Submitted:
19 February 2026
Posted:
27 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. The Oligodendrocyte Lineage: A Developmental Overview
1.2. The Microbiota-Gut-Brain Axis: A Paradigm Shift
1.3. Microbial Metabolites: The Molecular Messengers
- 1.
- 2.
- 3.
1.4. Scope and Objectives of This Review
- The pathways through which metabolites access the CNS
- Epigenetic mechanisms, particularly HDAC inhibition by SCFAs
- G-protein coupled receptor (GPCR) signaling cascades
- Metabolic support of myelin lipid biosynthesis
- Clinical implications for Multiple Sclerosis and other demyelinating disorders
- Therapeutic frontiers including postbiotics and dietary interventions
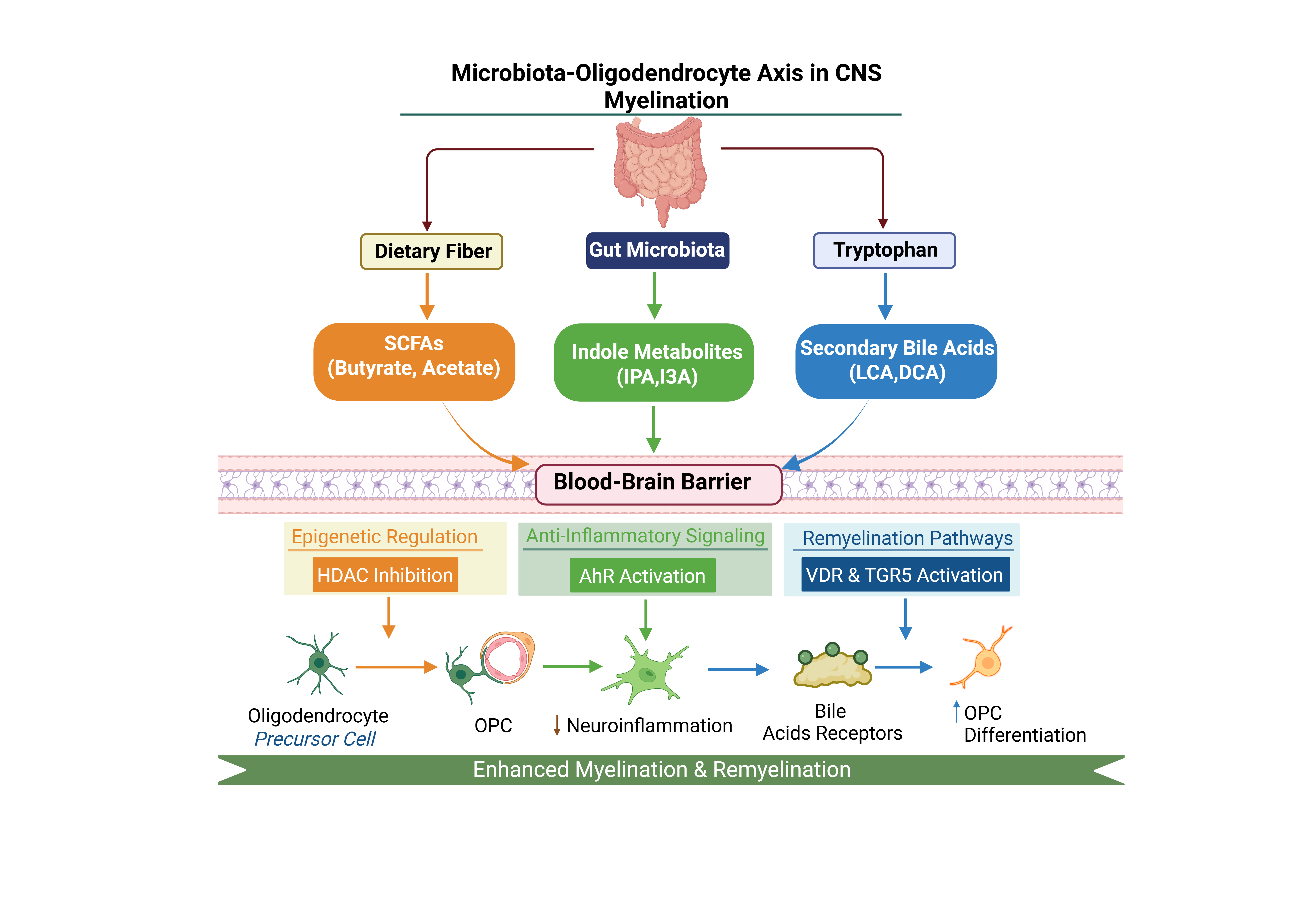
2. The Microbiota-Gut-Brain Axis: Pathways of Metabolite Delivery
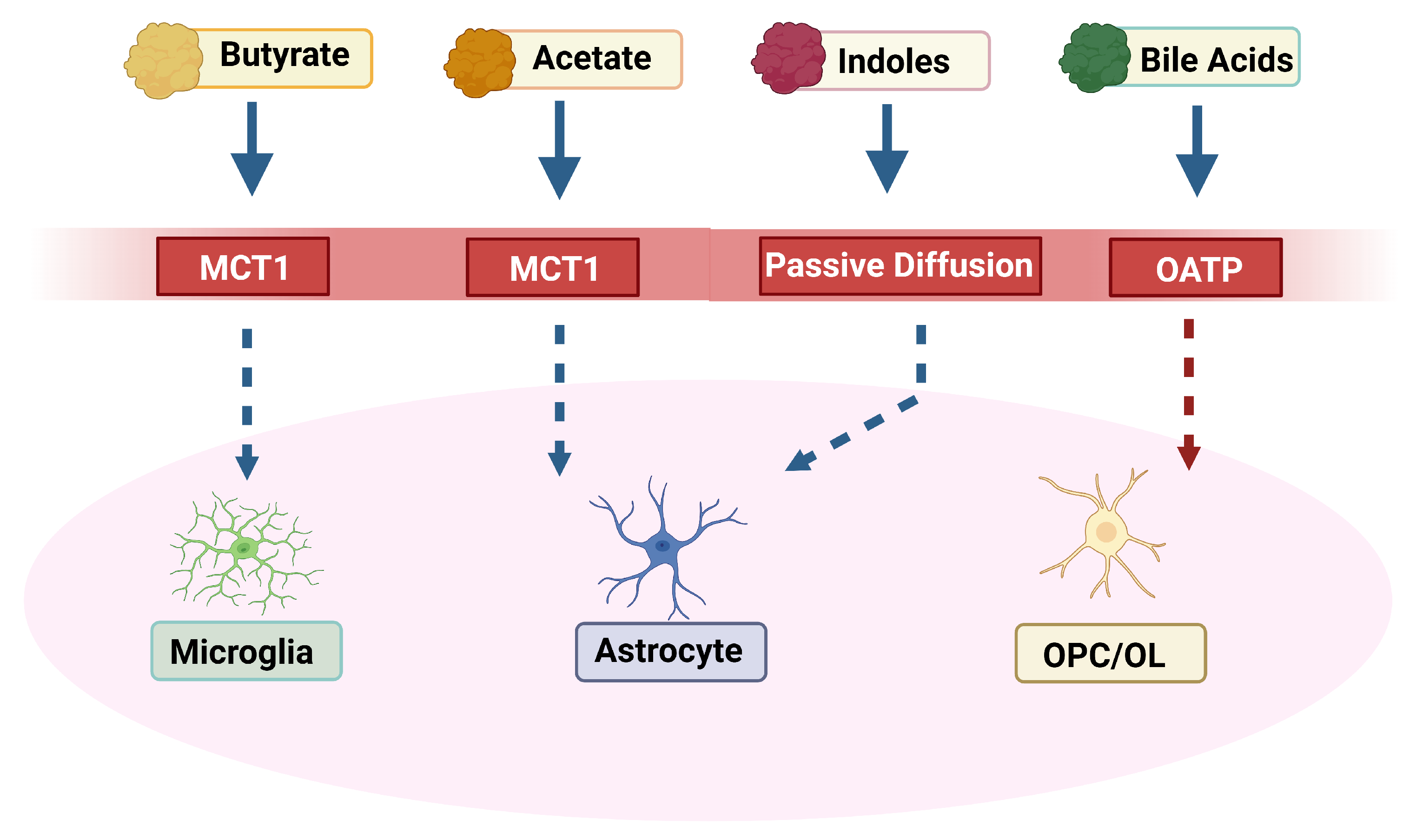
2.1. Intestinal Absorption and Systemic Distribution
2.2. Blood-Brain Barrier Penetration
2.3. Microglial and Astrocytic Intermediaries
2.3.1. Microglial Maturation and Function
2.3.2. Astrocyte-Oligodendrocyte Crosstalk
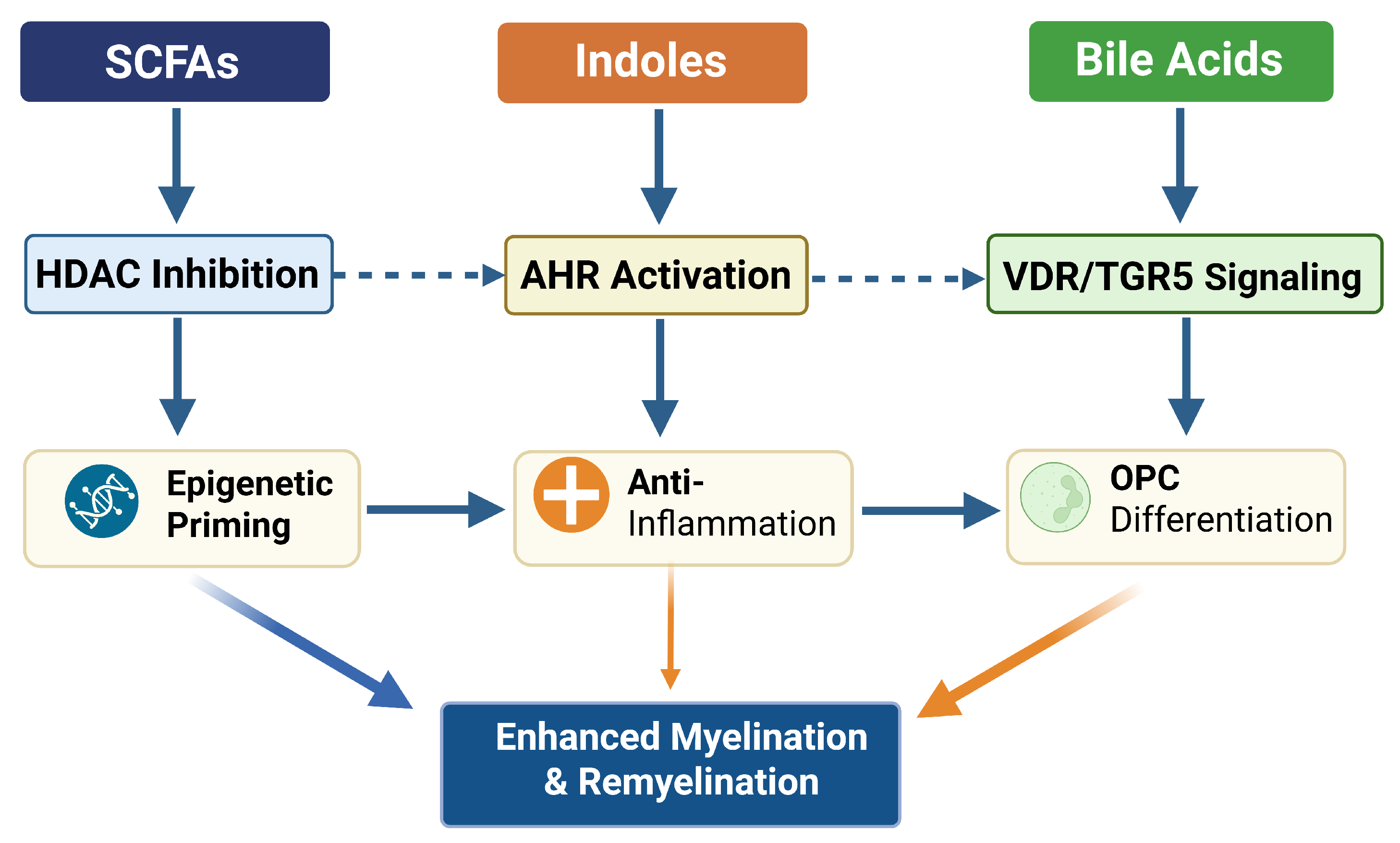
3. Short-Chain Fatty Acids: Epigenetic and Metabolic Regulation
3.1. SCFA Biosynthesis and Distribution
- Acetate: Produced via acetyl-CoA and the Wood-Ljungdahl pathway by diverse bacterial taxa including Bacteroides, Bifidobacterium, and Akkermansia [42].
- Propionate: Synthesized through the succinate, acrylate, and propanediol pathways, primarily by Bacteroidetes, Veillonella, and Propionibacterium [43].
3.2. Butyrate as a Histone Deacetylase Inhibitor

3.2.1. HDAC Function in Oligodendrocyte Development
3.2.2. Experimental Evidence for HDAC Inhibition in Myelination
- 1.
- 2.
- In vitro treatment of OPCs with butyrate or other HDAC inhibitors (valproic acid, trichostatin A) promotes expression of MBP and PLP [50].
- 3.
- Cuprizone-induced demyelination models show accelerated remyelination following butyrate supplementation [51].
- 4.
- Chromatin immunoprecipitation (ChIP) studies demonstrate increased histone H3 and H4 acetylation at myelin gene promoters following SCFA treatment [18].
3.3. G-Protein Coupled Receptor Signaling
3.3.1. GPR41 and GPR43
- GPR43: Highest affinity for acetate and propionate; expressed on microglia and immune cells
- GPR41: Highest affinity for propionate and butyrate; expressed on neurons and enteroendocrine cells
3.3.2. GPR109A (Hydroxycarboxylic Acid Receptor 2)
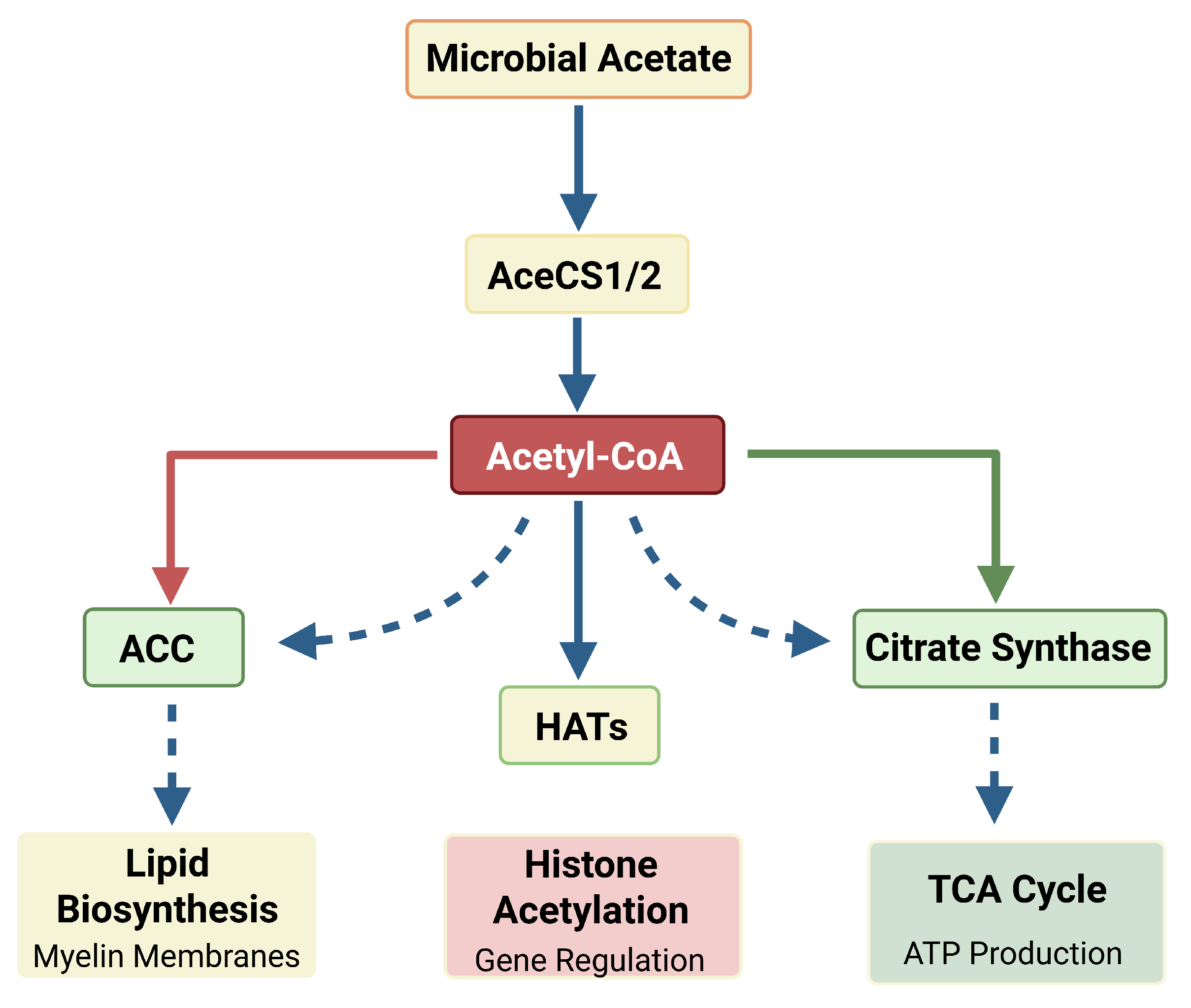
3.4. Acetate and the Acetyl-CoA Hub
3.4.1. Acetyl-CoA Synthetase Pathways
3.4.2. Implications for Myelination
3.5. Propionate: Immunomodulation and Beyond
3.5.1. Regulatory T Cell Induction
4. Tryptophan-Derived Metabolites and AhR Signaling
4.1. Microbial Tryptophan Metabolism
- 1.
- Indole pathway: Tryptophanase (TnaA) converts tryptophan to indole, which can be further modified to produce indole-3-aldehyde, indole-3-acetic acid, and indole-3-propionic acid (IPA) [68].
- 2.
- Tryptamine pathway: Decarboxylation produces tryptamine, a neuroactive amine [69].
- 3.
- Kynurenine pathway: Some bacteria possess indoleamine 2,3-dioxygenase (IDO) homologs that produce kynurenine metabolites [70].
4.2. The Aryl Hydrocarbon Receptor (AhR)
4.2.1. Mechanism of AhR Activation
4.2.2. AhR in Astrocytes: Suppression of Neuroinflammation
- AhR activation suppresses NF-B signaling in astrocytes
- Production of pro-inflammatory cytokines (IL-6, TNF-, CCL2) is reduced
- Astrocytes adopt a neuroprotective phenotype supporting oligodendrocyte survival
4.2.3. AhR in Microglia
4.3. Indole-3-Propionic Acid: A Potent Neuroprotectant
4.3.1. Antioxidant Properties
4.3.2. Pregnane X Receptor Activation
4.4. Clinical Relevance: Tryptophan Metabolism in MS
5. Secondary Bile Acids: Emerging Regulators of Remyelination
5.1. Bile Acid Metabolism and the Microbiota
- Deconjugation: Bile salt hydrolases remove glycine/taurine conjugates
- 7-dehydroxylation: Produces lithocholic acid (LCA) from chenodeoxycholic acid and deoxycholic acid (DCA) from cholic acid
- Epimerization and oxidation: Generate additional bile acid species
5.2. Bile Acid Receptors in the CNS
5.2.1. TGR5 (GPBAR1)
- Suppresses microglial inflammatory responses via cAMP elevation
- Reduces production of TNF- and IL-1
- Promotes neuroprotective microglial phenotypes
5.2.2. Vitamin D Receptor (VDR)
- Promotes OPC differentiation into mature oligodendrocytes
- Enhances expression of myelin genes
- Provides neuroprotection against inflammatory damage
5.3. Bile Acids and Remyelination
- 1.
- Tauroursodeoxycholic acid (TUDCA) administration in EAE models reduces demyelination and enhances OPC differentiation [86].
- 2.
- Ursodeoxycholic acid (UDCA) is being evaluated in clinical trials for progressive MS based on its neuroprotective and immunomodulatory properties [87].
- 3.
- MS patients show altered bile acid profiles with reduced secondary bile acids, suggesting dysbiosis-driven deficiency [88].
6. Metabolic Support of Axonal Integrity and Bioenergetics
6.1. The Monocarboxylate Transporter Shuttle
- 1.
- Oligodendrocytes take up glucose via GLUT1 or utilize glycogen stores
- 2.
- Glycolytic metabolism produces pyruvate, which is converted to lactate
- 3.
- MCT1 on the oligodendrocyte membrane exports lactate into the periaxonal space
- 4.
- Axonal MCT2 imports lactate for mitochondrial oxidation
6.2. Microbial Contribution to the Metabolic Axis
6.2.1. Direct Fuel Provision
6.2.2. Support of Lipid Synthesis
- Cholesterol biosynthesis (the mevalonate pathway)
- Fatty acid synthesis (via malonyl-CoA)
- Sphingolipid production (ceramide and galactocerebroside)
6.3. Mitochondrial Function and Oxidative Stress
- High iron content (required for myelin synthesis enzymes)
- Low levels of antioxidant enzymes (catalase, superoxide dismutase)
- Abundant polyunsaturated fatty acids in myelin membranes
7. Clinical Implications: Multiple Sclerosis and Demyelinating Disorders
7.1. Dysbiosis in Multiple Sclerosis
| Bacterial Taxon | Change in MS | Metabolic Impact |
|---|---|---|
| Faecalibacterium prausnitzii | Reduced butyrate | |
| Prevotella species | ↓ | Reduced propionate |
| Butyricimonas | ↓ | Reduced butyrate |
| Lactobacillus species | ↓ | Reduced indoles |
| Clostridium clusters IV/XIVa | ↓ | Reduced SCFAs/bile acids |
| Methanobrevibacter | ↑ | Altered fermentation |
| Akkermansia muciniphila | Variable | Context-dependent |
7.2. The Differentiation Block in Chronic Lesions
7.3. Modulation of Neuroinflammation
7.3.1. The Treg-Th17 Balance
- 1.
- 2.
- GPR43 activation: SCFA signaling through GPR43 on T cells promotes Treg differentiation [63].
- 3.
7.3.2. Microglial Polarization
7.4. Gut-Brain Axis in MS: Clinical Evidence

8. Experimental Models: Evidence from Germ-Free and Intervention Studies
8.1. Germ-Free Mouse Studies
8.1.1. Myelination Defects in GF Mice
- 1.
- 2.
- Transcriptomic changes: RNA-seq reveals downregulation of cholesterol biosynthesis genes (Hmgcr, Fdft1, Sqle) and myelin structural genes [17]
- 3.
- Altered white matter volume: MRI studies show reduced white matter in prefrontal regions [115]
- 4.
- OPC differentiation defects: Reduced numbers of mature CC1+ oligodendrocytes in corpus callosum [18]
8.1.2. Reversibility and Critical Windows
- Early colonization (weaning): Nearly complete rescue of myelin gene expression and white matter structure [17]
- Adult colonization: Partial rescue, suggesting a critical developmental window [18]
- SCFA supplementation alone: Substantial rescue even without live bacteria, demonstrating metabolite sufficiency [35]
8.2. Antibiotic Perturbation Studies
8.2.1. Effects of Antibiotic-Induced Dysbiosis
8.2.2. Recovery Following Antibiotic Cessation
8.3. Demyelination-Remyelination Models
8.3.1. Cuprizone Model
8.3.2. Experimental Autoimmune Encephalomyelitis (EAE)
| Model System | Manipulation | Myelination Outcome | Key References |
|---|---|---|---|
| Germ-free mice | Absence of microbiota | Hypomyelination (PFC) | [17,18] |
| Antibiotic-treated | Broad-spectrum ABx | Reduced myelin genes | [18,35] |
| Cuprizone + GF | Demyelination in GF | Delayed remyelination | [119] |
| Cuprizone + butyrate | SCFA supplementation | Accelerated repair | [51] |
| EAE + GF | Autoimmune model | Reduced severity | [123,124] |
| EAE + propionate | SCFA supplementation | Reduced severity; ↑ Tregs | [65,66] |
| EAE + probiotics | Live bacteria | Reduced severity | [126,169] |
8.4. Colonization Studies with Defined Communities
- Altered Schaedler Flora (ASF): An 8-member defined community that partially rescues GF phenotypes [128]
- Synthetic communities: Custom-designed consortia enriched in SCFA producers show enhanced myelination support [129]
- Monocolonization: Single-strain colonization identifies specific bacterial contributions [130]
9. Therapeutic Frontiers: From Probiotics to Precision Postbiotics
9.1. Dietary Interventions
9.1.1. High-Fiber Diets
9.1.2. Ketogenic and Low-Carbohydrate Diets
9.1.3. Mediterranean Diet
9.2. Probiotic and Synbiotic Approaches
9.2.1. Traditional Probiotics
9.2.2. Next-Generation Probiotics
9.2.3. Synbiotics
9.3. Postbiotic Supplementation

9.3.1. SCFA Supplementation
- Oral butyrate: Sodium butyrate or tributyrin (a butyrate prodrug) can be administered orally; shows efficacy in demyelination models [51]
- Oral propionate: Clinical trials in MS show increased Tregs and reduced relapse rates [66]
- Challenges: Rapid metabolism limits systemic exposure; enteric coating or prodrug formulations improve bioavailability [28]
9.3.2. Indole Derivatives
9.3.3. Bile Acid Therapeutics
9.4. Fecal Microbiota Transplantation
- Rationale: Restores entire microbial ecosystem including metabolite-producing capacity
- MS case reports: Anecdotal reports of improvement following FMT [152]
- Clinical trials: Phase I/II trials in MS are underway (ClinicalTrials.gov identifiers NCT03594487, NCT04203017)
- Challenges: Standardization, safety (infection risk), and donor selection remain obstacles [153]
9.5. Personalized Approaches and Biomarkers
10. Critical Discussion and Future Perspectives
10.1. Translational Challenges
10.1.1. Species Differences
10.1.2. Causality vs. Correlation
10.2. Mechanistic Gaps
10.2.1. Direct vs. Indirect Effects
10.2.2. Metabolite Synergies
10.2.3. Timing and Critical Windows
- 1.
- Can these windows be “reopened” in adults through pharmacological or environmental interventions?
- 2.
- Are there distinct windows for initial myelination versus remyelination after injury?
- 3.
- How do early-life antibiotic exposures affect long-term myelination capacity?
10.3. Future Directions
10.3.1. Advanced Imaging
10.3.2. Single-Cell Approaches
10.3.3. Organoid Systems
10.3.4. Clinical Trial Design
- Stratification by baseline microbiome composition
- Metabolomic endpoints (circulating SCFA, indole levels)
- Myelin-specific imaging outcomes
- Long-term follow-up for disability progression
11. Conclusions

Abbreviations
| AhR | Aryl Hydrocarbon Receptor |
| BBB | Blood-Brain Barrier |
| CNS | Central Nervous System |
| DCA | Deoxycholic Acid |
| EAE | Experimental Autoimmune Encephalomyelitis |
| FMT | Fecal Microbiota Transplantation |
| GF | Germ-Free |
| GPCR | G-Protein Coupled Receptor |
| HDAC | Histone Deacetylase |
| IPA | Indole-3-Propionic Acid |
| LCA | Lithocholic Acid |
| MBP | Myelin Basic Protein |
| MCT | Monocarboxylate Transporter |
| MS | Multiple Sclerosis |
| OL | Oligodendrocyte |
| OPC | Oligodendrocyte Precursor Cell |
| PLP | Proteolipid Protein |
| SCFA | Short-Chain Fatty Acid |
| TGR5 | Takeda G-Protein Receptor 5 |
| Treg | Regulatory T Cell |
| VDR | Vitamin D Receptor |
References
- Nave, K.A. Myelination and support of axonal integrity by glia. Nature 2010, 468(7321), 244–252. [Google Scholar] [CrossRef]
- Simons, M.; Nave, K.A. Oligodendrocytes: Myelination and axonal support. Cold Spring Harbor Perspectives in Biology 2015, 8(1), a020479. [Google Scholar] [CrossRef]
- Fünfschilling, U.; et al. Glycolytic oligodendrocytes maintain myelin and long-term axonal integrity. Nature 2012, 485(7399), 517–521. [Google Scholar] [CrossRef]
- Lee, Y.; et al. Oligodendroglia metabolically support axons and contribute to neurodegeneration. Nature 2012, 487(7408), 443–448. [Google Scholar] [CrossRef]
- Ronchi, G.; Pellegrino, D. Gut microbiota regulates optic nerve fiber myelination. Frontiers in Cell and Developmental Biology 2025, 13, 1526855. [Google Scholar] [CrossRef]
- Kessaris, N.; et al. Competing waves of oligodendrocytes in the forebrain and postnatal elimination of an embryonic lineage. Nature Neuroscience 2006, 9(2), 173–179. [Google Scholar] [CrossRef]
- Richardson, W.D.; et al. Oligodendrocyte wars. Nature Reviews Neuroscience 2006, 7(1), 11–18. [Google Scholar] [CrossRef]
- Emery, B. Regulation of oligodendrocyte differentiation and myelination. Science 2010, 330(6005), 779–782. [Google Scholar] [CrossRef] [PubMed]
- Zuchero, J.B.; et al. CNS myelin wrapping is driven by actin disassembly. Developmental Cell 2015, 34(2), 152–167. [Google Scholar] [CrossRef] [PubMed]
- Fancy, S.P.; et al. Myelin regeneration: A recapitulation of development? Annual Review of Neuroscience 2011, 34, 21–43. [Google Scholar] [CrossRef] [PubMed]
- Mitew, S.; et al. Mechanisms regulating the development of oligodendrocytes and central nervous system myelin. Neuroscience 2014, 276, 29–47. [Google Scholar] [CrossRef] [PubMed]
- Sender, R.; et al. Revised estimates for the number of human and bacteria cells in the body. PLoS Biology 2016, 14(8), e1002533. [Google Scholar] [CrossRef] [PubMed]
- Cryan, J.F.; et al. The microbiota-gut-brain axis. Physiological Reviews 2019, 99(4), 1877–2013. [Google Scholar] [CrossRef] [PubMed]
- Sharon, G.; et al. The central nervous system and the gut microbiome. Cell 2016, 167(4), 915–932. [Google Scholar] [CrossRef]
- Mayer, E.A.; et al. Gut microbes and the brain: Paradigm shift in neuroscience. Journal of Neuroscience 2015, 35(41), 13857–13867. [Google Scholar] [CrossRef]
- Dinan, T.G.; Cryan, J.F. Gut instincts: Microbiota as a key regulator of brain development, ageing and neurodegeneration. Journal of Physiology 2017, 595(2), 489–503. [Google Scholar] [CrossRef]
- Hoban, A.E.; et al. Regulation of prefrontal cortex myelination by the microbiota. Translational Psychiatry 2016, 6(4), e774. [Google Scholar] [CrossRef]
- Gacias, M.; et al. Microbiota-driven transcriptional changes in prefrontal cortex override genetic differences in social behavior. eLife 2016, 5, e13442. [Google Scholar] [CrossRef]
- Cummings, J.H.; et al. Short chain fatty acids in human large intestine, portal, hepatic and venous blood. Gut 1987, 28(10), 1221–1227. [Google Scholar] [CrossRef]
- Dalile, B.; et al. The role of short-chain fatty acids in microbiota-gut-brain communication. Nature Reviews Gastroenterology & Hepatology 2019, 16(8), 461–478. [Google Scholar] [CrossRef]
- Agus, A.; et al. Gut microbiota regulation of tryptophan metabolism in health and disease. Cell Host & Microbe 2018, 23(6), 716–724. [Google Scholar] [CrossRef] [PubMed]
- Roager, H.M.; Licht, T.R. Microbial tryptophan catabolites in health and disease. Nature Communications 2018, 9(1), 3294. [Google Scholar] [CrossRef] [PubMed]
- Wahlström, A.; et al. Intestinal crosstalk between bile acids and microbiota and its impact on host metabolism. Cell Metabolism 2016, 24(1), 41–50. [Google Scholar] [CrossRef] [PubMed]
- MahmoudianDehkordi, S.; et al. Altered bile acid profile associates with cognitive impairment in Alzheimer’s disease - An emerging role for gut microbiome. Alzheimer’s & Dementia 2019, 15(1), 76–92. [Google Scholar] [CrossRef]
- Sivaprakasam, S.; et al. Short-chain fatty acid transporters: Role in colonic homeostasis. Comprehensive Physiology 2017, 8(1), 299–314. [Google Scholar] [CrossRef]
- Borthakur, A.; et al. Regulation of monocarboxylate transporter 1 (MCT1) promoter by butyrate in human intestinal epithelial cells. American Journal of Physiology-Gastrointestinal and Liver Physiology 2008, 294(1), G241–G247. [Google Scholar] [CrossRef]
- Bloemen, J.G.; et al. Short chain fatty acids exchange across the gut and liver in humans measured at surgery. Clinical Nutrition 2009, 28(6), 657–661. [Google Scholar] [CrossRef]
- Boets, E.; et al. Systemic availability and metabolism of colonic-derived short-chain fatty acids in healthy subjects. American Journal of Physiology-Gastrointestinal and Liver Physiology 2017, 312(1), G37–G47. [Google Scholar] [CrossRef]
- Wikoff, W.R.; et al. Metabolomics analysis reveals large effects of gut microflora on mammalian blood metabolites. Proceedings of the National Academy of Sciences 2009, 106(10), 3698–3703. [Google Scholar] [CrossRef]
- Dawson, P.A.; et al. Bile acid transporters. Journal of Lipid Research 2009, 50(12), 2340–2357. [Google Scholar] [CrossRef]
- Braniste, V.; et al. The gut microbiota influences blood-brain barrier permeability in mice. Science Translational Medicine 2014, 6(263), 263ra158. [Google Scholar] [CrossRef] [PubMed]
- MacFabe, D.F. Short-chain fatty acid fermentation products of the gut microbiome: Implications in autism spectrum disorders. Microbial Ecology in Health and Disease 2012, 23, 19260. [Google Scholar] [CrossRef] [PubMed]
- Venkatesh, M.; et al. Symbiotic bacterial metabolites regulate gastrointestinal barrier function via the xenobiotic sensor PXR and Toll-like receptor 4. Immunity 2014, 41(2), 296–310. [Google Scholar] [CrossRef] [PubMed]
- Mertens, K.L.; et al. Bile acid signaling pathways from the enterohepatic circulation to the central nervous system. Frontiers in Neuroscience 2017, 11, 617. [Google Scholar] [CrossRef]
- Erny, D.; et al. Host microbiota constantly control maturation and function of microglia in the CNS. Nature Neuroscience 2015, 18(7), 965–977. [Google Scholar] [CrossRef]
- Lampron, A.; et al. Inefficient clearance of myelin debris by microglia impairs remyelinating processes. Journal of Experimental Medicine 2015, 212(4), 481–495. [Google Scholar] [CrossRef]
- Lloyd, A.F.; et al. Central nervous system regeneration is driven by microglia necroptosis and repopulation. Nature Neuroscience 2019, 22(7), 1046–1052. [Google Scholar] [CrossRef]
- Miron, V.E.; et al. M2 microglia and macrophages drive oligodendrocyte differentiation during CNS remyelination. Nature Neuroscience 2013, 16(9), 1211–1218. [Google Scholar] [CrossRef]
- Rothhammer, V.; et al. Type I interferons and microbial metabolites of tryptophan modulate astrocyte activity and CNS inflammation via the AhR. Nature Medicine 2016, 22(6), 586–597. [Google Scholar] [CrossRef]
- Orthmann-Murphy, J.L.; et al. Gap junctions couple astrocytes and oligodendrocytes. Journal of Molecular Neuroscience 2011, 35(1), 101–116. [Google Scholar] [CrossRef]
- Tress, O.; et al. Panglial gap junctional communication is essential for maintenance of myelin in the CNS. Journal of Neuroscience 2012, 32(22), 7499–7518. [Google Scholar] [CrossRef] [PubMed]
- Louis, P.; et al. The gut microbiota, bacterial metabolites and colorectal cancer. Nature Reviews Microbiology 2014, 12(10), 661–672. [Google Scholar] [CrossRef] [PubMed]
- Reichardt, N.; et al. Phylogenetic distribution of three pathways for propionate production within the human gut microbiota. The ISME Journal 2014, 8(6), 1323–1335. [Google Scholar] [CrossRef] [PubMed]
- Louis, P.; Flint, H.J. Diversity, metabolism and microbial ecology of butyrate-producing bacteria from the human large intestine. FEMS Microbiology Letters 2010, 294(1), 1–8. [Google Scholar] [CrossRef]
- Vital, M.; et al. Revealing the bacterial butyrate synthesis pathways by analyzing (meta) genomic data. mBio 2014, 5(2), e00889-14. [Google Scholar] [CrossRef]
- Davie, J.R. Inhibition of histone deacetylase activity by butyrate. The Journal of Nutrition 2003, 133(7), 2485S–2493S. [Google Scholar] [CrossRef]
- Waldecker, M.; et al. Inhibition of histone-deacetylase activity by short-chain fatty acids and some polyphenol metabolites formed in the colon. The Journal of Nutritional Biochemistry 2008, 19(9), 587–593. [Google Scholar] [CrossRef]
- Shen, S.; et al. Age-dependent epigenetic control of differentiation inhibitors is critical for remyelination efficiency. Nature Neuroscience 2008, 11(9), 1024–1034. [Google Scholar] [CrossRef]
- Conway, G.D.; et al. Histone deacetylase activity is required for human oligodendrocyte progenitor differentiation. Glia 2012, 60(12), 1944–1953. [Google Scholar] [CrossRef]
- Shen, S.; et al. Histone modifications affect timing of oligodendrocyte progenitor differentiation in the developing rat brain. Journal of Cell Biology 2005, 169(4), 577–589. [Google Scholar] [CrossRef]
- Chen, T.; et al. Butyrate suppresses demyelination and enhances remyelination. Journal of Neuroinflammation 2019, 16(1), 165. [Google Scholar] [CrossRef]
- Brown, A.J.; et al. The orphan G protein-coupled receptors GPR41 and GPR43 are activated by propionate and other short chain carboxylic acids. Journal of Biological Chemistry 2003, 278(13), 11312–11319. [Google Scholar] [CrossRef] [PubMed]
- Le Poul, E.; et al. Functional characterization of human receptors for short chain fatty acids and their role in polymorphonuclear cell activation. Journal of Biological Chemistry 2003, 278(28), 25481–25489. [Google Scholar] [CrossRef] [PubMed]
- Kimura, I.; et al. Short-chain fatty acids and ketones directly regulate sympathetic nervous system via G protein-coupled receptor 41 (GPR41). Proceedings of the National Academy of Sciences 2011, 108(19), 8030–8035. [Google Scholar] [CrossRef] [PubMed]
- Thangaraju, M.; et al. GPR109A is a G-protein–coupled receptor for the bacterial fermentation product butyrate and functions as a tumor suppressor in colon. Cancer Research 2009, 69(7), 2826–2832. [Google Scholar] [CrossRef]
- Fu, S.P.; et al. Anti-inflammatory effects of BHBA in both in vivo and in vitro Parkinson’s disease models are mediated by GPR109A-dependent mechanisms. Journal of Neuroinflammation 2015, 12(1), 9. [Google Scholar] [CrossRef]
- Mews, P.; et al. Acetyl-CoA synthetase regulates histone acetylation and hippocampal memory. Nature 2017, 546(7658), 381–386. [Google Scholar] [CrossRef]
- Camargo, N.; et al. Oligodendroglial myelination requires astrocyte-derived lipids. PLoS Biology 2017, 15(5), e1002605. [Google Scholar] [CrossRef]
- Wellen, K.E.; et al. ATP-citrate lyase links cellular metabolism to histone acetylation. Science 2009, 324(5930), 1076–1080. [Google Scholar] [CrossRef]
- Pietrocola, F.; et al. Acetyl coenzyme A: A central metabolite and second messenger. Cell Metabolism 2015, 21(6), 805–821. [Google Scholar] [CrossRef]
- Saher, G.; et al. Cholesterol in myelin biogenesis and hypomyelinating disorders. Biochimica et Biophysica Acta 2011, 1811(12), 698–703. [Google Scholar] [CrossRef]
- Mews, P.; et al. Alcohol metabolism contributes to brain histone acetylation. Nature 2019, 574(7780), 717–721. [Google Scholar] [CrossRef] [PubMed]
- Smith, P.M.; et al. The microbial metabolites, short-chain fatty acids, regulate colonic Treg cell homeostasis. Science 2013, 341(6145), 569–573. [Google Scholar] [CrossRef] [PubMed]
- Arpaia, N.; et al. Metabolites produced by commensal bacteria promote peripheral regulatory T-cell generation. Nature 2013, 504(7480), 451–455. [Google Scholar] [CrossRef] [PubMed]
- Haghikia, A.; et al. Dietary fatty acids directly impact central nervous system autoimmunity via the small intestine. Immunity 2015, 43(4), 817–829. [Google Scholar] [CrossRef]
- Duscha, A.; et al. Propionic acid shapes the multiple sclerosis disease course by an immunomodulatory mechanism. Cell 2020, 180(6), 1067–1080. [Google Scholar] [CrossRef]
- Gao, K.; et al. Tryptophan metabolism: A link between the gut microbiota and brain. Advances in Nutrition 2020, 11(3), 709–723. [Google Scholar] [CrossRef]
- Zelante, T.; et al. Tryptophan catabolites from microbiota engage aryl hydrocarbon receptor and balance mucosal reactivity via interleukin-22. Immunity 2013, 39(2), 372–385. [Google Scholar] [CrossRef]
- Williams, B.B.; et al. Discovery and characterization of gut microbiota decarboxylases that can produce the neurotransmitter tryptamine. Cell Host & Microbe 2014, 16(4), 495–503. [Google Scholar] [CrossRef]
- Gao, J.; et al. Impact of the gut microbiota on intestinal immunity mediated by tryptophan metabolism. Frontiers in Cellular and Infection Microbiology 2018, 8, 13. [Google Scholar] [CrossRef]
- Quintana, F.J.; Sherr, D.H. Aryl hydrocarbon receptor control of adaptive immunity. Pharmacological Reviews 2013, 65(4), 1148–1161. [Google Scholar] [CrossRef] [PubMed]
- Denison, M.S.; Nagy, S.R. Activation of the aryl hydrocarbon receptor by structurally diverse exogenous and endogenous chemicals. Annual Review of Pharmacology and Toxicology 2003, 43(1), 309–334. [Google Scholar] [CrossRef] [PubMed]
- Rothhammer, V.; et al. Microglial control of astrocytes in response to microbial metabolites. Nature 2018, 557(7707), 724–728. [Google Scholar] [CrossRef] [PubMed]
- Dodd, D.; et al. A gut bacterial pathway metabolizes aromatic amino acids into nine circulating metabolites. Nature 2017, 551(7682), 648–652. [Google Scholar] [CrossRef]
- Thorburne, S.K.; Bhagat, S. Low glutathione and high iron govern the susceptibility of oligodendroglial precursors to oxidative stress. Journal of Neurochemistry 1996, 67(3), 1014–1022. [Google Scholar] [CrossRef]
- Lim, C.K.; et al. Kynurenine pathway metabolomics predicts and provides mechanistic insight into multiple sclerosis progression. Scientific Reports 2017, 7(1), 41473. [Google Scholar] [CrossRef]
- Hartai, Z.; et al. Kynurenine metabolism in multiple sclerosis. Acta Neurologica Scandinavica 2005, 112(2), 93–96. [Google Scholar] [CrossRef]
- Chen, J.; et al. Multiple sclerosis patients have a distinct gut microbiota compared to healthy controls. Scientific Reports 2016, 6(1), 28484. [Google Scholar] [CrossRef]
- Jangi, S.; et al. Alterations of the human gut microbiome in multiple sclerosis. Nature Communications 2016, 7(1), 12015. [Google Scholar] [CrossRef]
- Ridlon, J.M.; et al. Bile salt biotransformations by human intestinal bacteria. Journal of Lipid Research 2006, 47(2), 241–259. [Google Scholar] [CrossRef]
- Gérard, P. Metabolism of cholesterol and bile acids by the gut microbiota. Pathogens 2013, 3(1), 14–24. [Google Scholar] [CrossRef] [PubMed]
- Keitel, V.; et al. The G-protein coupled bile salt receptor TGR5 is expressed in liver sinusoidal endothelial cells. Hepatology 2019, 70(3), 982–997. [Google Scholar] [CrossRef] [PubMed]
- Makishima, M.; et al. Vitamin D receptor as an intestinal bile acid sensor. Science 2002, 296(5571), 1313–1316. [Google Scholar] [CrossRef] [PubMed]
- Munger, K.L.; et al. Serum 25-hydroxyvitamin D levels and risk of multiple sclerosis. JAMA 2006, 296(23), 2832–2838. [Google Scholar] [CrossRef]
- Mokry, L.E.; et al. Vitamin D and risk of multiple sclerosis: A Mendelian randomization study. PLoS Medicine 2015, 12(8), e1001866. [Google Scholar] [CrossRef]
- Yanguas-Casás, N.; et al. TUDCA: An agonist of the bile acid receptor GPBAR1/TGR5 with anti-inflammatory effects in microglial cells. Journal of Cellular Physiology 2017, 232(8), 2231–2245. [Google Scholar] [CrossRef]
- Parry, G.J.; et al. Safety, tolerability, and cerebrospinal fluid penetration of ursodeoxycholic acid in patients with amyotrophic lateral sclerosis. Clinical Neuropharmacology 2010, 33(1), 17–21. [Google Scholar] [CrossRef]
- Bhargava, P.; et al. Bile acid metabolism is altered in multiple sclerosis and supplementation ameliorates neuroinflammation. Journal of Clinical Investigation 2020, 130(7), 3467–3482. [Google Scholar] [CrossRef]
- Moschen, A.R.; et al. Dietary factors: Major regulators of the gut’s microbiota. Gut & Liver 2012, 6(4), 411–416. [Google Scholar] [CrossRef]
- Dong, W.; et al. Sodium butyrate activates NRF2 to ameliorate diabetic nephropathy possibly via inhibition of HDAC. Journal of Endocrinology 2014, 223(1), 27–36. [Google Scholar] [CrossRef]
- Walton, C.; et al. Rising prevalence of multiple sclerosis worldwide: Insights from the Atlas of MS, third edition. Multiple Sclerosis Journal 2020, 26(14), 1816–1821. [Google Scholar] [CrossRef] [PubMed]
- Kuhlmann, T.; et al. Differentiation block of oligodendroglial progenitor cells as a cause for remyelination failure in chronic multiple sclerosis. Brain 2008, 131(7), 1749–1758. [Google Scholar] [CrossRef] [PubMed]
- Franklin, R.J. Why does remyelination fail in multiple sclerosis? Nature Reviews Neuroscience 2002, 3(9), 705–714. [Google Scholar] [CrossRef] [PubMed]
- Chang, A.; et al. Premyelinating oligodendrocytes in chronic lesions of multiple sclerosis. New England Journal of Medicine 2002, 346(3), 165–173. [Google Scholar] [CrossRef] [PubMed]
- Syed, Y.A.; et al. Inhibition of CNS remyelination by the presence of semaphorin 3A. Journal of Neuroscience 2011, 31(10), 3719–3728. [Google Scholar] [CrossRef]
- Fancy, S.P.; et al. Dysregulation of the Wnt pathway inhibits timely myelination and remyelination in the mammalian CNS. Genes & Development 2009, 23(13), 1571–1585. [Google Scholar] [CrossRef]
- Moyon, S.; et al. Demyelination causes adult CNS progenitors to revert to an immature state and express immune cues that support their migration. Journal of Neuroscience 2017, 37(5), 1197–1210. [Google Scholar] [CrossRef]
- Koreman, E.; et al. Chromatin remodeling and epigenetic regulation of oligodendrocyte myelination and myelin repair. Molecular and Cellular Neuroscience 2018, 87, 18–26. [Google Scholar] [CrossRef]
- Rone, M.B.; et al. Oligodendrogliopathy in multiple sclerosis: Low glycolytic metabolic rate promotes oligodendrocyte survival. Journal of Neuroscience 2016, 36(17), 4698–4707. [Google Scholar] [CrossRef]
- Schirmer, L.; et al. Neuronal vulnerability and multilineage diversity in multiple sclerosis. Nature 2019, 573(7772), 75–82. [Google Scholar] [CrossRef]
- Absinta, M.; et al. A lymphocyte–microglia–astrocyte axis in chronic active multiple sclerosis. Nature 2021, 597(7878), 709–714. [Google Scholar] [CrossRef] [PubMed]
- Lassmann, H. Multiple sclerosis pathology. Cold Spring Harbor Perspectives in Medicine 2018, 8(3), a028936. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Q.; et al. Gut dysbiosis and lack of short chain fatty acids in a Chinese cohort of patients with multiple sclerosis. Neurochemistry International 2019, 129, 104468. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; et al. Short-chain fatty acids induce both effector and regulatory T cells by suppression of histone deacetylases. Mucosal Immunology 2015, 8(1), 80–93. [Google Scholar] [CrossRef]
- Korn, T.; et al. IL-17 and Th17 cells. Annual Review of Immunology 2009, 27, 485–517. [Google Scholar] [CrossRef]
- Kleinewietfeld, M.; et al. Sodium chloride drives autoimmune disease by the induction of pathogenic TH17 cells. Nature 2013, 496(7446), 518–522. [Google Scholar] [CrossRef]
- Furusawa, Y.; et al. Commensal microbe-derived butyrate induces the differentiation of colonic regulatory T cells. Nature 2013, 504(7480), 446–450. [Google Scholar] [CrossRef]
- Nastasi, C.; et al. The effect of short-chain fatty acids on human monocyte-derived dendritic cells. Scientific Reports 2015, 5(1), 16148. [Google Scholar] [CrossRef]
- Kaisar, M.M.; et al. Butyrate conditions human dendritic cells to prime type 1 regulatory T cells via both histone deacetylase inhibition and G protein-coupled receptor 109A signaling. Frontiers in Immunology 2017, 8, 1429. [Google Scholar] [CrossRef]
- Ransohoff, R.M. A polarizing question: Do M1 and M2 microglia exist? Nature Neuroscience 2016, 19(8), 987–991. [Google Scholar] [CrossRef]
- Wenzel, T.J.; et al. Short-chain fatty acids (SCFAs) alone or in combination regulate select immune functions of microglia-like cells. Molecular and Cellular Neuroscience 2020, 105, 103493. [Google Scholar] [CrossRef] [PubMed]
- Berer, K.; et al. Gut microbiota from multiple sclerosis patients enables spontaneous autoimmune encephalomyelitis in mice. Proceedings of the National Academy of Sciences 2017, 114(40), 10719–10724. [Google Scholar] [CrossRef]
- Cekanaviciute, E.; et al. Gut bacteria from multiple sclerosis patients modulate human T cells and exacerbate symptoms in mouse models. Proceedings of the National Academy of Sciences 2017, 114(40), 10713–10718. [Google Scholar] [CrossRef] [PubMed]
- Tremlett, H.; et al. Gut microbiota in early pediatric multiple sclerosis: A case-control study. European Journal of Neurology 2016, 23(8), 1308–1321. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; et al. Microbiota influence the development of the brain and behaviors in C57BL/6J mice. PLoS ONE 2018, 13(8), e0201829. [Google Scholar] [CrossRef]
- Thion, M.S.; et al. Microbiome influences prenatal and adult microglia in a sex-specific manner. Cell 2018, 172(3), 500–516. [Google Scholar] [CrossRef]
- Desbonnet, L.; et al. Gut microbiota depletion from early adolescence in mice: Implications for brain and behaviour. Brain, Behavior, and Immunity 2015, 48, 165–173. [Google Scholar] [CrossRef]
- Leclercq, S.; et al. Low-dose penicillin in early life induces long-term changes in murine gut microbiota, brain cytokines and behavior. Nature Communications 2017, 8(1), 15062. [Google Scholar] [CrossRef]
- McMurran, C.E.; et al. The microbiota regulates murine inflammatory responses to toxin-induced CNS demyelination but has minimal impact on remyelination. Proceedings of the National Academy of Sciences 2019, 116(50), 25311–25321. [Google Scholar] [CrossRef]
- Langley, M.R.; et al. High fat diet consumption results in mitochondrial dysfunction, oxidative stress, and oligodendrocyte loss in the central nervous system. Biochimica et Biophysica Acta-Molecular Basis of Disease 2020, 1866(3), 165630. [Google Scholar] [CrossRef]
- Matsushima, G.K.; Morell, P. The neurotoxicant, cuprizone, as a model to study demyelination and remyelination in the central nervous system. Brain Pathology 2001, 11(1), 107–116. [Google Scholar] [CrossRef]
- Constantinescu, C.S.; et al. Experimental autoimmune encephalomyelitis (EAE) as a model for multiple sclerosis (MS). British Journal of Pharmacology 2011, 164(4), 1079–1106. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.K.; et al. Proinflammatory T-cell responses to gut microbiota promote experimental autoimmune encephalomyelitis. Proceedings of the National Academy of Sciences 2011, 108 (Suppl 1), 4615–4622. [Google Scholar] [CrossRef] [PubMed]
- Berer, K.; et al. Commensal microbiota and myelin autoantigen cooperate to trigger autoimmune demyelination. Nature 2011, 479(7374), 538–541. [Google Scholar] [CrossRef] [PubMed]
- Ochoa-Repáraz, J.; et al. A polysaccharide from the human commensal Bacteroides fragilis protects against CNS demyelinating disease. Mucosal Immunology 2010, 3(5), 487–495. [Google Scholar] [CrossRef]
- Mangalam, A.; et al. Human gut-derived commensal bacteria suppress CNS inflammatory and demyelinating disease. Cell Reports 2017, 20(6), 1269–1277. [Google Scholar] [CrossRef]
- Mizuno, M.; et al. The dual role of short fatty acid chains in the pathogenesis of autoimmune disease models. PLoS ONE 2017, 12(2), e0173032. [Google Scholar] [CrossRef]
- Dewhirst, F.E.; et al. Phylogeny of the defined murine microbiota: Altered Schaedler flora. Applied and Environmental Microbiology 1999, 65(8), 3287–3292. [Google Scholar] [CrossRef]
- Faith, J.J.; et al. Identifying gut microbe-host phenotype relationships using combinatorial communities in gnotobiotic mice. Science Translational Medicine 2014, 6(220), 220ra11. [Google Scholar] [CrossRef]
- Tan, J.; et al. The role of short-chain fatty acids in health and disease. Advances in Immunology 2016, 121, 91–119. [Google Scholar] [CrossRef]
- Atarashi, K.; et al. Treg induction by a rationally selected mixture of Clostridia strains from the human microbiota. Nature 2013, 500(7461), 232–236. [Google Scholar] [CrossRef] [PubMed]
- So, D.; et al. Dietary fiber intervention on gut microbiota composition in healthy adults: A systematic review and meta-analysis. The American Journal of Clinical Nutrition 2018, 107(6), 965–983. [Google Scholar] [CrossRef] [PubMed]
- Baxter, N.T.; et al. Dynamics of human gut microbiota and short-chain fatty acids in response to dietary interventions with three fermentable fibers. mBio 2019, 10(1), e02566-18. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, K.C.; et al. Diet quality is associated with disability and symptom severity in multiple sclerosis. Neurology 2018, 90(1), e1–e11. [Google Scholar] [CrossRef]
- Kim, D.Y.; et al. Inflammation-mediated memory dysfunction and effects of a ketogenic diet in a murine model of multiple sclerosis. PLoS ONE 2012, 7(5), e35476. [Google Scholar] [CrossRef]
- Storoni, M.; Plant, G.T. The therapeutic potential of the ketogenic diet in treating progressive multiple sclerosis. Multiple Sclerosis International 2015, 2015, 681289. [Google Scholar] [CrossRef]
- Brenton, J.N.; et al. Phase II study of ketogenic diets in relapsing multiple sclerosis: Safety, tolerability and potential clinical benefits. Journal of Neurology, Neurosurgery & Psychiatry 2019, 93(6), 637–644. [Google Scholar] [CrossRef]
- Swidsinski, A.; et al. Reduced mass and diversity of the colonic microbiome in patients with multiple sclerosis and their improvement with ketogenic diet. Frontiers in Microbiology 2017, 8, 1141. [Google Scholar] [CrossRef]
- Katz Sand, I.; et al. The role of diet in multiple sclerosis: Mechanistic connections and current evidence. Current Nutrition Reports 2018, 7(3), 150–160. [Google Scholar] [CrossRef]
- De Filippis, F.; et al. High-level adherence to a Mediterranean diet beneficially impacts the gut microbiota and associated metabolome. Gut 2016, 65(11), 1812–1821. [Google Scholar] [CrossRef]
- Tankou, S.K.; et al. A probiotic modulates the microbiome and immunity in multiple sclerosis. Annals of Neurology 2018, 83(6), 1147–1161. [Google Scholar] [CrossRef] [PubMed]
- Kouchaki, E.; et al. Clinical and metabolic response to probiotic supplementation in patients with multiple sclerosis: A randomized, double-blind, placebo-controlled trial. Clinical Nutrition 2017, 36(5), 1245–1249. [Google Scholar] [CrossRef] [PubMed]
- Salami, M.; et al. How probiotic bacteria influence the motor and mental behaviors as well as immunological and oxidative biomarkers in multiple sclerosis? A double blind clinical trial. Journal of Functional Foods 2019, 52, 8–13. [Google Scholar] [CrossRef]
- Mimee, M.; et al. Programming a human commensal bacterium, Bacteroides thetaiotaomicron, to sense and respond to stimuli in the murine gut microbiota. Cell Systems 2015, 1(1), 62–71. [Google Scholar] [CrossRef]
- Charbonneau, M.R.; et al. Developing a new class of engineered live bacterial therapeutics to treat human diseases. Nature Communications 2020, 11(1), 1738. [Google Scholar] [CrossRef]
- Depommier, C.; et al. Supplementation with Akkermansia muciniphila in overweight and obese human volunteers: A proof-of-concept exploratory study. Nature Medicine 2019, 25(7), 1096–1103. [Google Scholar] [CrossRef]
- Zmora, N.; et al. Personalized gut mucosal colonization resistance to empiric probiotics is associated with unique host and microbiome features. Cell 2018, 174(6), 1388–1405. [Google Scholar] [CrossRef]
- Venkatesh, M.; et al. Symbiotic bacterial metabolites regulate gastrointestinal barrier function via the xenobiotic sensor PXR and Toll-like receptor 4. Immunity 2014, 41(2), 296–310. [Google Scholar] [CrossRef]
- Konopelski, P.; et al. Indole-3-propionic acid, a tryptophan-derived bacterial metabolite, reduces weight gain in rats. Nutrients 2022, 14(7), 1507. [Google Scholar] [CrossRef]
- Busbee, P.B.; et al. Use of natural AhR ligands as potential therapeutic modalities against inflammatory disorders. Nutrition Reviews 2013, 71(6), 353–369. [Google Scholar] [CrossRef]
- Elia, A.E.; et al. Tauroursodeoxycholic acid in the treatment of patients with amyotrophic lateral sclerosis. European Journal of Neurology 2016, 23(1), 45–52. [Google Scholar] [CrossRef] [PubMed]
- Makkawi, S.; et al. Fecal microbiota transplantation associated with 10 years of stability in a patient with SPMS. Neurology-Neuroimmunology Neuroinflammation 2018, 5(4), e459. [Google Scholar] [CrossRef] [PubMed]
- Allegretti, J.R.; et al. The evolution of the use of faecal microbiota transplantation and emerging therapeutic indications. The Lancet 2019, 394(10196), 420–431. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, M.; et al. Mapping human microbiome drug metabolism by gut bacteria and their genes. Nature 2019, 570(7762), 462–467. [Google Scholar] [CrossRef]
- Sajad, M.; et al. Pyruvate Dehydrogenase-Dependent Metabolic Programming Affects the Oligodendrocyte Maturation and Remyelination. Molecular Neurobiology 2024, 61, 397–410. [Google Scholar]
- Xiao, L.; et al. A catalog of the mouse gut metagenome. Nature Biotechnology 2015, 33(10), 1103–1108. [Google Scholar] [CrossRef]
- Ventura-Antunes, L.; et al. Different scaling of white matter volume, cortical connectivity, and gyrification across rodent and primate brains. Frontiers in Neuroanatomy 2013, 7, 3. [Google Scholar] [CrossRef]
- Miller, D.J.; et al. Prolonged myelination in human neocortical evolution. Proceedings of the National Academy of Sciences 2012, 109(41), 16480–16485. [Google Scholar] [CrossRef]
- Storm-Larsen, C.; et al. Gut microbiota composition during a 12-week intervention with delayed-release dimethyl fumarate in multiple sclerosis. Multiple Sclerosis and Related Disorders 2020, 39, 101898. [Google Scholar] [CrossRef]
- Cantarel, B.L.; et al. Gut microbiota in multiple sclerosis: Possible influence of immunomodulators. Journal of Investigative Medicine 2015, 63(5), 729–734. [Google Scholar] [CrossRef]
- Wunsch, M.; et al. The enteric nervous system is a potential autoimmune target in multiple sclerosis. Acta Neuropathologica 2022, 144(5), 871–888. [Google Scholar] [CrossRef]
- Shoaie, S.; et al. Quantifying diet-induced metabolic changes of the human gut microbiome. Cell Metabolism 2015, 22(2), 320–331. [Google Scholar] [CrossRef] [PubMed]
- MacKay, A.L.; Laule, C. Magnetic resonance of myelin water: An in vivo marker for myelin. Brain Plasticity 2016, 2(1), 71–91. [Google Scholar] [CrossRef] [PubMed]
- Alexander, A.L.; et al. Diffusion tensor imaging of the brain. Neurotherapeutics 2007, 4(3), 316–329. [Google Scholar] [CrossRef] [PubMed]
- Marques, S.; et al. Oligodendrocyte heterogeneity in the mouse juvenile and adult central nervous system. Science 2016, 352(6291), 1326–1329. [Google Scholar] [CrossRef]
- Jäkel, S.; et al. Altered human oligodendrocyte heterogeneity in multiple sclerosis. Nature 2019, 566(7745), 543–547. [Google Scholar] [CrossRef]
- Madhavan, M.; et al. Induction of myelinating oligodendrocytes in human cortical spheroids. Nature Methods 2018, 15(9), 700–706. [Google Scholar] [CrossRef]
- Marton, R.M.; et al. Differentiation and maturation of oligodendrocytes in human three-dimensional neural cultures. Nature Neuroscience 2019, 22(3), 484–491. [Google Scholar] [CrossRef]
- Lavasani, S.; et al. A novel probiotic mixture exerts a therapeutic effect on experimental autoimmune encephalomyelitis mediated by IL-10 producing regulatory T cells. PLoS ONE 2010, 5(2), e9009. [Google Scholar] [CrossRef]
- Stacho, R.; Zucha, D.; Kirdajova, D.; Valihrach, L. Applications of Spatial Transcriptomics in Ischemic Stroke Research. American Journal of Pathology 2026. [Google Scholar] [CrossRef]
- Abulaban, A.A.; Al-Kuraishy, H.M.; et al. The Janus Face of Astrocytes in Multiple Sclerosis: Balancing Protection and Pathology. Brain Research Bulletin 2025, 226, 111356. [Google Scholar] [CrossRef]
- Jubair, H. Glial Cells in Dementia: From Cellular Dysfunction to Therapeutic Frontiers. Cell Transplantation 2026, 35, 09636897251414216. [Google Scholar] [CrossRef]
- Deng, X.; Zhao, M.; et al. New insights into acute ischemic stroke from the perspective of spatial omics. Theranostics 2025, 15(15), 7902–7924. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




