Submitted:
27 February 2026
Posted:
03 March 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Clinical Study
2.2. In Vitro Study
Cell Cultures and Treatments
2.3. Viability Assay
2.4. Proliferation Assay
2.5. Cytotoxicity Assay
2.6.Western Blot
2.7. Seahorse Metabolic Assay
2.8. Statistical Analysis
3. Results
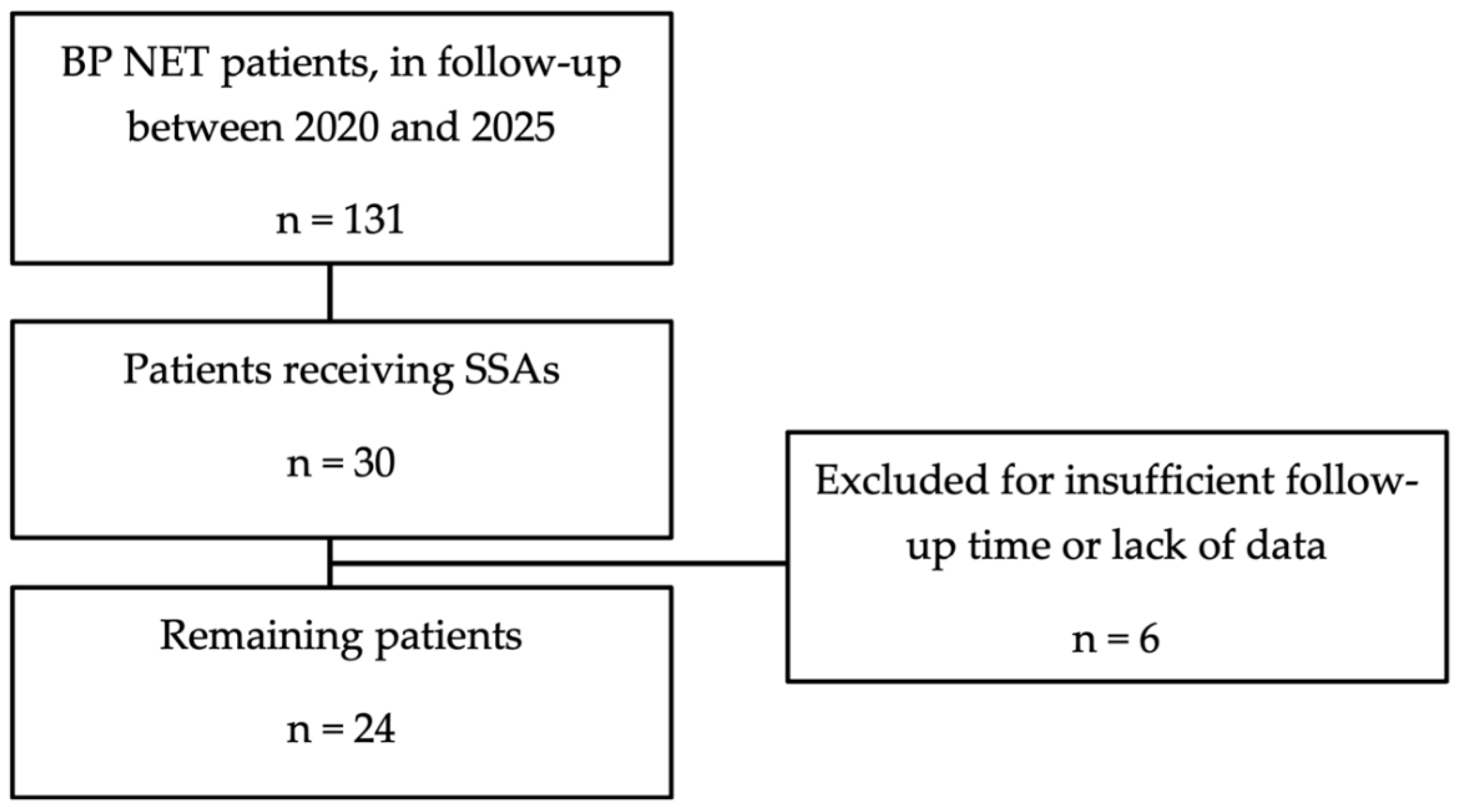
3.1. Clinical Investigation
3.1.1. Patients’ Characteristics
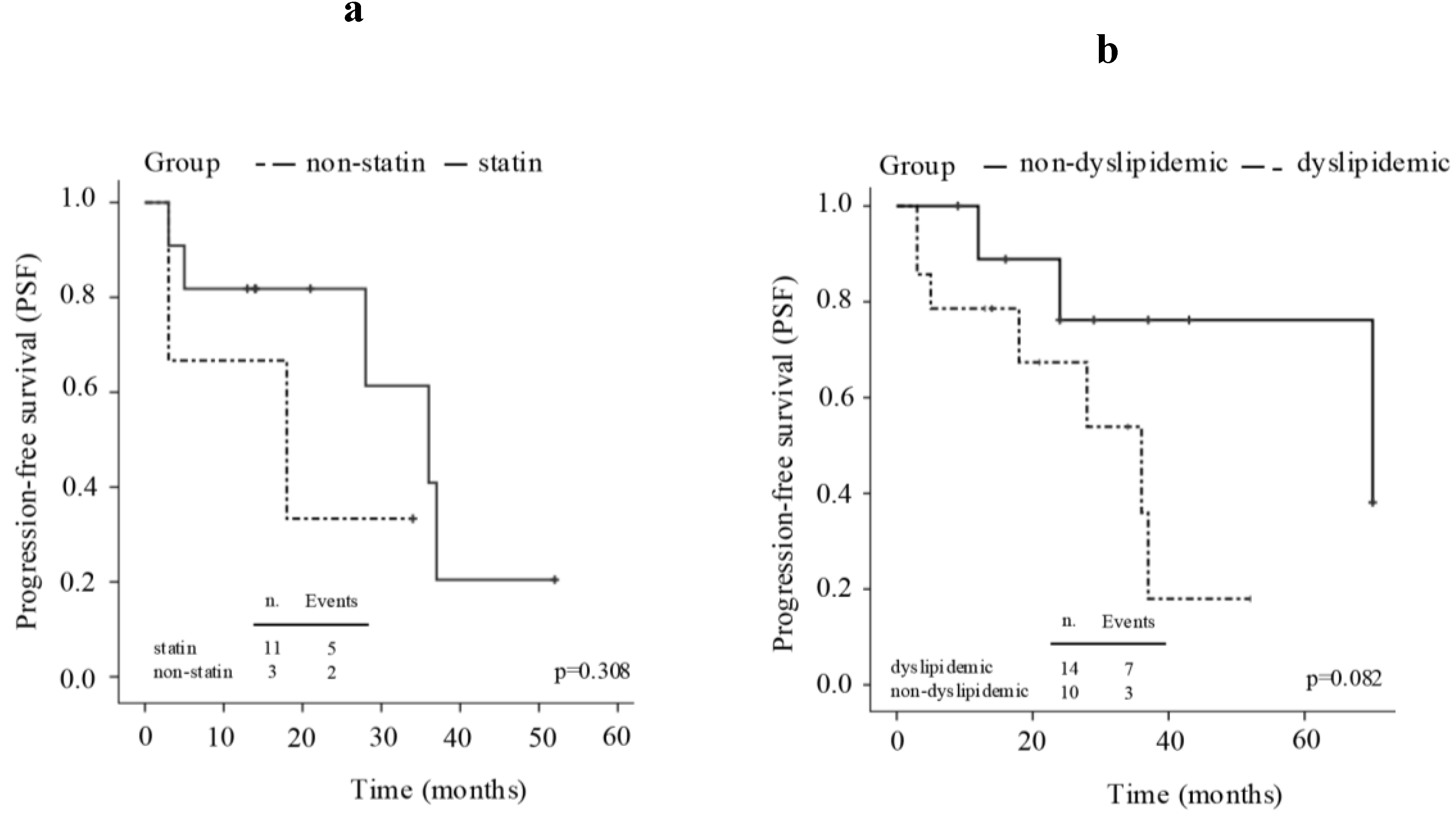
3.1.2. Clinical Outcome
3.2. In Vitro Investigations
3.2.1. Clinical Outcome Combined Lanreotide-Atorvastatin Treatment Reduces Cell Viability
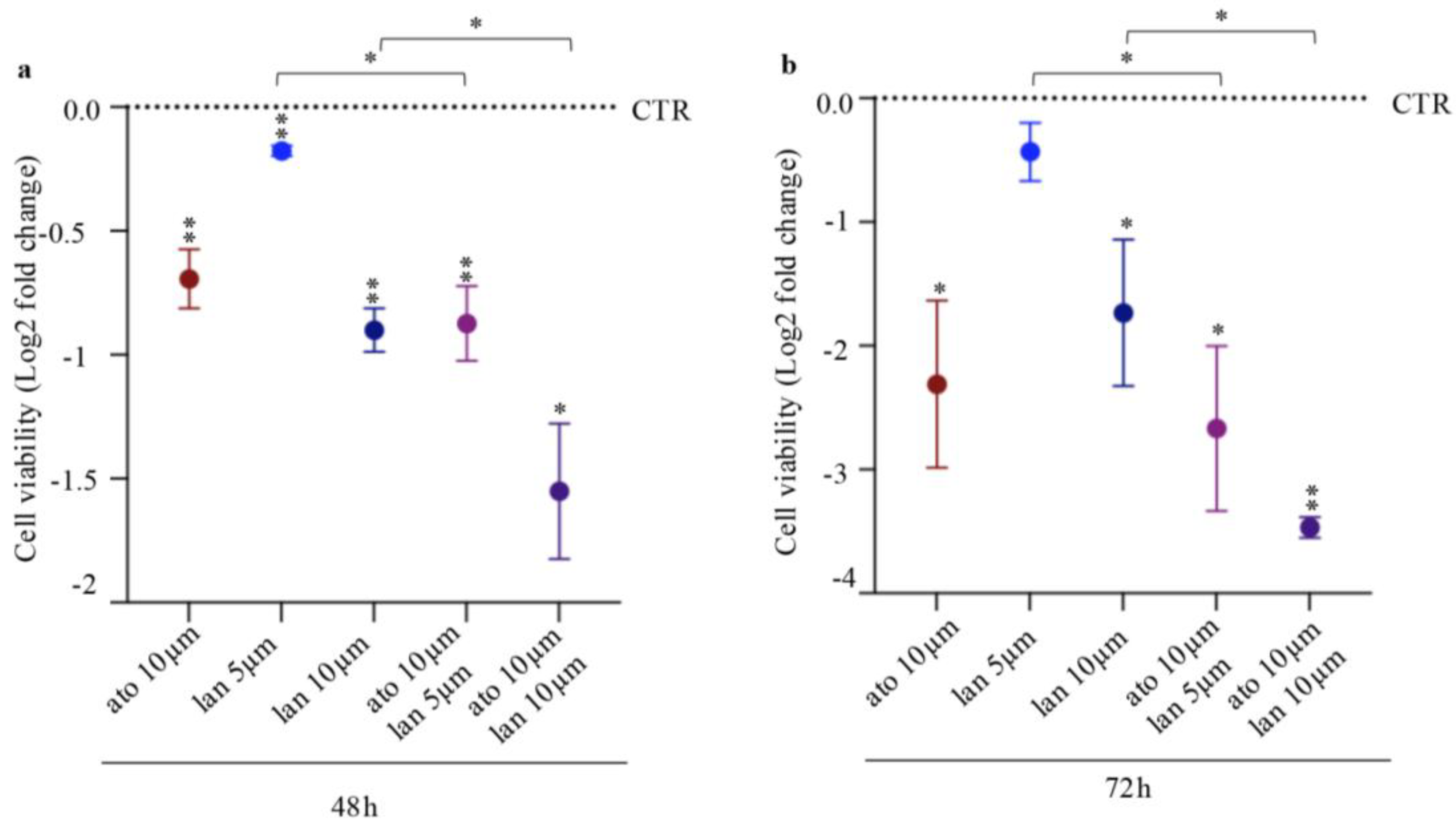
3.2.2. Reduced Proliferation in combined Lanreotide-Atorvastatin Treated Cells
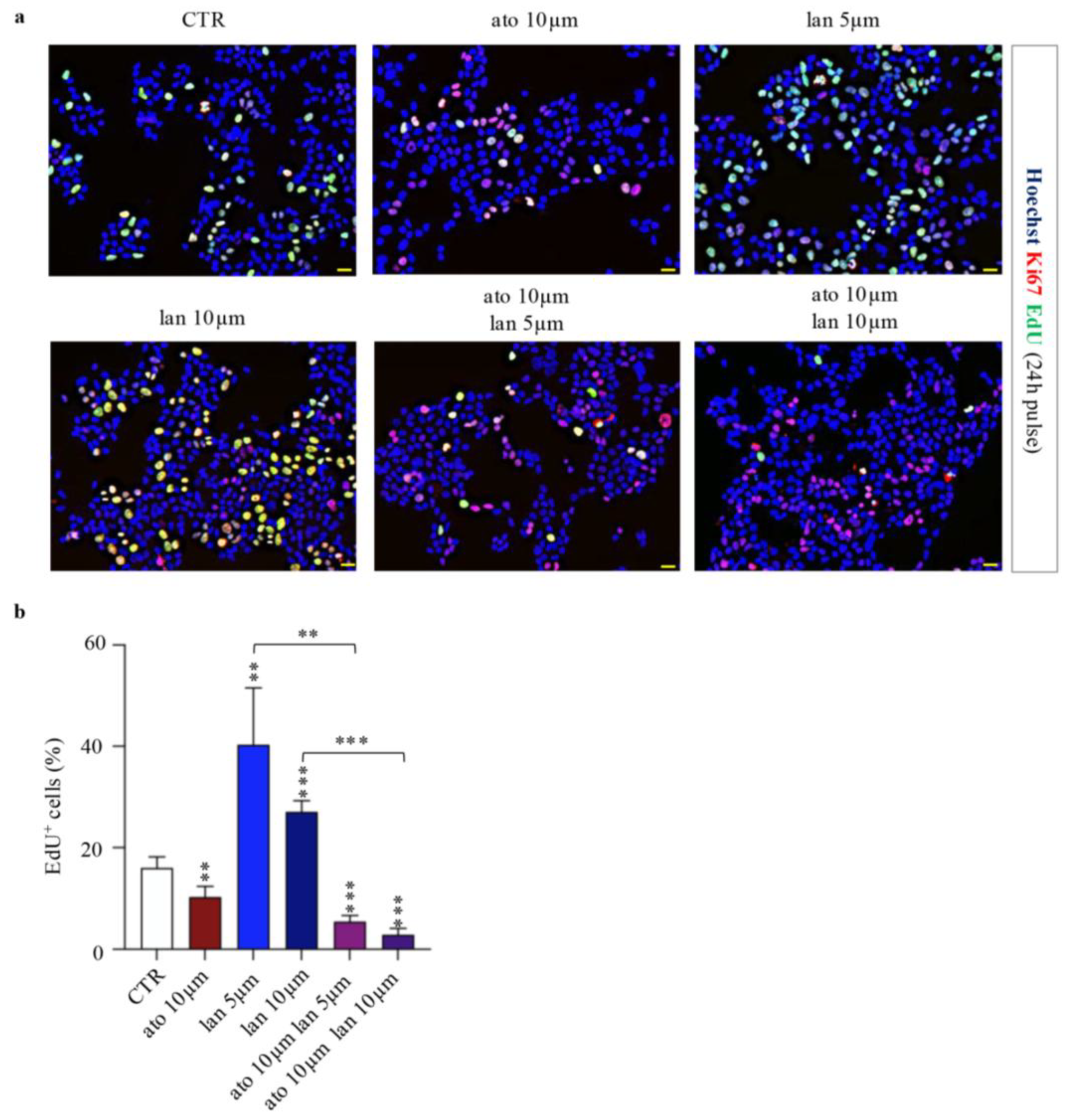
3.2.3. Enhanced Cell Death in Combined Lanreotide-Atorvastatin Treated Cells
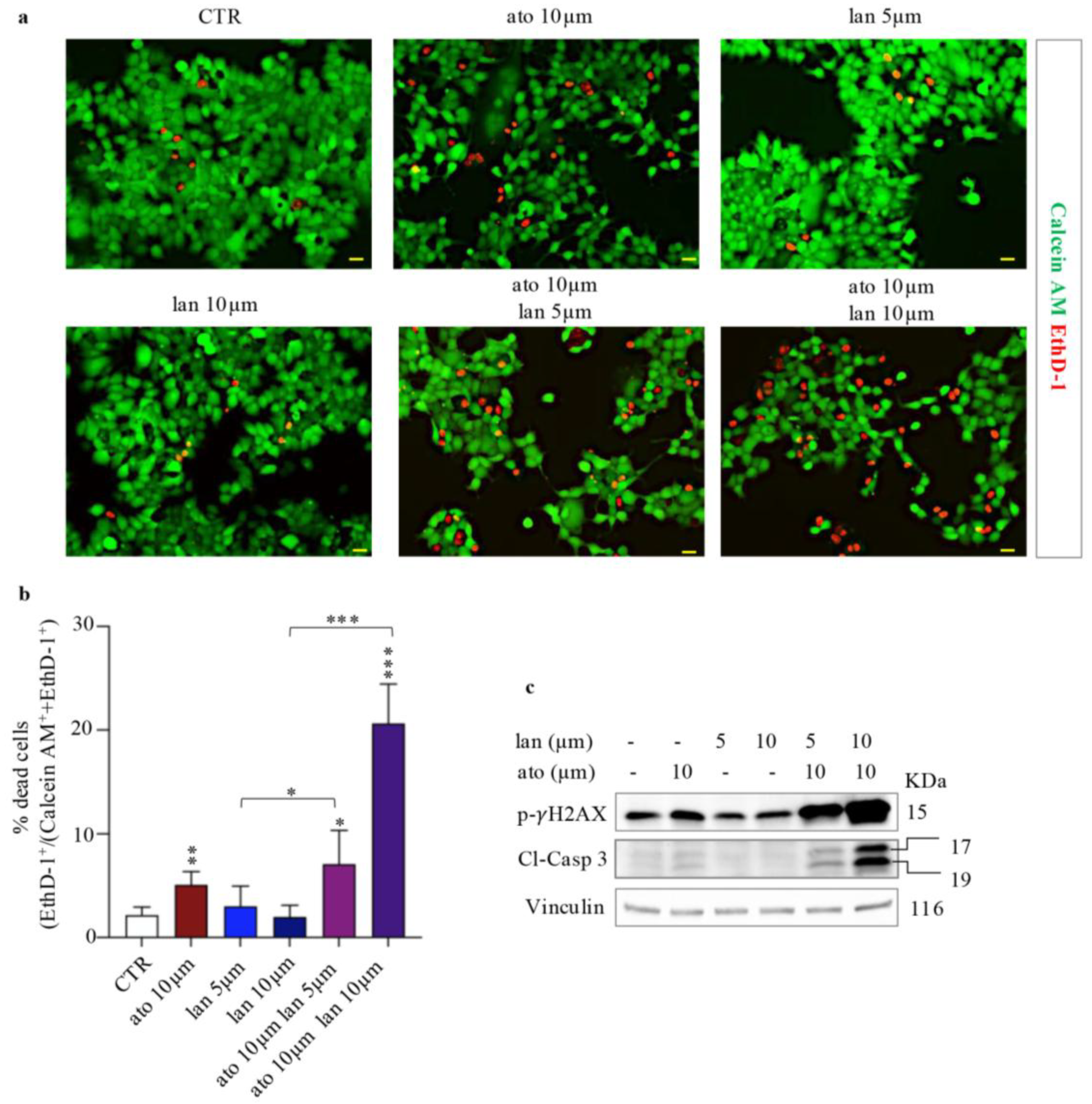
3.2.4. Impact of Lanreotide-Atorvastatin Treatment on Mitochondrial and Glycolytic ATP Production
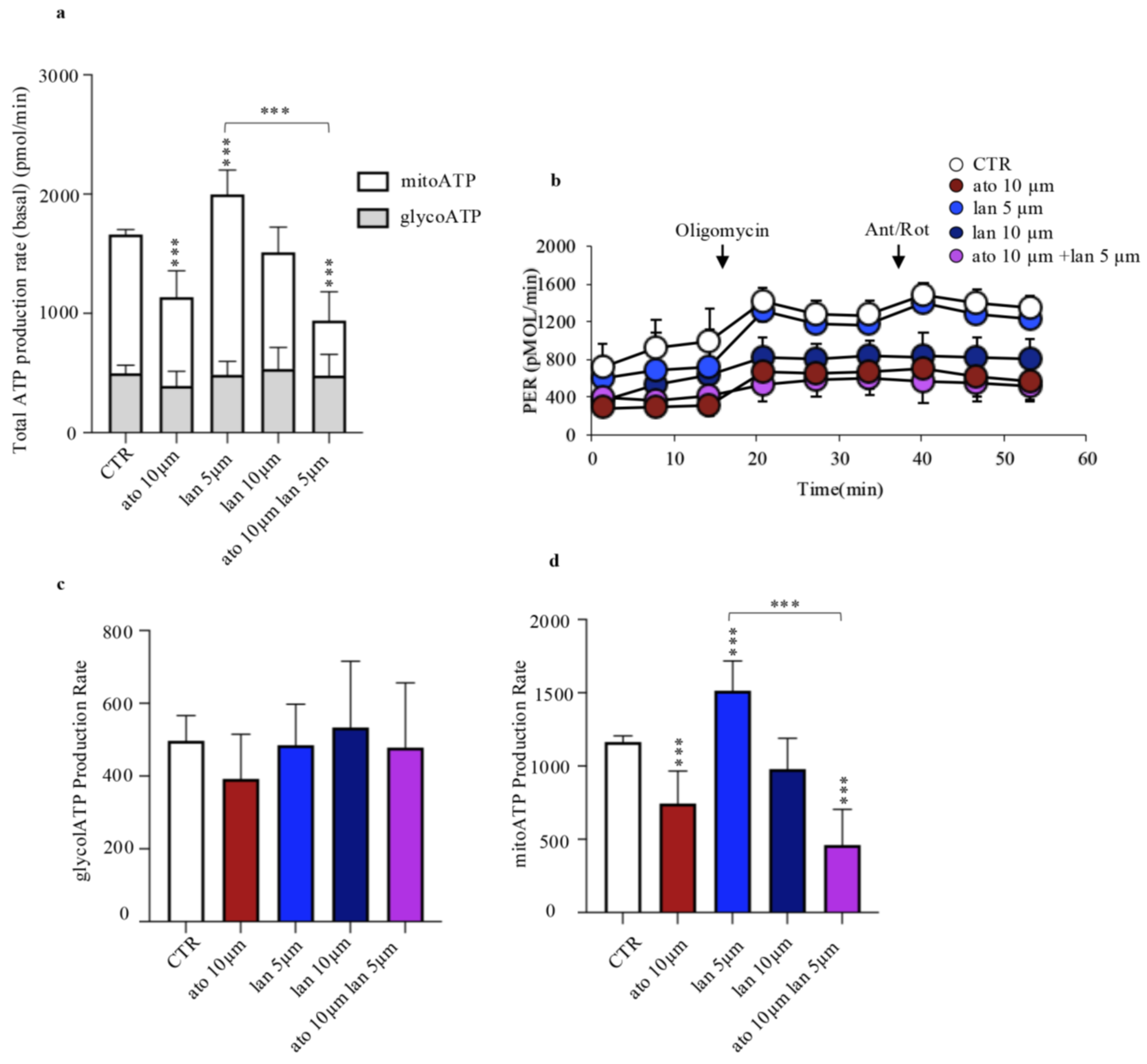
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| NETs | Neuroendocrine tumors |
| GEP | Gastroenteropancreatic |
| BP | Bronchopulmonary |
| SSTRs | Linear dichroism |
| PRRT | Peptide Receptor Radionuclide Therapy |
| PFS | Progression Free Survival |
| SSAs | Somatostatin Analogs |
| ATCC | American Type Culture Collection |
| OCR | Oxygen Consumption Rates |
| ECAR | Extracellular Acidification Rates |
| ATO | atorvastatin |
| LAN | lanreotide |
References
- Rindi, G; Mete, O; Uccella, S; Basturk, O; La Rosa, S; Brosens, LAA; et al. Overview of the 2022 WHO Classification of Neuroendocrine Neoplasms. Endocr Pathol 2022, 33(1), 115–54. [Google Scholar] [CrossRef]
- Dasari, A; Wallace, K; Halperin, DM; Maxwell, J; Kunz, P; Singh, S; et al. Epidemiology of Neuroendocrine Neoplasms in the US. JAMA Netw Open 2025, 8(6), e2515798. [Google Scholar]
- Davini, F; Gonfiotti, A; Comin, C; Caldarella, A; Mannini, F; Janni, A. Typical and atypical carcinoid tumours: 20-year experience with 89 patients. J Cardiovasc Surg (Torino) 2009, 50(6), 807–11. [Google Scholar]
- Faggiano, A. Long-acting somatostatin analogs and well differentiated neuroendocrine tumors: a 20-year-old story. J Endocrinol Invest. 2023, 47(1), 35–46. [Google Scholar]
- Rinke, A; Müller, H-H; Schade-Brittinger, C; Klose, K-J; Barth, P; Wied, M; et al. Placebo-Controlled, Double-Blind, Prospective, Randomized Study on the Effect of Octreotide LAR in the Control of Tumor Growth in Patients with Metastatic Neuroendocrine Midgut Tumors: A Report From the PROMID Study Group. Journal of Clinical Oncology 2009, 27(28), 4656–63. [Google Scholar] [CrossRef]
- Cives, M; Strosberg, JR. Gastroenteropancreatic Neuroendocrine Tumors. CA Cancer J Clin. 2018, 68(6), 471–87. [Google Scholar]
- Fazio, N; Buzzoni, R; Delle Fave, G; Tesselaar, ME; Wolin, E; Van Cutsem, E; et al. Everolimus in advanced, progressive, well-differentiated, non-functional neuroendocrine tumors: RADIANT -4 lung subgroup analysis. Cancer Sci. 2018, 109(1), 174–81. [Google Scholar] [CrossRef] [PubMed]
- Faggiano, A; Malandrino, P; Modica, R; Agrimi, D; Aversano, M; Bassi, V; et al. Efficacy and Safety of Everolimus in Extrapancreatic Neuroendocrine Tumor: A Comprehensive Review of Literature. Oncologist 2016, 21(7), 875–86. [Google Scholar]
- Abdalla, TSA; Klinkhammer-Schalke, M; Zeissig, SR; Tol, KK; Honselmann, KC; Braun, R; et al. Prognostic factors after resection of locally advanced non-functional pancreatic neuroendocrine neoplasm: an analysis from the German Cancer Registry Group of the Society of German Tumor Centers. J Cancer Res Clin Oncol. 2023, 149(11), 8535–43. [Google Scholar] [CrossRef] [PubMed]
- Marciello, F; Mercier, O; Ferolla, P; Scoazec, J-Y; Filosso, PL; Chapelier, A; et al. Natural History of Localized and Locally Advanced Atypical Lung Carcinoids after Complete Resection: A Joined French Italian Retrospective Multicenter Study. Neuroendocrinology 2018, 106(3), 264–73. [Google Scholar] [CrossRef] [PubMed]
- Vasseur, S; Guillaumond, F. Lipids in cancer: a global view of the contribution of lipid pathways to metastatic formation and treatment resistance. Oncogenesis 2022, 11(1), 46. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F. Dysregulated lipid metabolism in cancer. World J Biol Chem. 2012, 3(8), 167. [Google Scholar]
- Modica, R; La Salvia, A; Liccardi, A; Cozzolino, A; Di Sarno, A; Russo, F; et al. Dyslipidemia, lipid-lowering agents and neuroendocrine neoplasms: new horizons. Endocrine 2024, 85(2), 520–31. [Google Scholar] [CrossRef]
- Modica, R; La Salvia, A; Liccardi, A; Cannavale, G; Minotta, R; Benevento, E; et al. Lipid Metabolism and Homeostasis in Patients with Neuroendocrine Neoplasms: From Risk Factor to Potential Therapeutic Target. Metabolites 2022, 12(11), 1057. [Google Scholar] [CrossRef]
- Gallo, M; Muscogiuri, G; Pizza, G; Ruggeri, RM; Barrea, L; Faggiano, A; et al. The management of neuroendocrine tumours: A nutritional viewpoint. Crit Rev Food Sci Nutr. 2019, 59(7), 1046–57. [Google Scholar]
- Pyo, JH; Hong, SN; Min, B-H; Lee, JH; Chang, DK; Rhee, P-L; et al. Evaluation of the risk factors associated with rectal neuroendocrine tumors: a big data analytic study from a health screening center. J Gastroenterol. 2016, 51(12), 1112–21. [Google Scholar] [CrossRef]
- Duarte, JA; de Barros, ALB; Leite, EA. The potential use of simvastatin for cancer treatment: A review. Biomedicine & Pharmacotherapy 2021, 141, 111858. [Google Scholar] [CrossRef]
- Beckwitt, CH; Brufsky, A; Oltvai, ZN; Wells, A. Statin drugs to reduce breast cancer recurrence and mortality. Breast Cancer Research 2018, 20(1), 144. [Google Scholar] [CrossRef] [PubMed]
- Herrera-Martínez, AD; Pedraza-Arevalo, S; L-López, F; Gahete, MD; Gálvez-Moreno, MA; Castaño, JP; et al. Type 2 Diabetes in Neuroendocrine Tumors: Are Biguanides and Statins Part of the Solution? J Clin Endocrinol Metab. 2019, 104(1), 57–73. [Google Scholar] [CrossRef]
- Pusceddu, S; Vernieri, C; Di Maio, M; Marconcini, R; Spada, F; Massironi, S; et al. Metformin Use Is Associated With Longer Progression-Free Survival of Patients With Diabetes and Pancreatic Neuroendocrine Tumors Receiving Everolimus and/or Somatostatin Analogues. Gastroenterology 2018, 155(2), 479–489.e7. [Google Scholar] [CrossRef] [PubMed]
- Pusceddu, S; Vernieri, C; Prinzi, N; Torchio, M; Coppa, J; Antista, M; et al. The potential role of metformin in the treatment of patients with pancreatic neuroendocrine tumors: a review of preclinical to clinical evidence. Therap Adv Gastroenterol 2020, 13. [Google Scholar] [CrossRef]
- Faggiano, A; Russo, F; Zamponi, V; Sesti, F; Puliani, G; Modica, R; et al. Impact of dyslipidemia and lipid-lowering therapy with statins in patients with neuroendocrine tumors. J Neuroendocrinol. 2025, 37(2). [Google Scholar] [CrossRef]
- Koyuturk, M; Ersoz, M; Altiok, N. Simvastatin induces apoptosis in human breast cancer cells: p53 and estrogen receptor independent pathway requiring signalling through JNK. Cancer Lett. 2007, 250(2), 220–8. [Google Scholar] [CrossRef]
- Bjarnadottir, O; Romero, Q; Bendahl, P-O; Jirström, K; Rydén, L; Loman, N; et al. Targeting HMG-CoA reductase with statins in a window-of-opportunity breast cancer trial. Breast Cancer Res Treat 2013, 138(2), 499–508. [Google Scholar] [CrossRef] [PubMed]
- Nölting, S; Maurer, J; Spöttl, G; Aristizabal Prada, ET; Reuther, C; Young, K; et al. Additive Anti-Tumor Effects of Lovastatin and Everolimus In Vitro through Simultaneous Inhibition of Signaling Pathways. PLoS One 2015, 10(12), e0143830. [Google Scholar]
- Miyazawa, Y; Sekine, Y; Kato, H; Furuya, Y; Koike, H; Suzuki, K. Simvastatin Up-Regulates Annexin A10 That Can Inhibit the Proliferation, Migration, and Invasion in Androgen-Independent Human Prostate Cancer Cells. Prostate 2017, 77(4), 337–49. [Google Scholar]
- Vázquez-Borrego, MC; Fuentes-Fayos, AC; Herrera-Martínez, AD; Venegas-Moreno, E; L-López, F; Fanciulli, A; et al. Statins Directly Regulate Pituitary Cell Function and Exert Antitumor Effects in Pituitary Tumors. Neuroendocrinology 2020, 110(11–12), 1028–41. [Google Scholar] [CrossRef] [PubMed]
- Vernieri, C; Pusceddu, S; Fucà, G; Indelicato, P; Centonze, G; Castagnoli, L; et al. Impact of systemic and tumor lipid metabolism on everolimus efficacy in advanced pancreatic neuroendocrine tumors (pNETs). Int J Cancer 2019, 144(7), 1704–12. [Google Scholar] [CrossRef] [PubMed]
- Krug, S; Mordhorst, J-P; Moser, F; Theuerkorn, K; Ruffert, C; Egidi, M; et al. Correction: Interaction between somatostatin analogues and targeted therapies in neuroendocrine tumor cells. PLoS One 2020, 15(2), e0228905. [Google Scholar]
- von Hessert-Vaudoncourt, C; Lelek, S; Geisler, C; Hartung, T; Bröker, V; Briest, F; et al. Concomitant inhibition of PI3K/mTOR signaling pathways boosts antiproliferative effects of lanreotide in bronchopulmonary neuroendocrine tumor cells. Front Pharmacol 2024, 15. [Google Scholar] [CrossRef]
- Ungefroren, H; Künstner, A; Busch, H; Franzenburg, S; Luley, K; Viol, F; et al. Differential Effects of Somatostatin, Octreotide, and Lanreotide on Neuroendocrine Differentiation and Proliferation in Established and Primary NET Cell Lines: Possible Crosstalk with TGF-β Signaling. Int J Mol Sci. 2022, 23(24), 15868. [Google Scholar] [CrossRef]
- Abolghasemi, R; Ebrahimi-Barough, S; Bahrami, N; Ai, J. Atorvastatin Inhibits Viability and Migration of MCF7 Breast Cancer Cells. Asian Pac J Cancer Prev. 2022, 23(3), 867–75. [Google Scholar] [PubMed]
- Leoncini, E; Carioli, G; La Vecchia, C; Boccia, S; Rindi, G. Risk factors for neuroendocrine neoplasms: a systematic review and meta-analysis. Annals of Oncology 2016, 27(1), 68–81. [Google Scholar]
- Baudin, E; Caplin, M; Garcia-Carbonero, R; Fazio, N; Ferolla, P; Filosso, PL; et al. Lung and thymic carcinoids: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up☆. Annals of Oncology 2021, 32(4), 439–51. [Google Scholar] [CrossRef] [PubMed]
- Sidéris, L; Dubé, P; Rinke, A. Antitumor Effects of Somatostatin Analogs in Neuroendocrine Tumors. Oncologist 2012, 17(6), 747–55. [Google Scholar] [CrossRef] [PubMed]
- Michael, M; Garcia-Carbonero, R; Weber, MM; Lombard-Bohas, C; Toumpanakis, C; Hicks, RJ. The Antiproliferative Role of Lanreotide in Controlling Growth of Neuroendocrine Tumors: A Systematic Review. Oncologist 2017, 22(3), 272–85. [Google Scholar]
- Natalicchio, A; Marrano, N; Montagnani, M; Gallo, M; Faggiano, A; Zatelli, M; et al. Glycemic control and cancer outcomes in oncologic patients with diabetes: an Italian Association of Medical Oncology (AIOM), Italian Association of Medical Diabetologists (AMD), Italian Society of Diabetology (SID), Italian Society of Endocrinology (SIE), Italian Society of Pharmacology (SIF) multidisciplinary critical view. J Endocrinol Invest. 2024, 47(12), 2915–28. [Google Scholar]
- Ben-Shmuel, S; Rostoker, R; Scheinman, EJ; LeRoith, D. Metabolic Syndrome, Type 2 Diabetes, and Cancer: Epidemiology and Potential Mechanisms 2015, pp 355–72.
- Sciammarella, C; Luce, A; Riccardi, F; Mocerino, C; Modica, R; Berretta, M; et al. Lanreotide Induces Cytokine Modulation in Intestinal Neuroendocrine Tumors and Overcomes Resistance to Everolimus. Front Oncol. 2020, 10, 1047. [Google Scholar] [CrossRef]
- Fotouhi, O; Kjellin, H; Larsson, C; Hashemi, J; Barriuso, J; Juhlin, CC; et al. Proteomics Suggests a Role for APC-Survivin in Response to Somatostatin Analog Treatment of Neuroendocrine Tumors. J Clin Endocrinol Metab. 2016, 101(10), 3616–27. [Google Scholar]
- Vitale, G; Lupoli, G; Guarrasi, R; Colao, A; Dicitore, A; Gaudenzi, G; et al. Interleukin-2 and Lanreotide in the Treatment of Medullary Thyroid Cancer: In Vitro and In Vivo Studies. J Clin Endocrinol Metab. 2013, 98(10), E1567–74. [Google Scholar] [CrossRef]
- Friedman, R. Drug resistance in cancer: molecular evolution and compensatory proliferation. Oncotarget 2016, 7(11), 11746–55. [Google Scholar] [CrossRef] [PubMed]
- Amin, F; Fathi, F; Reiner, Ž; Banach, M; Sahebkar, A. The role of statins in lung cancer. Archives of Medical Science 2021. [Google Scholar] [CrossRef] [PubMed]
- Safari, M; Scotto, L; Litman, T; Petrukhin, LA; Zhu, H; Shen, M; et al. Novel Therapeutic Strategies Exploiting the Unique Properties of Neuroendocrine Neoplasms. Cancers (Basel) 2023, 15(20), 4960. [Google Scholar] [CrossRef] [PubMed]
| Patients’ characteristics |
Total (n=24) |
Non-dyslipidemic (n=10) |
Dyslipidemic (n=14) |
p-value |
Dyslipidemic without statins (n=3) |
Dyslipidemic with statins (n=11) |
p-value |
| Sex, No. (%) | |||||||
| Male | 7 (29.2%) | 3 | 4 | 1.00 | 1 | 3 | 0.837 |
| Female | 17 (70.8%) | 7 | 10 | 2 | 8 | ||
| Histology, No. (%) | |||||||
| Typical | 13 (54.2%) | 5 | 8 | 0.917 | 2 | 6 | 0.727 |
| Atypical | 7 (29.2%) | 3 | 4 | 1 | 3 | ||
| NOS | 4 (16.7%) | 2 | 2 | 0 | 2 | ||
| Tumor stage, No. (%)* | |||||||
| Loco-regional | 10 (41.7%) | 6 | 4 | 0.124 | 1 | 8 | 0.207 |
| Distant metastases | 14 (58.3%) | 4 | 10 | 2 | 3 | ||
| Ki67, No. (%) | |||||||
| ≤3% | 7 (29.2%) | 4 | 3 | 0.556 | * | 3 | |
| 4-20% | 13 (54.1%) | 6 | 7 | * | 7 | ||
| ≥21% | 1 (4%) | 0 | 1 | 1 | 0 | ||
| Somatostatin analogue, No. (%) | |||||||
| Octreotide LAR | 8 (33.3%) | 3 | 5 | 0.770 | 0 | 5 | 0.145 |
| Lanreotide | 16 (66.6%) | 7 | 9 | 3 | 6 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




