Submitted:
26 February 2026
Posted:
02 March 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Methods/Analytical Approach
Results, Discussion
Viruses and Nematodes
Distribution of C. elegans in Nature and Its Vectors
Passive Dispersion of Caenorhabditis Nematodes
Animal Carriers of Nematodes Caenorhabditis
The Theoretical Possibility of SARS-CoV-2 Spreading to New Ecological Niches and the Role of Nematodes
People infected with SARS-CoV-2 
|
Compost |
 |
Caenorhabditis nematodes in compost |  |
Animals, birds that carry nematodes |  |
New territories |  |
Animals in new territories far from the source of SARS-CoV-2 origins |
Conclusions
Conflict of Interest
References
- Blaxter, M; De Ley, P; Garey, JR; Liu, LX; Scheldeman, P; Vierstraete, A; et al. A molecular evolutionary framework for the phylum Nematoda. Nature 1998, 392(6671), 71–75. [Google Scholar] [CrossRef] [PubMed]
- De Ley, P; Blaxter, M. Systematic position and phylogeny. In The Biology of Nematodes; Lee, DL, Ed.; Taylor & Francis: London, 2002; pp. 1–30. [Google Scholar]
- Hodda, M. Phylum Nematoda: trends in species description, the documentation of diversity, and species richness estimation. Zootaxa 2011, 2861, 1–44. [Google Scholar] [CrossRef]
- Van Megen, H; van den Elsen, S; Holterman, M; Karssen, G; Mooyman, P; Bongers, T. A phylogenetic tree of nematodes based on about 1200 full-length small subunit ribosomal DNA sequences. Molecular Phylogenetics and Evolution 2009, 51(2), 314–329. [Google Scholar] [CrossRef]
- Lambshead, PJD. Recent developments in marine benthic biodiversity research. Oceanography and Marine Biology: An Annual Review 2004, 42, 1–70. [Google Scholar]
- Platt, HM; Warwick, RM. Free-living Marine Nematodes. Part I: British Enoplids; Cambridge University Press: Cambridge, 1983; p. 307 p. [Google Scholar]
- Decraemer, W; Hunt, DJ. Structure and classification. In Plant Nematology, 2nd ed.; Perry, RN, Moens, M, Eds.; CABI: Wallingford, 2013; pp. 3–39. [Google Scholar] [CrossRef]
- May, RM. How many species are there on Earth? Science 1988, 24d1(4872), 1441–1449. [Google Scholar] [CrossRef] [PubMed]
- Mora, C; Tittensor, DP; Adl, S; Simpson, AGB; Worm, B. How many species are there on Earth and in the ocean? PLoS Biology 2011, 9(8), e1001127. [Google Scholar] [CrossRef]
- Perry, RN; Moens, M. Plant Nematology; CABI: Wallingford, 2011. [Google Scholar]
- Anderson, RC. Nematode Parasites of Vertebrates: Their Development and Transmission, 2nd ed.; CABI Publishing: Wallingford, 2000; p. 650 p. [Google Scholar]
- Hotez, PJ; Brindley, PJ; Bethony, JM; King, CH; Pearce, EJ; Jacobson, J. Helminth infections: the great neglected tropical diseases. New England Journal of Medicine 2008, 358(10), 1010–1022. [Google Scholar] [CrossRef] [PubMed]
- Blaxter, M; Koutsovoulos, G. The evolution of parasitism in Nematoda. Parasitology 2015, 142(S1), S26–S39. [Google Scholar] [CrossRef] [PubMed]
- Brenner, S. The genetics of Caenorhabditis elegans. Genetics 1974, 77(1), 71–94. [Google Scholar] [CrossRef] [PubMed]
- Félix, MA; Braendle, C. The natural history of Caenorhabditis elegans. Current Biology 2010, 20(22), R965–R969. [Google Scholar] [CrossRef] [PubMed]
- Sommer, RJ. Pristionchus pacificus. In WormBook. The C. elegans Research Community; 2006. [Google Scholar] [CrossRef]
- Cutter, AD. Caenorhabditis evolution in the wild. Molecular Ecology 2015, 24(18), 4187–4201. [Google Scholar] [CrossRef]
- Markov, GV; Meyer, JM; Panda, O; Artyukhin, AB; Claaßen, MH; Witte, H; et al. Functional conservation and divergence of the Panagrellus redivivus genome. BMC Evolutionary Biology 2016, 16, 40. [Google Scholar] [CrossRef]
- WHO. Number of COVID-19 deaths reported to WHO (cumulative total). Reported COVID-19 deaths on 25.01. 2026. Available online: https://data.who.int/dashboards/covid19/deaths.
- WOAH, report from. 18 December 2025. Available online: https://www.fao.org/animal-health/situation-updates/emerging-zoonotic-coronaviruses-in-animals/en.
- Oude Munnink et al. (2021),Transmission of SARS-CoV-2 on mink farms between humans and mink and back to humans. Science 371, 172–177. doi:10.1126/science.abe5901; (Larsen C.S., Paludan S.R. Corona's new coat: SARS-CoV-2 in Danish minks and implications for travel medicine/ Travel Med Infect Dis. 2020 Nov 20;38:101922. [CrossRef]
- Hui-Ling, Yen; et al. Transmission of SARS-CoV-2 delta variant (AY.127) from pet hamsters to humans, leading to onward human-to-human transmission: a case study. Lancet 2022, 399(10329), 1070–1078. [Google Scholar] [CrossRef]
- Klestova. Possible spread of SARS-CoV-2 in domestic and wild animals and body temperature role. Virus Res. 2023, 327, 199066. [Google Scholar] [CrossRef] [PubMed]
- Xi E, Félix MA, Wang D. Beyond pathogenesis: the nematode immune network as the arbiter of a host–virus truce. Viruses. 2025;17(11):1485. doi:10.3390/v17111485 Available from: https://www.mdpi.com/1999-4915/17/11/1485.
- Huang, H.; et al. Expansion of the plant-parasitic nematode RNA viruses: unprecedented diversity, intron-bearing viruses, and cross-kingdom evolutionary links. npj Biofilms and Microbiomes 2025. [Google Scholar] [CrossRef]
- Abbafati, C.; et al. Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet 2020, 396, 1204–1222. [Google Scholar] [CrossRef]
- Quek S, Hadermann A, Wu Y, De Coninck L, Hegde S, Boucher JR, Cresswell J, Foreman E, Steven A, LaCourse EJ, Ward SA, Wanji S, Hughes GL, Patterson EI, Wagstaff SC, Turner JD, Parry RH, Kohl A, Heinz E, Bentum Otabil K, Matthijnssens J, Colebunders R, Taylor MJ. (2024). Diverse RNA viruses of parasitic nematodes can elicit antibody responses in vertebrate hosts. Nature Microbiology. 2024 Oct;9(10):2488–2505. doi:10.1038/s41564-024-01796-6. Epub 2024 Sep 4.
- Vieira, P.; Nemchinov, L. G. A novel species of RNA virus associated with root lesion nematode Pratylenchus penetrans. J. Gen. Virol. 2019, 100, 704–708. [Google Scholar] [CrossRef]
- Macfarlane, SA. Molecular determinants of the transmission of plant viruses by nematodes. Molecular Plant Pathology 2003, 4(3), 211–215. Available online: https://pubmed.ncbi.nlm.nih.gov/20569381/. [CrossRef] [PubMed]
- Sarwar, M. Chapter “Different nematodes and plasmodiophorids as vectors of plant viruses.”. In Plant Virus Diseases and Their Control; Available from; Elsevier, 2020; pp. 1–22. [Google Scholar] [CrossRef]
- Macfarlane, SA. Molecular determinants of the transmission of plant viruses by nematodes. Molecular Plant Pathology 2003, 4(3), 211–215. Available online: https://pubmed.ncbi.nlm.nih.gov/20569381/. [CrossRef]
- Huang, H; Mao, H; Wu, T; Xie, J; Zheng, J; Cheng, J; Fu, Y; Cai, Q; Lin, Y; Chen, T; Li, B; Yu, X; Hsiang, T; Jiang, D; Xiao, X. Expansion of the plant-parasitic nematode RNA viruses: unprecedented diversity, intron-bearing viruses, and cross-kingdom evolutionary links. npj Biofilms and Microbiomes. 2025. Available online: https://www.nature.com/articles/s41522-025-00867-8. [CrossRef]
- Gallet, R; Michalakis, Y; Blanc, S. Vector-transmission of plant viruses and constraints imposed by virus–vector interactions. Current Opinion in Virology 2018, 33, 144–150. [Google Scholar] [CrossRef] [PubMed]
- MacFarlane SA, Robinson DJ. Transmission of plant viruses by nematodes. In: Elliott SL, Blanford S, Thomas MB, editors. Microbe-Vector Interactions in Vector-Borne Diseases. Cambridge University Press; 2004.p. 127–148. Available from: https://www.cambridge.org/core/books/microbevector-interactions-in-vectorborne-diseases/transmission-of-plant-viruses-by-nematodes/0DE42B7342FC74AC26212FF5F562ACE1. p. 12.
- Schellenberger, P; Sauter, C; Lorber, B; Bron, P; Trapani, S; others. Structural insights into viral determinants of nematode-mediated Grapevine fanleaf virus transmission. PLoS Pathogens 2011. [Google Scholar] [CrossRef] [PubMed]
- Dheilly, N. M.; Lucas, P.; Blanchard, Y.; Rosario, K. A world of viruses nested within parasites: unraveling viral diversity within parasitic flatworms (Platyhelminthes). Microbiol. Spectr. 2022, 10, e0013822. [Google Scholar] [CrossRef]
- Vieira, P.; Subbotin, S. A.; Alkharouf, N.; Eisenback, J.; Nemchinov, L. G. Expanding the RNA virome of nematodes and other soil-inhabiting organisms. Virus Evol. 2022, 8, veac019. [Google Scholar] [CrossRef]
- Félix MA, Ashe A, Piffaretti J, Wu G, Nuez I, Bélicard T, et al. Natural and experimental infection of Caenorhabditis nematodes by novel viruses related to nodaviruses. PLoS Biology. 2011;9(1):e1000586. doi:10.1371/journal.pbio.1000586. Available from: https://journals.plos.org/plosbiology/article?id=10.1371/journal.pbio.1000586. e1000586.
- ICTV. Virus Taxonomy: 2023 Release; International Committee on Taxonomy of Viruses; Available online: https://ictv.global/taxonomy.
- Lefkowitz, EJ; Dempsey, DM; Hendrickson, RC; Orton, RJ; Siddell, SG; Smith, DB. Virus taxonomy: the database of the International Committee on Taxonomy of Viruses (ICTV). Nucleic Acids Research 2018, 46(D1), D708–D717. [Google Scholar] [CrossRef]
- Félix, M. A.; Wang, D. Natural viruses of Caenorhabditis nematodes. Annu. Rev. Genet. 2019, 53, 313–326. [Google Scholar] [CrossRef]
- Ashe, A; Bélicard, T; Le Pen, J; Sarkies, P; Frézal, L; Lehrbach, NJ; et al. A deletion polymorphism in the Caenorhabditis elegans RIG-I homolog disables viral RNA dicing and antiviral immunity. ЕLife 2013, 2, e00994. [Google Scholar] [CrossRef]
- Frézal, L; Félix, MA. Caenorhabditis elegans outside the Petri dish. Current Biology 2015, 25(19), R936–R949. [Google Scholar] [CrossRef]
- Wang, D; Pujol, N. C. elegans innate immunity against viruses. Current Opinion in Immunology 2016, 38, 47–53. [Google Scholar] [CrossRef]
- Pospischil, A; Hess, RG; Bachmann, PA. Light microscopy and ultrahistology of intestinal changes in pigs infected with epizootic diarrhoea virus (EVD): comparison with transmissible gastroenteritis (TGE) virus and porcine rotavirus infections. Zentralbl Veterinarmed B 1981, 28, 564–577. [Google Scholar] [CrossRef] [PubMed]
- Cox, E.; Pensaert, M. B.; Hooyberghs, J. Sites of replication of transmissible gastroenteritis virus in the intestinal tract of pigs. In Research in Veterinary Science; 1988; Vol. 44. pp. 342–348. [Google Scholar]
- Livanos, AE; Jha, D; Cossarini, F; Gonzalez-Reiche, AS; Tokuyama, M; Aydillo, T. Intestinal host response to SARS-CoV-2 infection and COVID-19 outcomes in patients with gastrointestinal symptoms. Gastroenterology 2021, 160(7), 2435–2450.e34. [Google Scholar] [CrossRef]
- Xiao, F; Tang, M; Zheng, X; Liu, Y; Li, X; Shan, H. Evidence for gastrointestinal infection of SARS-CoV-2. Gastroenterology 2020, 158(6), 1831–1833.e3. [Google Scholar] [CrossRef]
- Mithal, A; Hume, AJ; Lindstrom-Vautrin, J; Villacorta-Martin, C; Olejnik, J; Bullitt, E. Human pluripotent stem cell–derived intestinal organoids model SARS-CoV-2 infection revealing a common epithelial inflammatory response. Stem Cell Reports 2021, 16(4), 940–953. [Google Scholar] [CrossRef]
- Lamers, MM; Beumer, J; van der Vaart, J; Knoops, K; Puschhof, J; Breugem, TI; et al. SARS-CoV-2 productively infects human gut enterocytes. Science 2020, 369(6499), 50–54. [Google Scholar] [CrossRef] [PubMed]
- Franz, CJ; Renshaw, H; Frezal, L; Jiang, Y; Felix, MA; Wang, D. Orsay, Santeuil and Le Blanc viruses primarily infect intestinal cells in Caenorhabditis nematodes. J Virol. 2012, 86(14), 7686–7695. [Google Scholar] [CrossRef]
- Franz, CJ; Zhao, G; Félix, M-A; Wang, D. Complete genome sequence of Le Blanc virus, a third Caenorhabditis nematode-infecting virus. J.Virol. 2012, 86(21), 11940. [Google Scholar] [CrossRef]
- Bekal, S; Niblack, TL; Lambert, KN. Discovery and initial analysis of novel viral genomes in the soybean cyst nematode Heterodera glycines. J Gen Virol. 2011, 92 Pt 8, 1870–1879. [Google Scholar] [CrossRef]
- Bekal, S; Domier, LL; Gonfa, B; Lakhssassi, N; Niblack, TL; Lambert, KN. A novel flavivirus-like virus infecting the soybean cyst nematode Heterodera glycines. J Gen Virol. 2014, 95 Pt 6, 1273–1281. [Google Scholar] [CrossRef]
- Gammon, DB. Caenorhabditis elegans as an emerging model for virus-host interactions. J Virol. 2017, 91(23), e00509-17. [Google Scholar] [CrossRef]
- Ruark, CL; Gardner, M; Mitchum, MG; Davis, EL; Sit, TL. Soybean cyst nematode culture collections and field populations reveal high incidences of infection by viruses. PLoS ONE 2017, 12(1), e0171514. [Google Scholar] [CrossRef]
- Ruark, CL; Gardner, M; Mitchum, MG; Davis, EL; Sit, TL. Novel RNA viruses within plant parasitic cyst nematodes. PLOS ONE 2018, 13(3), e0193881. [Google Scholar] [CrossRef]
- Lin, XD; Zhou, XB; Guo, WP; Wang, W; Wang, MR; Tang, K; et al. Discovery and genomic characterization of a novel RNA virus, sugar beet cyst nematode virus 1, infecting Heterodera schachtii. J Gen Virol. 2018, 99(12), 1586–1592. [Google Scholar] [CrossRef]
- Vieira, P; Nemchinov, LG. A novel species of RNA virus associated with root lesion nematode Pratylenchus penetrans. J Gen Virol. 2019, 100(4), 704–708. [Google Scholar] [CrossRef]
- Frézal, L; Jung, H; Tahan, S; Wang, D; Félix, MA. Noda-like RNA viruses infecting Caenorhabditis: from persistence to pathogenesis. J Virol. 2019, 93(21), e01170-19. [Google Scholar] [CrossRef] [PubMed]
- Richaud, A; et al. Vertical transmission in Caenorhabditis nematodes of RNA molecules encoding a viral RNA-dependent RNA polymerase. Proc Natl Acad Sci USA 2019, 116(49), 24738–24747. [Google Scholar] [CrossRef] [PubMed]
- Williams, SH; et al. Discovery of two highly divergent negative-sense RNA viruses associated with the parasitic nematode, Capillaria hepatica, in wild mice. J Gen Virol. 2019, 100, 1350–1362. [Google Scholar] [CrossRef]
- Kud, J; Dahan, J; Orellana, GE; Dandurand, LM; Karasev, AV. A novel rhabdovirus associated with the Idaho population of potato c yst nematode Globodera pallida. Viruses 2022, 14(12), 2718. [Google Scholar] [CrossRef] [PubMed]
- Vieira, P; Subbotin, SA; Alkharouf, N; Eisenback, J; Nemchinov, LG. Expanding the RNA virome of nematodes and other soil-inhabiting organisms. Virus Evolution 2022, 8(1), veac019. [Google Scholar] [CrossRef] [PubMed]
- Jeong, D-E; Choi, JY; Lee, J; et al. DNA polymerase diversity reveals multiple incursions of Polintons during nematode evolution. Molecular Biology and Evolution 2023, 40(6), msad120. [Google Scholar] [CrossRef]
- Widen, SA; Feschotte, C; Malik, HS. Virus-like transposons cross species barriers and drive genome evolution. Science 2023, 380(6649), eabq1165. [Google Scholar] [CrossRef]
- Koonin, EV; Krupovic, M; Yutin, N. Evolution of double-stranded DNA viruses of eukaryotes: from bacteriophages to transposons and back. Microbiology and Molecular Biology Reviews 2015, 79(4), 448–470. [Google Scholar] [CrossRef]
- Liu, H; Fu, Y; Jiang, D; Li, G; Xie, J; Cheng, J. Widespread endogenization of densoviruses and parvoviruses in animal and plant genomes. Journal of Virology 2011, 85(19), 9863–9876. [Google Scholar] [CrossRef]
- Aeby, P; Spicher, A; Nigon, V. Structure and genomic organization of a proretrovirus-like dispersed genetic element (Tas) of Ascaris lumbricoides. Proceedings of the National Academy of Sciences of the USA 1986, 83(10), 3560–3564. [Google Scholar] [CrossRef]
- Lu, R.; Maduro, M.; Li, F.; Li, H.W.; Broitman-Maduro, G.; Li, W.X.; Ding, S.W. Аnimal virus replication and RNAi-mediated antiviral silencing in Caenorhabditis elegans. Nature 2005, 436(7053), 1040–1043. [Google Scholar] [CrossRef]
- Sterken, MG; Snoek, LB; Bosman, KJ; Daamen, J; Riksen, JAG; Bakker, J; Pijlman, GP; Kammenga, JE. A heritable antiviral RNAi response limits Orsay virus infection in Caenorhabditis elegans N2. PLoS ONE 2014, 9(2), e89760. [Google Scholar] [CrossRef] [PubMed]
- Frézal, L; Felix, MA; Wang, D. Natural viruses of Caenorhabditis nematodes. J Virol. 2019, 93(15), e00290-19. [Google Scholar] [CrossRef]
- Richaud, A; Frézal, L; Joffard, N; Felix, MA. A viral RdRp as a new transgenerational element in nematodes. Proc Natl Acad Sci U S A 2019, 116(7), 2475–2484. [Google Scholar] [CrossRef]
- Williams, T; Corrigan, RM; Meaden, S; et al. Divergent negative-sense RNA viruses associated with the parasitic nematode Capillaria hepatica. J Gen Virol. 2019, 100(7), 1026–1036. [Google Scholar] [CrossRef]
- Félix, MA; Braendle, C. Ecology of Caenorhabditis species. In The C. elegans Research Community; WormBook: Pasadena (CA), 2006. [Google Scholar] [CrossRef]
- Félix, MA; Braendle, C. The natural history of Caenorhabditis elegans. Curr Biol. 2010, 20(22), R965–R969. [Google Scholar] [CrossRef]
- Cassada, RC; Russell, RL. The dauerlarva, a post-embryonic developmental variant of the nematode Caenorhabditis elegans. Dev Biol. 1975, 46(2), 326–342. [Google Scholar] [CrossRef]
- Green, AJ; Brochet, AL; Kleyheeg, E; Soons, MB. Dispersal of plants and invertebrates by waterbirds: a review of current knowledge and future priorities. Freshwater Biology 2023, 68(1), 1–19. [Google Scholar] [CrossRef]
- Perez, DM; Greenway, R; Stier, T; Font-Massot, N; Pertzelan, A; Ding, SS. Towering behavior and collective dispersal in Caenorhabditis nematodes. Curr Biol. 2025, 35(12), 2980–2986.e4. [Google Scholar] [CrossRef] [PubMed]
- Petersen, C.; Krahn, A.; Leippe, M. The nematode Caenorhabditis elegans and diverse potential invertebrate vectors predominantly interact opportunistically. Frontiers in Ecology and Evolution 2023, 11, 1069056. [Google Scholar] [CrossRef]
- Braendle, C.; Paaby, A. B. Life history in Caenorhabditis elegans: from molecular genetics to evolutionary ecology. Genetics 2024, 228(3), iyae151. [Google Scholar] [CrossRef]
- Archer, H.; Deiparine, S.; Andersen, E. C. The nematode Caenorhabditis elegans and the terrestrial isopod Porcellio scaber likely interact opportunistically. PLoS ONE 2020, 15(6), e0235000. [Google Scholar] [CrossRef]
- Barrière, A; Félix, MA. Temporal dynamics and linkage disequilibrium in natural populations of C. Elegans. Genetics 2007, 176(2), 999–1011. [Google Scholar] [CrossRef]
- Félix, MA; Duveau, F. Population dynamics and habitat sharing of natural Caenorhabditis populations. BMC Biology 2012, 10, 59. [Google Scholar] [CrossRef] [PubMed]
- Williams B, Nelson M, McRobert S, Fingerut J. An easy and inexpensive method for determining the rate of individual phoretic events of nematodes. MicroPublication Biology. 2023 Sep 21;2023:10.17912/micropub.biology.000942. doi:10.17912/micropub.biology.000942.
- Kiontke, K; Félix, MA; Ailion, M; et al. A phylogeny and molecular barcodes for Caenorhabditis. BMC Evolutionary Biology 2011, 11, 339. [Google Scholar] [CrossRef]
- Pees, B; Johnke, J; Möhl, M; Hamerich, IK; Leippe, M; Petersen, C. Microbes to-go: slugs as source for Caenorhabditis elegans microbiota acquisition. Environ Microbiol. 2021, 23(11), 6721–6733. [Google Scholar] [CrossRef] [PubMed]
- Sudhaus, W; Kiontke, K. Comparison of the cryptic nematode species C. elegans and C. Briggsae. Journal of Nematology 2007, 39(1), 1–20. [Google Scholar]
- Cassada, RC; Russell, RL. The dauerlarva, a post-embryonic developmental variant of the nematode C. Elegans. Developmental Biology 1975, 46(2), 326–342. [Google Scholar] [CrossRef]
- Swinehart, A.; et al. Diet of a threatened rattlesnake (eastern massasauga) revealed by DNA metabarcoding. Ecology and Evolution 2023, 13, e10029. [Google Scholar] [CrossRef]
- Swinehart, A. Fecal Findings: Investigating Eastern Massasauga Rattlesnake Diet Using DNA Metabarcoding. MSc thesis, Grand Valley State University, 2021. Available online: https://scholarworks.gvsu.edu/theses/1008.
- Swinehart, А. Diet reconstruction of the eastern massasauga (Sistrurus catenatus) using fecal DNA metabarcoding reveals prey composition and non-target taxa. Ecology and Evolution 2023, 13(3), e9890. [Google Scholar] [CrossRef]
- Schaftenaar WGM, Dorrestein GM, Mensink JMCH, Cremers HJWM. An unusual infestation with rhabditid nematodes in a green tree monitor lizard (Varanus prasinus): diagnosis and treatment: a case report. In: Proceedings of the European Association of Zoo- and Wildlife Veterinarians Scientific Meeting, Paris; 2000. p. 67–69. Available from: https://www.researchgate.net/publication/46628105_An_unusual_infestation_with_rhabditid_nematodes_in_a_green_tree_monitor_lizard_Varanus_prasinus_diagnosis_and_treatment_a_case_report.
- Imai, D. M.; Nadler, S. A.; Brenner, D.; Donovan, T. A.; Pessier, A. P. Rhabditid nematode-associated ophthalmitis and meningoencephalomyelitis in captive Asian horned frogs (Megophrys montana). Journal of Veterinary Diagnostic Investigation 2009, 21(4), 568–573. [Google Scholar] [CrossRef] [PubMed]
- Kiontke, KC; Sudhaus, W. Ecology of Caenorhabditis species; WormBook, 2006; Available online: https://www.wormbook.org/chapters/www_ecology/ecology.html. [CrossRef]
- Frisch, D; Green, AJ; Figuerola, J. High dispersal capacity of a broad spectrum of aquatic invertebrates via waterbirds. Aquatic Sciences 2007, 69(4), 568–574. [Google Scholar] [CrossRef]
- Ptatscheck, C.; Traunspurger, W. The ability to get everywhere: dispersal modes of free-living, aquatic nematodes. Hydrobiologia 2020, - 847, 3519–3547. [Google Scholar] [CrossRef]
- Silva, G. G.; et al. Invertebrate dispersal by waterbird species in neotropical wetlands. Brazilian Journal of Biology 2021, 84(1), e250280. [Google Scholar] [CrossRef]
- Kenney, SJ; Anderson, GL; Williams, PL; Millner, PD; Beuchat, LR. Migration of Caenorhabditis elegans to manure and manure compost and potential for transport of Salmonella Newport to fruits and vegetables. Int J Food Microbiol. 2006, 106(1), 61–68. [Google Scholar] [CrossRef] [PubMed]
- WOAH, report from 31 July 2022. Available online: https://www.woah.org/app/uploads/2022/08/sars-cov-2-situation-report-15.pdf.
- McDuie, F.; Casazza, M. L.; Keiter, D.; Overton, C. T.; Herzog, M. P.; Feldheim, C. L.; Ackerman, J. T. Moving at the speed of flight: dabbling duck-movement rates and the relationship with electronic tracking interval. Wildlife Research 2019, 46(6), 533–543. [Google Scholar] [CrossRef]
- Chevallier, D.; et al. Influence of weather conditions on the flight of migrating black storks. Proceedings of the Royal Society B 2010, 277(1688), 2359–2366. [Google Scholar] [CrossRef]
- Alerstam, T.; Rosen, M.; Bäckman, J.; Ericson, P. G.; Hellgren, O. Flight speeds among bird species: allometric and phylogenetic effects. PLoS Biology 2007, 5(8), e197. [Google Scholar] [CrossRef] [PubMed]
- Newton, I. The Migration Ecology of Birds; Academic Press, 2008; ISBN 9780125173674. [Google Scholar]
- Hirose, R.; Itoh, Y.; Ikegaya, H.; Miyazaki, H.; Watanabe, N.; Yoshida, T.; Nakaya, T. Differences in environmental stability among SARS-CoV-2 variants of concern: both Omicron BA.1 and BA.2 have higher stability than the Wuhan strain. Clinical Microbiology and Infection 2022, 28(11), 1486–1491. [Google Scholar] [CrossRef]
- Wang, B.; Chen, L.; Sui, H.; Dong, X.; Huang, H.; Wang, X.; Wang, J. The environmental stability of SARS-CoV-2 variants Omicron BA.1 and BA.5 on the surfaces of widely used transport packaging materials. Microbiology Spectrum 2023, 11(3), e04881-22. [Google Scholar] [CrossRef]
- Chin, A. W. H.; Chu, J. T. S.; Perera, M. R. A.; Hui, K. P. Y.; Yen, H. L.; Chan, M. C. W.; Poon, L. L. M. Stability of SARS-CoV-2 in different environmental conditions. The Lancet Microbe 2020, 1(1), e10. [Google Scholar] [CrossRef] [PubMed]
- van Doremalen, N.; Bushmaker, T.; Morris, D. H.; Holbrook, M. G.; Gamble, A.; Williamson, B. N.; Tamin, A.; Harcourt, J. L.; Thornburg, N. J.; Gerber, S. I.; Lloyd-Smith, J. O.; de Wit, E.; Munster, V. J. Aerosol and surface stability of SARS-CoV-2 as compared with SARS-CoV-1. New England Journal of Medicine 2020, 382(16), 1564–1567. [Google Scholar] [CrossRef]
- Riddell, S.; Goldie, S.; Hill, A.; Eagles, D.; Drew, T. W. The effect of temperature on persistence of SARS-CoV-2 on common surfaces. Virology Journal 2020, 17, 145. [Google Scholar] [CrossRef]
- Jeong, H. W.; Kim, S. M.; Kim, J. Y.; Lee, E. Y.; Kim, H.; Kim, S. G. Viable SARS-CoV-2 in various specimens from COVID-19 patients. Clinical Microbiology and Infection 2020, 26(11), 1520–1524. [Google Scholar] [CrossRef] [PubMed]
- Ratnesar-Shumate, S.; Williams, G.; Green, B.; Krause, M.; Holland, B.; Wood, S.; Bohannon, J.; Boydston, J.; Freeburger, D.; Hooper, I.; Beck, K.; Yeager, J.; Altamura, L. A.; Biryukov, J.; Yolitz, J.; Schuit, M.; Wahl, V.; Hevey, M.; Dabisch, P. Simulated Sunlight Rapidly Inactivates SARS-CoV-2 on Surfaces. The Journal of Infectious Diseases 2020, 222(2), 214–222. [Google Scholar] [CrossRef]
- Bivins, A; Greaves, J; Fischer, R; Yinda, KC; Ahmed, W; Kitajima, M; Munster, VJ; Bibby, K. Persistence of SARS-CoV-2 in water and wastewater. Environ Sci Technol Lett. 2020, 7(12), 937–942. [Google Scholar] [CrossRef]
- Casanova, L; Rutala, WA; Weber, DJ; Sobsey, MD. Survival of surrogate coronaviruses in water. Water Res. 2009, 43(7), 1893–1898. [Google Scholar] [CrossRef] [PubMed]
- Bivins, A.; North, D.; Ahmad, A.; et al. Wastewater-based epidemiology: global collaborative to maximize contributions in the fight against COVID-19. Water Research 2020, 191, 116127. [Google Scholar] [CrossRef] [PubMed]
| Year | Virus/group | Genome type | Primary host (nematode) | Pathogenicity/effect | Source |
| 2011 | Orsay virus | (+)ssRNA, two-segment,, nodavirus-like | Caenorhabditis elegans | Intestinal infection, microvilli damage, decreased fertility | [38,51] |
| 2011 | Santeuil virus | (+)ssRNA, nodavirus-like | Caenorhabditis briggsae | Subclinical intestinal infection | [38,51] |
| 2011 | Le Blanc virus | (+)ssRNA, nodavirus-like | Caenorhabditis briggsae | Moderate pathogenicity, intestinal infection | [38,51,52] |
| 2011 | Novel RNA viruses in soybean cyst nematode | (+)ssRNA (flavi-like, picorna-like) | Heterodera glycines | First description of viruses in cyst-forming plant-parasitic nematodes | [53] |
| 2012 | Le Blanc virus (complete genome) | (+)ssRNA, nodavirus-like | Caenorhabditis briggsae | Genomic characteristics of the virus | [52] |
| 2013 | Orsay-like viruses (варианты) | (+)ssRNA | Caenorhabditis spp. | Antiviral immunity model (RNAi) | [42] |
| 2014 | Novel flavivirus-like nematode virus | (+)ssRNA | Heterodera glycines | Flavivirus-like lineage in nematodes | [54] |
| 2017 | Orsay-like viruses | (+)ssRNA | Caenorhabditis spp. | Used as a model for antivirus protection | [55] |
| 2017 | High incidence of viral infection in SCN populations | (+)ssRNA | Heterodera glycines | High prevalence of cryptic viral infections | [56] |
| 2018 | Novel RNA viruses (nematode-associated), diverse lineages | (+)ssRNA (picornavirales-like, nodavirus-like) | Plant-parasitic cyst nematodes (Heterodera glycines, Globodera pallida, G. rostochiensis) | Pathogenicity has not been established | [57] |
| 2018 | Sugar beet cyst nematode virus 1 (SBCNV1) | (+)ssRNA, picorna-like | Heterodera schachtii | Pathogenicity has not been established | [58] |
| 2019 | RLNV1 (picorna-like) | (+)ssRNA | Pratylenchus penetrans | Replication has been confirmed, but significant pathogenicity has not been described | [59] |
| 2019 | Noda-like RNA viruses diversity in Caenorhabditis | (+)ssRNA | Caenorhabditis spp. | Variability and evolution of Orsay-like viruses | [60] |
| 2019 | Vertically transmitted viral RNA elements | (+)ssRNA (RdRp-encoding) | Caenorhabditis spp. | Vertical transmission of viral-like RNAs | [61] |
| 2019 | Highly divergent negative-sense RNA viruses | (−)ssRNA | Capillaria hepatica | Confirmation of (−)ssRNA viruses in parasitic nematodes | [62] |
| 2022 | Rhabdovirus-like nematode virus (PcRV) | (−)ssRNA, non-segmented | Globodera pallida | Pathogenicity not established; possible impact on fitness is under discussion | [63] |
| 2022 | Diverse RNA viruses (picorna-like, noda-like, tombus-like, bunya-like и др.) | ssRNA (+) и ssRNA (−) | Soil-inhabiting nematodes (Pratylenchus, Globodera, Heterodera, Meloidogyne etc.) | Clinical pathogenicity has not been demonstrated; cryptic infections are suspected | [64] |
| 2024 | 91 RNA viruses (13 orders, 24 families) | ssRNA (+/−) и dsRNA | Parasitic nematodes (Brugia malayi, Onchocerca volvulus etc.) | Induces an antibody response in vertebrates; no evidence of productive replication has been detected | [27] |
| 2025 | Expansion of plant-parasitic nematode RNA viruses | RNA (ssRNA+, ssRNA−, dsRNA) | Free-living and parasitic nematodes | A review of the wide diversity of nematode viruses and their interactions with their hosts | [25] |
| Animal species | Movements/migrations (assessment) |
| Sistrurus catenatus (eastern massasauga) | Usually hundreds of meters – up to ~1–2 km during the season; rare dispersal movements between wintering grounds and summer habitats can reach ~3–5 km (exceptionally – up to ~8–10 km). |
| Varanus prasinus (green tree monitor lizard) | Mainly local movements within an individual's territory (tens to hundreds of meters); when dispersing juveniles and changing habitats, movements of up to ~1–3 km are possible, probably episodically. |
| Megophrys montana (Asian horned frog, captive) | Short distances: usually tens to hundreds of meters between ground shelters and breeding sites; in natural conditions, as a rule, do not exceed ~0.5–1 km per season, rarely up to ~1–2 km. |
| # | Birds:Latinname/English name | Frequency of propaguls found, % (s/n×100) /Comment | Approximate distance of movement/migration* |
| 1. | Amazonetta brasiliensis (Brazilian teal) | 0 % /Nematodes did not hatch * |
local migrations up to ≈ 500 km (partial migrations) |
| 2. | Anas flavirostris (Yellow-billed teal) | 23.5 (4/17)/There were isolated cases | migrations up to ≈ 1500 km (South American) |
| 3. | Spatula versicolor (Silver teal) | 0 %/were not recorded** | local/regional movements up to ≈ 800 km |
| 4. | Callonetta leucophrys (Ringed teal) | 6.9 (2/29)/Fixation of hatched nematodes | mainly local (≤ 500 km) |
| 5. | Coscoroba coscoroba (Coscoroba swan) | 48.1 (13/27) | migration up to ≈ 1500-2000 km |
| 6. | Dendrocygna viduata (White-faced whistling-duck) | 17.6 (6/34) | migration/movement up to ≈ 1000–2000 km |
| 7. | Chauna torquata (Southern screamer) | 16.7 (3/18) | local/regional up to ≈ 300–600 km |
| 8. | Fulica armillata (Red-gartered coot) | 0.0/не фиксирoвались ** | movements of up to ≈ 500-1000 km |
| 9. | Theristicus caudatus (Buff-necked ibis) | 14.3 (2/14) | Local/regional distance is ≈ 800 km |
| 10. | Theristicus caerulescens (Plumbeous ibis) | 0.0/ не фиксирoвались** | Local/regional distance is ≈ 800 km |
| 11. | Nycticorax nycticorax (Black-crowned night-heron) | 0.0/ не фиксирoвались** | Migration to ≈ 1500–3000 km in the new population |
| 12. | Egretta spp. (Little blue heron & Snowy egret) | 16.7 (1/6) | migration/movement up to ≈ 1000–2500 km |
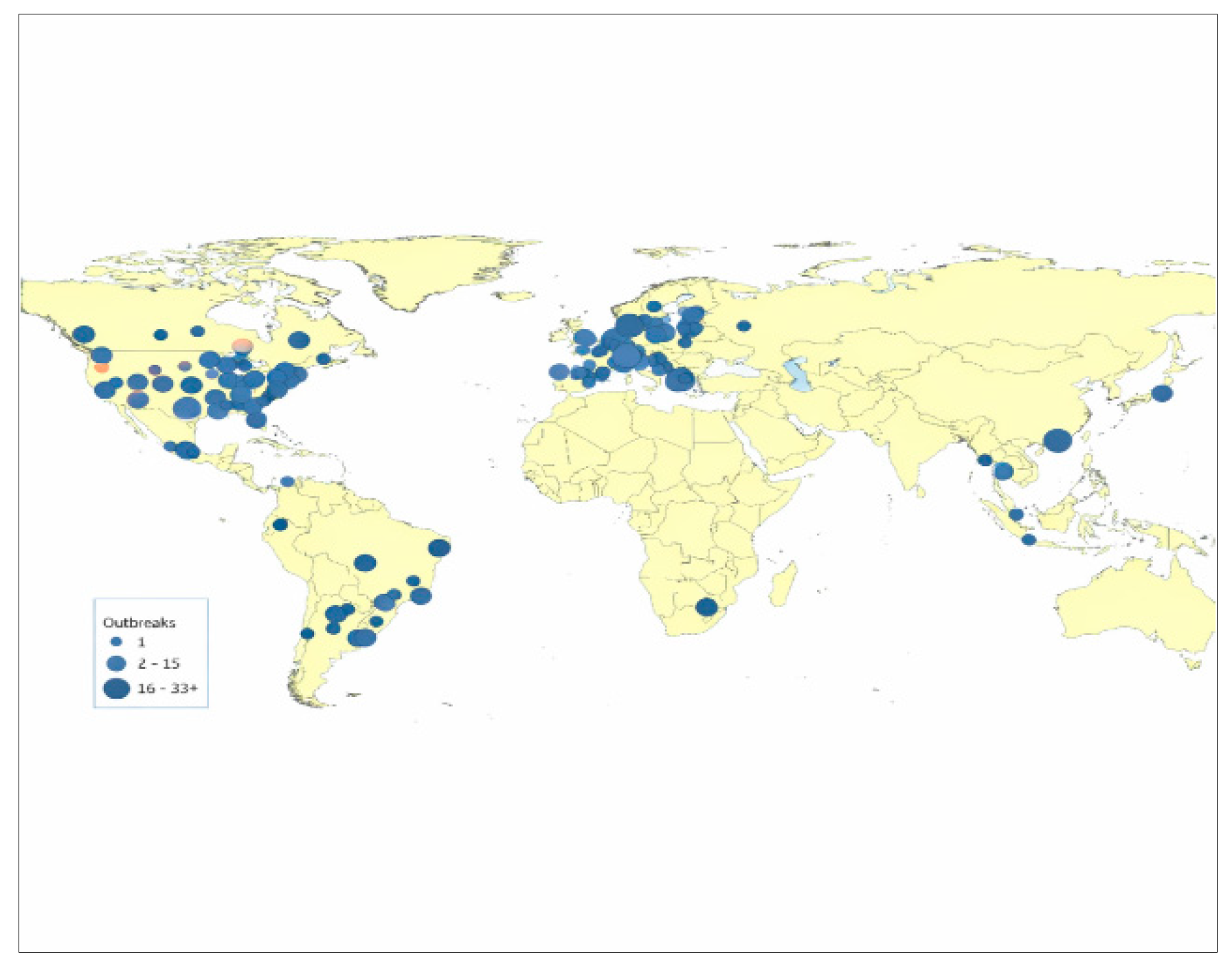
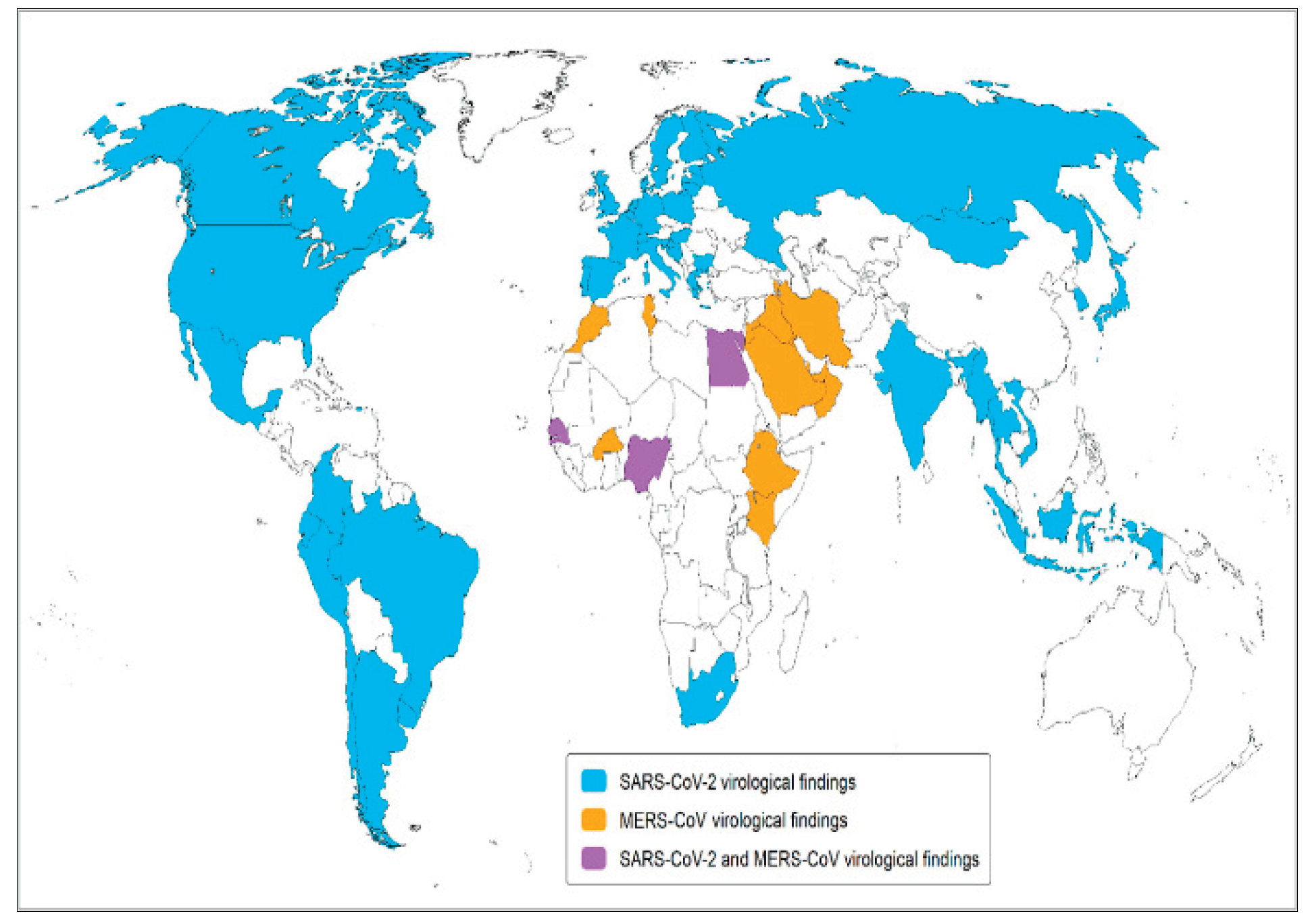
| # | Animal (English) | Scientific name (Latin) | Conditions under which SARS-CoV-2 was detected in animals | Natural geographic range/arial | Dispersal/migration capacity under natural conditions | Mobility category |
| 1. | Duck | Anas platyrhynchos | Backyards/farms (isolated cases) | Cosmopolitan (domestic ducks); wild ancestor—mallard (Anas platyrhynchos, Holarctic) | Max.: wild ducks usually migrate hundreds to thousands of km (up to 3–5 тис. km); domestic ducks are non-migratory and usually move locally (≤1–5 km), except for human-mediated transport |
Continental (≥500 km) |
| 2. | Puma | Puma concolor | Zoos/captivity | Americas (Canada–Patagonia) | Max.: up to > 1000 km; usually: Non-migratory; dispersal juveniles can be >1000 km | Continental (≥500 km) |
| 3. | Canadian lynx | Lynx canadensis | Zoos/captivity | Canada/Alaska/northern USA | Max.: n/a; usually: Non-migratory; long-distance settlements are possible in years of mining deficit | Continental (≥500 km) |
| 4. | Red fox | Vulpes vulpes | Wild populations and/or zoos (single) | Holarctic (native); Australia introduced |
Max.: ~1036 km (cumulative dispersal); usually: tens–hundreds km | Continental (≥500 km) |
| 5. | House finch | Haemorhous mexicanus | Wild populations (USA) | North America | Max.: seasonal movements up to ≥500 kmkm (partial migration); usually: tens–hundreds km | Regional (20–499 km) |
| 6. | Mule deer | Odocoileus hemionus | Wild populations (USA) | Western North America | Max.: up to~390 km; usually: Seasonal migration in some populations | Regional (20–499 km) |
| 7. | White-tailed deer | Odocoileus virginianus | Wild populations, massively in the USA/Canada | North America | Max.: usually about 10–50 km, sometimes >300 km; usually: Partial migration | Regional (20–499 km) |
| 8. | Lion | Panthera leo | Zoos/captivity | Africa (and a small population in India) | Max.: dispersal young males to ~200 km; usually: Non-migratory | Regional (20–499 km) |
| 9. | Red deer | Cervus elaphus | Zoos/captivity | Europe and West Asia (introductions) | Max.: 10–50 km (partial seasonal/altitude migration in some populations; usually: 1–20 km | Regional (20–499 km) |
| 10. | Antillean manatees | Trichechus manatus manatus | Wild populations/rehabilitation | Caribbean Basin, coastal waters | Max.: n/a; usually: Seasonal movements along the coast: tens–hundreds of km | Regional (20–499 km) |
| 11. | Asian small-clawed otter | Aonyx cinereus | Zoos/captivity | South and Southeast Asia | Max.: n/a; usually: Non-migratory; linear sections along waterbodies | Regional (20–499 km) |
| 12. | Eastern red bat | Lasiurus borealis | Wild populations (USA) | North America | Max.: n/a; usually: It migrates seasonally: hundreds of km | Regional (20–499 km) |
| 13. | Domestic American Mink | Domestic American mink (Neovison vison domesticus ) | Fur farms (Europe, North America, etc.), occasional escapes | North America (introduced/naturalized in Europe) | Max.: ~100 km; usually: up to ~50 km (more often along watercourses) | Regional (20–499 km) |
| 14. | Snow Leopard | Panthera uncia | Zoos/captivity | Central Asia (high mountains) | Max.: ~200 km ; usually: movements within the range | Regional (20–499 km) |
| 15. | Spotted hyena | Crocuta crocuta | Zoos/captivity | Sub-Saharan Africa | Max.: ~27 km/night (average); usually: active night movements tens of km | Regional (20–499 km) |
| 16. | Tiger | Panthera tigris | Zoos/captivity | Asia | Max.: ~315 km (long-range dispersal); usually: tens of kilometers dispersal juveniles | Regional (20–499 km) |
| 17. | White rhinoceros | Ceratotherium simum simum | Zoos/captivity | Southern Africa | Max.: ~25 km (distance from the release point/movements along the section); usually: tens of km | Regional (20–499 km) |
| 18. | Wild American Mink | Wild American mink (Neovison vison) | Wild populations (near farms/in the wild, singly) | North America; introduced в Еврoпе | Max.: ~100 km; usually: up to ~50 km (often along watercourses) | Regional (20–499 km) |
| 19. | Giant anteater | Myrmecophaga tridactyla | Zoos/rehabilitation/captivity | Central and South America | Max.: dispersal up to ~50–100 km; usually: daily movements 3–11 km | Regional (20–499 km) |
| 20. | Indian Leopard | Panthera pardus fusca | Zoos/captivity | Indian subcontinent | Max.: dispersal of young up to ~300 km; usually: movements within the range of tens of kilometers | Regional (20–499 km) |
| 21. | Mandrill | Mandrillus sphinx | Zoos/captivity | Equatorial Africa | Max.: seasonal nomadic movements groups up to ~50–100 km; usually: 5–10 km/day | Regional (20–499 km) |
| 22. | Maned wolf | Chrysocyon brachyurus | Zoos/captivity | Central South America (Cerrado/Pampas) | Max.: dispersal дo ~100–200 km; usually: nights movements 7–14 km | Regional (20–499 km) |
| 23. | Raccoon | Procyon lotor | Wild populations (USA) | North America; introduced in Europe/Japan | Max.: dispersal up to ~300 km; usually: tens of km | Regional (20–499 km) |
| 24. | Sheep | Ovis aries | Backyards/farms (isolated cases) | Cosmopolitan (domestic sheep) | Max.: Regional (20–499 km) movements herd up to ~100–300 km; usually: local | Regional (20–499 km) |
| 25. | Virginia opossum | Didelphis virginiana | Wild populations (USA) | North America | Max.: dispersal up to ~200 km; usually: movements within tens of kms | Regional (20–499 km) |
| 26. | Western lowland Gorilla | Gorilla gorilla gorilla | Zoos/holding facilities (human-to-animal transmission) | Central Africa (tropical forests) | Max.: seasonal nomadic movements groups up to ~50 km; usually: 2–10 km/day | Regional (20–499 km) |
| 27. | White-eared opossum | Didelphis albiventris | Wild populations (South America) | South America | Max.: dispersal дo ~150–200 km; usually: tens of km | Regional (20–499 km) |
| 28. | Wild Eurasian River Otter | Lutra lutra | Wild populations (sporadic detections) | Eurasia and North Africa | Max.: movements along rivers up to ~40–100 km | Regional (20–499 km) |
| 29. | Black- and brown headed Spider Monkey | Ateles fusciceps | Rehabilitation/captivity | Northwestern South America (Colombia/Ecuador) | Max.: n/a; usually: Non-migratory | Regional (20–50 km) |
| 30. | Brown rat | Rattus norvegicus | Synanthropic/anthropogenic populations (trapping/studies) | Cosmopolitan | Max.: n/a; usually: Non-migratory | Regional (1–10 km) |
| 31. | Common woolly monkey | Lagothrix lagothricha | Rehabilitation/captivity | Western/northern Amazonia | Max.: n/a; usually: Non-migratory | Regional (20–50 km) |
| 32. | Domestic Dog | Canis lupus familiaris | Domestic animals (human contact), cases in many countries | Cosmopolitan | Max.: n/a; usually: Non-migratory | Regional (5–20 km) |
| 33. | European fallow deer | Dama dama | Zoos/captivity | Europe/Western Asia (widely introduced) | Max.: n/a; Usually does not migrate; local seasonal movements | Regional (10–30 km) |
| 34. | Fishing cat | Prionailurus viverrinus | Zoos/captivity | South and Southeast Asia (wetlands) | Max.: n/a; usually: Non-migratory | Regional (10–30 km) |
| 35. | Goat domestic | Capra hircus | Farms/backyards (isolated cases) | Cosmopolitan (domestic goats) | Max.: n/a; usually: Non-migratory | Regional (5–20 km) |
| 36. | Gray brocket deer | Subulo gouazoubira | Zoos/captivity | Central and South America | Max.: n/a; usually: Non-migratory | Regional (10–30 km) |
| 37. |
South American squirrel monkey |
Saimiri sciureus | Zoos/captivity | Northern South America (Amazonia/Guiana) | Max.: n/a; usually: Non-migratory | Regional (20–50 km) |
| 38. | Pantanal cat | Leopardus braccatus | Zoos/captivity | Central South America (Pantanal/savannas) | Max.: n/a; usually: Non-migratory | Regional (10–30 km) |
| 39. | Pig | Sus scrofa domesticus | Farms/backyards (isolated cases) | Cosmopolitan (domestic pigs) | Max.: n/a; usually: Non-migratory | Regional (5–20 km) |
| 40. | domestic buffalo | Bubalus bubalis | Farms/backyards (isolated cases) | South Asia (domestic water buffalo is widespread) | Max.: n/a; usually: Non-migratory | Regional (5-30 km) |
| 41. | Coatimundi | Nasua nasua | Zoos/captivity | South America | Max.: n/a; usually: Non-migratory | Regional (5 -30 km) |
| 42. | White-fronted capuchin | Cebus unicolor | Captivity/rehabilitation | Western Amazonia (Peru, etc.) | Max.: n/a; usually: Non-migratory | Regional(20–50 km) |
| 43. | Eurasian beaver |
Castor fiber | Farms/captivity (single) | Europe and Northern Asia (reintroductions) | Max.: dispersal of young usually up to ~10 km (sometimes more); usually Non-migratory | Local (1–19 km |
| 44. | Hippopotamus | Hippopotamus amphibius | Zoos/captivity | Sub-Saharan Africa | Max.: up to 10 km; usually: Non-migratory; nights movements usually 2–10 km | Local (1–19 km) |
| 45. | large hairy armadillo | Chaetophractus villosus | Captivity/rehabilitation | South America (Pampas/steppe) | Max.: n/a; usually: Non-migratory | Local (1–5 km, limited) |
| 46. | Binturong | Arctictis binturong | Zoos/captivity | Southeast Asia | Max.: n/a; usually: Non-migratory; local movements | Local (1–5 km, limited) |
| 47. | Black-Tailed Marmoset | Mico melanurus | Wild populations (South America) | Центральная South America (Brazil/Bolivia/Paraguay) | Max.: n/a; usually: Non-migratory; local | Local (1–5 km, limited) |
| 48. | Cactus mouse | Peromyscus eremicus | Wild populations (USA/Mexico) | Southwestern USA and northern Mexico | Max.: n/a; usually: Non-migratory | Local (1–3 km, limited) |
| 49. | Cattle | Bos taurus taurus | Farms/backyards (isolated cases) | Cosmopolitan (domestic) | Max.: n/a; usually: Non-migratory | Local (1-20 km) |
| 50. | Chicken | (Gallus gallus domesticus) | Backyards/farms (isolated cases) | Cosmopolitan (domestic chickens) | Max.: n/a; usually: Non-migratory | Local (0.5–2 km, limited) |
| 51. | Domestic cat | Felis catus | Domestic animals (human contact), cases in many countries | Cosmopolitan (worldwide alongside humans) | Max.: n/a; usually: Non-migratory; usually km- (territory/free range) | Local (1–5 km / limited) |
| 52. | Domestic Ferret | Mustela furo | Domestic/laboratory animals; detected after contact with humans | Domesticated; kept in captivity worldwide | Max.: n/a; usually: Non-migratory | Local (1–3 km, limited) |
| 53. | Eastern cottontail | Sylvilagus floridanus | Wild populations (USA) | North America | Max.: n/a; usually: Non-migratory | Local (1–5 km, limited) |
| 54. | Eastern deer mouse | Peromyscus maniculatus | Wild populations (USA) | North America | Max.: n/a; usually: Non-migratory; small radius of activity | Local (1–5 km, limited) |
| 55. | European polecat | Mustela putorius | Wild populations (Europe) | Europe | Max.: ~5 km/day (linear movements within a few km); usually: no migrations, nomadic movements within the area | Local (1–15 km, limited) |
| 56. | Groundhog | Marmota monax | Wild populations (USA) | North America | Max.: n/a; usually: Non-migratory | Local (1–10 km) |
| 57. | Syrian hamster | Mesocricetus auratus | Pet hamsters/pet shops (e.g., Hong Kong), linked to trade | Native range: Syria; kept in captivity worldwide | Max.: n/a; usually: Non-migratory | Local (<1 km/ limited) |
| 58. | House mouse | Mus musculus | Synanthropic/anthropogenic populations (trapping/studies) | Cosmopolitan | Max.: n/a; usually: Non-migratory | Local (<0,5-2,0 km/ limited) |
| 59. | Lizard/ red-headed rock agama | Agama agama | Near a house/yard (single) | West and Central Africa | Max.: n/a; usually: Non-migratory; local movements | Local (0,5-2,0 km/ limited) |
| 60. | Domestic turkey | Meleagris gallopavo domesticus | Backyards/farms (isolated cases) | Cosmopolitan (domestic turkeys) | Max.: n/a; usually: Non-migratory | Local (1-5 km/ limited) |
| Virus variant/lineage | Which option does the data belong to | Temperature | Object / environment | Duration of infectivity | Source |
| Wuhan-Hu-1 (ранний штамм) | Direct evidence for an early pandemic variant | 4 °C | Liquid culture medium (without cells) | ≥14 days without significant loss of titer | [107] |
| Wuhan-Hu-1 | Direct evidence for an early pandemic variant | 22–25 °C | Liquid culture medium (without cells) | 3–7 days | [107] |
| Wuhan-Hu-1 | Direct evidence for an early pandemic variant | 37 °C | Liquid culture medium (without cells) | Loss of infectivity within 1–2 days | [107] |
| Wuhan-Hu-1 | Direct evidence for an early pandemic variant | 70 °C | Liquid culture medium (without cells) | Inactivation in ~5 min | [107] |
| Wuhan-Hu-1 | Direct evidence for an early pandemic variant | 21–23 °C | Plastic, stainless steel | Up to 72 hours | [108] |
| Wuhan-Hu-1 | Direct evidence for an early pandemic variant | 20 °C | Glass, polymer | Up to 28 days | [109] |
| Wuhan-Hu-1 | Direct evidence for an early pandemic variant | 30 °C | Glass, polymer | Up to 7 days | [109] |
| Wuhan-Hu-1 | Direct evidence for an early pandemic variant | 40 °C | Glass, polymer | <24 hours | [109] |
| Wuhan-Hu-1 | Direct evidence for an early pandemic variant | 4 °C | Skin (human skin model) | ≥14 days | [105] |
| Wuhan-Hu-1 | Direct evidence for an early pandemic variant | 22 °C | Skin (human skin model) | Up to 96 hours | [105] |
| Wuhan-Hu-1 | Direct evidence for an early pandemic variant | 37 °C | Skin (human skin model) | ~8 hours | [105] |
| Alpha (B.1.1.7) | Прямые данные для варианта Alpha | 22 °C | Plastic | Up to 7 days | [105] |
| Delta (B.1.617.2) | Прямые данные для варианта Delta | 22 °C | Plastic | Up to 7 days | [105] |
| Omicron (BA.1) | Прямые данные для варианта Omicron | 22 °C | Plastic | Up to 7-9 days (higher than Wuhan) | [105] |
| Wuhan-Hu-1 | Direct evidence for an early pandemic variant | 20–25 °C | Human feces | Infectious virus for up to 2-3 days | [110] |
| Wuhan-Hu-1 | Direct evidence for an early pandemic variant | 20–25 °C | Human urine | Infectious virus for up to 3-4 days | [110] |
| Wuhan-Hu-1 | Direct evidence for an early pandemic variant | 4 °C | Aquatic* environment | ≥7 days | [112] |
| Wuhan-Hu-1 | Direct evidence for an early pandemic variant | 20 °C | Aquatic* environment | 1–3 days | [112,114] |
| Wuhan-Hu-1 | Direct evidence for an early pandemic variant | 20–25 °C + УФ | Surfaces | Rapid inactivation (minutes–hours) | [111] |
| Wuhan-Hu-1 | Direct evidence for an early pandemic variant | 4–20 °C | Organic matrices (feces, sludge, wastewater - similar to cold compost) | Hours–days, at 4°C possibly up to several days | [112] |
| Wuhan-Hu-1 | Direct evidence for an early pandemic variant | 50–70 °C | Organic matrices/active compost | Rapid inactivation (minutes to tens of minutes) | [107] |
| Bird species | Approximate flight speed of a bird | Theoretical time for 400 km without stops |
| Mallard (Anas platyrhynchos) | ~82–83 km/h | ~4,5–5 hours |
| House finch (Haemorhous mexicanus) | ~~25–40 km/h (estimated) | ~10–16 hours |
| № | Bird species | Max. distance (km) | Average speed (km/h) | Flight time (hours) |
| 1 | Amazonetta brasiliensis | 500 | ~70 | ~7,1 h |
| 2 | Anas flavirostris | 1500 | ~75 | ~20 h |
| 3 | Spatula versicolor | 800 | ~75 | ~10,7 h |
| 4 | Callonetta leucophrys | 500 | ~70 | ~7,1 h |
| 5 | Coscoroba coscoroba | 2000 | ~75 | ~26,7 h |
| 6 | Dendrocygna viduata | 2000 | ~70 | ~28,6 h |
| 7 | Chauna torquata | 600 | ~65 | ~9,2 h |
| 8 | Fulica armillata | 1000 | ~70 | ~14,3 h |
| 9 | Theristicus caudatus | 800 | ~60 | ~13,3 h |
| 10 | Theristicus caerulescens | 800 | ~60 | ~13,3 h |
| 11 | Nycticorax nycticorax | 3000 | ~65 | ~46,2 h |
| 12 | Egretta spp. | 2500 | ~65 | ~38,5 h |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




