Submitted:
01 March 2026
Posted:
02 March 2026
You are already at the latest version
Abstract
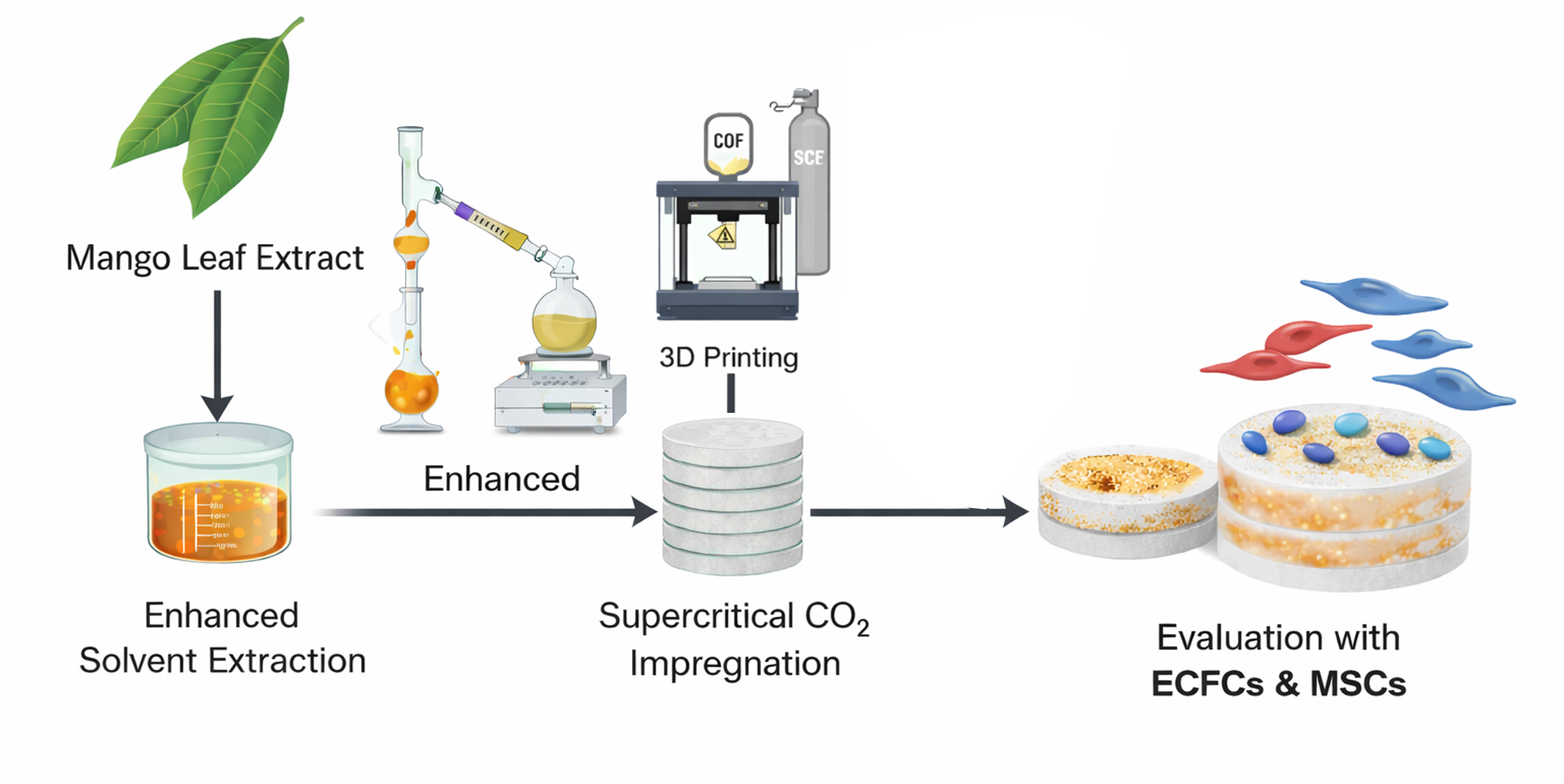
Poly(lactic acid) (PLA) devices can be functionalized with plant derived bioactives to introduce antioxidant activity while maintaining manufacturability and cytocompatibility. Here, a polyphenol rich mango leaf extract (MLE) was obtained by enhanced solvent extraction and incorporated into PLA using supercritical carbon dioxide assisted impregnation. Two manufacturing sequences were compared: impregnation after three dimensional (3D) printing of discs and impregnation of filaments prior to printing. Extract yield and radical scavenging capacity were quantified, and impregnation efficiency was assessed as a function of pressure and temperature. Biological performance was evaluated using adipose tissue derived endothelial colony forming cells (ECFCs) and adipose tissue derived mesenchymal stromal cells (MSCs), cultured separately and in co culture on functionalized substrates. Impregnation after printing provided higher and more reproducible loading while preserving disc geometry, whereas impregnation before printing promoted swelling and printing associated deformation that compromised structural fidelity. Cell based analyses supported improved adhesion, spatial distribution and proliferative status on discs produced by impregnation after printing under low temperature and high pressure conditions, without evidence of selective loss of either population in co culture by flow cytometry. These results support post print supercritical impregnation as a robust route to generate antioxidant, cell supportive PLA scaffolds from agricultural by products with potential relevance for vascular oriented biomedical applications.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Raw Material and Chemicals
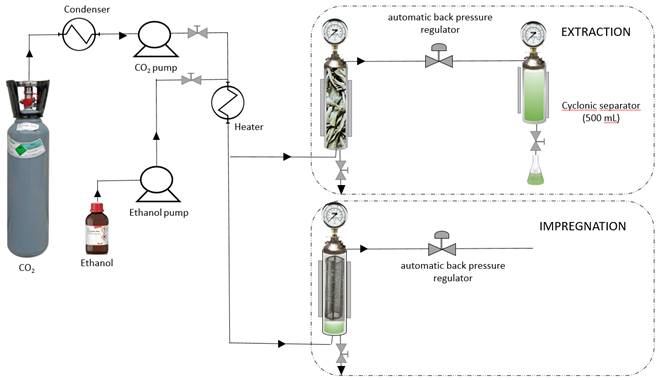
2.2. Enhance Solvent Extraction (ESE)
2.3. Characterization of the Extracts
2.4. 3D Printing
2.5. Impregnation at High Pressure (SCI)
2.6. Characterization of the Impregnated Polymers
2.7. Isolation and Culture of ECFCs and MSCs
2.8. Cell Viability
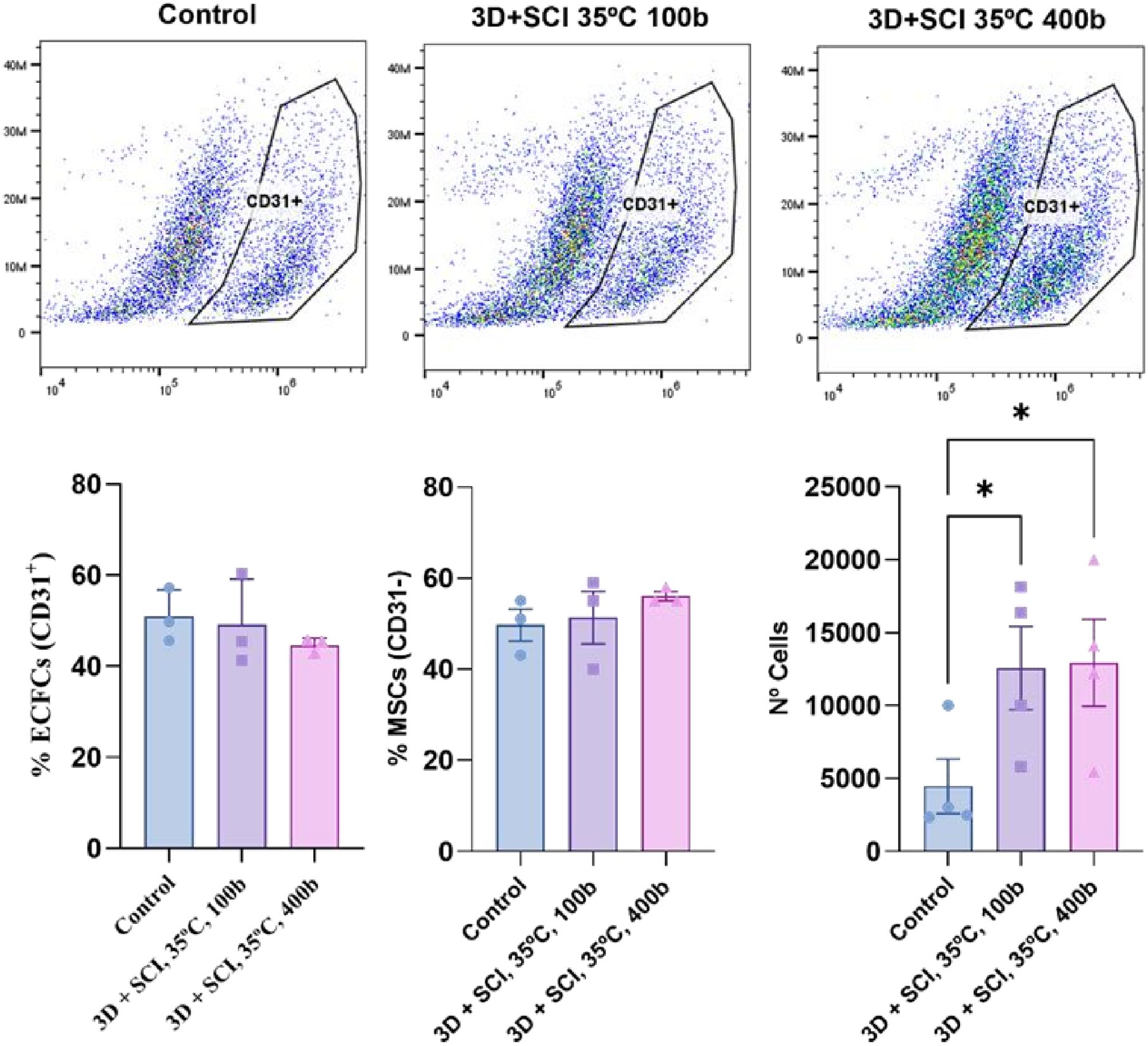
2.9. Flow Cytometry Based Quantification of ECFCs and MSCs
2.10. Proliferation Assay
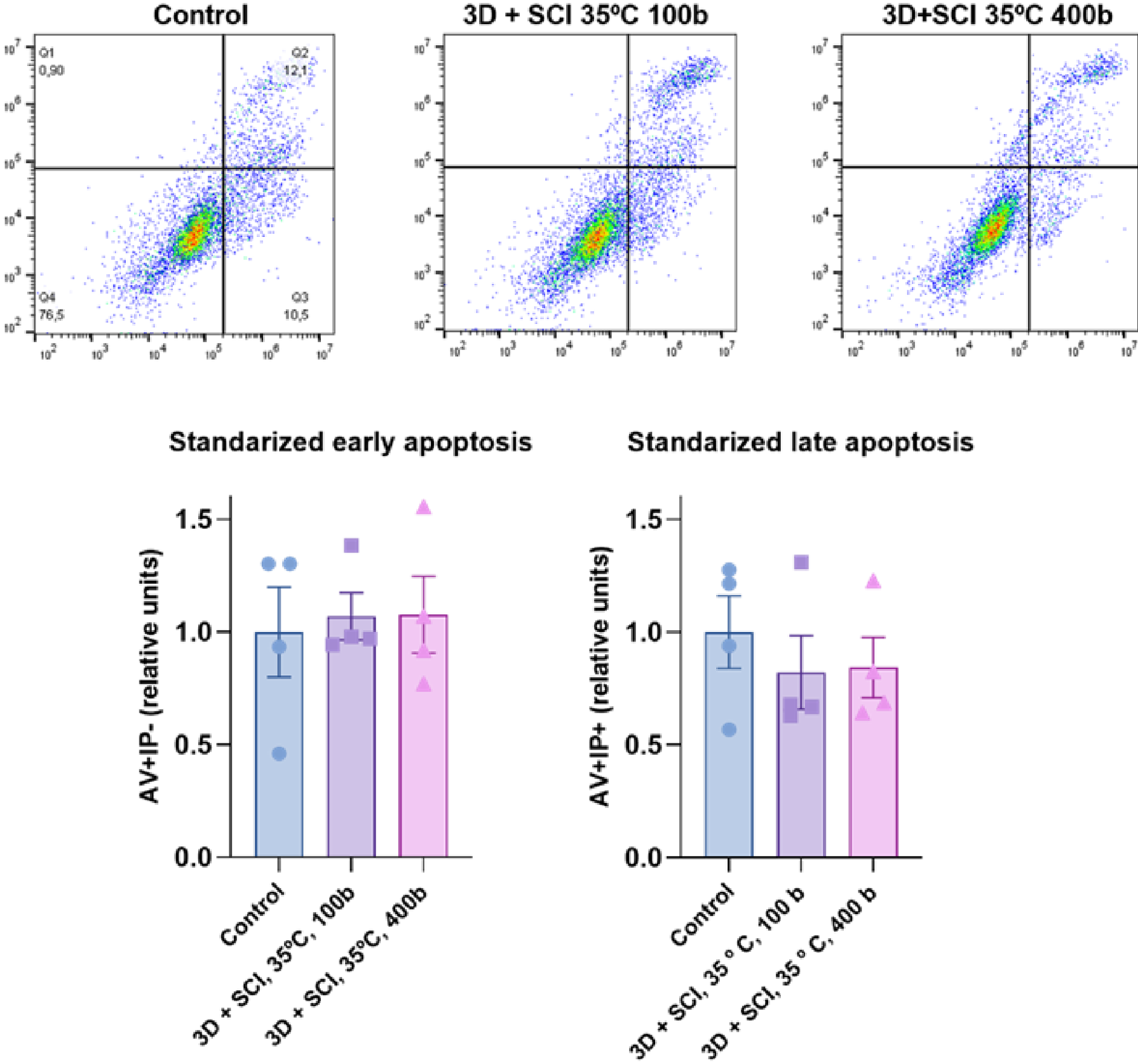
2.11. Apoptosis Assay
2.12. Statistical Analysis
3. Results and Discussion
3.1. Characterization of Mango Leaf Extract
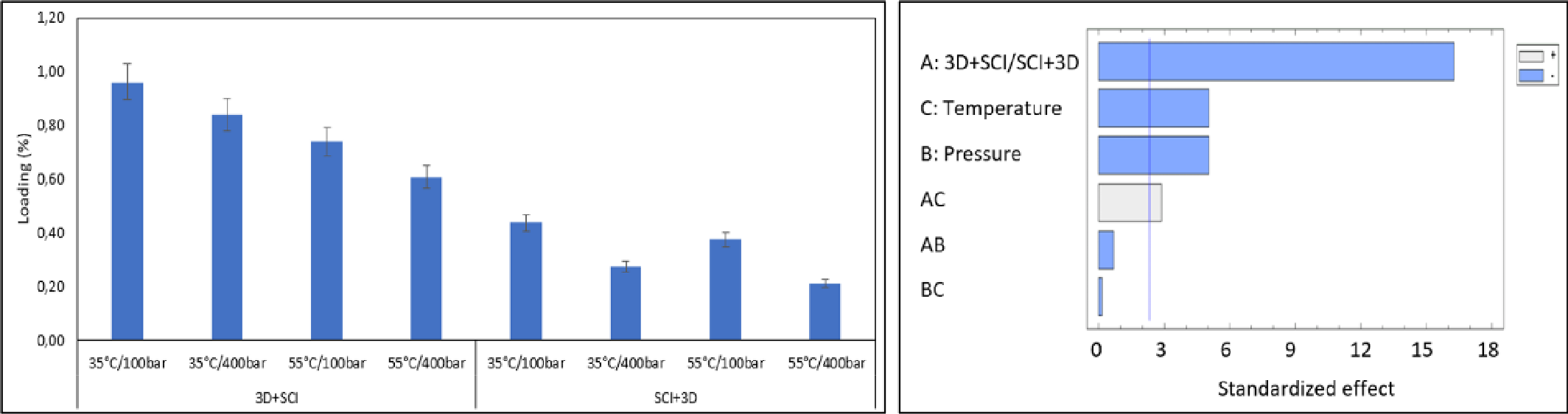
3.2. Supercritical Impregnation
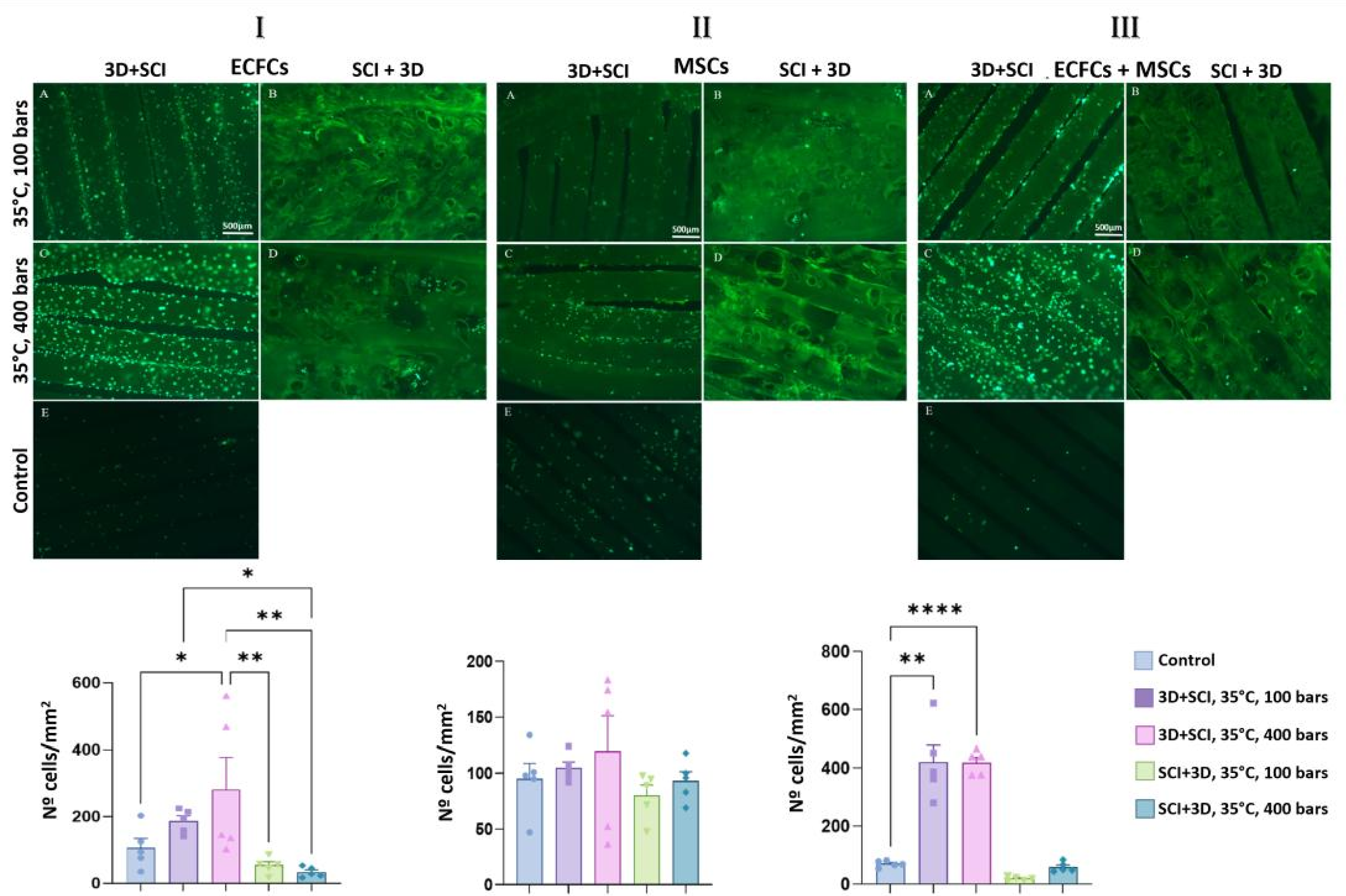
3.3. Cell Viability
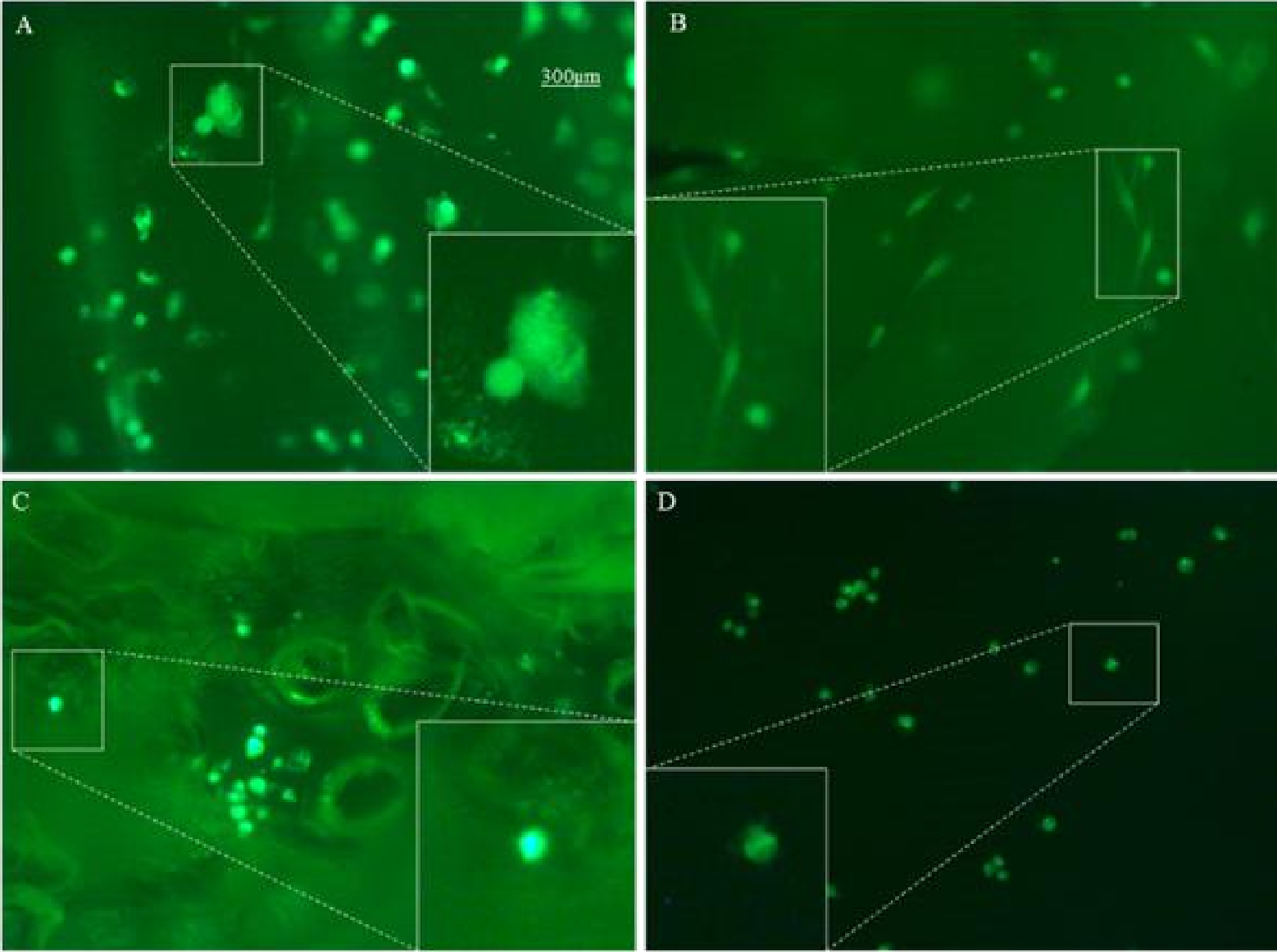
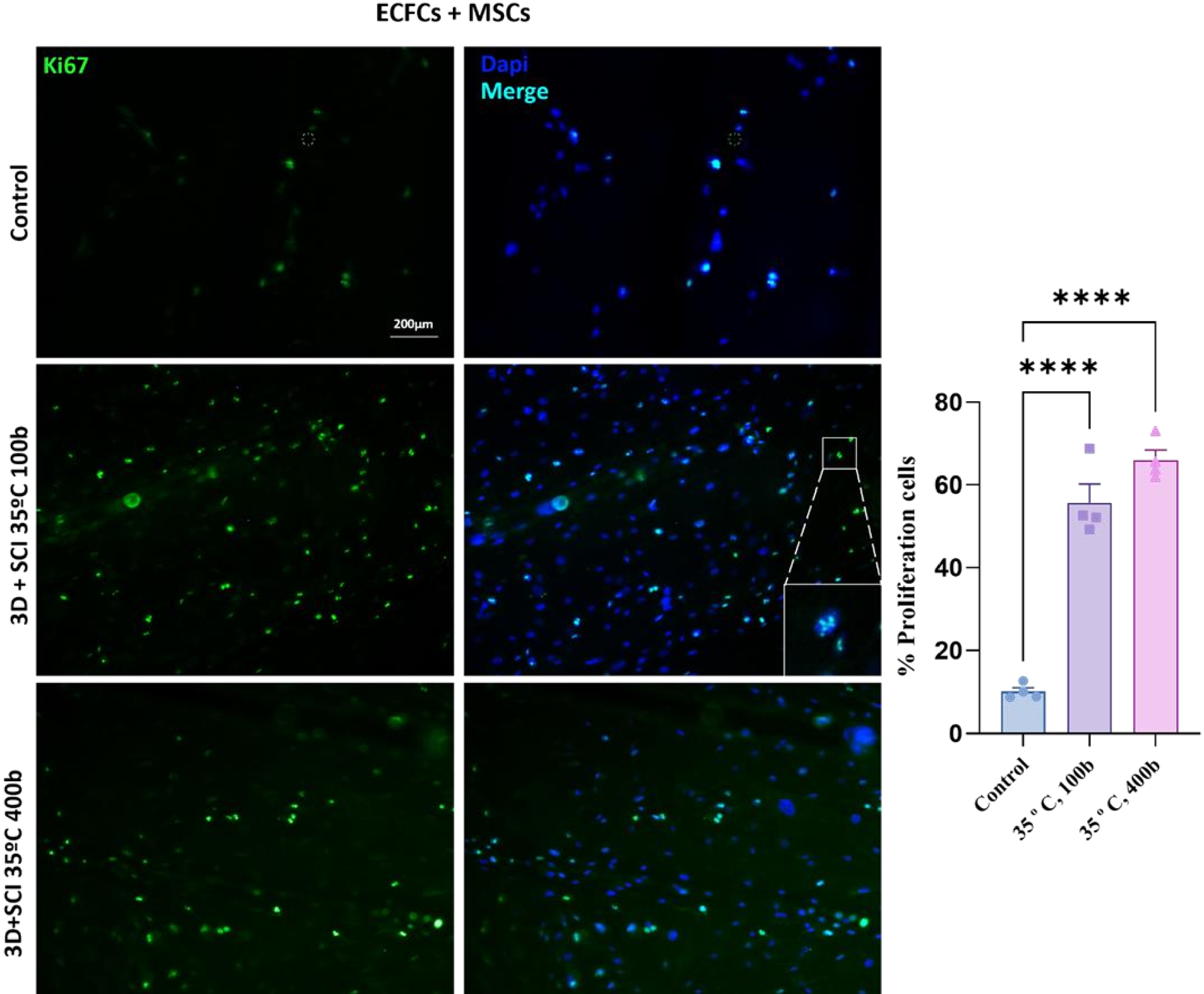
3.4. Proliferation, Cell Type Identification, and Apoptosis Assays
4. Conclusions
Author Contributions
Funding
Informed Consent Statement
Conflicts of Interest
Abbreviations
| AAI | Antioxidant activity index |
| AN V | Annexin V |
| CO2 | Carbon dioxide |
| DAPI | 4′,6 diamidino 2 phenylindole |
| DPPH | 2,2 diphenyl 1 picrylhydrazyl |
| ECFCs | Endothelial colony forming cells |
| EGM 2 | Endothelial Growth Medium 2 |
| FBS | Fetal bovine serum |
| GPS | Glutamine penicillin streptomycin |
| IC50 | Half maximal inhibitory concentration |
| MLE | Mango leaf extract |
| MSCs | Mesenchymal stromal cells |
| PBS | Phosphate buffered saline |
| PBT | PBS with Triton X 100 |
| PI | Propidium iodide |
| PLA | Poly(lactic acid) |
| PFA | Paraformaldehyde |
| SCI | Supercritical impregnation |
References
- Ulery, B.D.; Nair, L.S.; Laurencin, C.T. Biomedical Applications of Biodegradable Polymers. J. Polym. Sci. B Polym. Phys. 2011, 49.
- Tabernero, A.; González-Garcinuño, Á.; Cardea, S.; Martín del Valle, E. Supercritical Carbon Dioxide and Biomedicine: Opening the Doors towards Biocompatibility. Chemical Engineering Journal 2022, 444, 136615. [CrossRef]
- Liu, S.; Wu, G.; Zhang, X.; Yu, J.; Liu, M.; Zhang, Y.; Wang, P.; Yin, X.; Zhang, J.; Li, F.; et al. Preparation and Properties of Poly (Lactic Acid) (PLA) Suture Loaded with PLA Microspheres Enclosed Drugs (PM-Ds). Journal of the Textile Institute 2019, 110, 1596–1605. CTYPE:STRING:JOURNAL. [CrossRef]
- Luo, G.; Zhang, Y.; Chen, P.; Wu, F.; Shi, M.; Ma, Y.; Wang, X. Tailoring Osteo-Immunomodulatory Micro-Environments via a Bioactive 3D PLA Scaffold to Potentiate Regenerative Healing. Colloids Surf. B Biointerfaces 2025, 253, 114711. [CrossRef]
- Liu, Q.; Bi, C.; Hu, H.; Zhang, Z.; Zhang, B. Reduction of Amylose/Amylopectin Ratio Improves the Molecular Orientation and Performance of Three-Dimensional-Printed Thermoplastic Starch/Polylactic Acid Intestinal Stents. Int. J. Biol. Macromol. 2025, 308, 142419. [CrossRef]
- Faal, M.; Faal, M.; Ahmadi, T.; Dehgan, F. Fabrication and Evaluation of Polylactic Acid-Curcumin Containing Carbon Nanotubes (CNTs) Wound Dressing Using Electrospinning Method with Experimental and Computational Approaches. Scientific Reports 2025 15:1 2025, 15, 13398-. [CrossRef]
- Shi, Y.; Li, D.; Hill, C.; Yang, L.; Sheerin, E.D.; Pilliadugula, R.; Wang, J.J.; Boland, J.; Xiao, L. Micro and Nano Plastics Release from a Single Absorbable Suture into Simulated Body Fluid. J. Hazard. Mater. 2024, 466, 133559. [CrossRef]
- Seegobin, N.; Abdalla, Y.; Li, G.; Murdan, S.; Shorthouse, D.; Basit, A.W. Optimising the Production of PLGA Nanoparticles by Combining Design of Experiment and Machine Learning. Int. J. Pharm. 2024, 667, 124905. [CrossRef]
- Tong, L.; Shi, G.; Liu, Q.; Qian, Z.; Li, J.; Zhang, K.; Zhu, Y.; Fang, Y.; Sha, L.; Bai, L.; et al. Fabrication and Evaluation of 3D Printed PLGA/NHA/GO Scaffold for Bone Tissue Engineering. Scientific Reports 2025 15:1 2025, 15, 12446-. [CrossRef]
- Lin, Y.; Zhao, L.; Jin, H.; Gu, Q.; Lei, L.; Fang, C.; Pan, X. Multifunctional Applications of Silk Fibroin in Biomedical Engineering: A Comprehensive Review on Innovations and Impact. Int. J. Biol. Macromol. 2025, 309, 143067. [CrossRef]
- Peiravi, M.; Sherafat, Z.; Sani, M.; Azarpira, N. In Vitro and in Vivo Assessment of 3D-Printed PCL/PLA/ZnO Nanocomposite Scaffolds for Osteoarthritis Treatment. Composites Communications 2025, 57, 102432. [CrossRef]
- Teng, Y.; Wang, X.; Song, L.; Yang, J.; Hou, S.; Lv, Q.; Jiang, L.; Guan, Y.; Shi, J. 3D Printed Polycaprolactone/Poly (L-Lactide-Co-ϵ-Caprolactone) Composite Ureteral Stent with Biodegradable and Antibacterial Properties. Biomedical Materials 2025, 20, 025026. [CrossRef]
- Demartis, S.; Picco, C.J.; Larrañeta, E.; Korelidou, A.; Islam, R.; Coulter, J.A.; Giunchedi, P.; Donnelly, R.F.; Rassu, G.; Gavini, E. Evaluating the Efficacy of Rose Bengal-PVA Combinations within PCL/PLA Implants for Sustained Cancer Treatment. Drug Delivery and Translational Research 2024 15:5 2024, 15, 1770–1785. [CrossRef]
- Obiweluozor, F.O.; Kayumov, M.; Kwak, Y.; Cho, H.J.; Park, C.H.; Park, J. kyu; Jeong, Y.J.; Lee, D.W.; Kim, D.W.; Jeong, I.S. Rapid Remodeling Observed at Mid-Term in-Vivo Study of a Smart Reinforced Acellular Vascular Graft Implanted on a Rat Model. Journal of Biological Engineering 2022 17:1 2023, 17, 1-. [CrossRef]
- Stramiello, J.A.; Mohammadzadeh, A.; Ryan, J.; Brigger, M.T. The Role of Bioresorbable Intraluminal Airway Stents in Pediatric Tracheobronchial Obstruction: A Systematic Review. Int. J. Pediatr. Otorhinolaryngol. 2020, 139, 110405. [CrossRef]
- Pandey, A.; Adama, N.; Adjallé, K.; Blais, J.F. Sustainable Applications of Polyhydroxyalkanoates in Various Fields: A Critical Review. Int. J. Biol. Macromol. 2022, 221, 1184–1201. [CrossRef]
- Ren, Z.W.; Wang, Z.Y.; Ding, Y.W.; Dao, J.W.; Li, H.R.; Ma, X.; Yang, X.Y.; Zhou, Z.Q.; Liu, J.X.; Mi, C.H.; et al. Polyhydroxyalkanoates: The Natural Biopolyester for Future Medical Innovations. Biomater. Sci. 2023, 11, 6013–6034. [CrossRef]
- Santhamoorthy, M.; Kim, S.C. A Review of the Development of Biopolymer Hydrogel-Based Scaffold Materials for Drug Delivery and Tissue Engineering Applications. Gels 2025, Vol. 11, 2025, 11. [CrossRef]
- Scaffaro, R.; Botta, L.; Sanfilippo, M.; Gallo, G.; Palazzolo, G.; Puglia, A.M. Combining in the Melt Physical and Biological Properties of Poly(Caprolactone) and Chlorhexidine to Obtain Antimicrobial Surgical Monofilaments. Applied Microbiology and Biotechnology 2012 97:1 2012, 97, 99–109. [CrossRef]
- Iqbal, J.; Gunn, J.; Serruys, P.W. Coronary Stents: Historical Development, Current Status and Future Directions. Br. Med. Bull. 2013, 106, 193–211. [CrossRef]
- Soluri, A.; Hui, A.; Jones, L. Delivery of Ketotifen Fumarate by Commercial Contact Lens Materials. Optom. Vis. Sci. 2012, 89, 1140–1149. [CrossRef]
- Davies, O.R.; Lewis, A.L.; Whitaker, M.J.; Tai, H.; Shakesheff, K.M.; Howdle, S.M. Applications of Supercritical CO2 in the Fabrication of Polymer Systems for Drug Delivery and Tissue Engineering. Adv. Drug Deliv. Rev. 2008, 60, 373–387. [CrossRef]
- Beckman, E.J. Supercritical and Near-Critical CO2 in Green Chemical Synthesis and Processing. J. Supercrit. Fluids 2004, 28, 121–191. [CrossRef]
- Dendoncker, K.; Putzeys, G.; Nieuwenhuizen, T.; Voet, P.; Lambrecht, S.; Bertrand, M.; Valster, H.; Croes, K. Struggling with a Cefazolin Impregnation Protocol of Bone Chips. Cell and Tissue Banking 2025 26:2 2025, 26, 11-. [CrossRef]
- do Nascimento, U.S.; Neves, C.K.; Santos, E.F. dos; Ezegbe, C.A.; Rezende, L.R.; Marchi, J.; Lombello, C.B.; Maia-Obi, L.P. Ibuprofen Loaded Gelatin-Siloxane Gels Prepared by Supercritical CO2-Assisted Impregnation/Deposition for Controlled Drug Release and Tissue Engineering. J. Supercrit. Fluids 2025, 218, 106511. [CrossRef]
- Yamamoto, K.; Ushiki, I. Impregnation of Non-Steroidal Anti-Inflammatory Drugs (Ibuprofen and Ketoprofen) onto Mesoporous Silica SBA-15 Using Supercritical CO2. Journal of CO2 Utilization 2025, 92, 103022. [CrossRef]
- Erdogan, H.; Gungor, B.; Suner, S.S.; Silan, C.; Saraydin, S.U.; Saraydin, D.; Ayyala, R.S.; Sahiner, N. Moxifloxacin-Impregnated Contact Lenses for Treatment of Keratitis in Rabbit Eyes. Polym. Adv. Technol. 2025, 36, e70134. [CrossRef]
- Machado, N.D.; Cejudo-Bastante, C.; Goñi, M.L.; Gañán, N.A.; Casas-Cardoso, L.; Mantell-Serrano, C. Screening of the Supercritical Impregnation of Olea Europaea Leaves Extract into Filaments of Thermoplastic Polyurethane (TPU) and Polylactic Acid (PLA) Intended for Biomedical Applications. Antioxidants 2022, Vol. 11, 2022, 11. [CrossRef]
- Verano-Naranjo, L.; Cejudo-Bastante, C.; Casas, L.; Martínez de la Ossa, E.; Mantell, C. Use of Winemaking By-Products for the Functionalization of Polylactic Acid for Biomedical Applications. Antioxidants 2023, Vol. 12, 2023, 12. [CrossRef]
- Khumpook, T.; Saenphet, S.; Tragoolpua, Y.; Saenphet, K. Anti-Inflammatory and Antioxidant Activity of Thai Mango (Mangifera Indica Linn.) Leaf Extracts. Comparative Clinical Pathology 2018 28:1 2018, 28, 157–164. [CrossRef]
- Jiang, T.; Han, F.; Gao, G.; Liu, M. Mangiferin Exert Cardioprotective and Anti-Apoptotic Effects in Heart Failure Induced Rats. Life Sci. 2020, 249, 117476. [CrossRef]
- Sánchez-Gomar, I.; Benítez-Camacho, J.; Cejudo-Bastante, C.; Casas, L.; Moreno-Luna, R.; Mantell, C.; Durán-Ruiz, M.C. Pro-Angiogenic Effects of Natural Antioxidants Extracted from Mango Leaf, Olive Leaf and Red Grape Pomace over Endothelial Colony-Forming Cells. Antioxidants 2022, Vol. 11, 2022, 11. [CrossRef]
- Rosales, J.M.; Cejudo, C.; Verano, L.; Casas, L.; Mantell, C.; Martínez de la Ossa, E.J. Supercritical Impregnation of PLA Filaments with Mango Leaf Extract to Manufacture Functionalized Biomedical Devices by 3D Printing. Polymers 2021, Vol. 13, 2021, 13. [CrossRef]
- Fernández-Ponce, M.T.; Casas, L.; Mantell, C.; Martínez De La Ossa, E.J. Use of high pressure techniques to produce Mangifera indica L. leaf extracts enriched in potent antioxidant phenolic compounds. Innov. Food Sci. Emerg. Technol Food Funct. 2015, 29, 94-106. [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a Free Radical Method to Evaluate Antioxidant Activity. LWT - Food Science and Technology 1995, 28, 25–30. [CrossRef]
- Scherer, R.; Godoy, H.T. Antioxidant Activity Index (AAI) by the 2,2-Diphenyl-1-Picrylhydrazyl Method. Food Chem. 2009, 112, 654–658. [CrossRef]
- Lin, R.Z.; Chen, Y.C.; Moreno-Luna, R.; Khademhosseini, A.; Melero-Martin, J.M. Transdermal Regulation of Vascular Network Bioengineering Using Aphotopolymerizable Methacrylated Gelatin Hydrogel. Biomaterials 2013, 34, 6785–6796. [CrossRef]
- Grosso, P.; Cejudo, C.; Sánchez-Gomar, I.; Durán-Ruiz, M.a. C.; Moreno-Luna, R.; Casas, L.; Pereyra, C.; Mantell, C. Supercritical Impregnation of Mango Leaf Extract into PLA 3D-Printed Devices and Evaluation of Their Biocompatibility with Endothelial Cell Cultures. Polymers 2022, Vol. 14, 2022, 14. [CrossRef]
- Mustafa, A.; Turner, C. Pressurized Liquid Extraction as a Green Approach in Food and Herbal Plants Extraction: A Review. Anal. Chim. Acta 2011, 703, 8–18. [CrossRef]
- Fernández-Ponce, M.T.; López-Biedma, A.; Sánchez-Quesada, C.; Casas, L.; Mantell, C.; Gaforio, J.J.; Martínez De La Ossa, E.J. Selective Antitumoural Action of Pressurized Mango Leaf Extracts against Minimally and Highly Invasive Breast Cancer. Food Funct. 2017, 8, 3610–3620. [CrossRef]
- Zhang, J.; Wu, H. Valorization of Bioactive Compounds from Food By-Products Using Supercritical Fluid Extraction: A Technological and Industrial Perspective. Food Chem. 2025, 484, 144277. [CrossRef]
- de Araujo, E.J.S.; Martínez, J. Scenarios, Prospects, and Challenges Related to Supercritical Fluid Impregnation in the Food Industry: A Scoping Review (2018–2023). Biofuels, Bioproducts and Biorefining 2024, 18, 2091–2115. [CrossRef]
- Reed, K.E.; Westphale, E.M.; Larson, D.M.; Wang, H.Z.; Veenstra, R.D.; Beyer, E.C. Molecular Cloning and Functional Expression of Human Connexin37, an Endothelial Cell Gap Junction Protein. J. Clin. Invest. 1993, 91, 997–1004. [CrossRef]
- Brighton, C.T.; Hunt, R.M. Early Histological and Ultrastructural Changes in Medullary Fracture Callus. Journal of Bone and Joint Surgery - Series A 1991, 73. [CrossRef]
- Ismail, M.B.; Rajendran, P.; Zahra, H.M.A.; Veeraraghavan, V.P. Mangiferin Inhibits Apoptosis in Doxorubicin-Induced Vascular Endothelial Cells via the Nrf2 Signaling Pathway. International Journal of Molecular Sciences 2021, Vol. 22, 2021, 22. [CrossRef]
- Tu, J.B.; Ma, R.Z.; Dong, Q.; Jiang, F.; Hu, X.Y.; Li, Q.Y.; Pattar, P.; Zhang, H. Induction of Apoptosis in Infantile Hemangioma Endothelial Cells by Propranolol. Exp. Ther. Med. 2013, 6, 574–578. [CrossRef]
- Azizah, A.H.; Nik Ruslawati, N.M.; Swee Tee, T. Extraction and Characterization of Antioxidant from Cocoa By-Products. Food Chem. 1999, 64, 199–202. [CrossRef]
- Goodman, S.B.; Yao, Z.; Keeney, M.; Yang, F. The Future of Biologic Coatings for Orthopaedic Implants. Biomaterials 2013, 34, 3174–3183. [CrossRef]
- Jiang, T.; Han, F.; Gao, G.; Liu, M. Mangiferin Exert Cardioprotective and Anti-Apoptotic Effects in Heart Failure Induced Rats. Life Sci. 2020, 249, 117476. [CrossRef]


| Yield extraction (%) | 10.83 ± 0.91 |
| IC50 (µg/mL) | 9.74 ± 0.03 |
| AAI | 2.36 ± 0.01 |
| Phenolic compounds (g/100 g dried extract) | |
| gallic acid | 2.57 ± 0.04 |
| mangiferin | 9.08 ± 0.02 |
| iriflophenone 3-C-β-d-glucoside | 11.12 ± 1.03 |
| iriflophenone 3-C-(2-O-galloyl)-β-d-glucoside | 2.72 ± 0.05 |
| quercetin 3-d-galactoside | 1.87 ± 0.08 |
| quercetin 3-β-d-glucoside | 1.52 ± 0.02 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




