Submitted:
27 February 2026
Posted:
28 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction and Clinical Significance
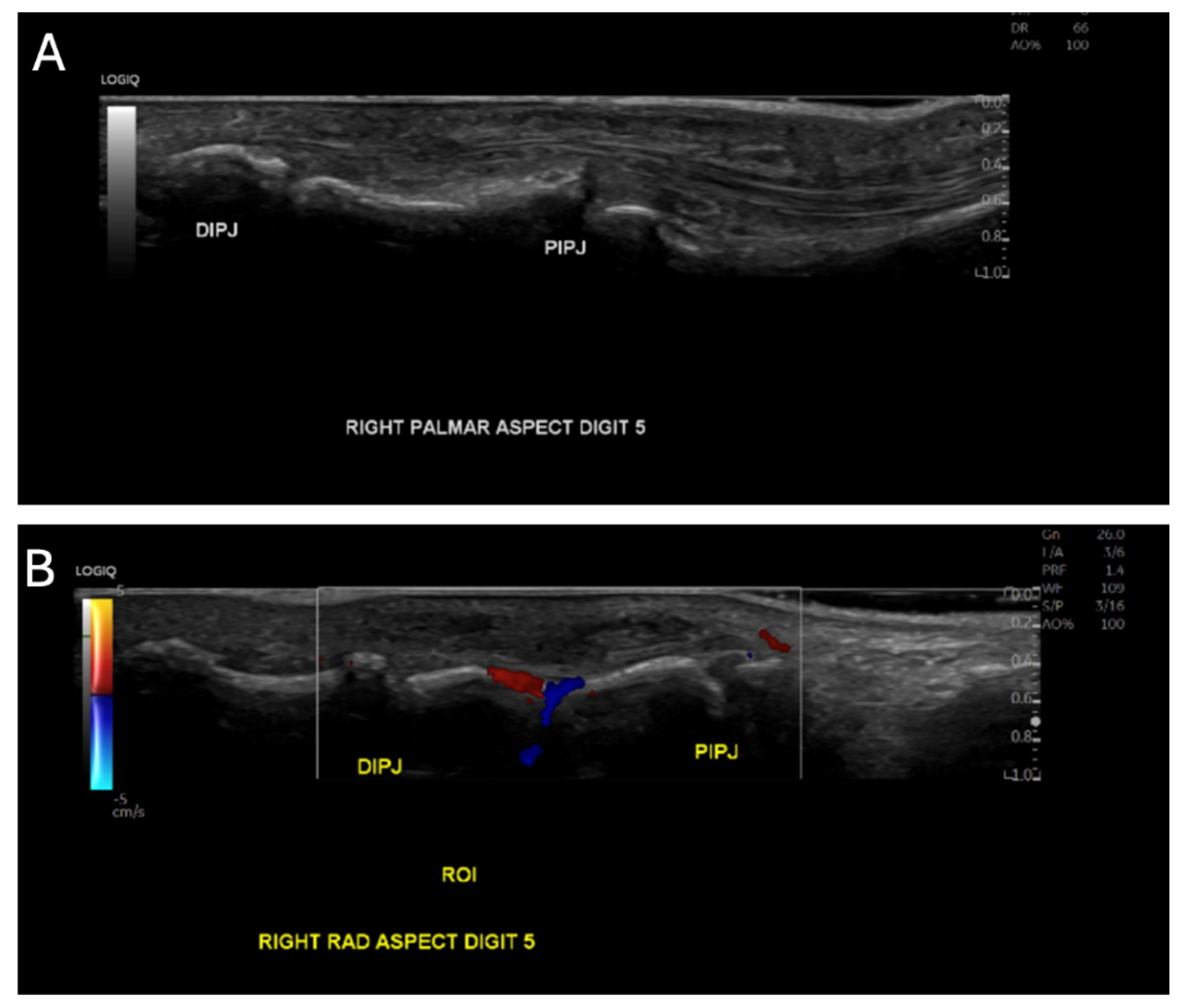
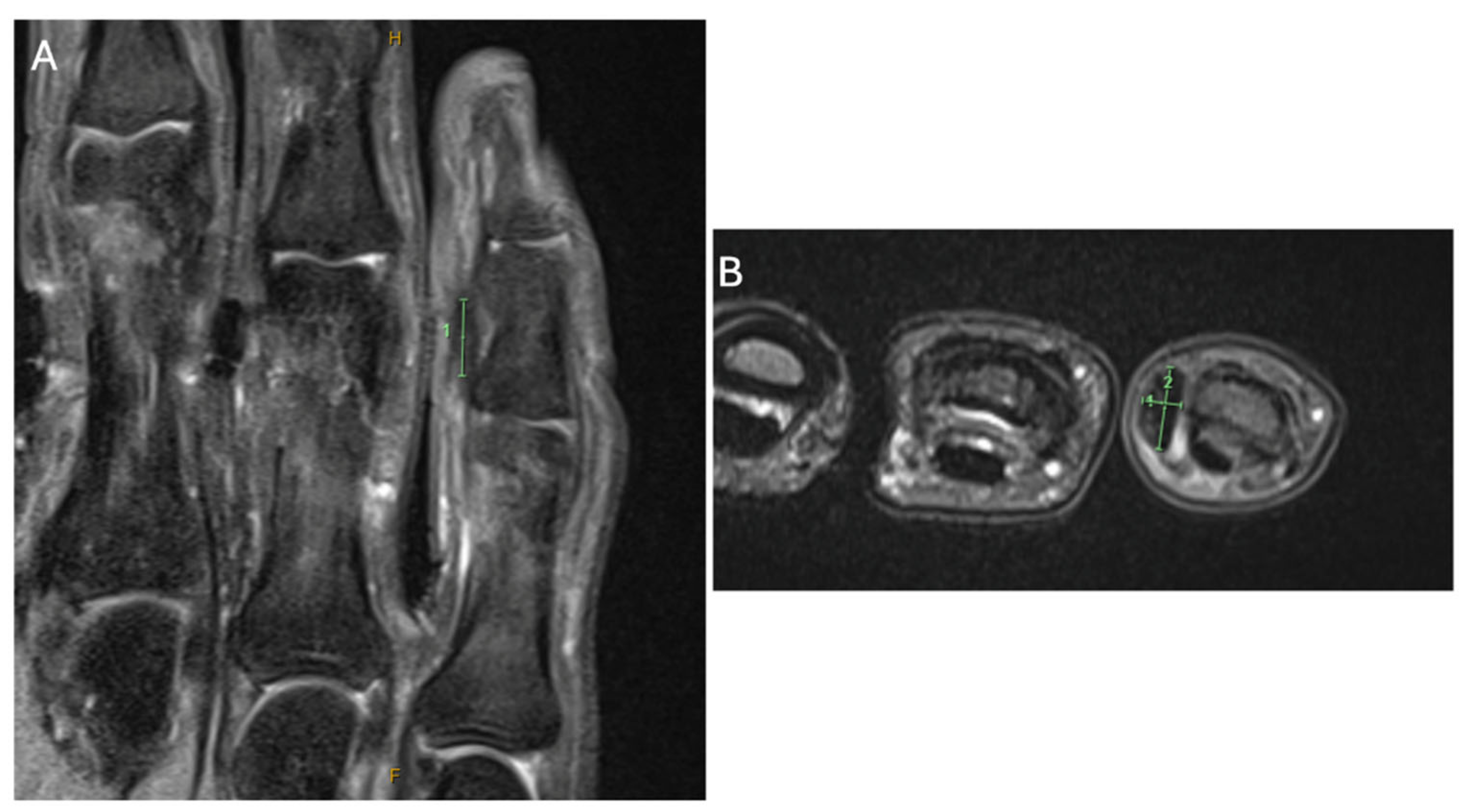
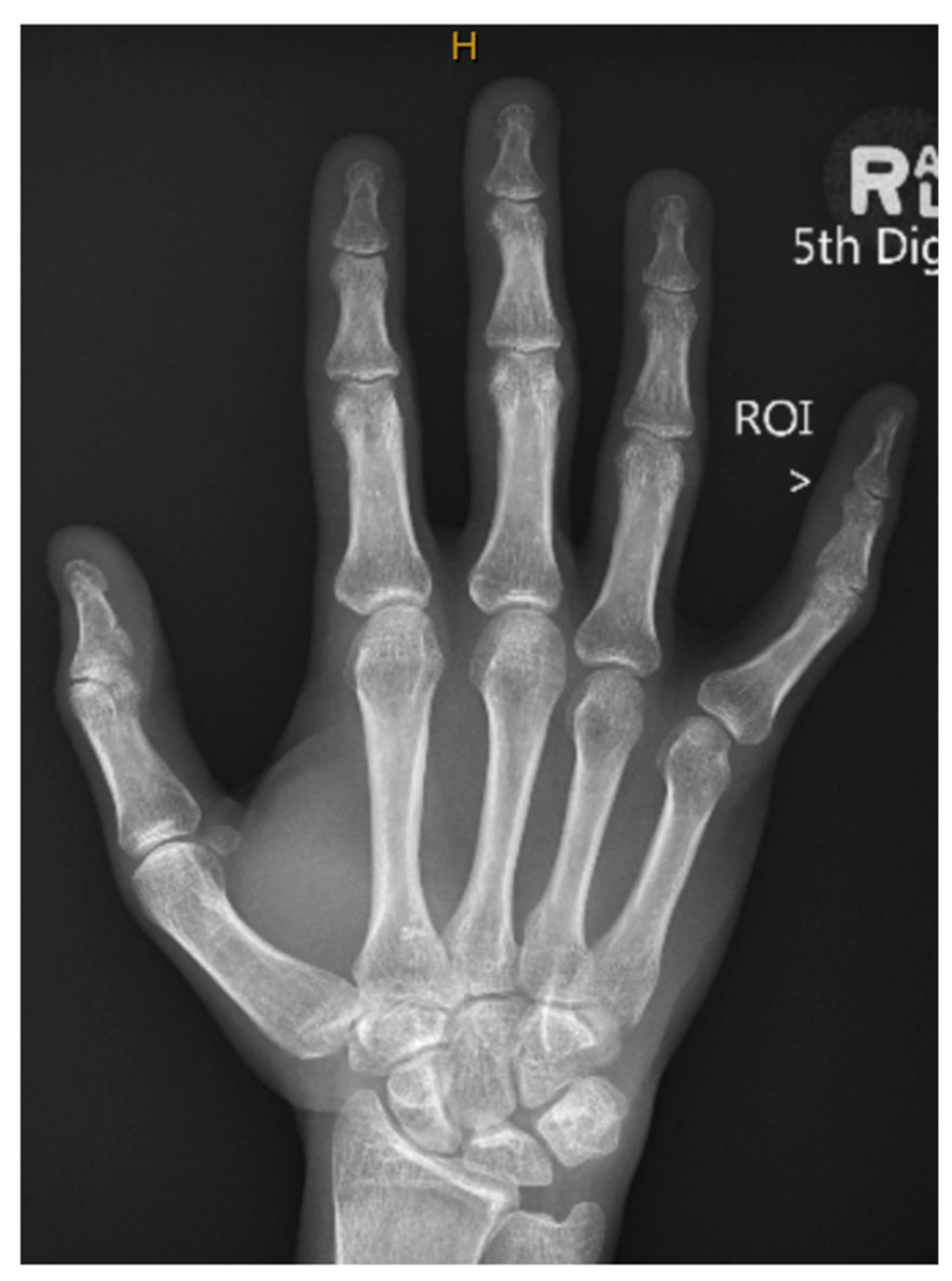
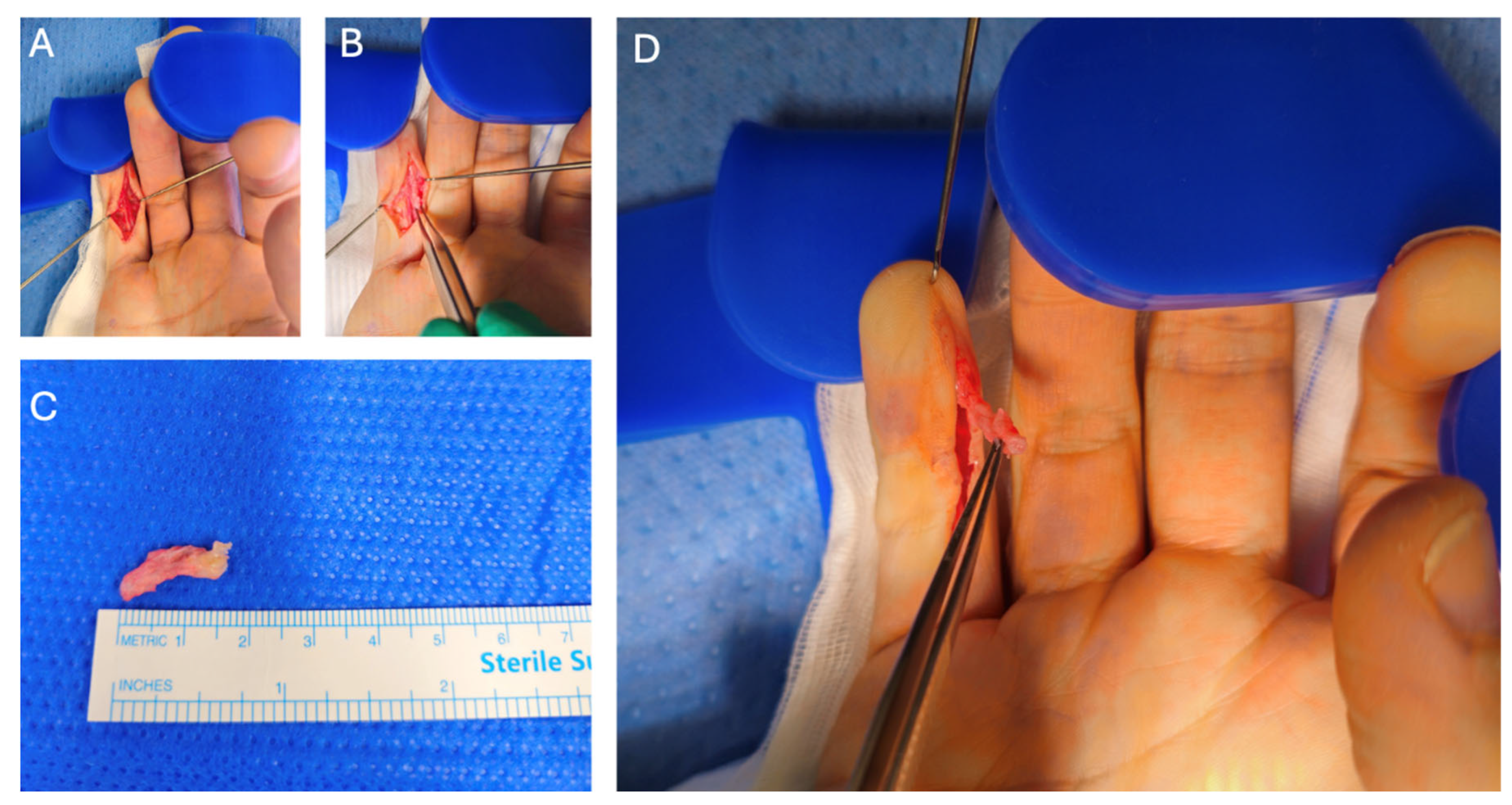
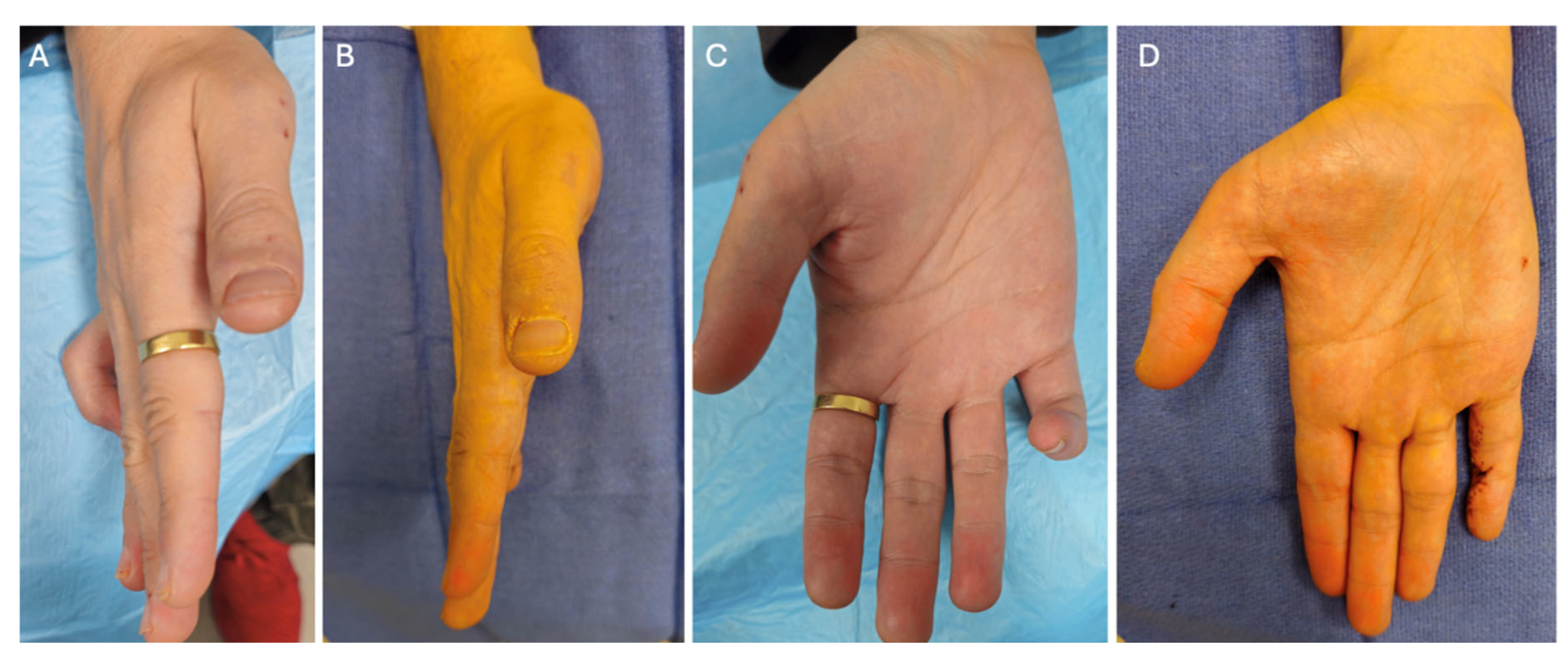
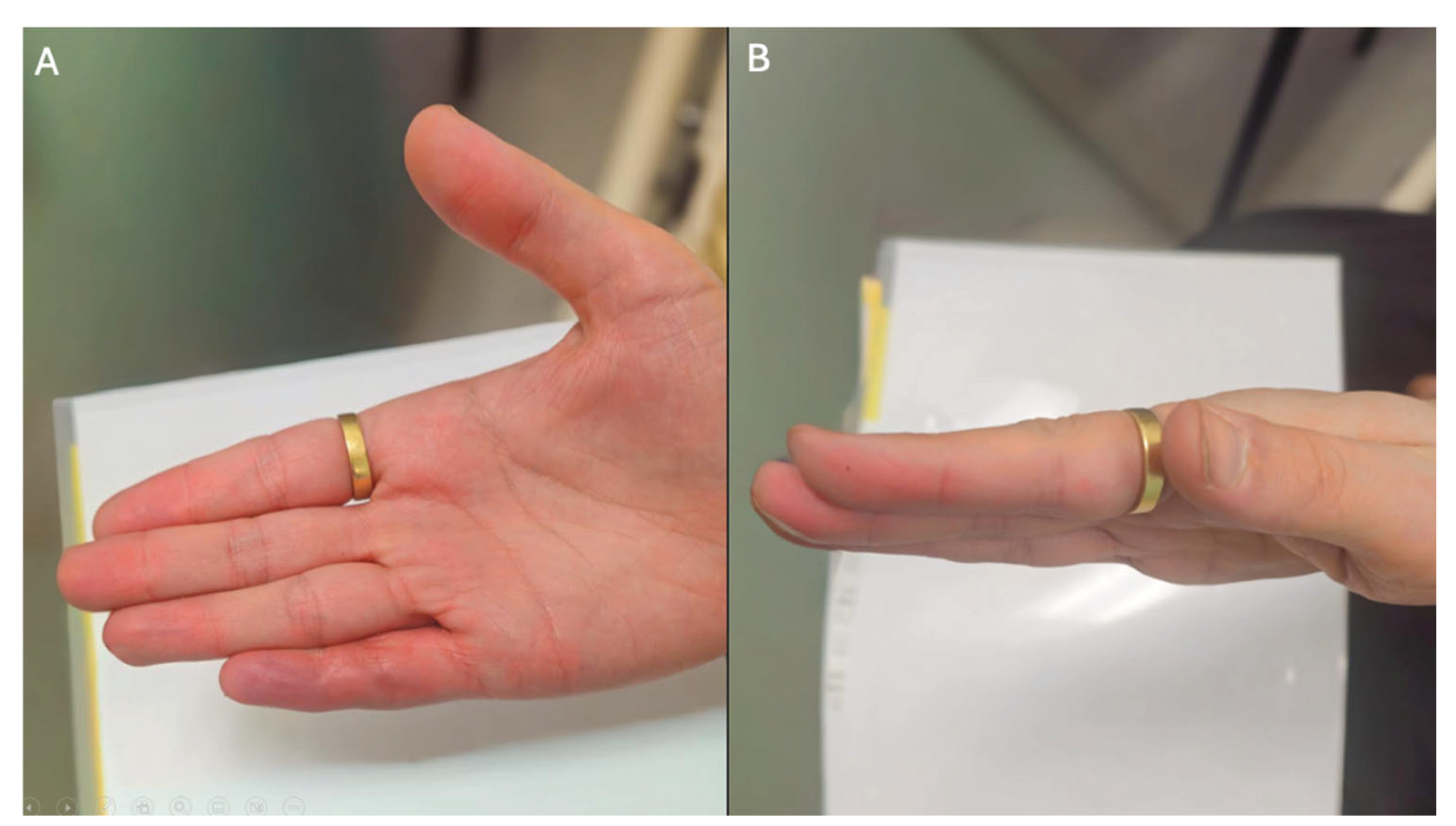
2. Case Presentation
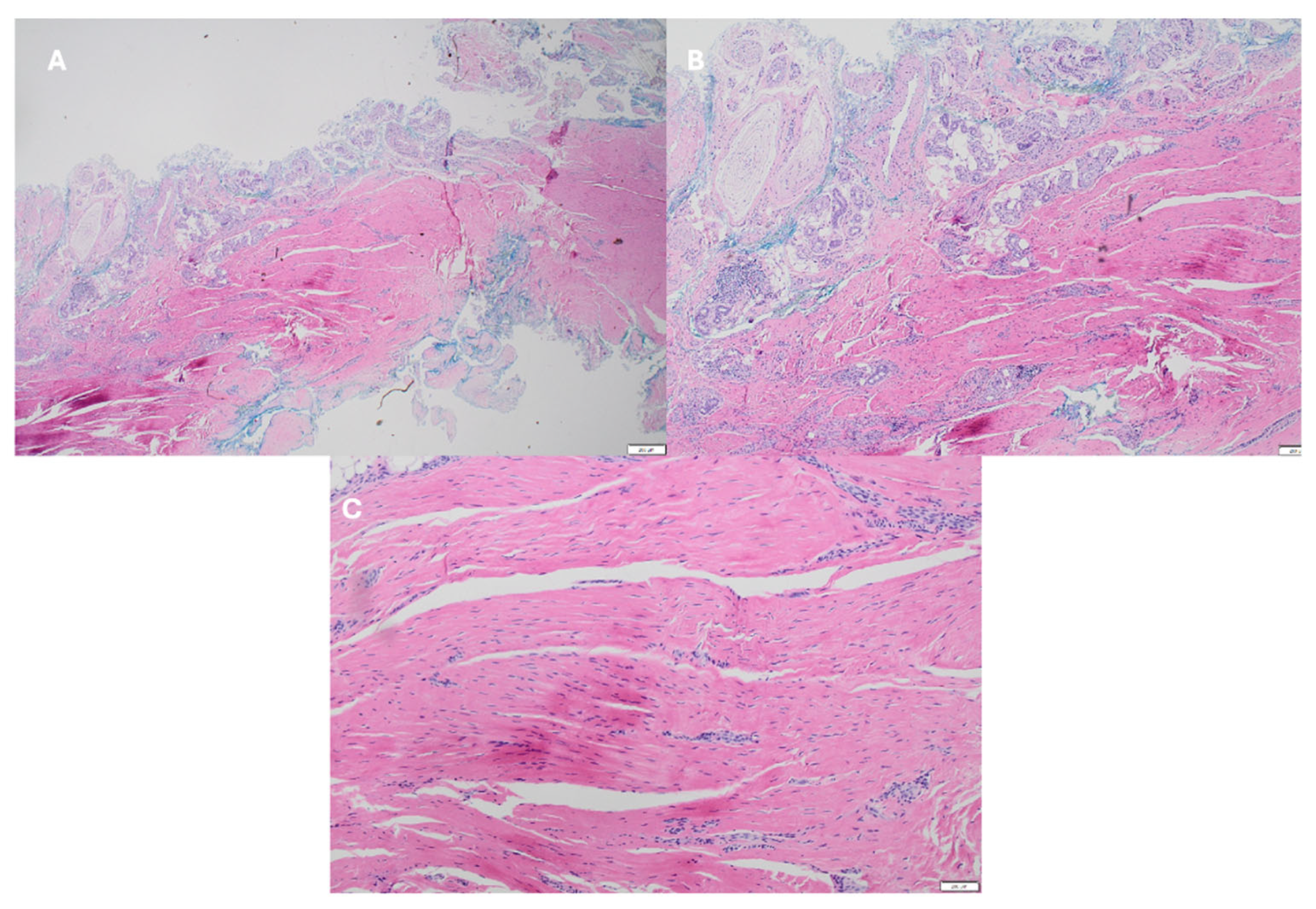
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| MDPI | Multidisciplinary Digital Publishing Institute |
| DOAJ | Directory of open access journals |
| TLA | Three letter acronym |
| LD | Linear dichroism |
References
- Hindocha S: Risk Factors, Disease Associations, and Dupuytren Diathesis. Hand Clin. 2018, 34, 307–314. [CrossRef]
- Black, EM; Blazar, PE. Dupuytren disease: an evolving understanding of an age-old disease. J Am Acad Orthop Surg. 2011, 19, 746–757. [Google Scholar] [CrossRef] [PubMed]
- Khaliq F and Orji C: Dupuytren's Contracture: A Review of the Literature. Cureus 2024, 16, 74945–20241202. [CrossRef]
- Sarkar, M; Kashyap, N; Madabhavi, I. Dupuytren’s disease: A review. Chinese Journal of Plastic and Reconstructive Surgery 2024, 6, 142–148. [Google Scholar] [CrossRef]
- Dutta, A; Jayasinghe, G; Deore, S; et al. Dupuytren’s Contracture - Current Concepts. Journal of Clinical Orthopaedics and Trauma. 2020, 11, 590–596. [Google Scholar] [CrossRef] [PubMed]
- Bellonias, EC. Nancarrow JD: Two unusual cases of distal interphalangeal joint Dupuytren's contracture. Br J Plast Surg. 1991, 44, 602–603. [Google Scholar] [CrossRef] [PubMed]
- Rayan, GM. Dupuytren disease: Anatomy, pathology, presentation, and treatment. J Bone Joint Surg Am. 2007, 89, 189–198. [Google Scholar] [CrossRef] [PubMed]
- Rao, K; Shariff, Z; Howcroft, AJ. Dupuytren's contracture of the distal interphalangeal joint: a rare presentation. J Hand Surg Br. 2006, 31, 694–695. [Google Scholar] [CrossRef] [PubMed]
- Saleh, WR; Horii, E. Hirata H: Dupuytren's disease confined to the interphalangeal joints: a case report. Hand Surg. 2009, 14, 69–71. [Google Scholar] [CrossRef] [PubMed]
- Karasoy, Ismail; Dogan, Özgür; Kati, Yusuf Alper; Akinic, Metin. Isolated Dupuytren's disease of the interphalangeal joints:a case report. Hand and Microsurgery 2012, 1, 65–67. [Google Scholar] [CrossRef]
- Mehdi, SK; King, JD; Keshtvarz, S. : Isolated Small Finger Distal Interphalangeal Joint Dupuytren's Contracture. Case Rep Orthop. 2019, 2019, 7183739–20191104. [Google Scholar] [CrossRef] [PubMed]
- Leibovic SJ: Normal and Pathologic Anatomy of Dupuytren Disease. Hand Clin. 2018, 34, 315–329. [CrossRef] [PubMed]
- Hauck, RM; Camp, L; Ehrlich, HP; et al. Pulp nonfiction: microscopic anatomy of the digital pulp space. Plast Reconstr Surg. 2004, 113, 536–539. [Google Scholar] [CrossRef] [PubMed]
- Stanley, EA; Cavallo, A. Dupuytren's contracture: involvement of the distal interphalangeal joint - a case series and anatomical dissection. ANZ Journal of Surgery 2020, 90, 91–92. [Google Scholar] [CrossRef] [PubMed]
- Simón-Pérez, C; Rodríguez-Mateos, JI; Maestro, IA; et al. Long-term recurrence of Dupuytren's disease treated with clostridium histolitycum collagenase. Surgical treatment and anatomopathological study. Arch Orthop Trauma Surg. 2024, 144, 2085–2091. [Google Scholar] [CrossRef] [PubMed]
- Hindocha, S; Stanley, JK; Watson, JS. Bayat A: Revised Tubiana's staging system for assessment of disease severity in Dupuytren's disease-preliminary clinical findings. Hand (N Y 2008, 3, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Jung, J; Kim, GW; Lee, B; et al. Integrative genomic and transcriptomic analysis of genetic markers in Dupuytren’s disease. BMC Medical Genomics 2019, 12, 98. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



