Submitted:
25 February 2026
Posted:
27 February 2026
You are already at the latest version
Abstract

Keywords:
1. Introduction
2. Materials and Methods
2.1. Ethics, Design and Setting
2.2. Participants
2.3. Interviews and Data Collection
2.4. Demographic and Clinical Information
2.5. Instruments for Psychiatric Evaluation
2.6. Quality of Life
2.7. Statistical Analysis
3. Results
4. Discussion
4.1. Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability
Conflicts of Interest
Abbreviations
| CBCL | Child Behavior Checklist |
| CDI | Children’s Depression Inventory |
| SCARED | Screen for Child Anxiety Related Disorders |
| CRIES-13 | Screen |
| SCA | Children’s Revised Impact of Events Scale-13 |
| PedsQL | Pediatric Quality of Life Inventory |
| HRQOL | health-related quality of life |
| PLT | Pediatric liver transplantation |
| DSM | Diagnostic and Statistical Manual of Mental Disorders |
| AST | aspartate aminotransferase |
| ALT | alanine aminotransferase |
| GGT | gamma-glutamyl transferase |
References
- Kwong, A.J.; Ebel, N.H.; Kim, W.R.; Lake, J.R.; Smith, J.M.; Schladt, D.P.; Schnellinger, E.M.; Handarova, D.; Weiss, S.; Cafarella, M. OPTN/SRTR 2021 annual data report: liver. Am. J. Transplant. 2023, 23, S178–S263. [Google Scholar] [CrossRef]
- Goss, J.A.; Shackleton, C.R.; Mcdiarmid, S.V.; Maggard, M.; Swenson, K.; Seu, P.; Vargas, J.; Martin, M.; Ament, M.; Brill, J. Long-term results of pediatric liver transplantation: an analysis of 569 transplants. Ann. Surg. 1998, 228, 411–420. [Google Scholar] [CrossRef]
- Ünay, M.; Önder, A.; Gizli Çoban, Ö.; Atalay, A.; Sürer Adanir, A.; Artan, R.; Özatalay, E. Psychopathology, quality of life, and related factors in pediatric liver transplantation candidates and recipients. Pediatr. Transplant. 2020, 24, e13633. [Google Scholar] [CrossRef]
- Taylor, R.; Franck, L.; Gibson, F.; Donaldson, N.; Dhawan, A. Study of the factors affecting health-related quality of life in adolescents after liver transplantation. Am. J. Transplant. 2009, 9, 1179–1188. [Google Scholar] [CrossRef]
- Düken, M.E.; Yayan, E.H. Psychosocial conditions of children after liver transplant: Post-traumatic stress, depression, and anxiety. J. Pediatr. Nurs. 2024, 75, e75–e80. [Google Scholar] [CrossRef] [PubMed]
- Huang, M.; Hou, Y.; Li, W.; Wang, G.; Gu, G.; Xia, Q. Mental health in children with living donor liver transplantation: A propensity score-matched analysis. Child Adolesc. Psychiatry Ment. Health 2022, 16, 94. [Google Scholar] [CrossRef]
- Taş, B.Y.; Özbaran, B. Psychiatric comorbidities in pediatric organ transplantation: Current findings and clinical approaches. Eur. Transplant. Res. 2025, 1, 25–33. [Google Scholar]
- Parmar, A.; Vandriel, S.M.; Ng, V.L. Health-related quality of life after pediatric liver transplantation: a systematic review. Liver Transpl. 2017, 23, 361–374. [Google Scholar] [CrossRef]
- Mohammad, S.; Sundaram, S.S.; Mason, K.; Lobritto, S.; Martinez, M.; Turmelle, Y.P.; Bucuvalas, J.; Feng, S.; Alonso, E.M. Improvements in disease-specific health-related quality of life of pediatric liver transplant recipients during immunosuppression withdrawal. Liver Transpl. 2021, 27, 735–746. [Google Scholar] [CrossRef] [PubMed]
- Hager, A.; Mager, D.; Robert, C.; Nicholas, D.; Gilmour, S. Health-related quality of life 10 years after liver transplantation: a longitudinal retrospective review. Diagnostics 2021, 11, 111. [Google Scholar] [CrossRef] [PubMed]
- Gritti, A.; Sicca, F.; Di Sarno, A.M.; Di Cosmo, N.; Vajro, S.; Vajro, P. Emotional and behavioral problems after pediatric liver transplantation: A quantitative assessment. Pediatr. Transplant. 2006, 10, 205–209. [Google Scholar] [CrossRef]
- Marangoz, Y.; Özçay, F.; Haberal, M. Association of Liver Transplant and Attention-Deficit Hyperactivity Disorder: A Single-Center Experience. Exp. Clin. Transplant. 2023. Online ahead of print. [Google Scholar] [CrossRef]
- Uçgun, T.; Koyuncu, İ.E.; Koç, E.; Kılınç, B.Ş.; Sarıdağ, K.N.K. Identification of Psychosocial Issues in Pediatric Patients Undergoing or Waiting for Organ Transplant: A Systematic Review. Exp. Clin. Transplant. 2024, 22, 132–138. [Google Scholar] [CrossRef]
- Kovacs, M. Children’s Depression Inventory (CDI and CDI 2); John Wiley & Sons, 2014. [Google Scholar]
- Monga, S.; Birmaher, B.; Chiappetta, L.; Brent, D.; Kaufman, J.; Bridge, J.; Cully, M. Screen for child anxiety-related emotional disorders (SCARED): Convergent and divergent validity. Depress. Anxiety 2000, 12, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Perrin, S.; Meiser-Stedman, R.; Smith, P. The Children’s Revised Impact of Event Scale (CRIES): Validity as a screening instrument for PTSD. Behav. Cogn. Psychother. 2005, 33, 487–498. [Google Scholar] [CrossRef]
- Achenbach, T.M.; Rescorla, L. Child behavior checklist for ages 6-18; University of Vermont: Burlington, VT, 2001. [Google Scholar]
- Desai, A.D.; Zhou, C.; Stanford, S.; Haaland, W.; Varni, J.W.; Mangione-Smith, R.M. Validity and responsiveness of the pediatric quality of life inventory (PedsQL) 4.0 generic core scales in the pediatric inpatient setting. JAMA Pediatr. 2014, 168, 1114–1121. [Google Scholar] [CrossRef] [PubMed]
- Sorensen, L.G.; Neighbors, K.; Martz, K.; Zelko, F.; Bucuvalas, J.; Alonso, E.M. Cognitive and academic outcomes after pediatric liver transplantation: Functional Outcomes Group (FOG) results. Am. J. Transplant. 2011, 11, 303–311. [Google Scholar] [CrossRef]
- Cousino, M.K.; Hazen, R.A. Parenting stress among caregivers of children with chronic illness: a systematic review. J. Pediatr. Psychol. 2013, 38, 809–828. [Google Scholar] [CrossRef]
- Yalcinkaya, A.; Yalcinkaya, R.; Sardh, F.; Landegren, N. Immune dynamics throughout life in relation to sex hormones and perspectives gained from gender-affirming hormone therapy. Front. Immunol. 2025, 15, 1501364. [Google Scholar] [CrossRef]
- Ng, V.L.; Alonso, E.M.; Bucuvalas, J.C.; Cohen, G.; Limbers, C.A.; Varni, J.W.; Mazariegos, G.; Magee, J.; Mcdiarmid, S.V.; Anand, R. Health status of children alive 10 years after pediatric liver transplantation performed in the US and Canada: report of the studies of pediatric liver transplantation experience. J. Pediatr. 2012, 160, 820–826.e823. [Google Scholar] [CrossRef]
- Vimalesvaran, S.; Souza, L.N.; Deheragoda, M.; Samyn, M.; Day, J.; Verma, A.; Vilca-Melendez, H.; Rela, M.; Heaton, N.; Dhawan, A. Outcomes of adults who received liver transplant as young children. EClinicalMedicine 2021, 38, 100987. [Google Scholar] [CrossRef] [PubMed]
- Ala, S.; Ramos-Campos, F.; Relva, I.C. Symptoms of post-traumatic stress and mental health in a sample of university students: The mediating role of resilience and psychological well-being. Eur. J. Investig. Health Psychol. Educ. 2024, 14, 2262–2281. [Google Scholar] [CrossRef]
- Marshall, A.F.; Mandac, C.; Qin, F.; Boothroyd, D.; Nasr, A.S. Effect of Age at Liver Transplant on Anxiety, Depression, and Quality of Life Among Adolescents. Prog. Transplant. 2024, 34, 192–198. [Google Scholar] [CrossRef] [PubMed]
- Birmaher, B.; Brent, D.A.; Chiappetta, L.; Bridge, J.; Monga, S.; Baugher, M. Psychometric properties of the Screen for Child Anxiety Related Emotional Disorders (SCARED): a replication study. J. Am. Acad. Child Adolesc. Psychiatry 1999, 38, 1230–1236. [Google Scholar] [CrossRef] [PubMed]
- Fredericks, E.M.; Magee, J.C.; Opipari-Arrigan, L.; Shieck, V.; Well, A.; Lopez, M.J. Adherence and health-related quality of life in adolescent liver transplant recipients. Pediatr. Transplant. 2008, 12, 289–299. [Google Scholar] [CrossRef] [PubMed]
| Age of child, years | 14 (11 - 16) |
|---|---|
| Sex | |
| Male | 28 (56.0%) |
| Female | 22 (44.0%) |
| Height, cm | 155.5 (135 - 165) |
| Weight, kg | 45.10 ± 15.94 |
| Body mass index, kg/m2 | 19.38 ± 4.16 |
| Comorbidity | 2 (4.0%) |
| Immigrant | 7 (14.0%) |
| Going to school | 36 (72.0%) |
| Age of mother, years | 38.10 ± 5.98 |
| Education status of mother | |
| Literate | 17 (34.0%) |
| Primary school | 17 (34.0%) |
| Secondary school | 7 (14.0%) |
| High school | 5 (10.0%) |
| Associate degree | 3 (6.0%) |
| Bachelor degree | 1 (2.0%) |
| Age of father, years | 42.08 ± 6.03 |
| Education status of father | |
| Literate | 8 (16.0%) |
| Primary school | 13 (26.0%) |
| Secondary school | 17 (34.0%) |
| High school | 8 (16.0%) |
| Associate degree | 1 (2.0%) |
| Bachelor degree | 3 (6.0%) |
| Chronic disease in family | 10 (20.0%) |
| Age at transplantation, years | 5 (2 - 11) |
| Time since transplantation, years | 7.44 (2.25 - 11.67) |
| Type of donor | |
| Living | 44 (88.0%) |
| Deceased | 6 (12.0%) |
| Immunosuppressive drug | |
| Tacrolimus | 43 (86.0%) |
| Everolimus | 7 (14.0%) |
| Transplantation complication | 0 (0.0%) |
| Rejection attack, last year | 0 (0.0%) |
| Hospitalization, last year | 2 (4.0%) |
| AST, U/L | 32 (25 - 58) |
| ALT, U/L | 27 (16 - 71) |
| Total bilirubin, mg/dL | 0.60 (0.42 - 0.88) |
| GGT, U/L | 27.5 (17 - 93) |
| Psychiatric follow-up | 1 (2.0%) |
|---|---|
| Psychiatric diagnosis | 0 (0.0%) |
| Type of interview | |
| Face to face | 30 (60.0%) |
| Online | 20 (40.0%) |
| CBCL Total score | 43.5 (36 - 57) |
| CDI score | 8.14 ± 5.47 |
| Normal (<15) | 42 (84.0%) |
| Mild depression (15-19) | 6 (12.0%) |
| Moderate depression (20-24) | 2 (4.0%) |
| Severe depression (≥25) | 0 (0.0%) |
| SCARED score | |
| Panic/Somatic | 1 (0 - 2) |
| ≥7 | 4 (8.0%) |
| Generalized Anxiety | 0 (0 - 2) |
| ≥9 | 4 (8.0%) |
| Separation Anxiety | 3 (1 - 5) |
| ≥5 | 15 (30.0%) |
| Social Anxiety | 4.40 ± 2.84 |
| ≥8 | 5 (10.0%) |
| School Avoidance | 0 (0 - 1) |
| ≥3 | 5 (13.9%) |
| Total | 10 (6 - 12) |
| ≥25 | 6 (12.0%) |
| CRIES-13 score | |
| Intrusion | 1.5 (0 - 5) |
| Avoidance | 2.5 (0 - 7) |
| Arousal | 3.5 (1 - 7) |
| Total | 8 (3 - 19) |
| PedsQL score | |
| Physical Health | 71.88 (53.13 - 93.75) |
| Emotional Functioning | 92.5 (85 - 100) |
| Social Functioning | 80 (65 - 100) |
| School Functioning | 63.29 ± 19.25 |
| Psychosocial Health | 78.90 ± 13.14 |
| Total | 75.71 ± 14.57 |
| PedsQL | |||||||
|---|---|---|---|---|---|---|---|
| Physical Health | Emotional Functioning | Social Functioning | School Functioning | Psychosocial Health | Total | ||
| CBCL Total | r | -0.417‡ | -0.778‡ | -0.406‡ | -0.140‡ | -0.608‡ | -0.607‡ |
| p | 0.003 | <0.001 | 0.003 | 0.422 | <0.001 | <0.001 | |
| CDI | r | -0.365‡ | -0.688‡ | -0.284‡ | -0.197† | -0.467† | -0.446† |
| p | 0.009 | <0.001 | 0.045 | 0.257 | 0.001 | 0.001 | |
| SCARED Panic/Somatic | r | -0.278‡ | -0.678‡ | -0.177‡ | 0.101‡ | -0.390‡ | -0.397‡ |
| p | 0.051 | <0.001 | 0.219 | 0.565 | 0.005 | 0.004 | |
| SCARED Generalized Anxiety | r | -0.275‡ | -0.757‡ | -0.211‡ | 0.084‡ | -0.539‡ | -0.439‡ |
| p | 0.053 | <0.001 | 0.142 | 0.630 | <0.001 | 0.001 | |
| SCARED Separation Anxiety | r | -0.147‡ | -0.514‡ | -0.213‡ | -0.110‡ | -0.370‡ | -0.318‡ |
| p | 0.310 | <0.001 | 0.137 | 0.529 | 0.008 | 0.025 | |
| SCARED Social Anxiety | r | -0.511‡ | -0.516‡ | -0.304‡ | -0.223† | -0.480† | -0.582† |
| p | <0.001 | <0.001 | 0.032 | 0.198 | <0.001 | <0.001 | |
| SCARED School Avoidance | r | -0.373‡ | -0.595‡ | -0.099‡ | -0.157‡ | -0.409‡ | -0.457‡ |
| p | 0.025 | <0.001 | 0.566 | 0.367 | 0.013 | 0.005 | |
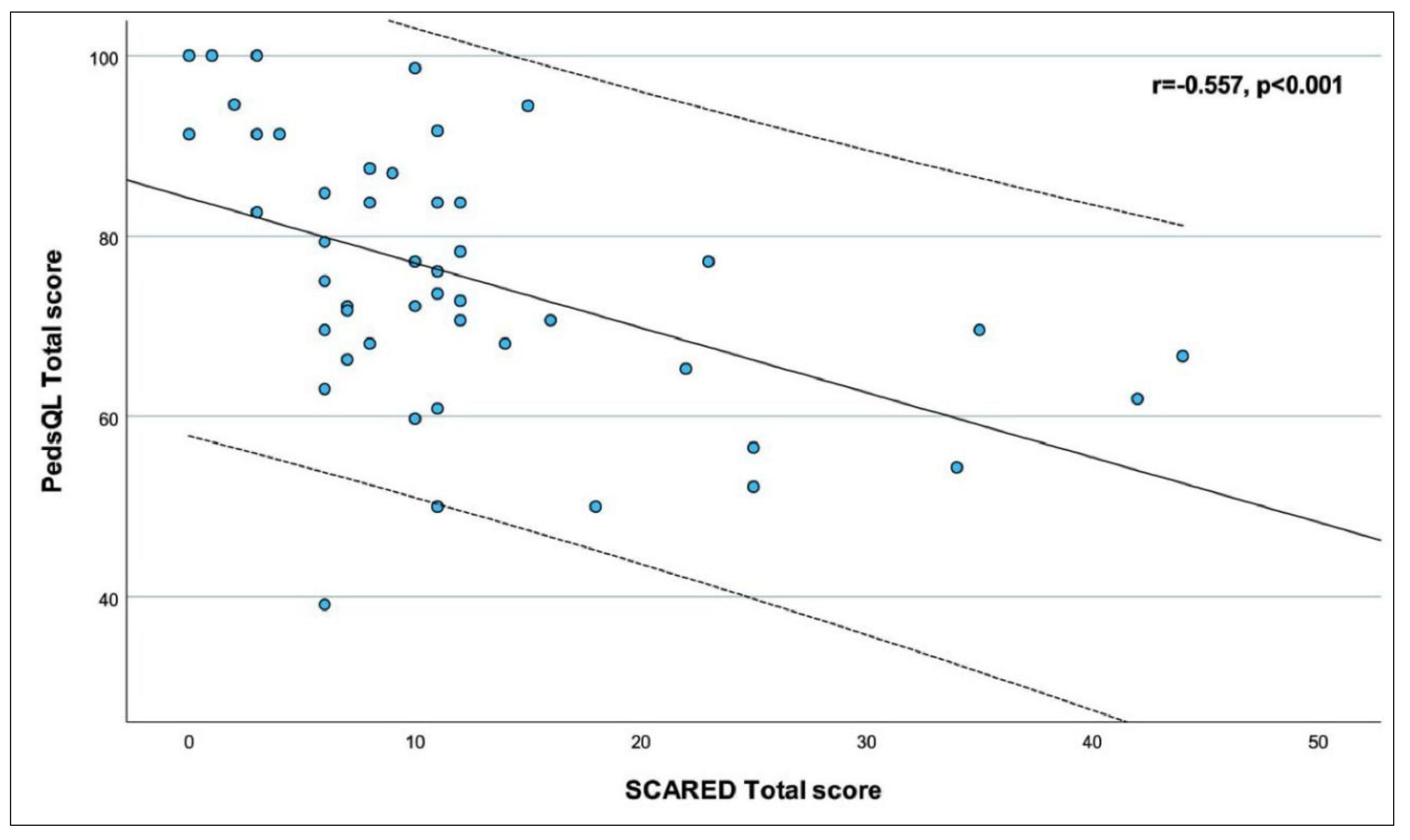
| SCARED Total | r | -0.403‡ | -0.745‡ | -0.312‡ | -0.087‡ | -0.510‡ | -0.557‡ |
| p | 0.004 | <0.001 | 0.028 | 0.620 | <0.001 | <0.001 | |
| CRIES-13 Intrusion | r | -0.169‡ | -0.642‡ | -0.320‡ | -0.075‡ | -0.522‡ | -0.400‡ |
| p | 0.239 | <0.001 | 0.023 | 0.667 | <0.001 | 0.004 | |
| CRIES-13 Avoidance | r | -0.216‡ | -0.436‡ | -0.376‡ | -0.181‡ | -0.470‡ | -0.396‡ |
| p | 0.132 | <0.001 | 0.007 | 0.297 | 0.001 | 0.004 | |
| CRIES-13 Arousal | r | -0.434‡ | -0.665‡ | -0.225‡ | -0.165‡ | -0.544‡ | -0.575‡ |
| p | 0.002 | <0.001 | 0.117 | 0.342 | <0.001 | <0.001 | |
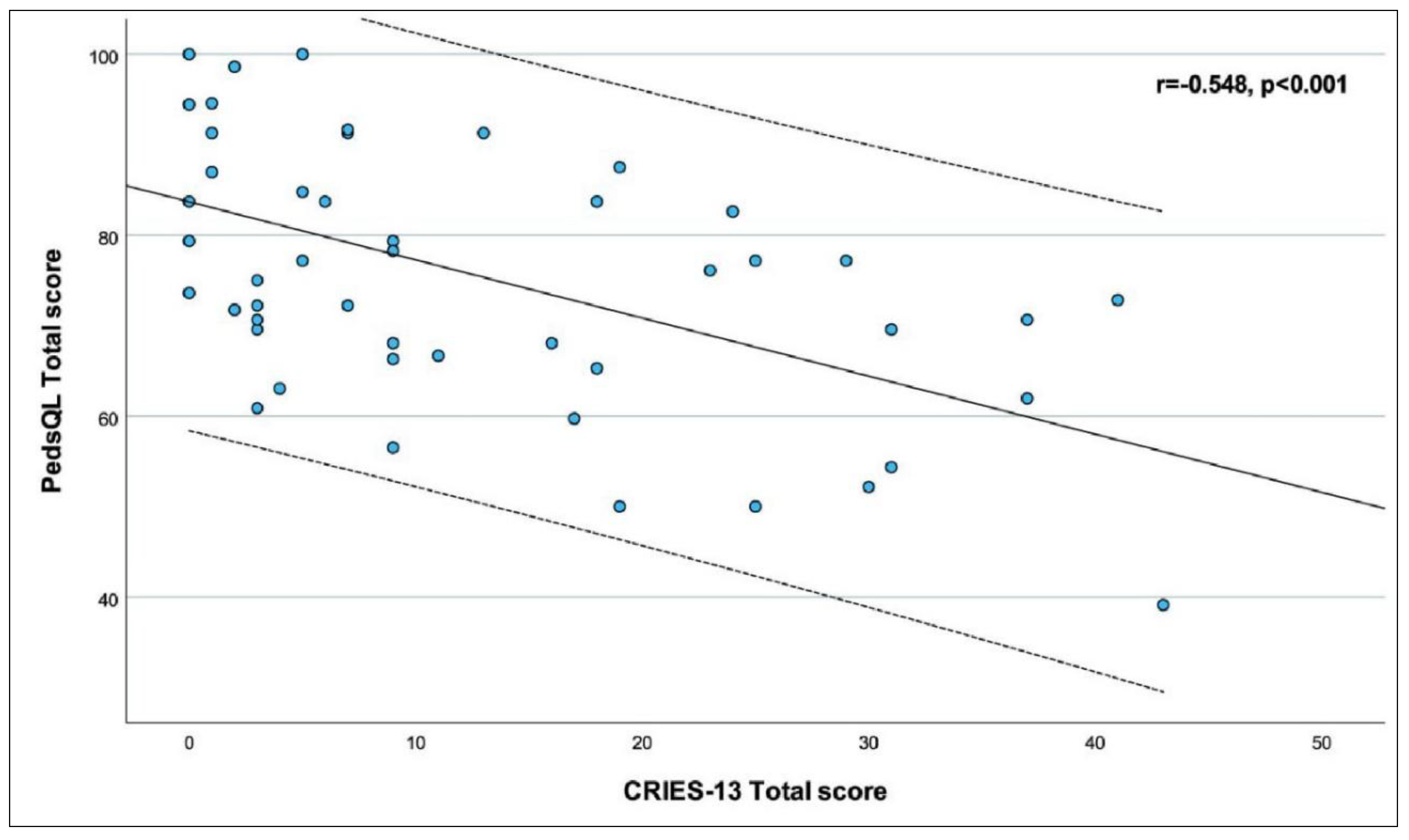
| CRIES-13 Total | r | -0.336‡ | -0.676‡ | -0.340‡ | -0.207‡ | -0.605‡ | -0.548‡ |
| p | 0.017 | <0.001 | 0.016 | 0.234 | <0.001 | <0.001 | |
| Univariable | Multivariable (a) | |||||
|---|---|---|---|---|---|---|
| Unstandardized coefficients (95% CI) | Standardized coefficients | p | Unstandardized coefficients (95% CI) | Standardized coefficients | p | |
| Age of child, years | 1.400 (0.084 - 2.715) | 0.295 | 0.038 | 0.081 | ||
| Sex, Female | -4.719 (-13.042 - 3.604) | -0.162 | 0.260 | |||
| Body mass index, kg/m2 | 0.283 (-0.731 - 1.297) | 0.081 | 0.577 | |||
| Comorbidity, Yes | -11.494 (-32.598 - 9.610) | -0.156 | 0.279 | |||
| Immigrant, Yes | 0.670 (-11.395 - 12.735) | 0.016 | 0.912 | |||
| Going to school, Yes | -2.047 (-11.353 - 7.259) | -0.064 | 0.660 | |||
| Age of mother, years | 0.259 (-0.443 - 0.962) | 0.107 | 0.461 | |||
| Education status of mother | 0.114 (-3.123 - 3.352) | 0.010 | 0.944 | |||
| Age of father, years | -0.069 (-0.774 - 0.637) | -0.029 | 0.846 | |||
| Education status of father | -0.150 (-3.419 - 3.119) | -0.013 | 0.927 | |||
| Chronic disease in family, Yes | -10.134 (-20.180 - -0.088) | -0.281 | 0.048 | 0.072 | ||
| Age at transplantation, years | -0.197 (-1.060 - 0.667) | -0.066 | 0.649 | |||
| Time since transplantation, years | 0.777 (-0.049 - 1.603) | 0.263 | 0.065 | |||
| Type of donor, Deceased | -10.081 (-22.628 - 2.467) | -0.227 | 0.113 | |||
| Immunosuppressive drug, Everolimus | -1.406 (-13.466 - 10.654) | -0.034 | 0.816 | |||
| Hospitalization, last year | -19.421 (-40.031 - 1.189) | -0.264 | 0.064 | |||
| AST | 0.031 (-0.136 - 0.197) | 0.053 | 0.714 | |||
| ALT | 0.035 (-0.051 - 0.122) | 0.118 | 0.414 | |||
| Bilirubin | -0.320 (-1.602 - 0.961) | -0.072 | 0.618 | |||
| GGT | 0.007 (-0.046 - 0.061) | 0.040 | 0.783 | |||
| Psychiatric follow-up, Yes | -24.020 (-53.103 - 5.063) | -0.233 | 0.103 | |||
| Type of interview, Online | 7.661 (-0.591 - 15.914) | 0.260 | 0.068 | |||
| CBCL Total score | -0.408 (-0.611 - -0.206) | -0.505 | <0.001 | 0.279 | ||
| CDI score | -1.189 (-1.881 - -0.497) | -0.446 | 0.001 | 0.475 | ||
| SCARED Total score | -0.719 (-1.088 - -0.350) | -0.493 | <0.001 | -0.431 (-0.824 - -0.038) | -0.295 | 0.032 |
| CRIES-13 Total score | -0.642 (-0.928 - -0.356) | -0.546 | <0.001 | -0.471 (-0.787 - -0.154) | -0.400 | 0.004 |
| Adjusted R2 | - | 0.337 | ||||
| Regression model | - | F=13.436, p<0.001 | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.





