Submitted:
19 February 2026
Posted:
28 February 2026
You are already at the latest version
Abstract
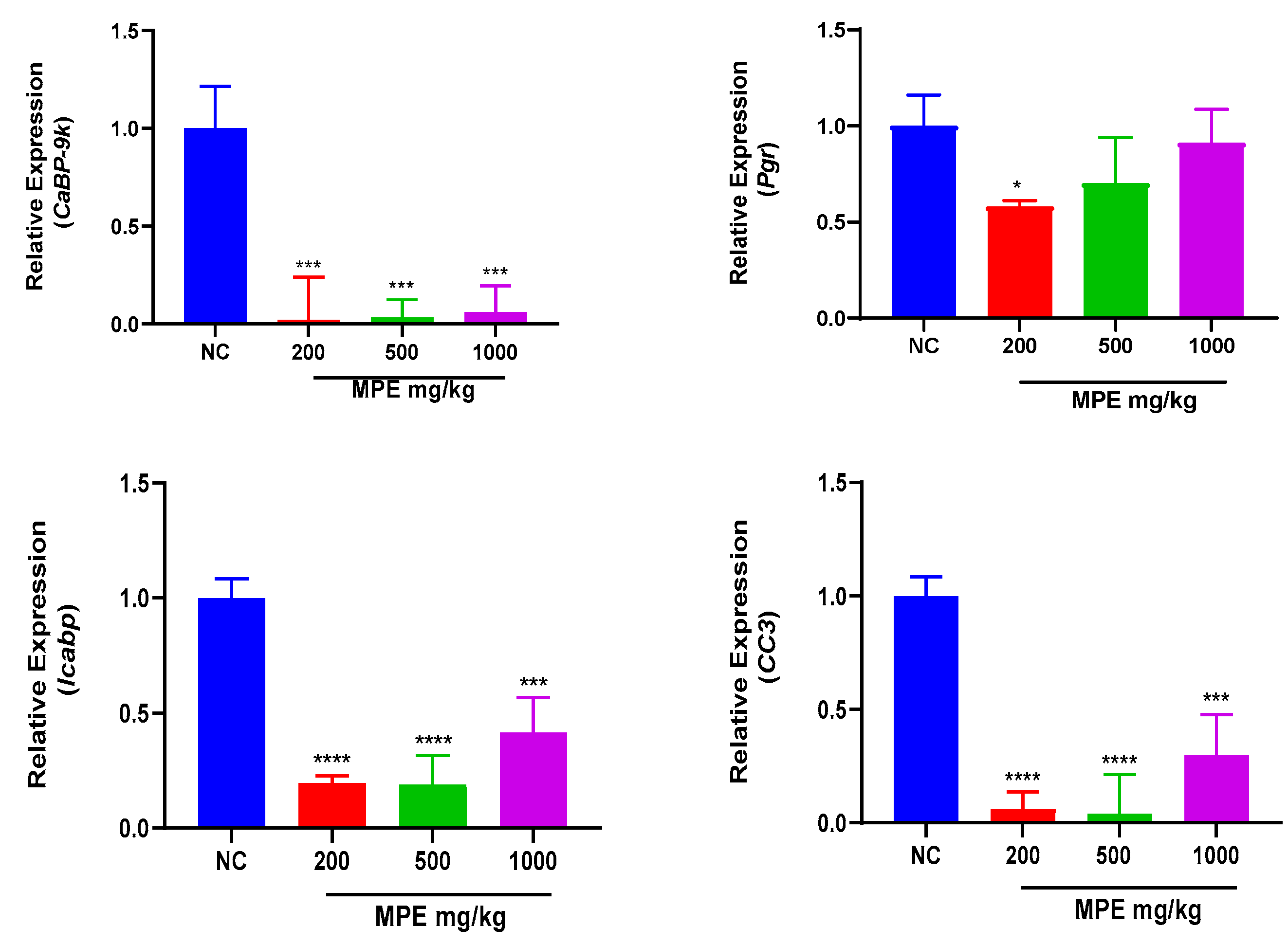
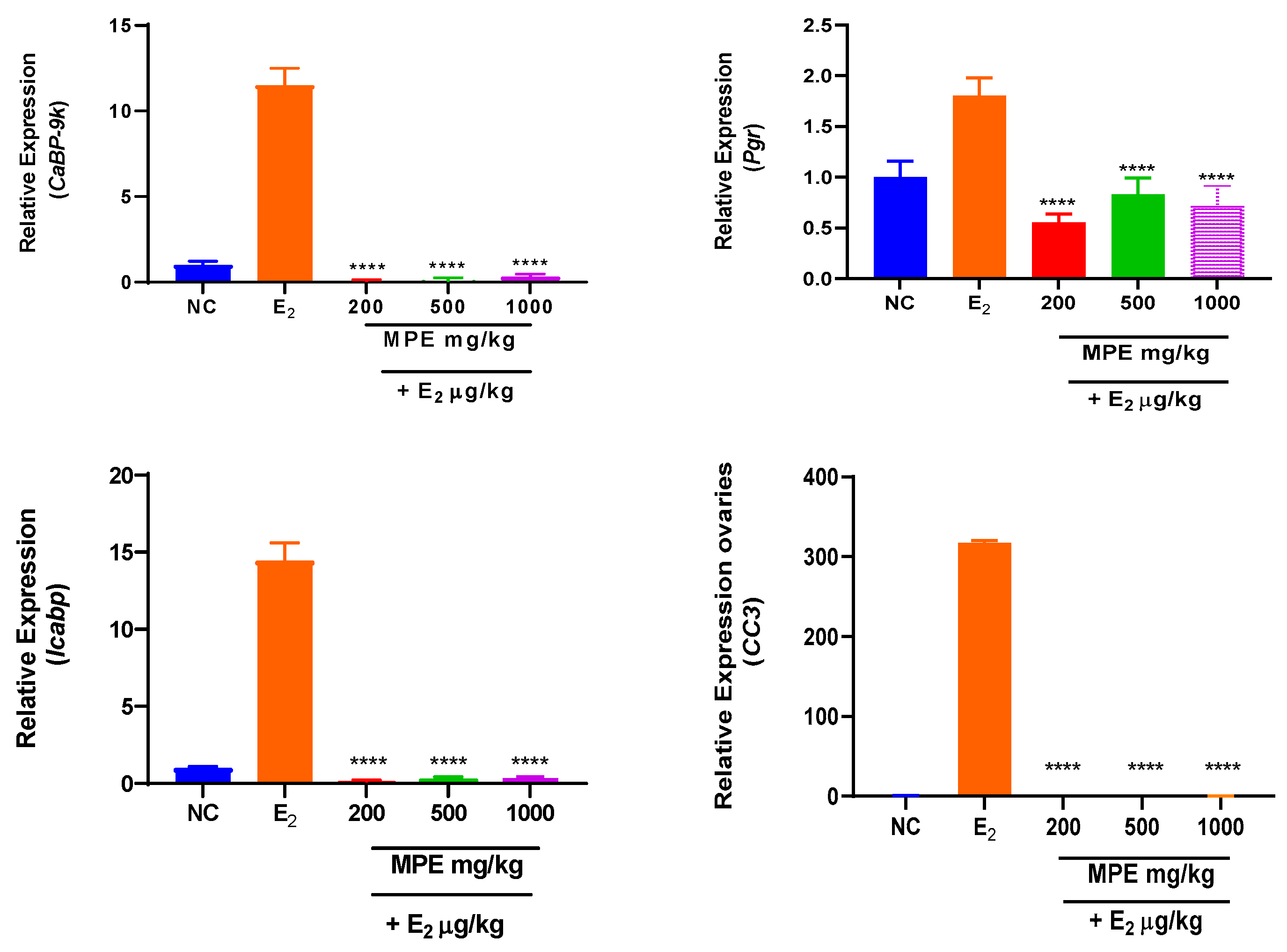
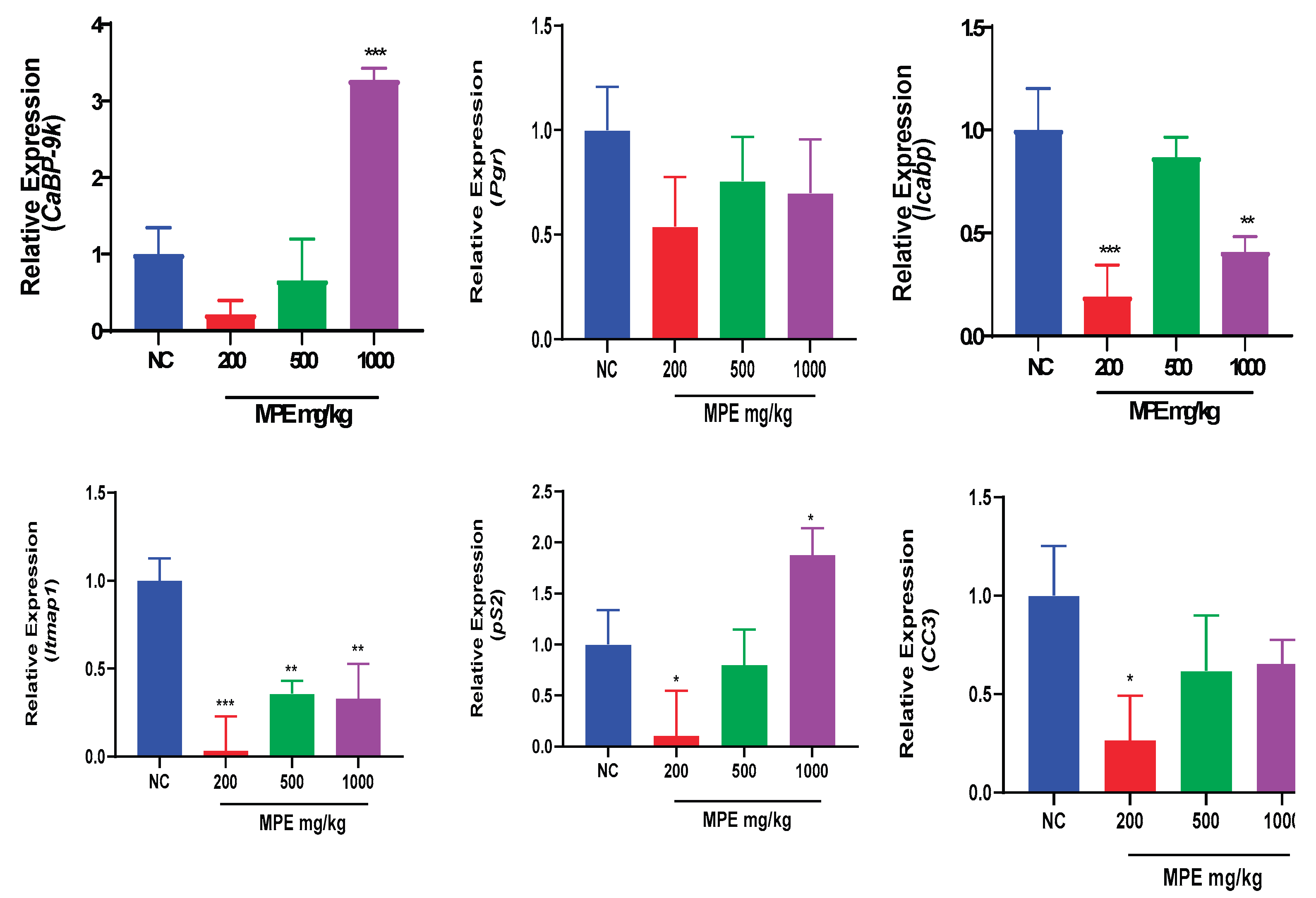
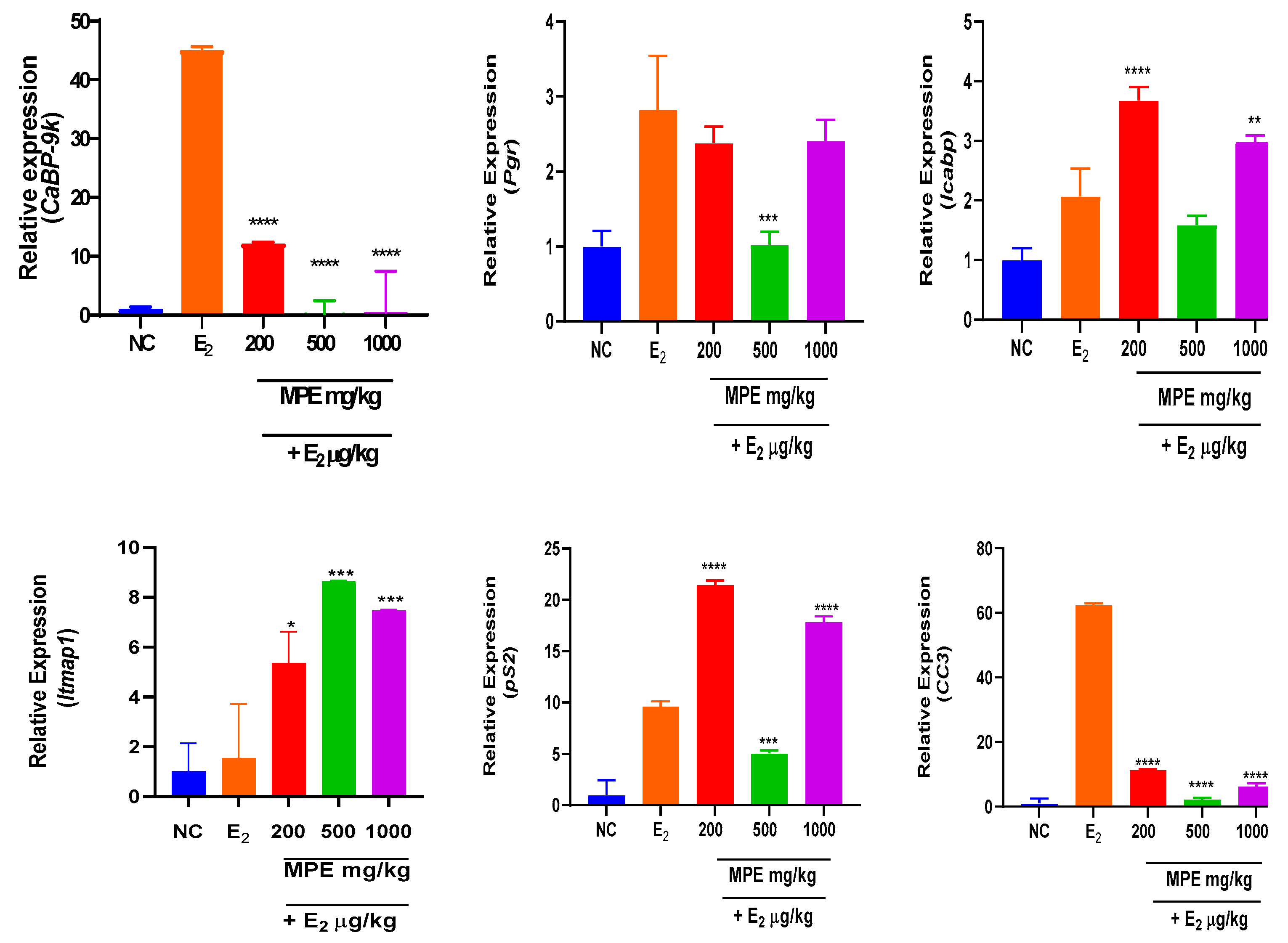
Mentha pulegium L. contains phytochemicals known to bind to estrogen receptors and modulate estrogenic effects. The study investigated the effects of Mentha pulegium L. (MPE) on uterine and ovarian tissues in immature rats, focusing on the transcriptional endpoint-related estrogenic activity. Female Sprague Dawley rats were treated with varying doses of MPE alone or in combination with estradiol for seven days. Gene expression analysis was performed using reverse transcriptase-quantitative polymerase chain reaction (RT-qPCR) to evaluate the effect of MPE on estrogen-responsive biomarkers: Calbindin-D9k (CaBP-9k), Progesterone receptor (Pgr), Trefoil factor 1 (pS2), Intestinal calcium-binding protein integral (Icabp), Integral membrane-associated protein-1 (Itmap1) and Complement component 3 (CC3) genes. In ovarian tissues, MPE treatment down-regulated CaBP-9k and Icapb expression, with CC3 showing significant down-regulation in the 200 mg/kg group. Treatments with MPE and estradiol significantly reduced the expression of all estrogen-responsive genes compared to estradiol treatment. In uterine tissues, 1000 mg/kg MPE up-regulated CaBP-9k and pS2 expression significantly but down-regulated Icapb, CC3, pS2, and Itmap across all treatment groups significantly. Combined estradiol treatment with MPE (500 and 1000) mg/kg showed significantly low CaBP-9k and CC3 expressions. Increased expression of Icapb, Itmap, and pS2 was observed when combined estradiol treatments with MPE (500 and 1000) mg/kg were compared to estradiol treatment. MPE influenced the expression of specific genes in the uterus and ovaries and thus may act as an endocrine disruptor with multiple mechanisms of action, highlighting its potential complexity in modulating estrogenic responses.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Setting
2.2. Plant Material and Extraction
2.3. Animals and Experimental Design
2.4. RNA Isolation and Synthesis of Complementary DNA (cDNA)
2.5. Quantitative Polymerase Chain Reaction (qPCR)
2.6. Relative Gene Expression and Statistical Analysis
3. Results
3.1. Effects of MPE and E2 Treatments on Expression Levels of mRNA Transcripts in Ovarian Tissues
3.2. Effects of MPE and E2 Treatments on Expression Levels of mRNA Transcripts in Uterine Tissues
4. Discussion
5. Conclusions
6. Recommendation
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Acknowledgments
Abbreviations
| CaBP-9k | Calbindin-D9k |
| Pgr | Progesterone |
| pS2 | Trefoil factor 1 |
| Icabp | Intestinal Calcium Binding Protein |
| Itmap1 | Integral membrane- associated protein -1 |
| CC3 | Complement Component 3 |
References
- Hilton, HN; Clarke, CL; Graham, JD. Estrogen and progesterone signalling in the normal breast and its implications for cancer development. Mol Cell Endocrinol. 2018, 466, 2–14. [Google Scholar] [CrossRef] [PubMed]
- Fertuck, KC; Eckel, JE; Gennings, C; Zacharewski, TR. Identification of temporal patterns of gene expression in the uteri of immature, ovariectomized mice following exposure to ethynylestradiol. Physiol Genomics 2004, 15(84), 127–41. [Google Scholar] [CrossRef]
- Zingue, S; Ntsa, DM; Magne Nde, CB; Michel, T; Ndinteh, DT; Clyne, C; et al. Lupeol, the major compound of the dichloromethane extract of Millettia macrophylla Benth (Fabaceae), displays estrogenic effects in ovariectomized rats. Phyther Res. 2019, 33(4), 949–57. [Google Scholar] [CrossRef]
- Kim, SH; Park, MJ. Effects of phytoestrogen on sexual development. Korean J Pediatr. 2012, 55(8), 265–71. [Google Scholar] [CrossRef]
- Jawaid, T; Awasthi, A; Kamal, M. Estrogenic activity of a hydro-alcoholic extract of Bambusa arundinaceae leaves on female wistar rats. Journal of Advanced Pharmaceutical Technology and Research 2015, 6, 19–24. [Google Scholar]
- El-Gazar, AA; Emad, AM; Ragab, GM; Rasheed, DM. Mentha pulegium L. (Pennyroyal, Lamiaceae) Extracts Impose Abortion or Fetal-Mediated Toxicity in Pregnant Rats; Evidenced by the Modulation of Pregnancy Hormones, MiR-520, MiR-146a, TIMP-1 and MMP-9 Protein Expressions, Inflammatory State, Certain Relate. Toxins (Basel) 2022, 14(5), 347. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, B; Marques, A; Ramos, C; Batista, I; Serrano, C; Matos, O; et al. European pennyroyal (Mentha pulegium) from Portugal: Chemical composition of essential oil and antioxidant and antimicrobial properties of extracts and essential oil. Ind Crops Prod. 2012, 36(1), 81–7. [Google Scholar] [CrossRef]
- Casiglia, S; Bruno, M; Fontana, G; Senatore, F. Chemical composition of the essential oil of Mentha pulegium growing wild in Sicily and its activity on microorganisms affecting historical art crafts. Nat Prod Commun. 2017, 12(8), 1311–5. [Google Scholar] [CrossRef]
- Salem, N; Sriti, J; Bachrouch, O; Msaada, K; Khammassi, S; Hammami, M; et al. Phenological stage effect on phenolic composition and repellent potential of Mentha pulegium against Tribolium castaneum and Lasioderma serricorne. Asian Pac J Trop Biomed. 2018, 8(4), 207–16. [Google Scholar] [CrossRef]
- Mondher, B. Comparative study of the chemical properties and composition of the Mentha Pulegium L. of Algerian origin and that of Tunisian origin. Int J Agric Sci Food Technol. 2020, 6(1), 030–6. [Google Scholar] [CrossRef]
- Sallah, L; Coffie, PN; Babatunde, DM; Danquah, CA; Baah, FK; Mante, PK; et al. Anti-implantation and uterotonic properties of Mentha pulegium L. in female Sprague-Dawley rats. 2024, 10(10), 346–55. [Google Scholar] [CrossRef]
- Fatih, B; Madani, K; Chibane, M; Duez, P. Chemical Composition and Biological Activities of Mentha Species. In: Aromatic and Medicinal Plants - Back to Nature. InTech; 2017. p. 47–79.
- Ouakouak, H; Chohra, M; Denane, M. Chemical Composition, Antioxidant Activities of the Essential Oil of Mentha pulegium L, South East of Algeria. Int Lett Nat Sci. 2015, 39, 49–55. [Google Scholar] [CrossRef]
- Soukaina, K; Safa, Z; Soukaina, H; Hicham, C; Bouchra, C. Choline chloride-based deep eutectic solvents (NADES): Potential use as green extraction media for polyphenols from Mentha pulegium, antioxidant activity, and antifungal activity. Microchem J 2024, 199, 110174. [Google Scholar] [CrossRef]
- Soares, PMG; De Freitas Pires, A; De Souza, EP; Assreuy, AMS; Criddle, DN. Relaxant effects of the essential oil of Mentha pulegium L. in rat isolated trachea and urinary bladder. J Pharm Pharmacol. 2012, 64(12), 1777–84. [Google Scholar] [CrossRef]
- Aires, A; Marrinhas, E; Carvalho, R; Dias, C; Saavedra, MJ. Phytochemical composition and antibacterial activity of hydroalcoholic extracts of pterospartum tridentatum and mentha pulegium against staphylococcus aureus isolates. Biomed Res Int. 2016, 2016. [Google Scholar] [CrossRef]
- Yusuf, U; Muhammad, M. Antibacterial Properties of Mentha pulegium. South Asian Res J Appl Med Sci. 2019, 01(02), 43–5. [Google Scholar] [CrossRef]
- El-Naggar, HM; Osman, AR. Enhancing growth and bioactive metabolites characteristics in Mentha pulegium L. via silicon nanoparticles during in vitro drought stress. BMC Plant Biol. 2024, 24(1), 1–16. [Google Scholar] [CrossRef]
- Brahmi, F; Khodir, M; Chibane, M; Pierre, D. Chemical Composition and Biological Activities of Mentha Species. In: Aromatic and Medicinal Plants - Back to Nature. 2017. p. 47–79.
- Kelidari, HR; Alipanah, H; Roozitalab, G; Ebrahimi, M; Osanloo, M. Anticancer effect of solid-lipid nanoparticles containing mentha longifolia and mentha pulegium essential oils: In vitro study on human melanoma and breast cancer cell lines. Biointerface Res Appl Chem. 2022, 12(2), 2128–37. [Google Scholar]
- Gerenutti, M; Modesto, L; Carrara, VA; Magalhães, SA; Paulo, S. Maternal exposure to aqueous extract of Mentha pulegium L. inducing toxicity to embryo development in rats. African J Pharm Pharmacol. 2014, 8(22), 609–14. [Google Scholar]
- Assaggaf, H; El Hachlafi, N; Elbouzidi, A; Taibi, M; Benkhaira, N; El Kamari, F; et al. Unlocking the combined action of Mentha pulegium L. essential oil and Thym honey: In vitro pharmacological activities, molecular docking, and in vivo anti-inflammatory effect. Heliyon 2024, 10(11), e31922. [Google Scholar] [CrossRef] [PubMed]
- Sun, L; Yu, T; Guo, J; Zhang, Z; Hu, Y; Xiao, X; et al. The estrogenicity of methylparaben and ethylparaben at doses close to the acceptable daily intake in immature Sprague-Dawley rats. Sci Rep. 2016, 6(1), 1–9. [Google Scholar] [CrossRef]
- Jeong, SY; Chang, M; Choi S ho; Oh, SR; Wu, HH; Zhu, Y; et al. Estrogenic effects of phytoestrogens derived from Flemingia strobilifera in MCF-7 cells and immature rats. Vol. 41, Archives of Pharmacal Research. Pharmaceutical Society of Korea; 2018. 519–529 p.
- Naciff, JM; Overmann, GJ; Torontali, SM; Carr, GJ; Tiesman, JP; Daston, GP. Impact of the phytoestrogen content of laboratory animal feed on the gene expression profile of the reproductive system in the immature female rat. Environ Health Perspect. 2004, 112(15), 1519–26. [Google Scholar] [CrossRef]
- Daston, GP; Naciff, JM. Gene expression changes related to growth and differentiation in the fetal and juvenile reproductive system of the female rat: Evaluation of microarray results. Reprod Toxicol. 2005, 19(3 SPEC. ISS.), 381–94. [Google Scholar] [CrossRef] [PubMed]
- Daston, GP; Naciff, JM. Predicting developmental toxicity through toxicogenomics. Birth Defects Res Part C - Embryo Today Rev. 2010, 90(2), 110–7. [Google Scholar] [CrossRef] [PubMed]
- Wall, EH; Hewitt, SC; Case, LK; Lin, CY; Korach, KS; Teuscher, C. The role of genetics in estrogen responses: A critical piece of an intricate puzzle. FASEB J. 2014, 28(12), 5042–54. [Google Scholar] [CrossRef] [PubMed]
- Heikaus, S; Winterhager, E; Traub, O; Grümmer, R. Responsiveness of endometrial genes connexin26, connexin43, C3 and clusterin to primary estrogen, selective estrogen receptor modulators, phyto- and xenoestrogens. J Mol Endocrinol. 2002, 29(2), 239–49. [Google Scholar] [CrossRef]
- Tang, ZR; Zhang, R; Lian, ZX; Deng, SL; Yu, K. Estrogen-receptor expression and function in female reproductive disease. Cells 2019, 8(10), 1–15. [Google Scholar] [CrossRef]
- Hopert, AC; Bayer, A; Frank, K; Strunck, E; Wünsche, W; Vollmer, G. Characterization of estrogenicity of phytoestrogens in an endometrial-derived experimental model. Environ Health Perspect. 1998, 106(9), 581–6. [Google Scholar] [CrossRef]
- Basu, P; Maier, C. Phytoestrogens and breast cancer: In vitro anticancer activities of isoflavones, lignans, coumestans, stilbenes and their analogs and derivatives. Biomed Pharmacother. 2018, 107(August), 1648–66. [Google Scholar] [CrossRef]
- Kajita, S; Suzuki, E; Saegusa, M. Progesterone receptor expression correlates with estrogen receptor. 2012;(December 2011):22–32.
- Zalewski, A; Cecchini, EL; Deroo, BJ. Expression of extracellular matrix components is disrupted in the immature and adult estrogen receptor β-null mouse ovary. PLoS One 2012, 7(1). [Google Scholar] [CrossRef]
- Lee, EB; Praveen Chakravarthi, V; Wolfe, MW; Karim Rumi, MA. ERβ regulation of gonadotropin responses during folliculogenesis. Int J Mol Sci. 2021, 22(19). [Google Scholar] [CrossRef]
- Rasier, G; Parent, AS; Gérard, A; Lebrethon, MC; Bourguignon, JP. Early maturation of gonadotropin-releasing hormone secretion and sexual precocity after exposure of infant female rats to estradiol or dichlorodiphenyltrichloroethane. Biol Reprod. 2007, 77(4), 734–42. [Google Scholar] [CrossRef] [PubMed]
- Mayer, C; Acosta-Martinez, M; Dubois, SL; Wolfe, A; Radovick, S; Boehm, U; et al. Timing and completion of puberty in female mice depend on estrogen receptor α-signaling in kisspeptin neurons. Proc Natl Acad Sci U S A 2010, 107(52), 22693–8. [Google Scholar] [CrossRef]
- Hewitt, SC; Couse, JF; Korach, KS. Estrogen receptor knockout mice: What their phenotypes reveal about mechanisms of estrogen action. Breast Cancer Res. 2000, 2(5), 345–52. [Google Scholar] [CrossRef]
- Xu, XL; Huang, ZY; Yu, K; Li, J; Fu, XW; Deng, SL. Estrogen Biosynthesis and Signal Transduction in Ovarian Disease. Front Endocrinol (Lausanne) 2022, 13(March), 1–14. [Google Scholar] [CrossRef]
- Chauvin, S; Cohen-Tannoudji, J; Guigon, CJ. Estradiol Signaling at the Heart of Folliculogenesis: Its Potential Deregulation in Human Ovarian Pathologies. Int J Mol Sci. 2022, 23(1). [Google Scholar] [CrossRef]
- Diep, CH; Ahrendt, H; Lange, CA. HHS Public Access Author manuscript . Author manuscript; available in PMC 2017 October 01. Published in final edited form as: Steroids Steroids . 2016 October ; 114: 48–58. Progesterone induces progesterone receptor gen. 2016;48–58. [CrossRef]
- Ing, NH; Tornesi, MB. Estradiol up-regulates estrogen receptor and progesterone receptor gene expression in specific ovine uterine cells. Biol Reprod. 1997, 56(5), 1205–15. [Google Scholar] [CrossRef] [PubMed]
- Peluso, JJ; Pru, JK. Non-canonical progesterone signaling in granulosa cell function. Reproduction 2014, 147(5), 169–78. [Google Scholar] [CrossRef]
- Shin, YY; Kang, EJ; Jeong, JS; Kim, MJ; Jung, EM; Jeung, EB; et al. Pregnenolone as a potential candidate for hormone therapy for female reproductive disorders targeting ERβ. Mol Reprod Dev. 2019, 86(1), 109–17. [Google Scholar] [CrossRef]
- Park, OK; Mayo, KE. Transient expression of progesterone receptor messenger RNA in ovarian granuiosa cells after the preovulatory luteinizing hormone surge. Mol Endocrinol. 1991, 5(7), 967–78. [Google Scholar] [CrossRef]
- Dey, A; Dhadhal, S; Maharjan, R; Nagar, PS; Nampoothiri, L. Partially purified non-polar phytocomponents from Aloe barbadensis Mill. gel restores metabolic and reproductive comorbidities in letrozole-induced polycystic ovary syndrome rodent model- an “in-vivo” study. J Ethnopharmacol. 2022, 291, 115161. [Google Scholar] [CrossRef]
- Liew, SH; Sarraj, MA; Drummond, AE; Findlay, JK. Estrogen-Dependent Gene Expression in the Mouse Ovary. 2011;6(2).
- Moon, B; Kim, W; Park, CH; Oh, SM. Ginkgo biloba extract (EGb761) did not express estrogenic activity in an immature rat uterotrophic assay. Vol. 33, Environmental Health and Toxicology. 2018. e2018016 p.
- Christakos, S; Mady, LJ; Dhawan, P. Calbindin-D28K and Calbindin-D9K and the Epithelial Calcium Channels TRPV5 and TRPV6. Fourth Edi. Vol. 1, Vitamin D: Fourth Edition. Elsevier Inc.; 2018. 343–359 p.
- Sim, JY; Jung, EM; Yoo, YM; Choi, KC; Jeung, EB. Transcriptional and translational expression of calbindin-D9k in the duodenum, kidney and uterus of a female canine model. J Vet Sci. 2010, 11(1), 15–9. [Google Scholar] [CrossRef]
- Mortlock, S; Mckinnon, B; Montgomery, GW. Genetic Regulation of Transcription in the Endometrium in Health and Disease. 2022, 3, 1–13. [Google Scholar] [CrossRef]
- Nephew, KP; Long, X; Osborne, E; Burke, KA; Ahluwalia, A; Bigsby, RM. Effect of estradiol on estrogen receptor expression in rat uterine cell types. Biol Reprod. 2000, 62(1), 168–77. [Google Scholar] [CrossRef]
- Wang, T; Wang, Y; Zhuang, X; Luan, F; Zhao, C; Cordeiro, MNDS. Interaction of coumarin phytoestrogens with ERα and ERβ: A molecular dynamics simulation study. Molecules 2020, 25(5). [Google Scholar] [CrossRef]
- Heneweer, M; Houtman, R; Poortman, J; Groot, M; Maliepaard, C; Peijnenburg, A. Estrogenic effects in the immature rat uterus after dietary exposure to ethinylestradiol and zearalenone using a systems biology approach. Toxicol Sci. 2007, 99(1), 303–14. [Google Scholar] [CrossRef]
- Yang, H; Nguyen, TT; An, BS; Choi, KC; Jeung, EB. Synergistic effects of parabens on the induction of calbindin-D9k gene expression act via a progesterone receptor-mediated pathway in GH3 cells. Hum Exp Toxicol. 2012, 31(2), 134–44. [Google Scholar] [CrossRef]
- Lee, JS; Kim, YK; Yang, H; Kang, HY; Ahn, C; Jeung, EB. Two faces of the estrogen metabolite 2-methoxyestradiol in vitro and in vivo. Mol Med Rep. 2015, 12(4), 5375–82. [Google Scholar] [CrossRef]
- Choi, KC; Leung, PCK; Jeung, EB. Biology and physiology of Calbindin-D9k in female reproductive tissues: Involvement of steroids and endocrine disruptors. Reprod Biol Endocrinol. 2005, 3, 1–8. [Google Scholar] [CrossRef]
- Sadigh, AR; Mihanfar, A; Fattahi, A; Latifi, Z; Akbarzadeh, M; Hajipour, H; et al. S100 protein family and embryo implantation. J Cell Biochem. 2019, 120(12), 19229–44. [Google Scholar] [CrossRef]
- Abd-Elhakim, YM; Mohammed, AT; Ali, HA. Impact of subchronic exposure to triclosan and/or fluoride on estrogenic activity in immature female rats: The expression pattern of calbindin-D9k and estrogen receptor α genes. J Biochem Mol Toxicol. 2018, 32(2), 6–13. [Google Scholar] [CrossRef]
- Dang, VH; Choi, KC; Jeung, EB. Tetrabromodiphenyl ether (BDE 47) evokes estrogenicity and calbindin-D9k expression through an estrogen receptor-mediated pathway in the uterus of immature rats. Toxicol Sci. 2007, 97(2), 504–11. [Google Scholar] [CrossRef]
- Grimm, SL; Hartig, SM; Edwards, DP. Review: Progesterone Receptor Signaling Mechanisms AC NU. J Mol Biol. 2016. [Google Scholar] [CrossRef]
- Marquardt, RM; Kim, TH; Shin, JH; Jeong, JW. Progesterone and estrogen signaling in the endometrium: What goes wrong in endometriosis? Int J Mol Sci. 2019, 20(15). [Google Scholar] [CrossRef]
- An, BH; Jeong, H; Zhou, W; Liu, X; Kim, S; Jang, CY; et al. Evaluation of the Biological Activity of Opuntia ficus indica as a Tissue- and Estrogen Receptor Subtype-Selective Modulator. Phyther Res. 2016, 30(6), 971–80. [Google Scholar] [CrossRef]
- Pinto, MP; Medina, RA; Owen, GI. 2-Methoxyestradiol and Disorders of Female Reproductive Tissues. Horm Cancer 2014, 5(5), 274–83. [Google Scholar] [CrossRef]
- Kasik, JW. A cDNA cloned from pregnant mouse uterus exhibits temporo-spatial expression and predicts a novel protein. Biochem J. 1998, 950, 947–50. [Google Scholar] [CrossRef]
- Imamura, T; Asada, M; Vogt, SK; Rudnick, DA; Lowe, ME; Muglia, LJ. Protection from pancreatitis by the zymogen granule membrane protein integral membrane-associated protein-1. J Biol Chem. 2002, 277(52), 50725–33. [Google Scholar] [CrossRef]
- Zekri, N; Amalich, S; Boughdad, A; Alaoui El Belghiti, M; Zair, T. Phytochemical study and insecticidal activity of Mentha pulegium L. oils from Morocco against Sitophilus Oryzae. Mediterr J Chem. 2013, 2(4), 607–19. [Google Scholar] [CrossRef]
- Zaid, SSM; Othman, S; Kassim, NM. Protective role of Mas Cotek (Ficus deltoidea) against the toxic effects of bisphenol A on morphology and sex steroid receptor expression in the rat uterus. Biomed Pharmacother 2021, 140. [Google Scholar] [CrossRef]
- Jung, EM; An, BS; Yang, H; Choi, KC; Jeung, EB. Biomarker genes for detecting estrogenic activity of endocrine disruptors via estrogen receptors. Int J Environ Res Public Health 2012, 9(3), 698–711. [Google Scholar] [CrossRef] [PubMed]
- Diel, P; Hertrampf, T; Seibel, J; Laudenbach-Leschowsky, U; Kolba, S; Vollmer, G. Combinatorial effects of the phytoestrogen genistein and of estradiol in uterus and liver of female Wistar rats. J Steroid Biochem Mol Biol. 2006, 102, 60–70. [Google Scholar] [CrossRef] [PubMed]
 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





