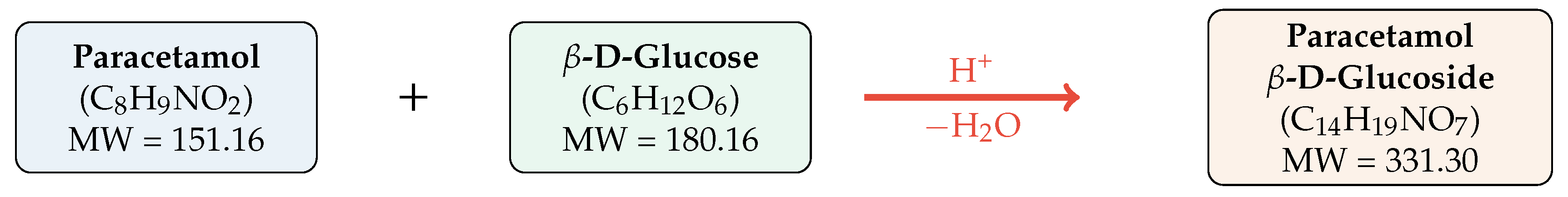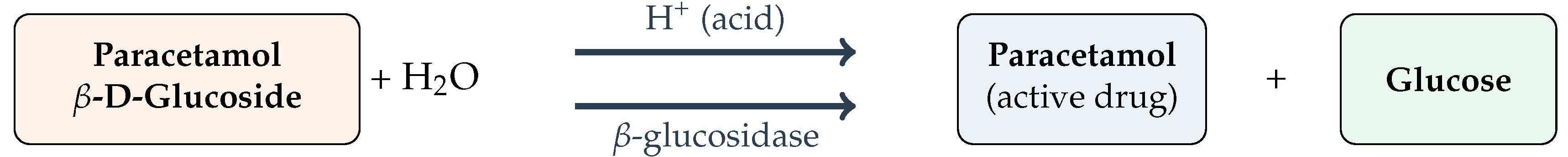Figure 1.
Synthesis of paracetamol -D-glucoside by acid-catalyzed glycosidation. Paracetamol reacts with -D-glucose under acid catalysis, releasing one water molecule.
Figure 1.
Synthesis of paracetamol -D-glucoside by acid-catalyzed glycosidation. Paracetamol reacts with -D-glucose under acid catalysis, releasing one water molecule.
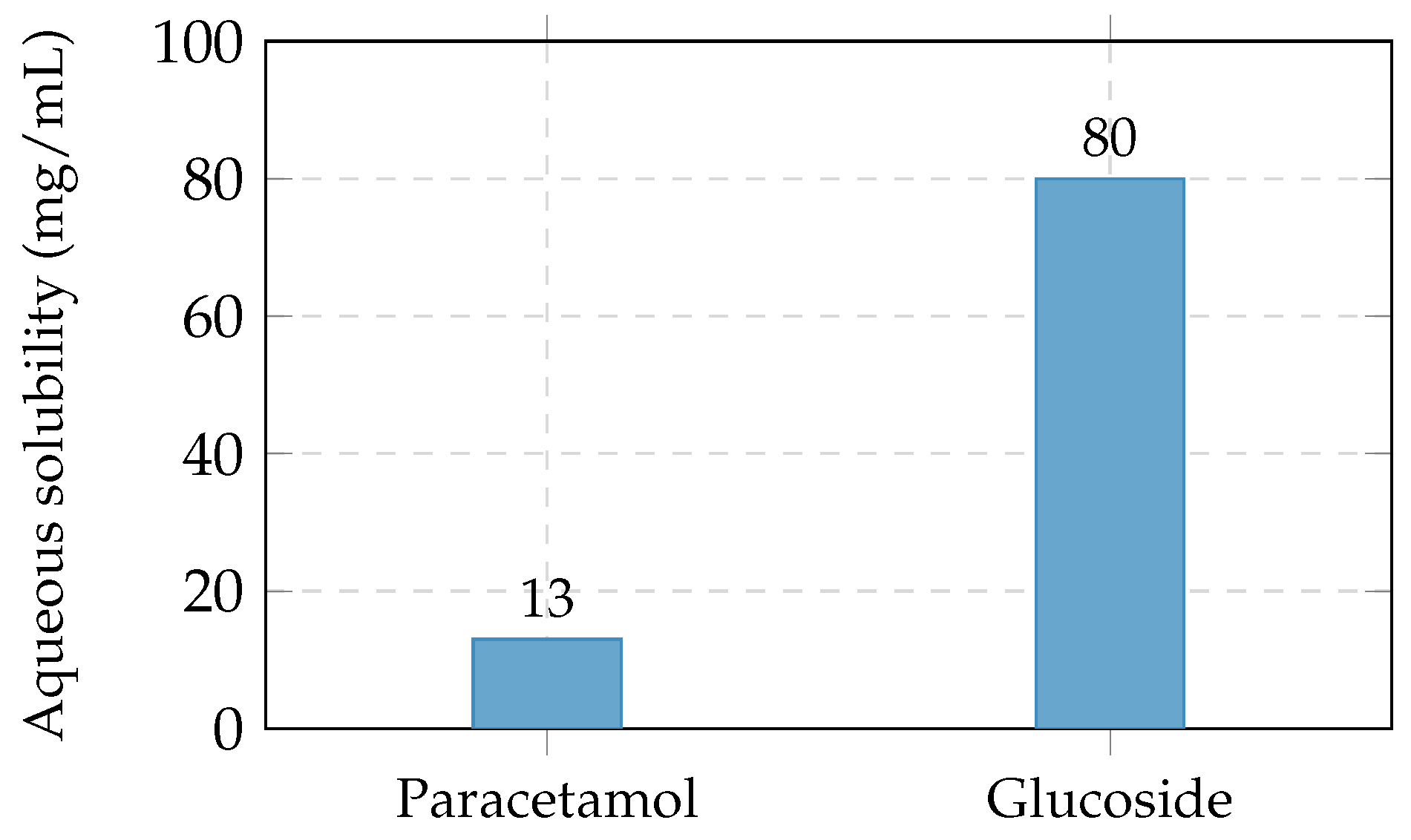
Figure 2.
Water solubility comparison at 25°C. Glucose attachment increases solubility approximately 6-fold.
Figure 2.
Water solubility comparison at 25°C. Glucose attachment increases solubility approximately 6-fold.
Figure 3.
Hydrolysis of paracetamol -D-glucoside by either acid catalysis or -glucosidase, regenerating free paracetamol and glucose.
Figure 3.
Hydrolysis of paracetamol -D-glucoside by either acid catalysis or -glucosidase, regenerating free paracetamol and glucose.
Figure 4.
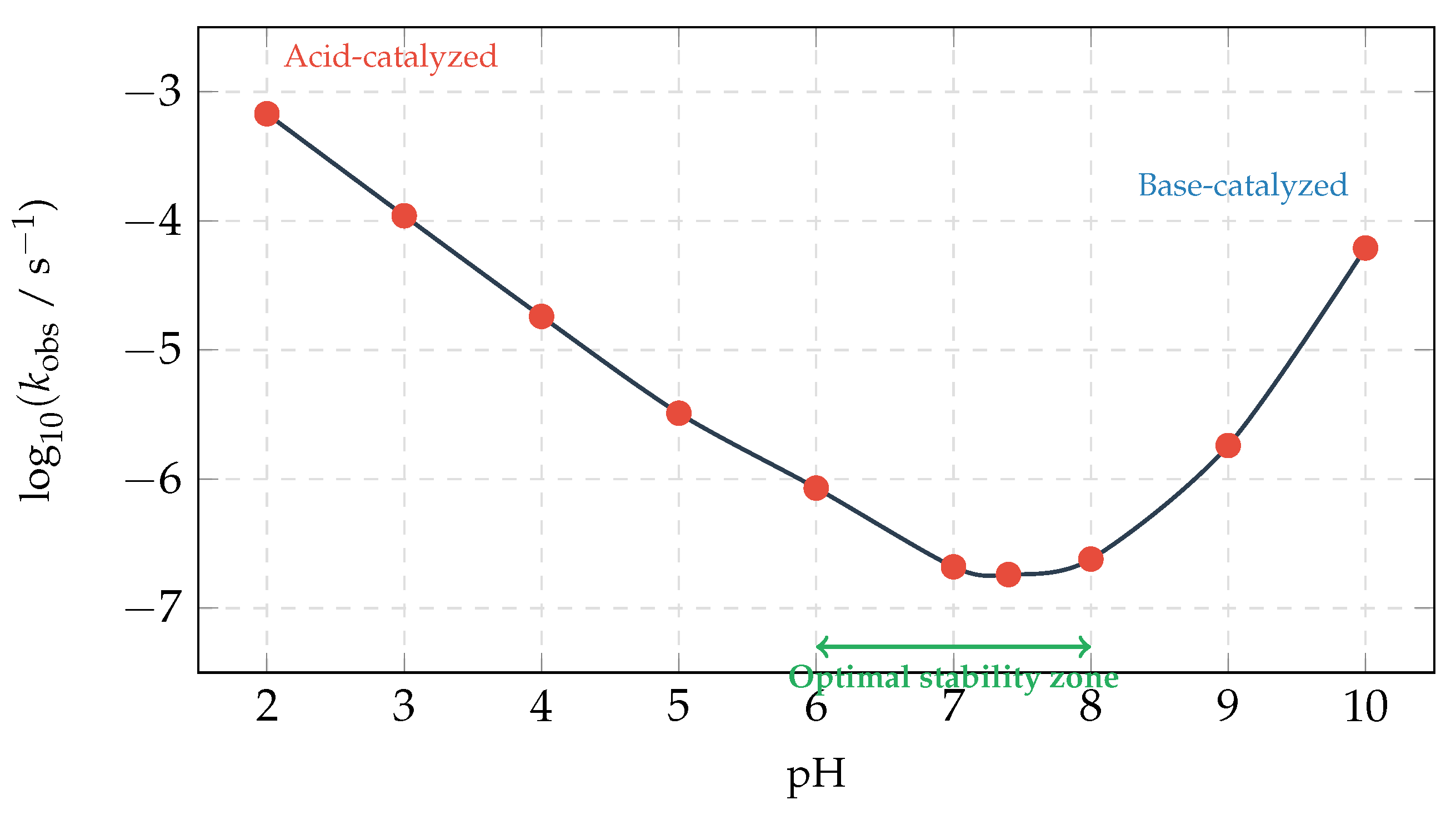
2D pH–rate profile showing the characteristic V-shape. Maximum stability occurs at pH 6–8.
Figure 4.
2D pH–rate profile showing the characteristic V-shape. Maximum stability occurs at pH 6–8.
Figure 5.
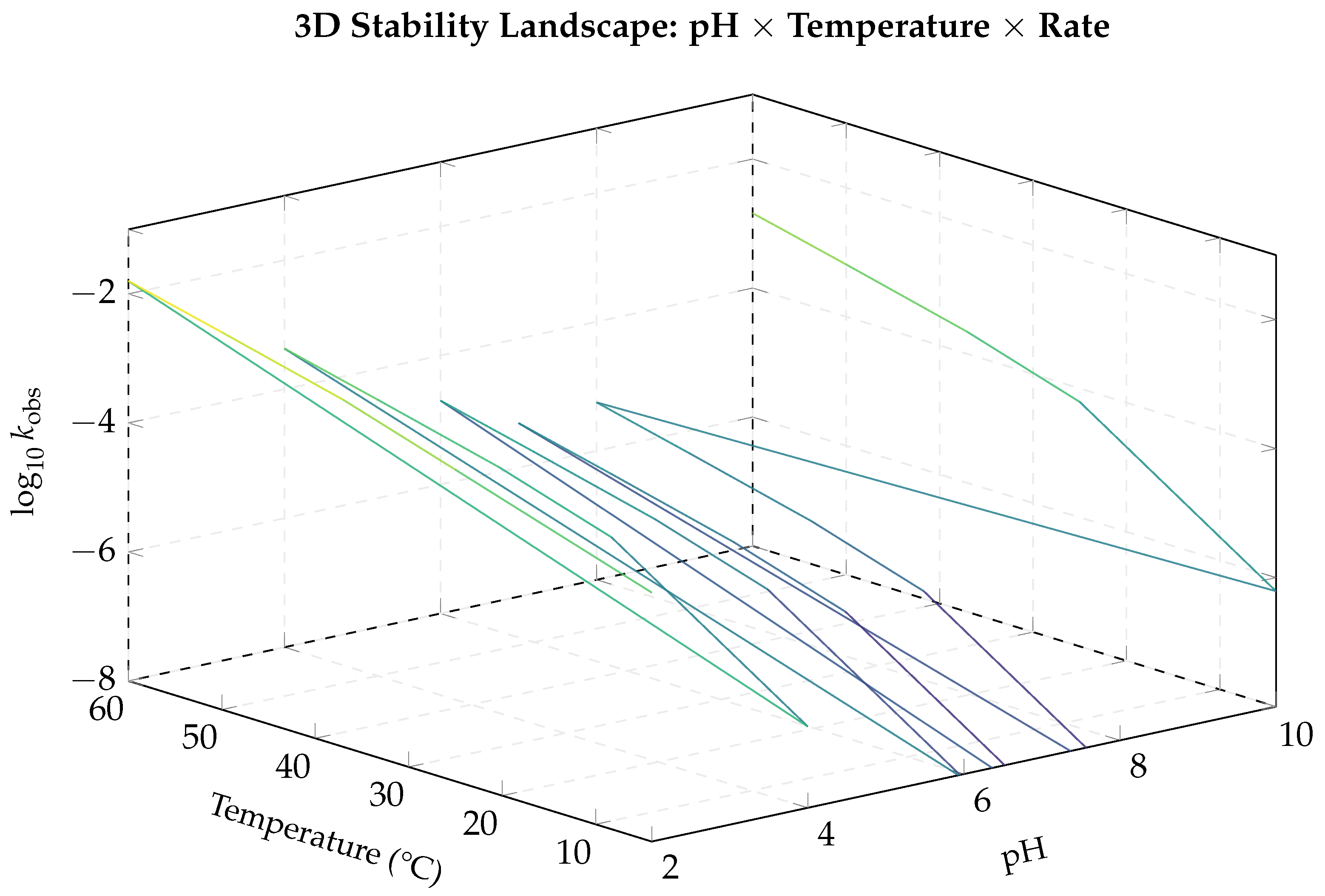
3D stability landscape showing how the breakdown rate constant () varies simultaneously with pH and temperature. The deep valley at pH 6–8 and low temperature represents the region of maximum stability. The steep walls at low pH and high temperature indicate rapid degradation zones. This visualisation guides formulation design: the optimal operating window sits in the valley floor.
Figure 5.
3D stability landscape showing how the breakdown rate constant () varies simultaneously with pH and temperature. The deep valley at pH 6–8 and low temperature represents the region of maximum stability. The steep walls at low pH and high temperature indicate rapid degradation zones. This visualisation guides formulation design: the optimal operating window sits in the valley floor.
Figure 8.
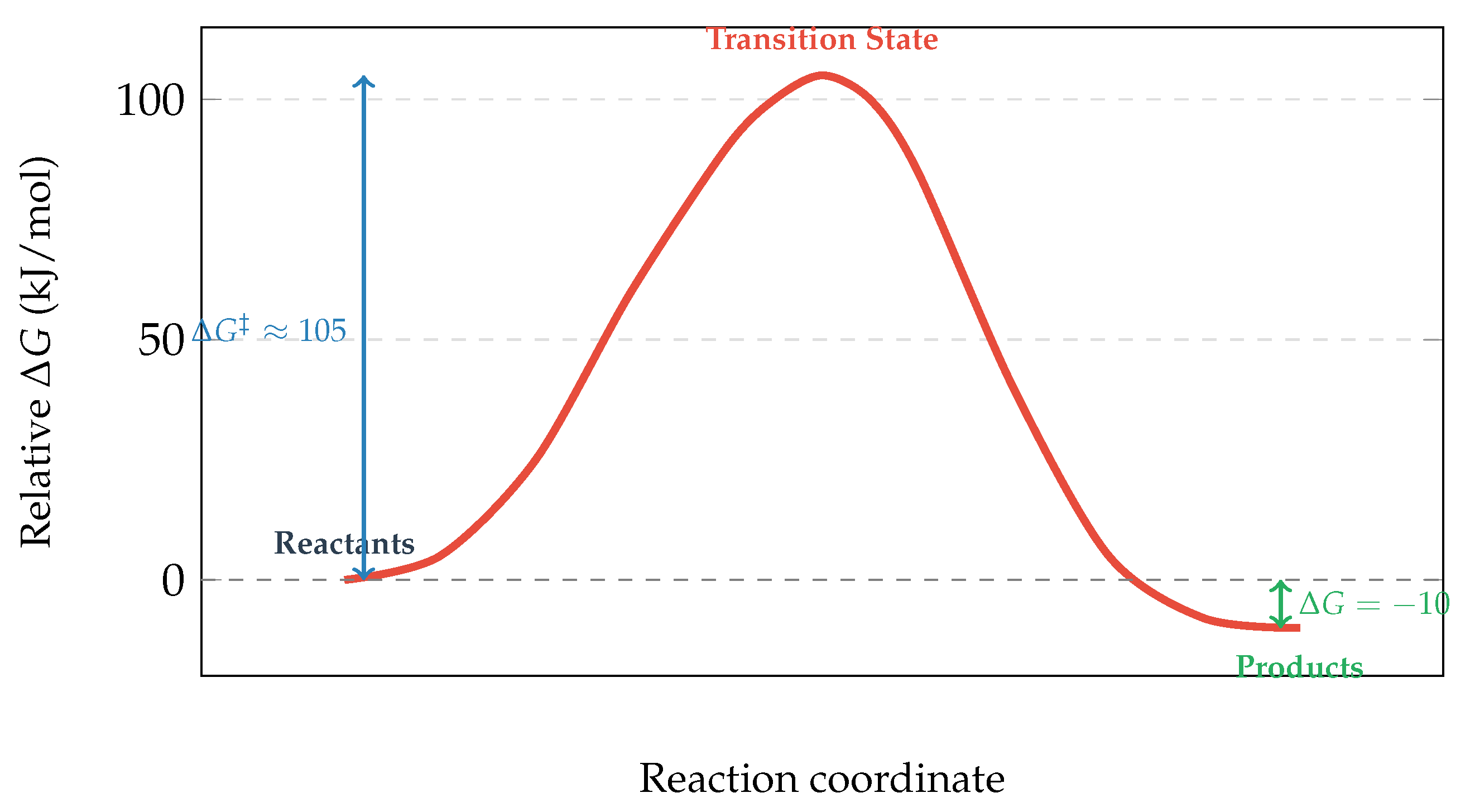
2D Gibbs free energy profile at pH 7.4, 25°C. The large barrier ( kJ/mol) provides kinetic stability despite thermodynamic favourability ( kJ/mol).
Figure 8.
2D Gibbs free energy profile at pH 7.4, 25°C. The large barrier ( kJ/mol) provides kinetic stability despite thermodynamic favourability ( kJ/mol).
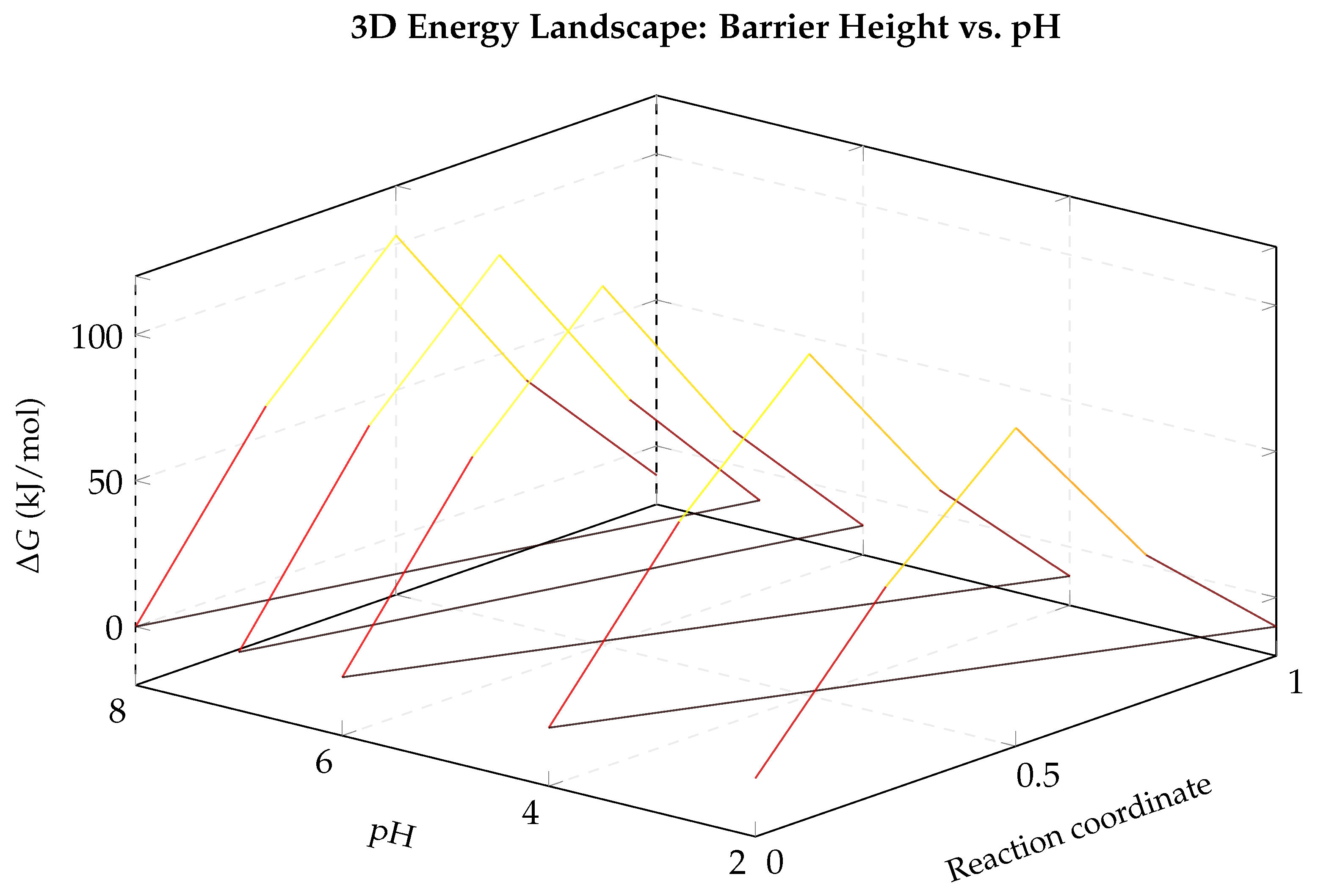
Figure 9.
3D energy landscape showing how the activation barrier varies with both reaction progress and pH. At low pH (front face), the barrier is lower (89 kJ/mol), enabling faster hydrolysis. At neutral pH (back face), the barrier rises to 105 kJ/mol, providing kinetic stability. All pathways lead to the same product valley at kJ/mol.
Figure 9.
3D energy landscape showing how the activation barrier varies with both reaction progress and pH. At low pH (front face), the barrier is lower (89 kJ/mol), enabling faster hydrolysis. At neutral pH (back face), the barrier rises to 105 kJ/mol, providing kinetic stability. All pathways lead to the same product valley at kJ/mol.
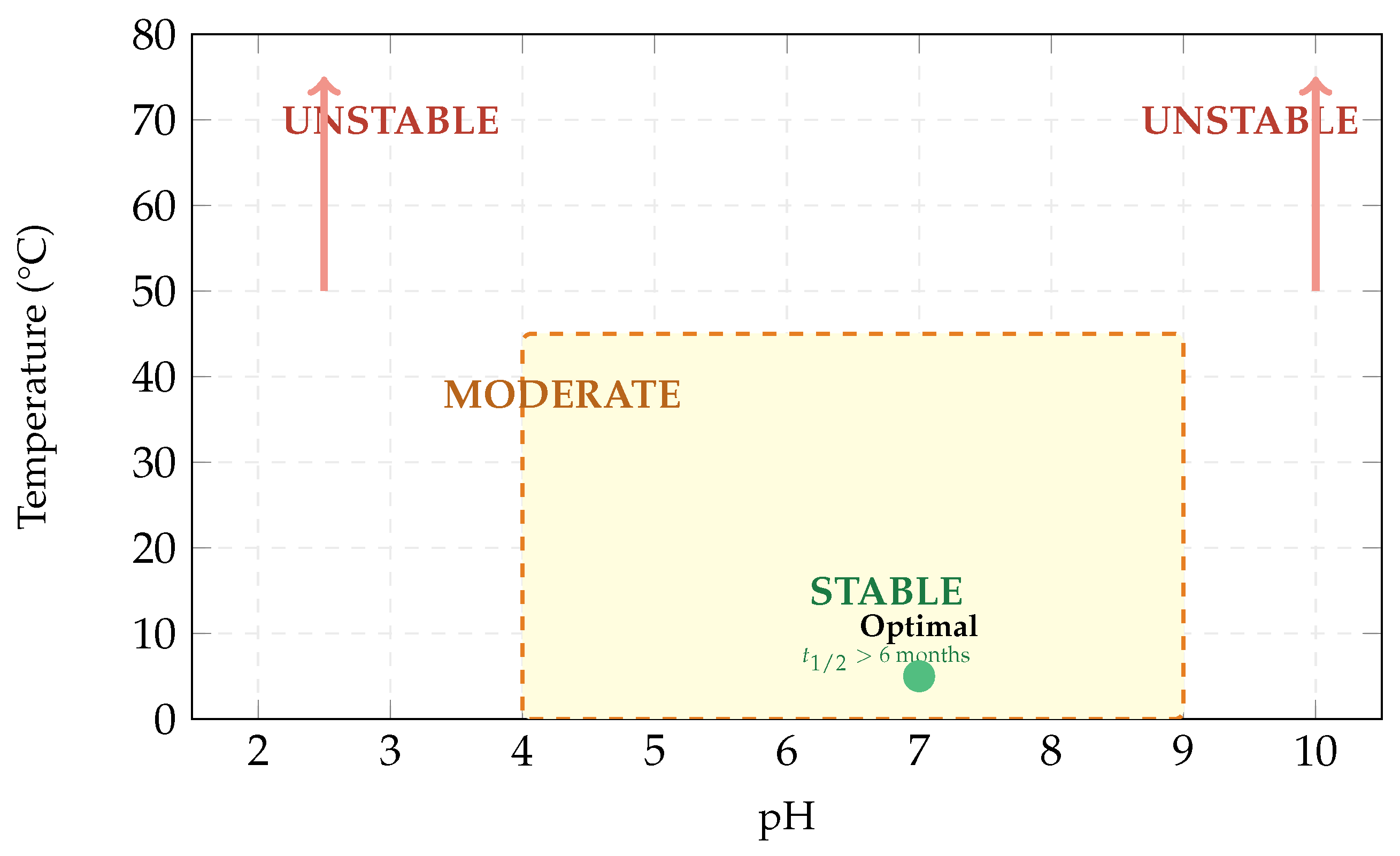
Figure 13.
2D stability map. Green zone: stable ( months). Yellow dashed: moderate. Outside: unstable.
Figure 13.
2D stability map. Green zone: stable ( months). Yellow dashed: moderate. Outside: unstable.
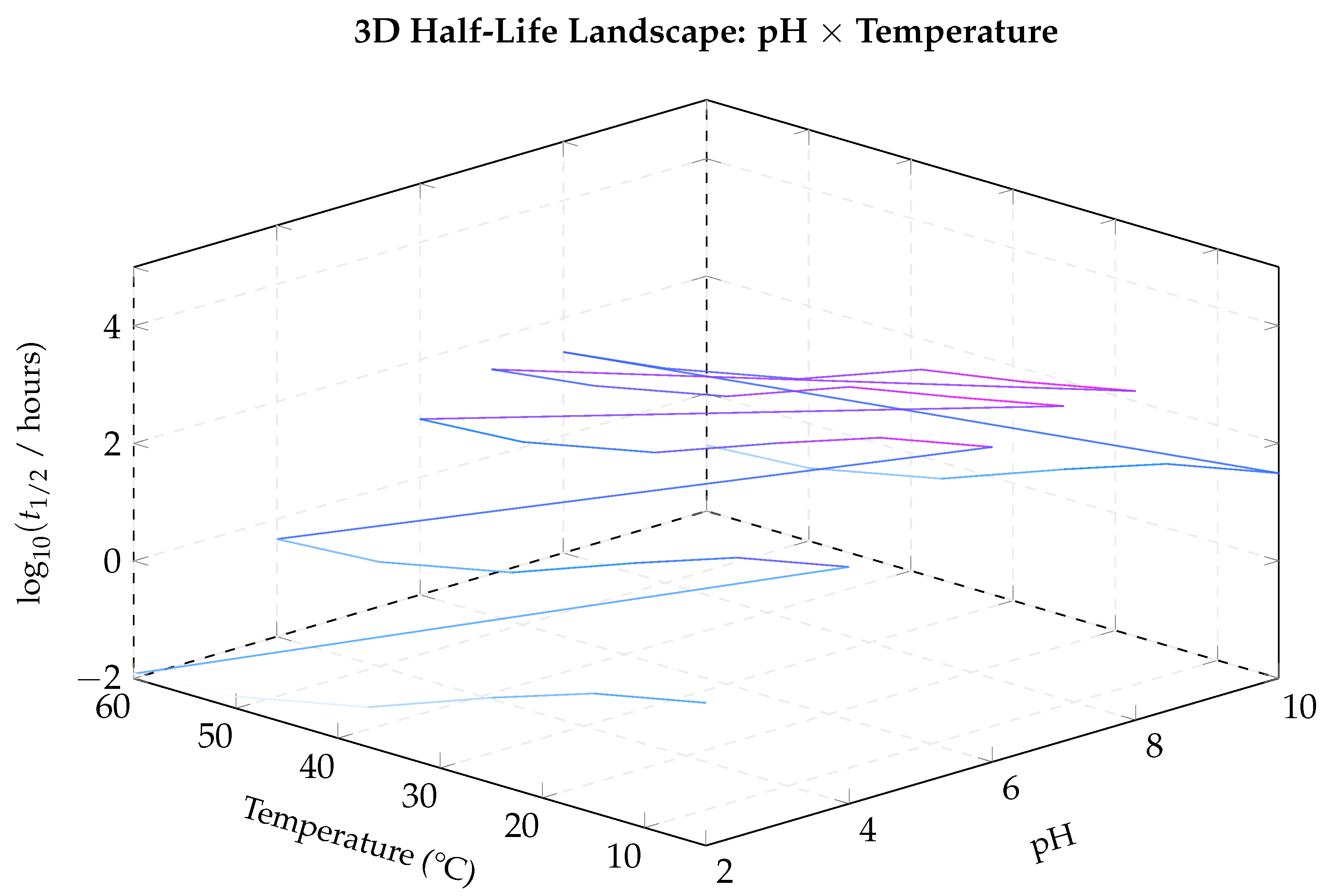
Figure 14.
3D half-life landscape. The elevated plateau at neutral pH and low temperature represents the long-lived stability zone (peak: , equivalent to ∼5000 hours). The steep drop-off at low pH or high temperature shows how quickly stability is lost outside the optimal window.
Figure 14.
3D half-life landscape. The elevated plateau at neutral pH and low temperature represents the long-lived stability zone (peak: , equivalent to ∼5000 hours). The steep drop-off at low pH or high temperature shows how quickly stability is lost outside the optimal window.
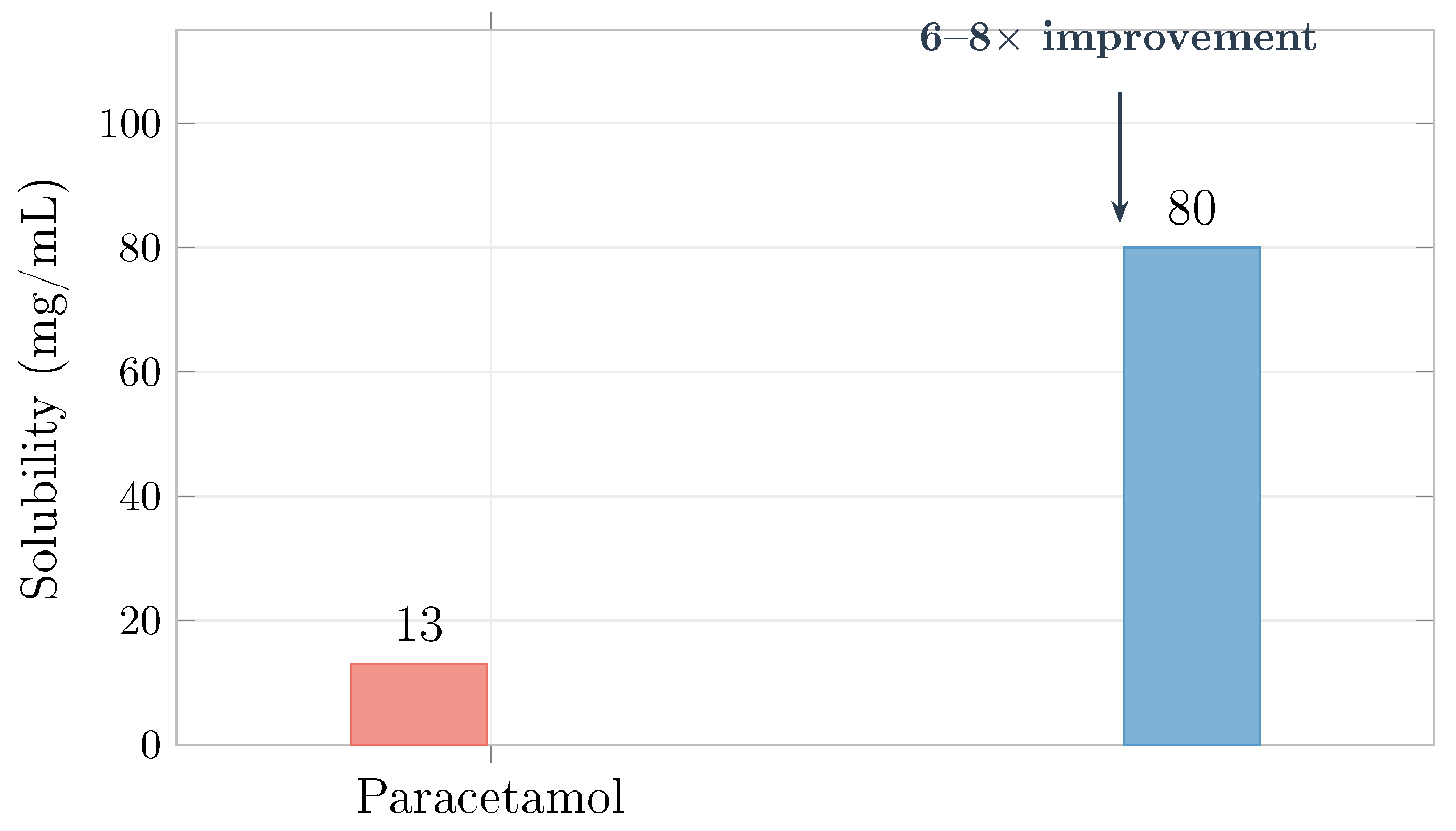
Figure 15.
Aqueous solubility at 25°C. Glucose attachment yields a 6–8-fold improvement, eliminating the dissolution bottleneck that limits conventional paracetamol formulations.
Figure 15.
Aqueous solubility at 25°C. Glucose attachment yields a 6–8-fold improvement, eliminating the dissolution bottleneck that limits conventional paracetamol formulations.
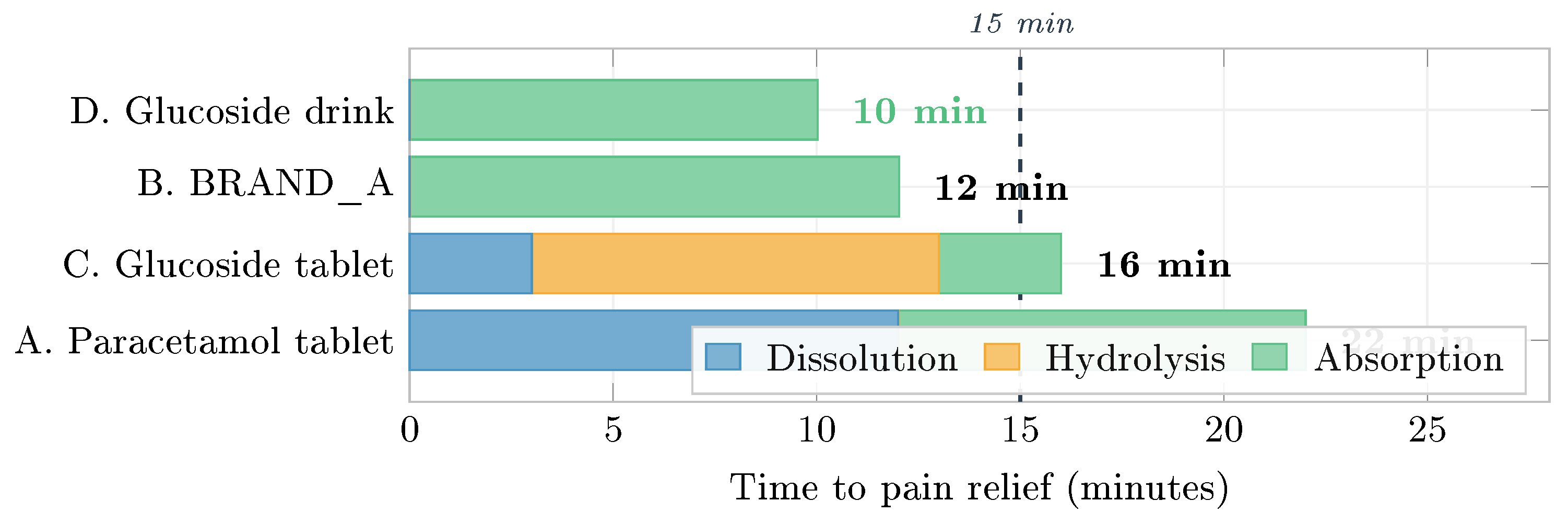
Figure 16.
Timeline to pain-relief onset (500 mg paracetamol-equivalent). Each bar decomposes into dissolution, hydrolysis, and absorption to onset. Pathway D eliminates both dissolution and hydrolysis ex vivo, achieving pain control in ∼10 min. Dashed line marks 15 min.
Figure 16.
Timeline to pain-relief onset (500 mg paracetamol-equivalent). Each bar decomposes into dissolution, hydrolysis, and absorption to onset. Pathway D eliminates both dissolution and hydrolysis ex vivo, achieving pain control in ∼10 min. Dashed line marks 15 min.
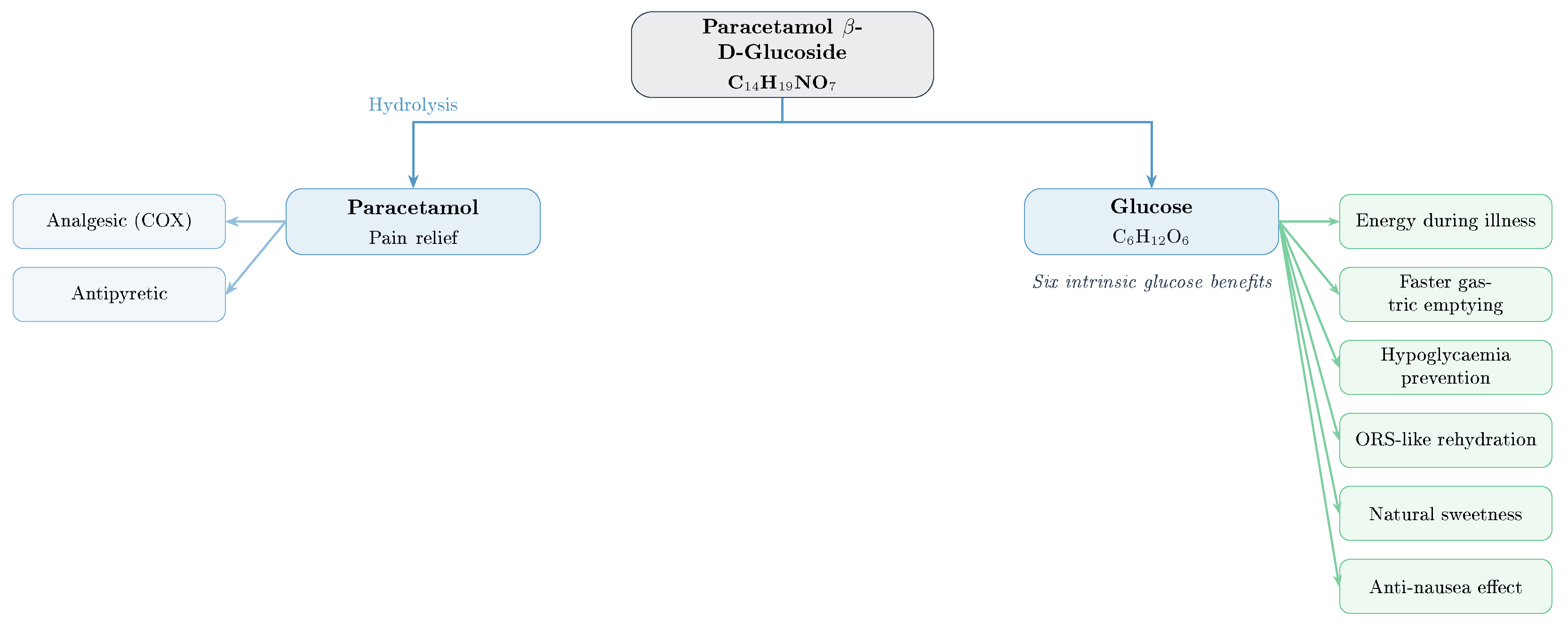
Figure 17.
Dual-action therapeutic architecture. Hydrolysis yields paracetamol (analgesic/antipyretic) and glucose (six intrinsic therapeutic benefits). Unlike conventional formulations where sugar is an optional excipient, glucose is a stoichiometric product of prodrug activation.
Figure 17.
Dual-action therapeutic architecture. Hydrolysis yields paracetamol (analgesic/antipyretic) and glucose (six intrinsic therapeutic benefits). Unlike conventional formulations where sugar is an optional excipient, glucose is a stoichiometric product of prodrug activation.
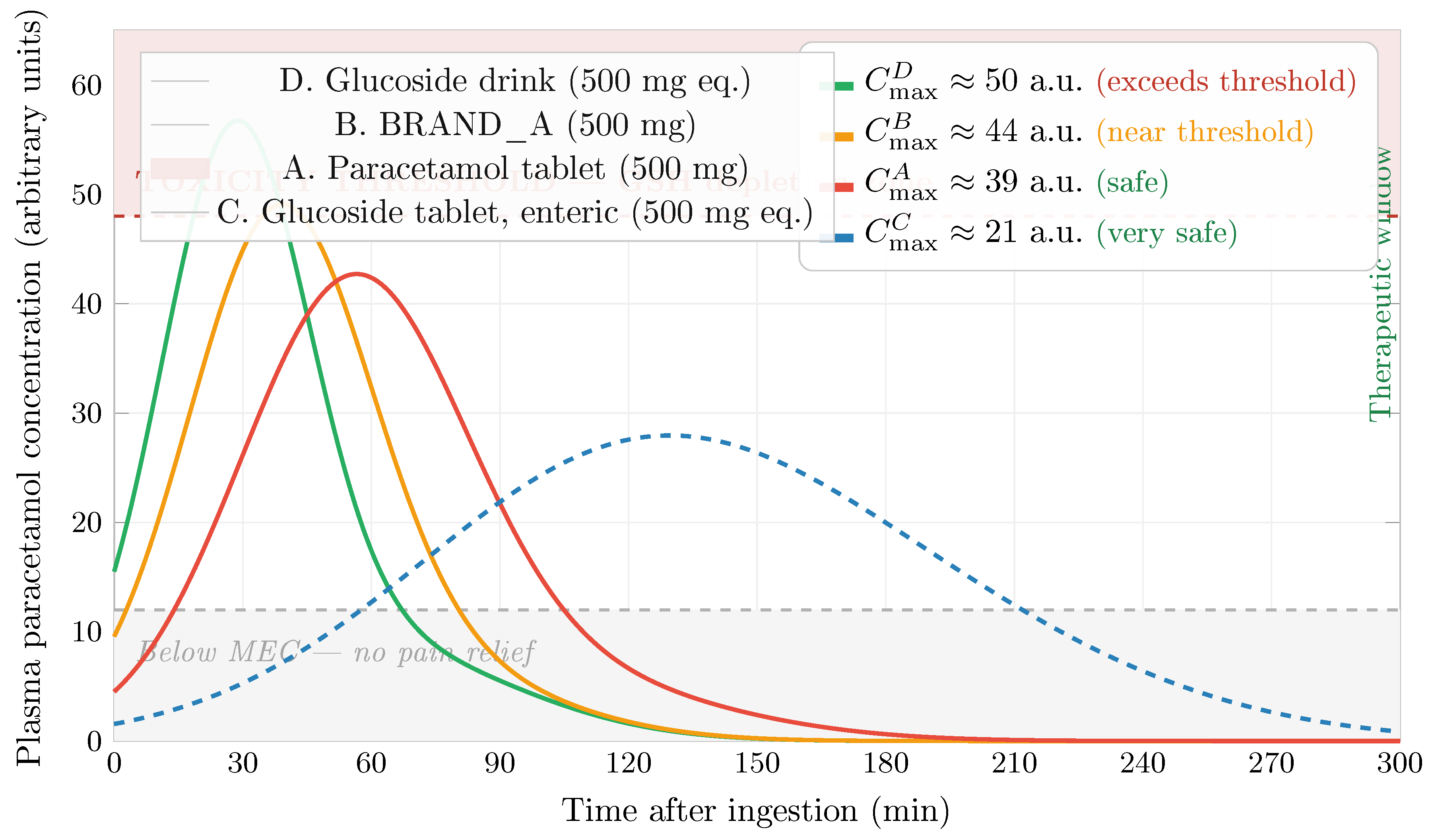
Figure 18.
Schematic plasma concentration–time profiles at 500 mg paracetamol-equivalent dose (illustrative; clinical validation pending). The shaded red zone above 48 a.u. represents the region where NAPQI generation may exceed glutathione (GSH) detoxification capacity. At 500 mg, Pathway D () breaches the toxicity threshold, Pathway B () approaches it, while Pathways A and C remain safely within the therapeutic window. MEC = minimum effective concentration.
Figure 18.
Schematic plasma concentration–time profiles at 500 mg paracetamol-equivalent dose (illustrative; clinical validation pending). The shaded red zone above 48 a.u. represents the region where NAPQI generation may exceed glutathione (GSH) detoxification capacity. At 500 mg, Pathway D () breaches the toxicity threshold, Pathway B () approaches it, while Pathways A and C remain safely within the therapeutic window. MEC = minimum effective concentration.
Table 1.
Aqueous solubility comparison at 25°C.
Table 1.
Aqueous solubility comparison at 25°C.
| Property |
Paracetamol |
Glucoside |
| Solubility (mg/mL) |
12–14 |
75–85 |
| Solubility (mM) |
79–93 |
225–256 |
| Fold improvement |
1.0× |
6–8× |
Table 2.
Pseudo-first-order rate constants and half-lives as a function of pH (25°C).
Table 2.
Pseudo-first-order rate constants and half-lives as a function of pH (25°C).
| pH |
(s−1) |
Half-life |
Stability |
| 2.0 |
|
17 min |
Very poor |
| 3.0 |
|
1.75 h |
Poor |
| 4.0 |
|
10.7 h |
Moderate |
| 5.0 |
|
60 h |
Good |
| 6.0 |
|
227 h |
Excellent |
| 7.0 |
|
914 h |
Excellent |
| 7.4 |
|
1078 h |
Excellent |
| 8.0 |
|
801 h |
Excellent |
| 9.0 |
|
111 h |
Good |
| 10.0 |
|
3.1 h |
Poor |
Table 3.
Temperature dependence of hydrolysis at pH 2.0.
Table 3.
Temperature dependence of hydrolysis at pH 2.0.
| Temp. (°C) |
(s−1) |
Half-life |
Q10
|
| 4 |
|
2.67 h |
— |
| 15 |
|
1.07 h |
2.5 |
| 25 |
|
17 min |
3.8 |
| 37 |
|
4.8 min |
3.5 |
| 50 |
|
1.6 min |
3.0 |
| 60 |
|
43 s |
2.3 |
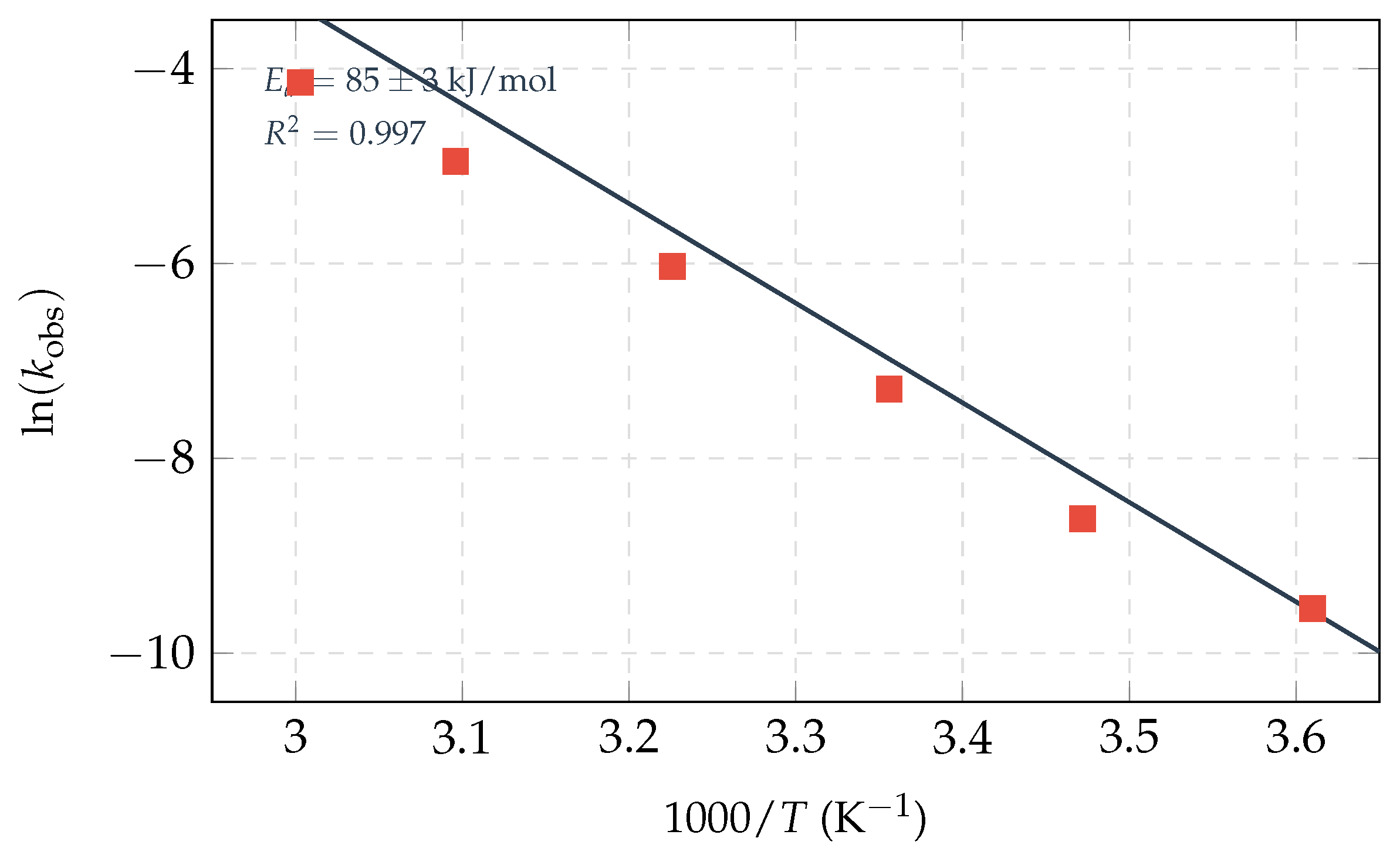
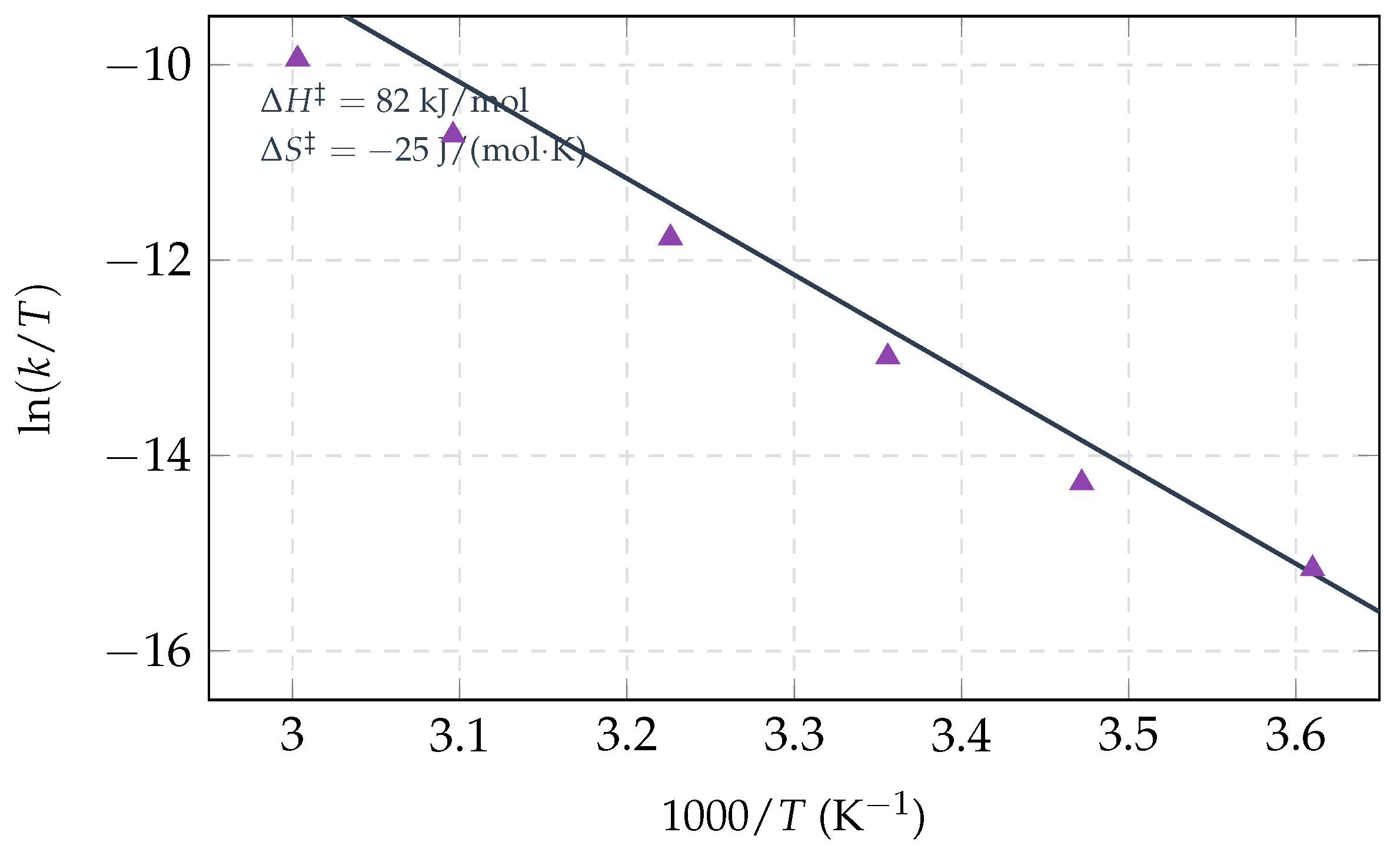
Table 4.
Thermodynamic activation parameters (pH 2.0, acid-catalyzed hydrolysis).
Table 4.
Thermodynamic activation parameters (pH 2.0, acid-catalyzed hydrolysis).
| Parameter |
Value |
Physical meaning |
|
kJ/mol |
Energy input to reach transition state |
|
J/(molK) |
Transition state is more ordered |
|
kJ/mol |
Total energy barrier at 25°C |
Table 5.
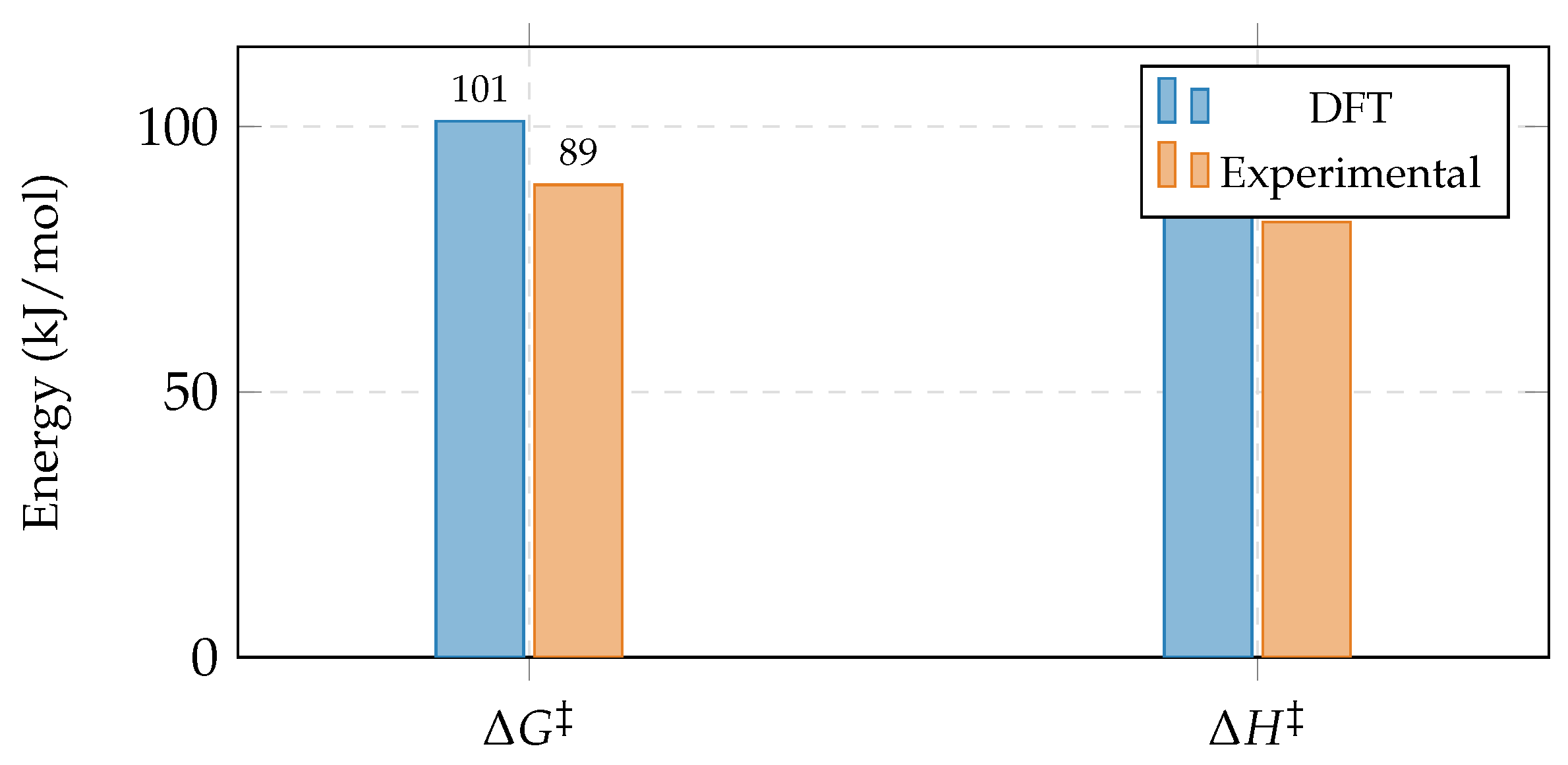
Comparison of DFT-calculated and experimental activation parameters.
Table 5.
Comparison of DFT-calculated and experimental activation parameters.
| Parameter |
DFT (B3LYP) |
Experimental |
Deviation |
|
(kJ/mol) |
|
|
+13% |
|
(kJ/mol) |
|
|
+16% |
|
(J/(molK)) |
|
|
+12% |
| Bond length (C–O, TS) |
2.05 Å |
— |
— |
| Charge on C1 (TS) |
+0.52 e |
— |
— |
Table 6.
Michaelis-Menten parameters for -glucosidase hydrolysis (pH 7.0, 37°C).
Table 6.
Michaelis-Menten parameters for -glucosidase hydrolysis (pH 7.0, 37°C).
| Parameter |
Almond enzyme |
Human recombinant |
|
(mM) |
|
|
|
(mol/min/mg) |
|
|
|
(s−1) |
|
|
|
(M−1s−1) |
|
|
| Rate enhancement |
-fold vs. uncatalyzed |
Table 7.
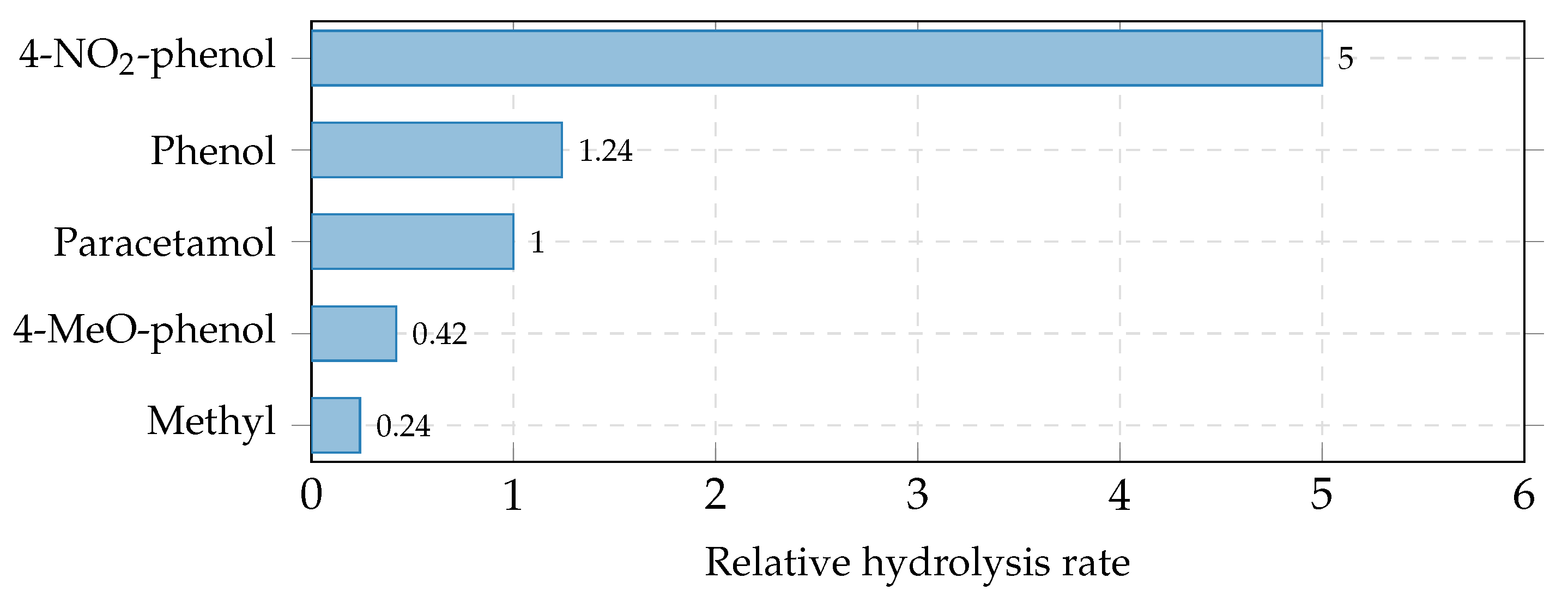
Hydrolysis rates of structurally related glucosides (pH 2.0, 37°C).
Table 7.
Hydrolysis rates of structurally related glucosides (pH 2.0, 37°C).
| Compound |
(M−1s−1) |
Relative rate |
| Methyl -D-glucoside |
|
0.24 |
| 4-Methoxyphenol glucoside |
|
0.42 |
| Paracetamol glucoside |
|
1.00 |
| Phenol glucoside |
|
1.24 |
| 4-Nitrophenol glucoside |
|
5.00 |
Table 9.
Regulatory compliance mapping for paracetamol -D-glucoside development.
Table 9.
Regulatory compliance mapping for paracetamol -D-glucoside development.
| Standard |
Scope |
Application in this study |
| ICH Q1A(R2) |
Stability testing of new drug substances |
Long-term (25°C/60% RH, 12 mo), intermediate (30°C/65% RH, 6 mo), and accelerated (40°C/75% RH, 6 mo) conditions applied |
| ICH Q1B |
Photostability testing |
UV-A/visible light exposure at 1.2 million luxh applied; amber packaging recommended |
| ICH Q2(R1) |
Analytical method validation |
HPLC method validated for specificity, linearity (), accuracy (98–102%), precision (RSD < 2%), LOD, LOQ |
| ICH Q3B(R2) |
Impurities in new drug products |
Individual impurities < 0.5%; total impurities < 1.0% |
| ICH Q6A |
Specifications for new drug substances |
Acceptance criteria defined: assay 90–110%, degradation products, water content, pH, microbial limits |
| ICH Q8(R2) |
Pharmaceutical development |
Quality-by-design approach applied to formulation optimisation |
| ICH Q9 |
Quality risk management |
Risk assessment for pH-sensitive degradation and cold-chain failures |
| ICH Q10 |
Pharmaceutical quality system |
Stability monitoring integrated into quality system framework |
Table 10.
ICH Q1A(R2) stability study design with sampling schedule.
Table 10.
ICH Q1A(R2) stability study design with sampling schedule.
| Study type |
Temperature |
Humidity |
Duration |
Sampling points |
| Long-term |
25 ± 2°C |
60 ± 5% RH |
12 months |
0, 3, 6, 9, 12 mo |
| Intermediate |
30 ± 2°C |
65 ± 5% RH |
6 months |
0, 3, 6 mo |
| Accelerated |
40 ± 2°C |
75 ± 5% RH |
6 months |
0, 1, 2, 3, 6 mo |
Table 11.
HPLC analytical method validation summary per ICH Q2(R1).
Table 11.
HPLC analytical method validation summary per ICH Q2(R1).
| Validation parameter |
Criterion |
Result |
| Specificity |
Baseline resolution from degradants |
Achieved () |
| Linearity range |
10–200% of target |
|
| Accuracy (recovery) |
98–102% |
99.3–101.1% |
| Precision (intra-day) |
RSD < 2% |
RSD = 0.8% |
| Precision (inter-day) |
RSD < 2% |
RSD = 1.2% |
| LOD |
— |
0.1 g/mL |
| LOQ |
— |
0.3 g/mL |
| Robustness |
±2% organic, ±0.2 pH |
Meets criteria |
Table 12.
Pharmacopoeial compliance assessment.
Table 12.
Pharmacopoeial compliance assessment.
| Pharmacopoeia |
Applicable test |
Status and notes |
| USP 〈1225〉 |
Method validation |
HPLC method meets all USP requirements for specificity, linearity, accuracy, and precision |
| USP 〈621〉 |
Chromatography |
C18 method compliant with system suitability criteria (, 0.8–1.5) |
| USP 〈1150〉 |
Pharmaceutical stability |
Stability-indicating method confirmed by forced degradation |
| BP Appendix XII |
Stability testing |
Conditions and acceptance criteria aligned with BP requirements |
| Ph. Eur. 2.2.29 |
UV/Vis spectrophotometry |
nm confirmed; Beer-Lambert linear range validated |
| Ph. Eur. 5.4 |
Residual solvents |
Pyridine (Class 2): limit 200 ppm; ethanol (Class 3): limit 5000 ppm |
Table 14.
Forced degradation study results and mass balance.
Table 14.
Forced degradation study results and mass balance.
| Stress |
Conditions |
Duration |
Degradation |
Mass balance |
Products identified |
| Acid |
pH 2.0, 37°C |
2 h |
28% |
97.3% |
Paracetamol + glucose |
| Base |
pH 10.0, 37°C |
24 h |
22% |
96.8% |
Paracetamol + glucose |
| Heat |
60°C, dry |
7 d |
15% |
98.1% |
Paracetamol + glucose |
| Oxidation |
3% H2O2, RT |
24 h |
8% |
95.2% |
Quinone derivatives |
| Light |
1.2M luxh |
7 d |
5% |
97.9% |
Minor coloured products |
Table 17.
Definitions of the four oral delivery pathways analysed.
Table 17.
Definitions of the four oral delivery pathways analysed.
| Path |
Description |
Commercial analogue |
Formulation type |
|
A |
Conventional paracetamol solid tablet swallowed with water |
Pana_Brand_B, Tyl_Brand_C |
Standard compressed tablet |
|
B |
Paracetamol powder pre-dissolved in hot water before ingestion |
BRAND_A |
Sachet / powder for solution |
|
C |
Paracetamol -D-glucoside uncoated tablet swallowed with water |
Research prodrug (tablet) |
Uncoated compressed tablet |
|
D |
Paracetamol -D-glucoside sachet dissolved in hot acidified water; hydrolysis occurs ex vivo in the cup |
Research prodrug (drink) |
Sachet with citric acid |
Table 18.
Stage-by-stage comparison of the four oral delivery pathways. Shading indicates relative speed: fastest, moderate, slowest.
Table 18.
Stage-by-stage comparison of the four oral delivery pathways. Shading indicates relative speed: fastest, moderate, slowest.
| Stage |
A. Tablet |
B. BRAND_A |
C. Glucoside tablet |
D. Glucoside drink |
State before
ingestion |
Solid tablet |
Paracetamol in solution |
Solid tablet |
Paracetamol + glucose in solution |
Dissolution in
gastric fluid |
Slow: 8–15 min |
Already done |
Fast: 2–5 min |
Already done |
Conversion to
active drug |
Not required |
Not required |
min |
Already done |
Drug state at
absorption site |
Gradually dissolving |
Full solution |
Solution after hydrolysis |
Full solution |
Glucose
co-delivery |
None |
Nonea
|
Released in stomach |
Pre-released |
|
a Unless sugar is added as an excipient — not intrinsic to the chemistry. |
Table 19.
Estimated onset of pain relief for each delivery pathway at a 500 mg paracetamol-equivalent dose.
Table 19.
Estimated onset of pain relief for each delivery pathway at a 500 mg paracetamol-equivalent dose.
| Path |
Pathway |
Dissolution
(min) |
Conversion
(min) |
Est. onset
(min) |
Rank |
| A |
Paracetamol tablet |
12 |
0 |
22 |
4th |
| B |
BRAND_A (pre-dissolved) |
0 |
0 |
12 |
2nd |
| C |
Glucoside tablet |
3 |
10 |
16 |
3rd |
|
D |
Glucoside drink |
0 |
0 |
10 |
1st |
Table 20.
Stoichiometric glucose yield from paracetamol -D-glucoside hydrolysis at standard dose equivalents.
Table 20.
Stoichiometric glucose yield from paracetamol -D-glucoside hydrolysis at standard dose equivalents.
Paracetamol
dose (mg) |
Glucoside
required (mg) |
Glucose
released (mg) |
Caloric content |
| 250 |
548 |
298 |
∼1.2 kcal |
| 500 |
1096 |
596 |
∼2.4 kcal |
| 1000 |
2192 |
1192 |
∼4.8 kcal |
Table 21.
BRAND_A vs. glucoside drink: intrinsic vs. extrinsic formulation components.
Table 21.
BRAND_A vs. glucoside drink: intrinsic vs. extrinsic formulation components.
| Component |
B. BRAND_A |
D. Glucoside drink |
| Active drug |
Added as powder |
Released from prodrug✓ |
| Sweetness |
Artificial (aspartame / sucralose) |
Glucose from hydrolysis✓ |
| Energy provision |
Not intrinsically provided |
Glucose (∼0.3–0.6 g/dose)✓ |
| Flavour |
Synthetic lemon flavour |
Citric acid as catalyst and flavour✓ |
| Rehydration |
Water only |
Glucose-enhanced absorption (ORS)✓ |
| Artificial additives |
Required |
Minimal |
| Molecular efficiency |
1 molecule = 1 function |
1 molecule = drug + energy + sweetness + rehydration |
Table 22.
Comprehensive property comparison across all four delivery pathways. Scores on a 1–5 scale (5 = best). Bold indicates the best score in each row.
Table 22.
Comprehensive property comparison across all four delivery pathways. Scores on a 1–5 scale (5 = best). Bold indicates the best score in each row.
| Property |
A. Tablet |
B. BRAND_A |
C. Gluc. tab. |
D. Gluc. drink |
| Speed of onset |
2 |
4 |
3 |
5 |
| Energy co-delivery |
1 |
1 |
3 |
5 |
| Rehydration support |
1 |
3 |
1 |
5 |
| Nausea tolerance |
2 |
3 |
2 |
4 |
| Palatability |
2 |
3 |
2 |
5 |
| Child-friendliness |
1 |
3 |
1 |
5 |
| Convenience |
5 |
2 |
5 |
2 |
| Injectable potential |
1 |
1 |
3 |
4 |
| Controlled release |
1 |
1 |
5 |
1 |
| Shelf life |
5 |
4a
|
3 |
4b
|
| Simplicity |
5 |
3 |
4 |
3 |
| Cost |
5 |
4 |
3 |
3 |
| Total score |
31 |
32 |
35 |
46 |
|
aBRAND_A sachet shelf life; must be dissolved fresh. bGlucoside sachet: 2–3 years solid shelf life; dissolved fresh before use. |
Table 23.
Recommended dosage adjustments to maintain a.u. (20% safety margin below the toxicity threshold) for each pathway at a 500 mg reference dose.
Table 23.
Recommended dosage adjustments to maintain a.u. (20% safety margin below the toxicity threshold) for each pathway at a 500 mg reference dose.
| Path |
Pathway |
Standard
dose (mg) |
(a.u.) |
Adjusted
dose (mg) |
Safety
margin |
| D |
Glucoside drink |
500 |
50 |
380 |
20% |
| B |
BRAND_A |
500 |
44 |
430 |
20% |
| A |
Paracetamol tablet |
500 |
39 |
490 |
20% |
|
C |
Glucoside (enteric) |
500 |
21 |
500 |
>56% |
Table 24.
Recommended dosing framework for each delivery pathway at the 500 mg dose level, balancing efficacy, speed, and hepatic safety.
Table 24.
Recommended dosing framework for each delivery pathway at the 500 mg dose level, balancing efficacy, speed, and hepatic safety.
| Path |
Pathway |
Recommended
dose (mg) |
Est. onset
(min) |
Best suited for |
| D |
Glucoside drink |
380 |
∼10 |
Acute flu/fever; rapid relief |
| B |
BRAND_A |
430 |
∼12 |
Home cold/flu management |
| A |
Paracetamol tablet |
500a
|
∼22 |
Standard pain, portability |
|
C |
Glucoside (enteric) |
500 |
∼60 |
Hepatic-risk; sustained release; colonic targeting |
|
aReduce to 250 mg in patients with compromised hepatic glutathione reserves (chronic alcohol use, malnutrition, hepatic impairment). |
Table 25.
Clinical scenario–pathway mapping. The glucoside platform (Pathways C and D) covers the widest range of clinical needs.
Table 25.
Clinical scenario–pathway mapping. The glucoside platform (Pathways C and D) covers the widest range of clinical needs.
| Clinical scenario |
Optimal |
Rationale |
| Acute pain + flu symptoms |
D |
Fastest onset at 380 mg; glucose energy and rehydration |
| Fast relief on the go |
C |
Portable tablet, faster dissolution than conventional |
| Standard mild pain |
A |
Proven, lowest cost, widest availability |
| Paediatric fever |
D |
Sweet taste, glucose energy, easy to drink, rehydration |
| Elderly / poor appetite |
D |
Energy provision, rehydration, nausea reduction |
| High-dose IV / injectable |
C/D |
6–8× solubility enables concentrated solutions |
| Hepatotoxicity-risk patients |
C |
Gradual release; at <44% of threshold |
| Colonic-targeted delivery |
C |
-glucosidase in colonic bacteria |
| Sustained-release regimens |
C |
Flat plasma profile over extended duration |