] [PubMed]
- Bibulić, P.; Rončević, I.; Varga, K.; Mihalić, Z.; Vančik, H. Structure and Topochemistry of Azodioxide Oligomers in Solid State. J. Mol. Struct. 2016, 1104, 85–90. [Google Scholar] [CrossRef]
- Gowenlock, B.G.; Richter-Addo, G.B. Dinitroso and Polynitroso Compounds. Chem. Soc. Rev. 2005, 34, 797. [Google Scholar] [CrossRef] [PubMed]
- Rowan, S.J.; Cantrill, S.J.; Cousins, G.R.L.; Sanders, J.K.M.; Stoddart, J.F. Dynamic Covalent Chemistry. Angew. Chem. Int. Ed. 2002, 41, 898–952. [Google Scholar] [CrossRef]
- Beaudoin, D.; Maris, T.; Wuest, J.D. Constructing Monocrystalline Covalent Organic Networks by Polymerization. Nat. Chem. 2013, 5, 830–834. [Google Scholar] [CrossRef]
- Nath, B.; Li, W.-H.; Huang, J.-H.; Wang, G.-E.; Fu, Z.; Yao, M.-S.; Xu, G. A New Azodioxy-Linked Porphyrin-Based Semiconductive Covalent Organic Framework with I2 Doping-Enhanced Photoconductivity. CrystEngComm 2016, 18, 4259–4263. [Google Scholar] [CrossRef]
- Huskić, I.; Halasz, I.; Friščić, T.; Vančik, H. Mechanosynthesis of Nitrosobenzenes: A Proof-of-Principle Study in Combining Solvent-Free Synthesis with Solvent-Free Separations. Green Chem. 2012, 14, 1597. [Google Scholar] [CrossRef]
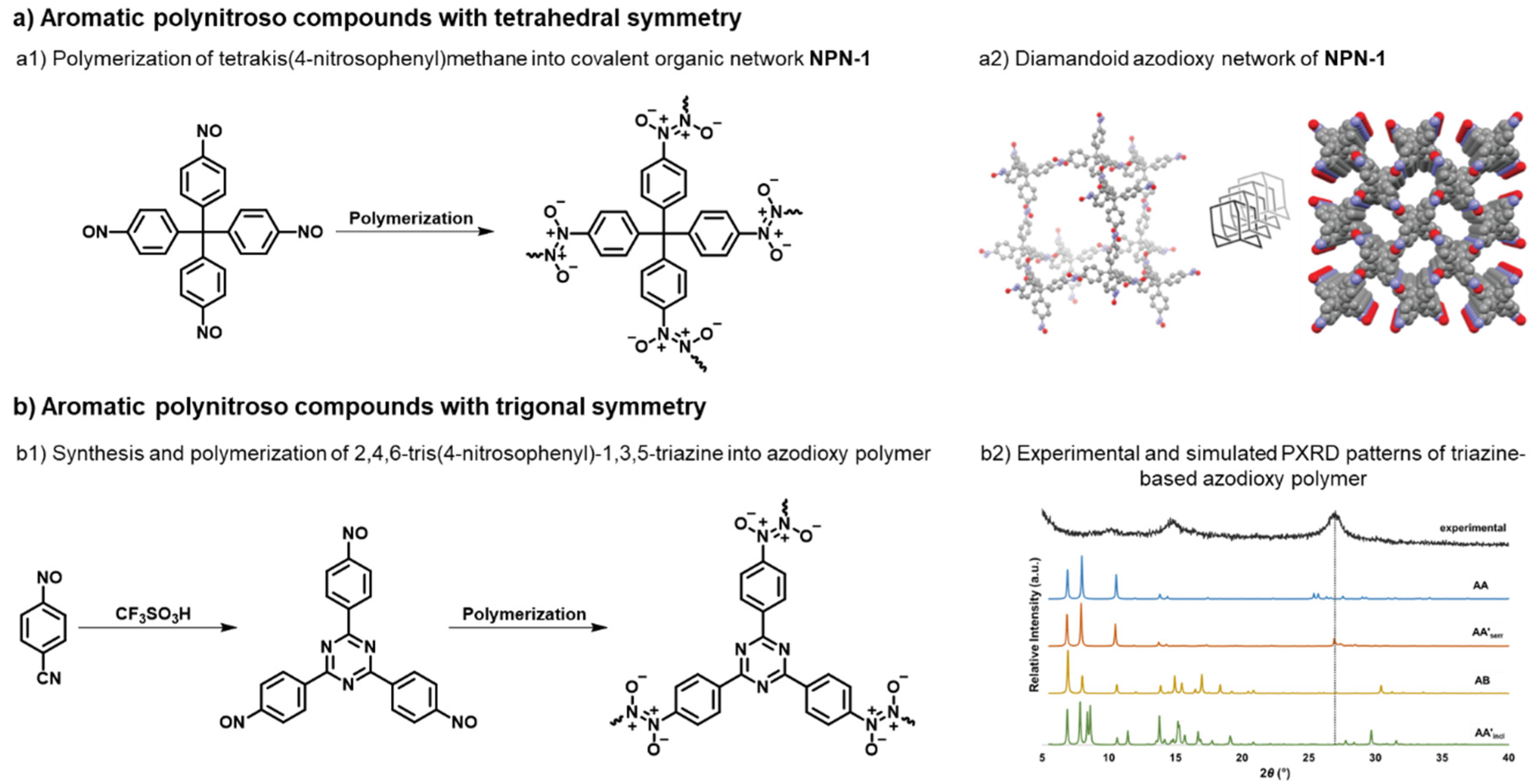
- Cindro, N.; Car, Ž.; Petrović Peroković, V.; Borovina, M.; Panić, B.; Kodrin, I.; Biljan, I. Synthesis of Aromatic Polynitroso Compounds: Towards Functional Azodioxy-Linked Porous Polymers. Heliyon 2023, 9, e21781. [Google Scholar] [CrossRef]
- Panić, B.; Frey, T.; Borovina, M.; Konopka, K.; Sambolec, M.; Kodrin, I.; Biljan, I. Synthesis and Characterization of Benzene- and Triazine-Based Azo-Bridged Porous Organic Polymers. Polymers (Basel). 2023, 15, 229. [Google Scholar] [CrossRef]
- Yang, Z.; Zhang, H.; Yu, B.; Zhao, Y.; Ma, Z.; Ji, G.; Han, B.; Liu, Z. Azo-Functionalized Microporous Organic Polymers: Synthesis and Applications in CO2 Capture and Conversion. Chem. Commun. 2015, 51, 11576–11579. [Google Scholar] [CrossRef]
- Ji, G.; Yang, Z.; Zhang, H.; Zhao, Y.; Yu, B.; Ma, Z.; Liu, Z. Hierarchically Mesoporous o-Hydroxyazobenzene Polymers: Synthesis and Their Applications in CO2 Capture and Conversion. Angew. Chem. 2016, 128, 9837–9841. [Google Scholar] [CrossRef]
- Bera, R.; Ansari, M.; Alam, A.; Das, N. Triptycene, Phenolic-OH, and Azo-Functionalized Porous Organic Polymers: Efficient and Selective CO2 Capture. ACS Appl. Polym. Mater. 2019, 1, 959–968. [Google Scholar] [CrossRef]
- Buyukcakir, O.; Je, S.H.; Park, J.; Patel, H.A.; Jung, Y.; Yavuz, C.T.; Coskun, A. Systematic Investigation of the Effect of Polymerization Routes on the Gas-Sorption Properties of Nanoporous Azobenzene Polymers. Chem. Eur. J. 2015, 21, 15320–15327. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Lee, H.J.; Lim, S.Y.; Kim, B.G.; Choi, J.W. Combined CO2 -Philicity and Ordered Mesoporosity for Highly Selective CO2 Capture at High Temperatures. J. Am. Chem. Soc. 2015, 137, 7210–7216. [Google Scholar] [CrossRef]
- Dang, Q.-Q.; Wang, X.-M.; Zhan, Y.-F.; Zhang, X.-M. An Azo-Linked Porous Triptycene Network as an Absorbent for CO2 and Iodine Uptake. Polym. Chem. 2016, 7, 643–647. [Google Scholar] [CrossRef]
- Varga, K.; Biljan, I.; Tomišić, V.; Mihalić, Z.; Vančik, H. Quantum Chemical Calculations of Monomer–Dimer Equilibria of Aromatic C -Nitroso Compounds. J. Phys. Chem. A 2018, 122, 2542–2549. [Google Scholar] [CrossRef]
- Biljan, I.; Vančik, H. Aromatic C-Nitroso Compounds: From Solid-State Reactivity to New Materials. ChemistrySelect 2025, 10. [Google Scholar] [CrossRef]
- Borovina, M.; Petrović Peroković, V.; Panić, B.; Biljan, I. Structural Characterization of 1,2-Bis(4-Cyanophenyl)Diazene Oxide and Its Cyclotrimerization to a Triazine-Linked Polymer. ACS Omega 2026. [Google Scholar] [CrossRef]
- Erba, A.; Desmarais, J.K.; Casassa, S.; Civalleri, B.; Donà, L.; Bush, I.J.; Searle, B.; Maschio, L.; Edith-Daga, L.; Cossard, A.; et al. CRYSTAL23: A Program for Computational Solid State Physics and Chemistry. J. Chem. Theory Comput. 2023, 19, 6891–6932. [Google Scholar] [CrossRef] [PubMed]
- Ran, Y.A.; Sharma, S.; Balestra, S.R.G.; Li, Z.; Calero, S.; Vlugt, T.J.H.; Snurr, R.Q.; Dubbeldam, D. RASPA3: A Monte Carlo Code for Computing Adsorption and Diffusion in Nanoporous Materials and Thermodynamics Properties of Fluids. J. Chem. Phys. 2024, 161. [Google Scholar] [CrossRef]
- Chandra, S.; Kundu, T.; Kandambeth, S.; BabaRao, R.; Marathe, Y.; Kunjir, S.M.; Banerjee, R. Phosphoric Acid Loaded Azo (−N═N−) Based Covalent Organic Framework for Proton Conduction. J. Am. Chem. Soc. 2014, 136, 6570–6573. [Google Scholar] [CrossRef]
- Ge, R.; Hao, D.; Shi, Q.; Dong, B.; Leng, W.; Wang, C.; Gao, Y. Target Synthesis of an Azo (N═N) Based Covalent Organic Framework with High CO 2 -over-N 2 Selectivity and Benign Gas Storage Capability. J. Chem. Eng. Data 2016, 61, 1904–1909. [Google Scholar] [CrossRef]
- Huang, J.; Golomb, M.J.; Kavanagh, S.R.; Tolborg, K.; Ganose, A.M.; Walsh, A. Band Gap Opening from Displacive Instabilities in Layered Covalent-Organic Frameworks. J. Mater. Chem. A 2022, 10, 13500–13507. [Google Scholar] [CrossRef]
- Huang, J.; Shin, S.-J.; Tolborg, K.; Ganose, A.M.; Krenzer, G.; Walsh, A. Room-Temperature Stacking Disorder in Layered Covalent-Organic Frameworks from Machine-Learning Force Fields. Mater. Horiz. 2023, 10, 2883–2891. [Google Scholar] [CrossRef]
- Koo, B.T.; Dichtel, W.R.; Clancy, P. A Classification Scheme for the Stacking of Two-Dimensional Boronate Ester-Linked Covalent Organic Frameworks. J. Mater. Chem. 2012, 22, 17460. [Google Scholar] [CrossRef]
- Lukose, B.; Kuc, A.; Heine, T. The Structure of Layered Covalent-Organic Frameworks. Chem. Eur. J. 2011, 17, 2388–2392. [Google Scholar] [CrossRef] [PubMed]
- Pütz, A.M.; Terban, M.W.; Bette, S.; Haase, F.; Dinnebier, R.E.; Lotsch, B. V. Total Scattering Reveals the Hidden Stacking Disorder in a 2D Covalent Organic Framework. Chem. Sci. 2020, 11, 12647–12654. [Google Scholar] [CrossRef]
- Kang, C.; Zhang, Z.; Usadi, A.K.; Calabro, D.C.; Baugh, L.S.; Yu, K.; Wang, Y.; Zhao, D. Aggregated Structures of Two-Dimensional Covalent Organic Frameworks. J. Am. Chem. Soc. 2022, 144, 3192–3199. [Google Scholar] [CrossRef]
- Emmerling, S.T.; Schuldt, R.; Bette, S.; Yao, L.; Dinnebier, R.E.; Kästner, J.; Lotsch, B. V. Interlayer Interactions as Design Tool for Large-Pore COFs. J. Am. Chem. Soc. 2021, 143, 15711–15722. [Google Scholar] [CrossRef] [PubMed]
- Wan, Y.; Wang, L.; Xu, H.; Wu, X.; Yang, J. A Simple Molecular Design Strategy for Two-Dimensional Covalent Organic Framework Capable of Visible-Light-Driven Water Splitting. J. Am. Chem. Soc. 2020, 142, 4508–4516. [Google Scholar] [CrossRef]
- Zhou, Z.-B.; Tian, P.-J.; Yao, J.; Lu, Y.; Qi, Q.-Y.; Zhao, X. Toward Azo-Linked Covalent Organic Frameworks by Developing Linkage Chemistry via Linker Exchange. Nat. Commun. 2022, 13, 2180. [Google Scholar] [CrossRef] [PubMed]
- Spitler, E.L.; Koo, B.T.; Novotney, J.L.; Colson, J.W.; Uribe-Romo, F.J.; Gutierrez, G.D.; Clancy, P.; Dichtel, W.R. A 2D Covalent Organic Framework with 4.7-nm Pores and Insight into Its Interlayer Stacking. J. Am. Chem. Soc. 2011, 133, 19416–19421. [Google Scholar] [CrossRef] [PubMed]
| Compound |
Average surface area (m2 g−1) |
CO2 uptake (mg g−1) |
| TPB-azo (AA)a | 1957 | 19 |
| TPB-azo (AB)a | 2462 | 65 |
| TPB-azoxy (AA)a | 1830 | 28 |
| TPB-azoxy (AB)a | 2284 | 73 |
| TPB-azodioxy (AA)a | 1691 | 31 |
| TPB-azodioxy (AB)a | 1949 | 118 |
| TPT-azo (AA)a | 1828 | 14 |
| TPT-azo (AB)a | 2411 | 92 |
| TPT-azoxy (AA)a | 1704 | 19 |
| TPT-azoxy (AB)a | 2077 | 73 |
| TPT-azodioxy (AA)a | 1596 | 21 |
| TPT-azodioxy (AB)a | 1635 | 44 |
| TPA-azo (AA)b | 1858 | 23 |
| TPA-azo (AB)b | 2327 | 153 |
| TPA-azo (AA’incl)b | 1395 | 46 |
| TPA-azo (AA’serr)b | 1911 | 28 |
| TPA-azoxy (AA)b | 1686 | 38 |
| TPA-azoxy (AB)b | 2051 | 169 |
| TPA-azodioxy (AA)b | 1501 | 48 |
| TPA-azodioxy (AB)b | 1570 | 177 |
| TPP-azo (AA)b | 1880 | 18 |
| TPP-azo (AB)b | 1894 | 31 |
| TPP-azo (AA’incl)b | 132 | 37 |
| TPP-azo (AA’serr)b | 1783 | 18 |
| TPP-azoxy (AA)b | 1588 | 21 |
| TPP-azoxy (AA’incl)b | 194 | 37 |
| TPP-azodioxy (AA)b | 1636 | 27 |
| TPP-azodioxy (AA’incl)b | 342 | 68 |
| AZO-Bc | 1957 | 20 |
| AZO-B-PPDc | 2237 | 19 |
| AZO-B-BZDc | 2306 | 19 |
| AZO-Tc | 1828 | 14 |
| AZO-T-PPDc | 2127 | 14 |
| AZO-T-BZDc | 2207 | 15 |
| APP-1d | 1969 | 13 |
| APP-2d | 2246 | 18 |
| APP-3d | 1931 | 15 |
| APP-4d | 1934 | 20 |
| APP-5d | 6777 | 49 |
| APP-6d | 7567 | 49 |
| APP-7d | 1332 | 23 |
| APP-8d | 1332 | 23 |
| APP-BP-OH (AA)e | 1750 | 25 |
| APP-BP-OH (AB)e | 2716 | 85 |
| APP-BP-OH (AA’ serr)e | 1894 | 213 |
| APP-AQe | 1699 | 23 |
| APP-BP-Mee | 1805 | 20 |
| APP-Ph-Mee | 1660 | 23 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




