Submitted:
26 February 2026
Posted:
27 February 2026
You are already at the latest version
Abstract
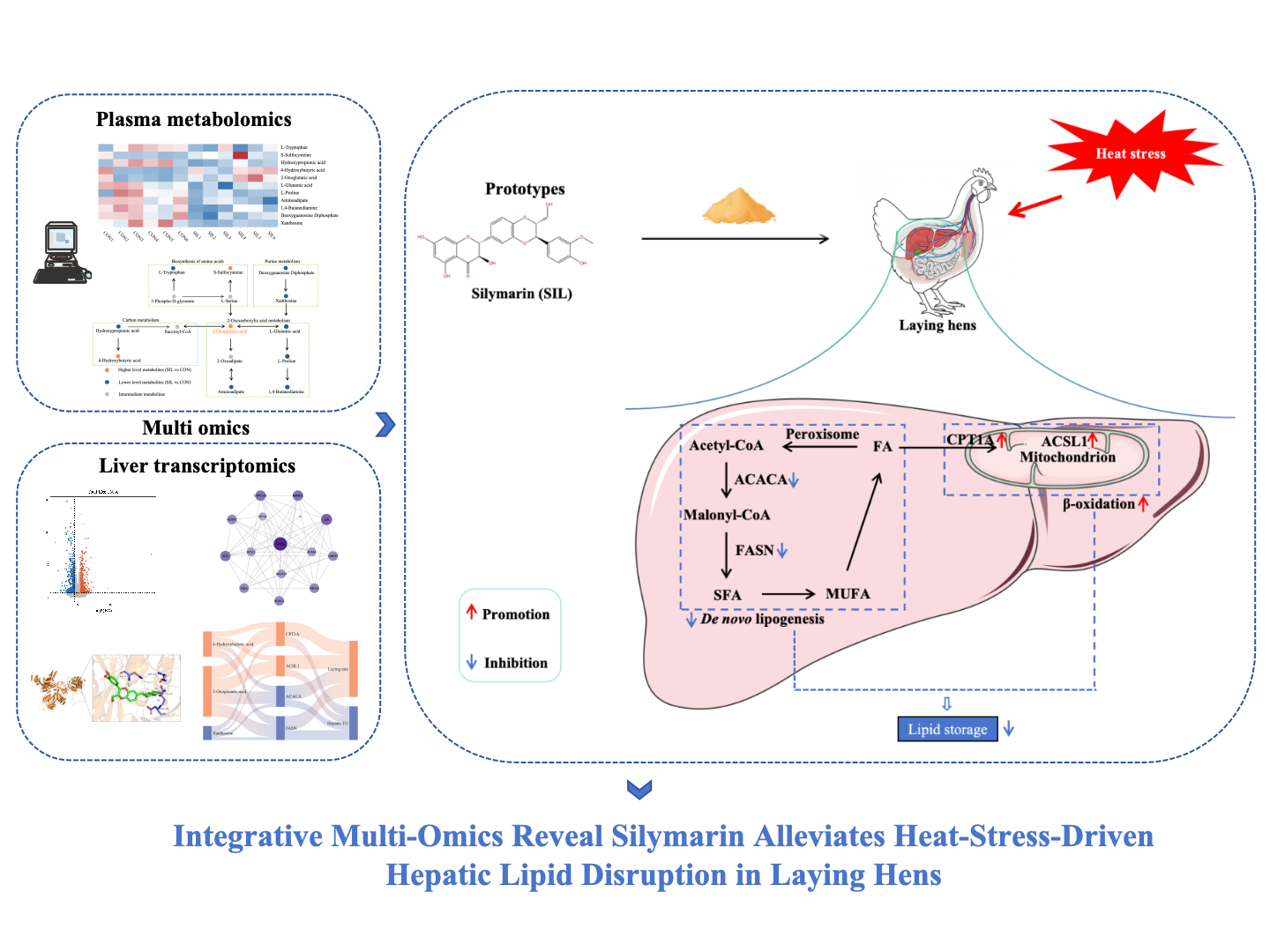
Keywords:
1. Introduction
2. Materials and Methods
2.1. Breeding and Management of Animals
2.2. Egg Quality
2.3. Sample Collection
2.4. Plasma Biochemistry and Metabolomics
Liver Lipid Content and Antioxidant Activity
2.5. Liver Transcriptomics
2.6. Quantitative Polymerase Chain Reaction
2.7. Molecular Docking
2.8. Western Blot
2.9. Statistical Analysis
3. Results
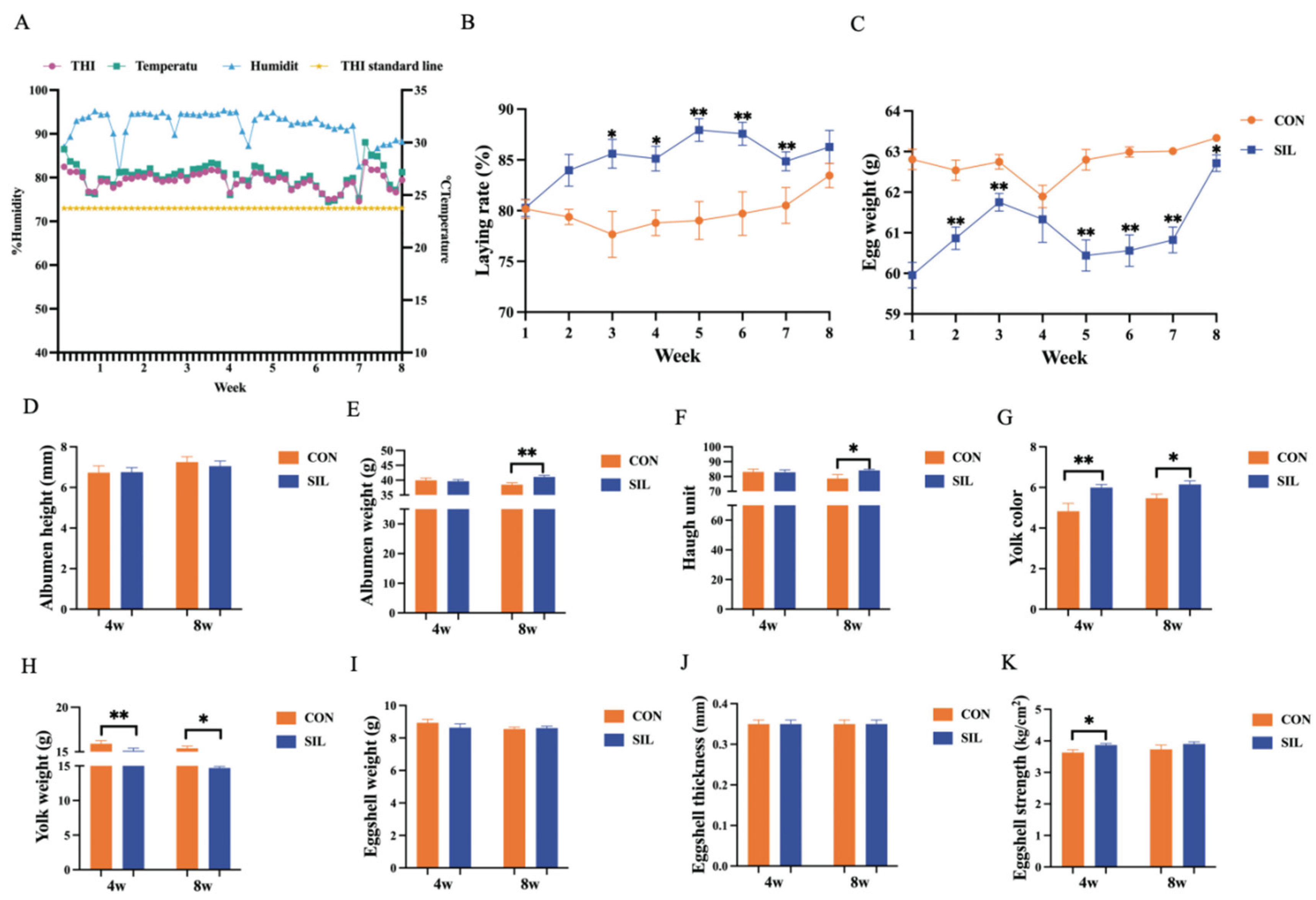
3.1. Silymarin Enhances Laying Rate and Egg Quality of Laying Hens Under Heat Stress
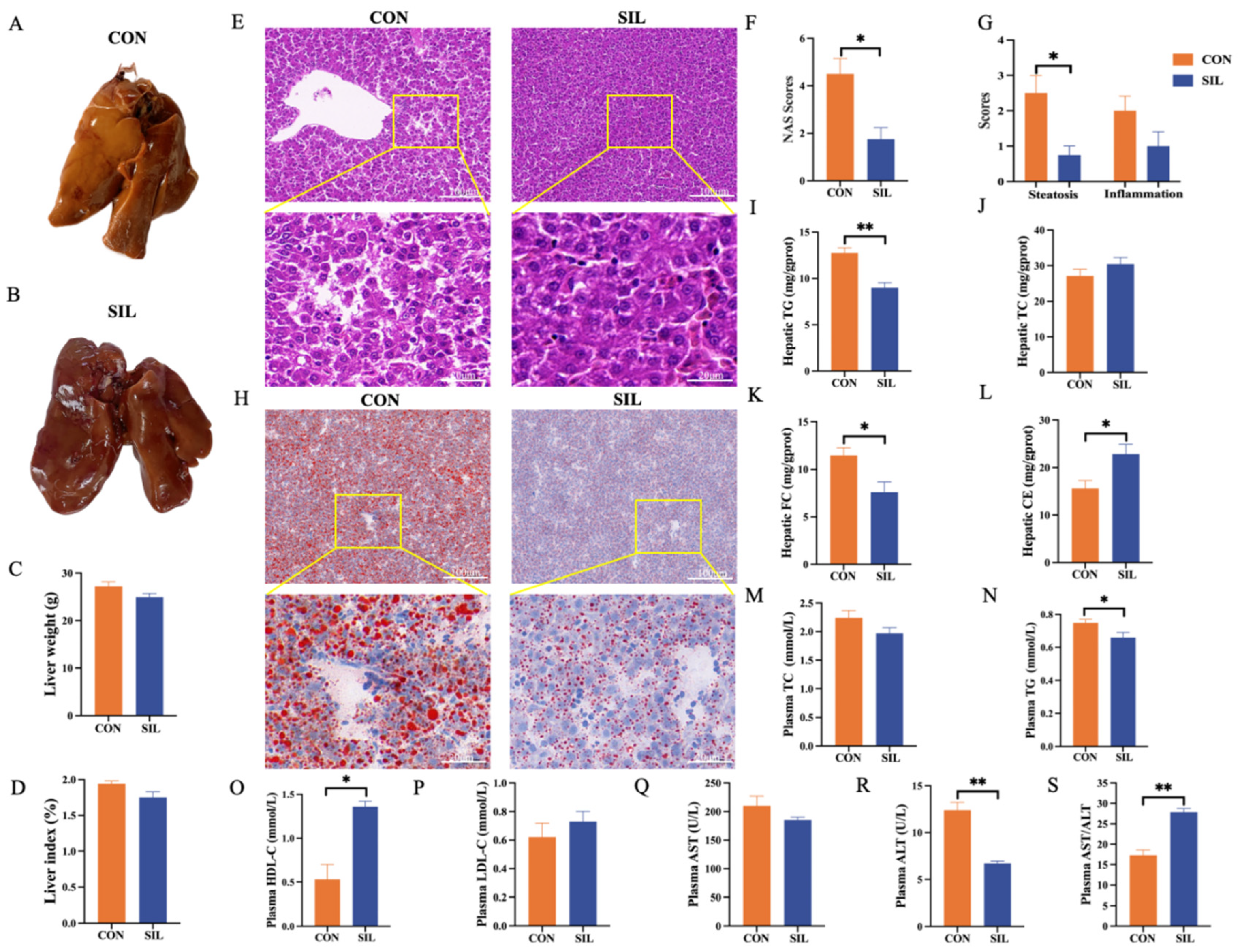
3.2. Silymarin Alleviates Hepatic Steatosis in Laying Hens Under Heat Stress
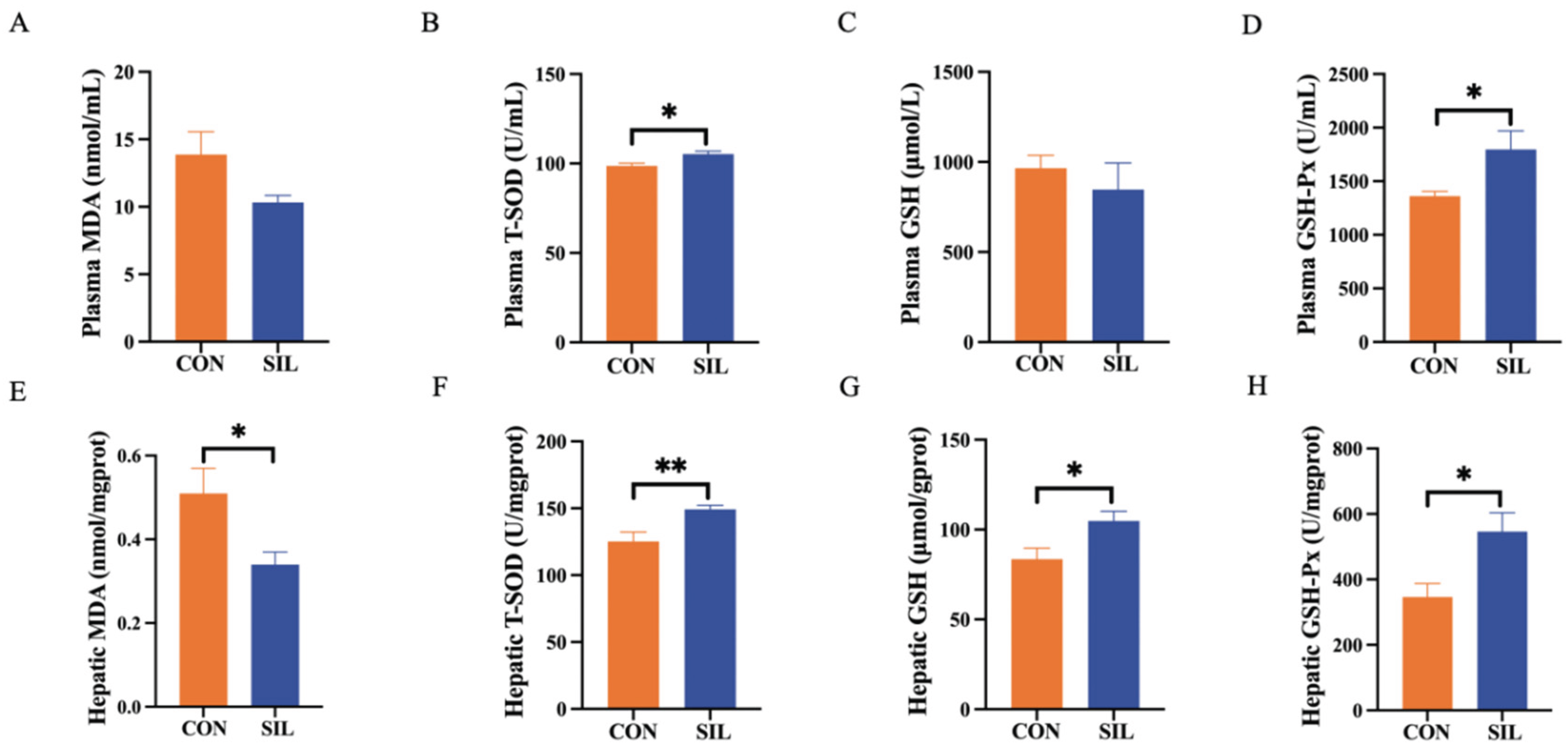
3.3. Silymarin Enhances the Antioxidant Capacity of Laying Hens
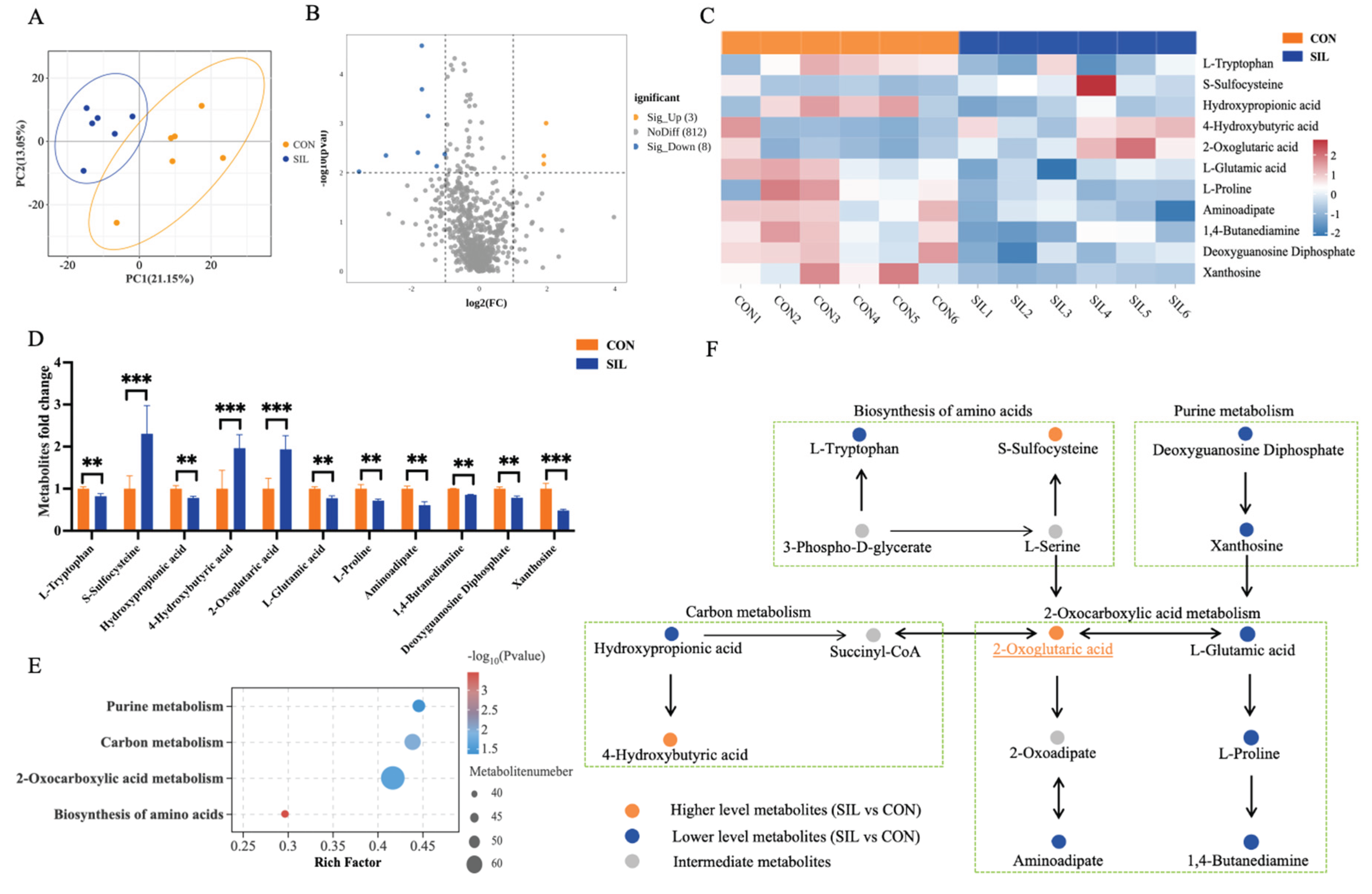
3.4. Silymarin Alters Plasma Metabolites
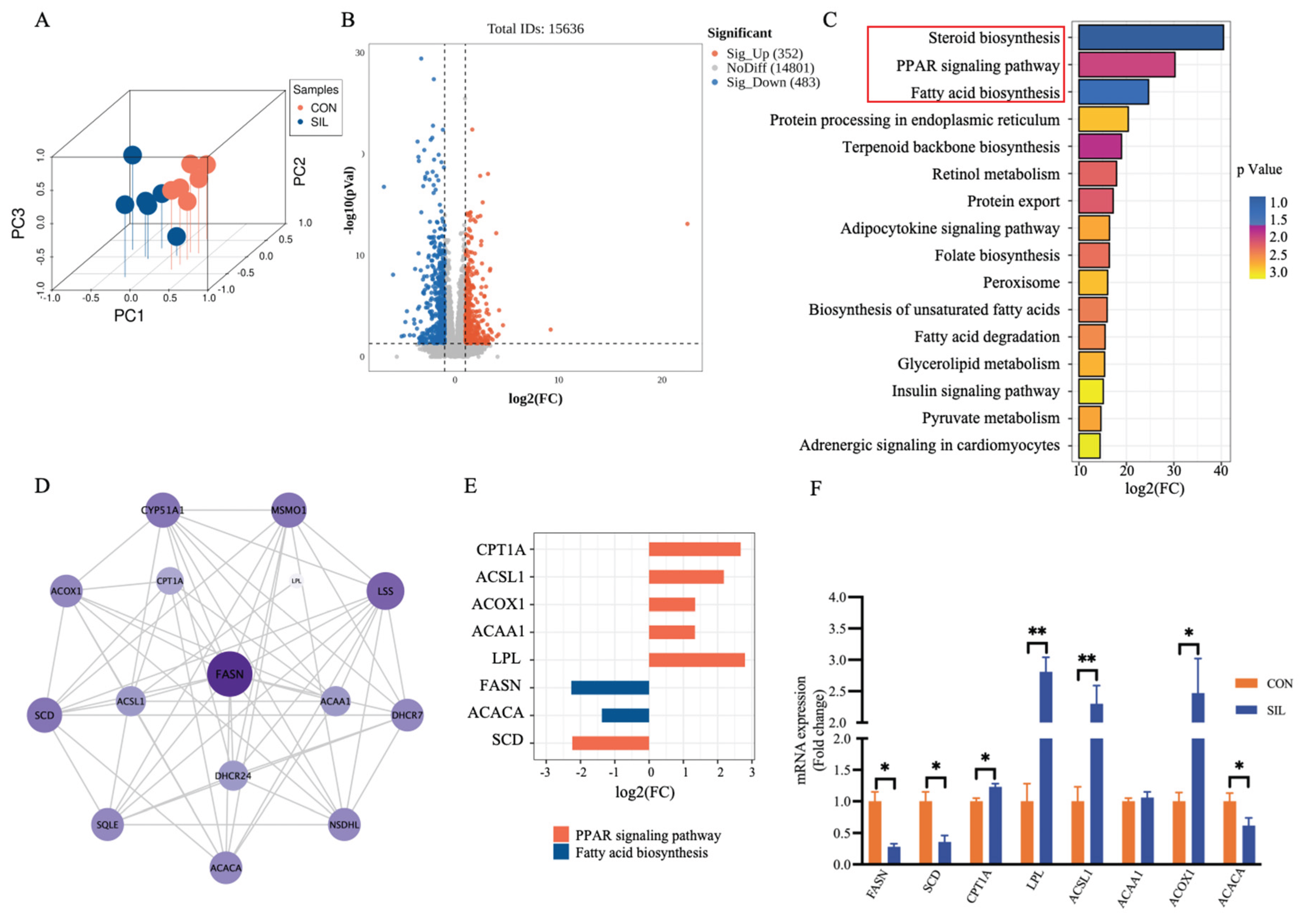
3.5. Silymarin Regulates Liver Lipid Metabolism
3.6. Silymarin Suppresses Hepatic De Novo Lipogenesis and Promotes Fatty Acid β-Oxidation
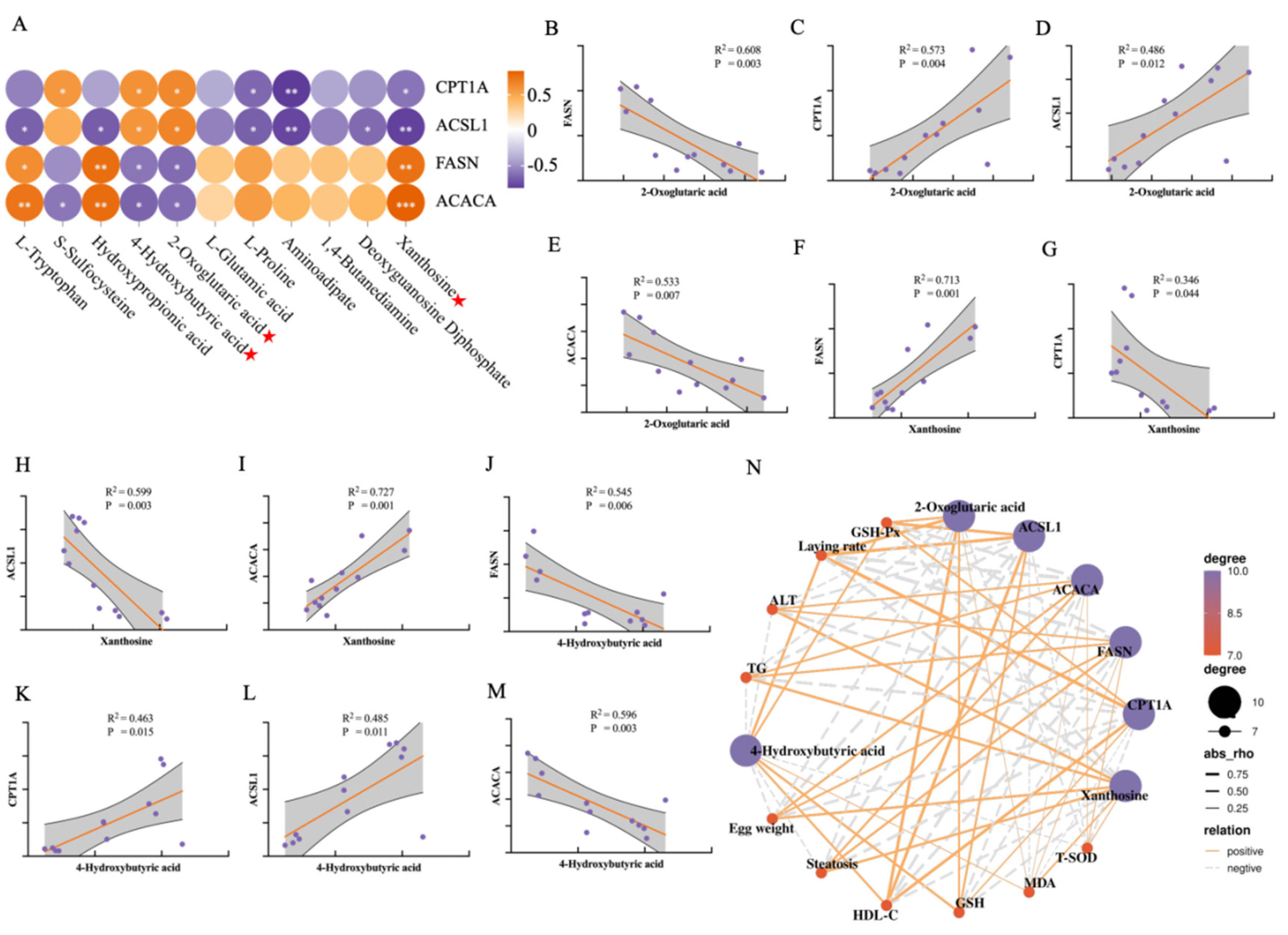
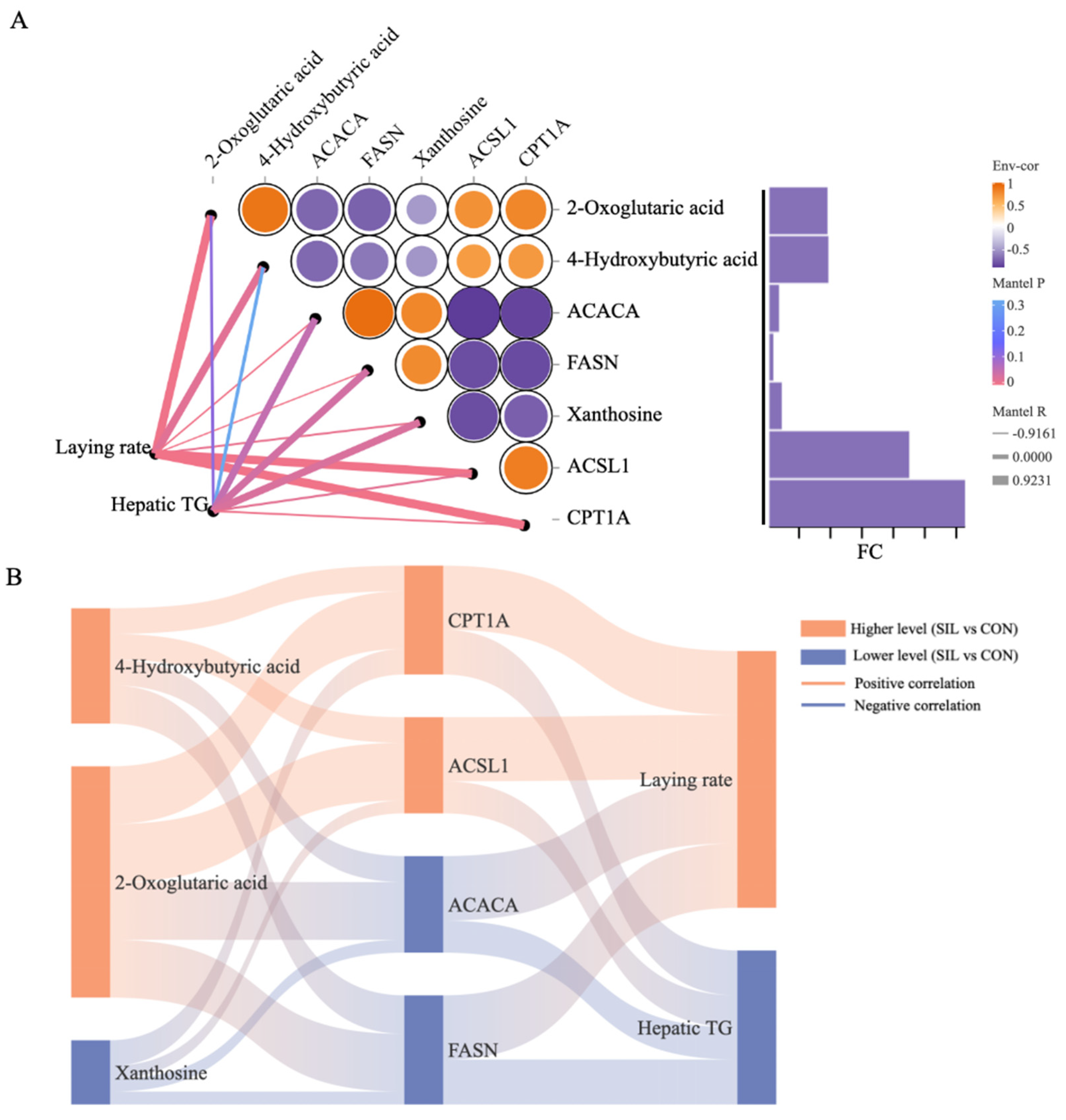
3.7. Correlation Analysis
4. Discussion
5. Conclusion
Supplementary Materials
Author Contributions
Funding
Ethics approval and consent to participate
Data Availability Statement
Conflicts of Interest
References
- He, X.; Lu, Z.; Ma, B.; Zhang, L.; Li, J.; Jiang, Y.; Zhou, G.; Gao, F. Chronic heat stress damages small intestinal epithelium cells associated with the adenosine 5'-monophosphate-activated protein kinase pathway in broilers. J. Agric. Food Chem. 2018, 66, 7301–7309. [Google Scholar] [CrossRef]
- Mangan, M.; Siwek, M. Strategies to combat heat stress in poultry production-A review. J. Anim. Physiol. Anim. Nutr. 2024, 108, 576–595. [Google Scholar] [CrossRef] [PubMed]
- Li, G.M.; Liu, L.P.; Yin, B.; Liu, Y.Y.; Dong, W.W.; Gong, S.; Zhang, J.; Tan, J.H. Heat stress decreases egg production of laying hens by inducing apoptosis of follicular cells via activating the FasL/Fas and TNF-α systems. Poult. Sci. 2020, 99, 6084–6093. [Google Scholar] [CrossRef] [PubMed]
- Ma, B.; Xing, T.; Li, J.; Zhang, L.; Jiang, Y.; Gao, F. Chronic heat stress causes liver damage via endoplasmic reticulum stress-induced apoptosis in broilers. Poult. Sci. 2022, 101, 102063. [Google Scholar] [CrossRef] [PubMed]
- Tesakul, S.; Mitsuwan, W.; Morita, Y.; Kitpipit, W. Effects of heat stress on egg performance in laying hens under hot and humid conditions. Vet. World 2025, 18, 851–858. [Google Scholar] [CrossRef]
- Gao, J.; Ren, H.; Wu, X.; Zou, C.; He, B.; Ma, W. Dietary glycerol monolaurate mitigates heat stress-induced disruption of intestinal homeostasis and hepatic lipid metabolism in laying hens. Stress Biol. 2025, 5, 49. [Google Scholar] [CrossRef]
- Abdel-Moneim, A.E.; Shehata, A.M.; Khidr, R.E.; Paswan, V.K.; Ibrahim, N.S.; El-Ghoul, A.A.; Aldhumri, S.A.; Gabr, S.A.; Mesalam, N.M.; Elbaz, A.M.; Elsayed, M.A.; Wakwak, M.M.; Ebeid, T.A. Nutritional manipulation to combat heat stress in poultry - A comprehensive review. J. Therm. Biol. 2021, 98, 102915. [Google Scholar] [CrossRef]
- Nanto-Hara, F.; Ohtsu, H. In laying hens, chronic heat stress-induced renal fibrosis is potentially promoted by indoxyl sulfate. Sci. Rep. 2024, 14, 23213. [Google Scholar] [CrossRef]
- Yin, C.; Zhou, C.; Shi, Y.; Ge, Y.; Gao, X.; Wu, C.; Xu, Z.; Huang, C.; Hu, G.; Liu, P.; et al. Effects and potential mechanism of dietary vitamin C supplementation on hepatic lipid metabolism in growing laying hens under chronic heat stress. J. Anim. Sci. 2023, 101, skad308. [Google Scholar] [CrossRef]
- Shakeri, M.; Oskoueian, E.; Le, H.H.; Shakeri, M. Strategies to combat heat stress in broiler chickens: Unveiling the roles of selenium, vitamin E and vitamin C. Vet. Sci. 2020, 7, 71. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhuang, Y.; Shi, Y.; Xu, Z.; Zhou, C.; Guo, L.; Liu, P.; Wu, C.; Hu, R.; Hu, G.; et al. Effects of N-acetyl-l-cysteine on heat stress-induced oxidative stress and inflammation in the hypothalamus of hens. J. Therm. Biol. 2021, 98, 102927. [Google Scholar] [CrossRef] [PubMed]
- Gillessen, A.; Schmidt, H.H. Silymarin as supportive treatment in liver diseases: A narrative review. Adv. Ther. 2020, 37, 1279–1301. [Google Scholar] [CrossRef] [PubMed]
- Lima, R.L.S.; Menegatto, M.B.D.S.; Almeida, L.T.; Magalhães, J.C.; Ferraz, A.C.; Magalhães, C.L.B. Silymarin exerts antioxidant and antiviral effects on Zika virus infection. J. Virol. Methods 2025, 335, 115133. [Google Scholar] [CrossRef] [PubMed]
- Stephen-Robert, J.M.; Peddha, M.S.; Srivastava, A.K. Effect of silymarin and quercetin in a miniaturized scaffold in Wistar rats against non-alcoholic fatty liver disease. ACS Omega 2021, 6, 20735–20745. [Google Scholar] [CrossRef]
- Guo, Y.; Xu, Y.; Wang, D.; Yang, S.; Song, Z.; Li, R.; He, X. Dietary silymarin improves performance by altering hepatic lipid metabolism and cecal microbiota function and its metabolites in late laying hens. J. Anim. Sci. Biotechnol. 2024, 15, 100. [Google Scholar] [CrossRef]
- Faryadi, S.; Sheikhahmadi, A.; Farhadi, A.; Nourbakhsh, H. Evaluating the therapeutic effect of different forms of silymarin on liver status and expression of some genes involved in fat metabolism, antioxidants and anti-inflammatory in older laying hens. Vet. Med. Sci. 2024, 10, e70025. [Google Scholar] [CrossRef]
- Gholizadeh, H.; Torki, M.; Mohammadi, H. Production performance, egg quality and some blood parameters of heat-stressed laying hens as affected by dietary supplemental Vit B6, Mg and Zn. Vet. Med. Sci. 2022, 8, 681–694. [Google Scholar] [CrossRef]
- Cornescu, G.M.; Panaite, T.D.; Untea, A.E.; Varzaru, I.; Saracila, M.; Dumitru, M.; Vlaicu, P.A.; Gavris, T. Mitigation of heat stress effects on laying hens' performances, egg quality, and some blood parameters by adding dietary zinc-enriched yeasts, parsley, and their combination. Front. Vet. Sci. 2023, 10, 1202058. [Google Scholar] [CrossRef]
- Khan, S.U.; Jeon, Y.H.; Kim, I.H. Dietary inclusion of micelle silymarin enhances egg production, quality, and lowers blood cholesterol in Hy-line brown laying hens. J. Anim. Physiol. Anim. Nutr. 2024, 108, 1038–1045. [Google Scholar] [CrossRef]
- Nanto-Hara, F.; Yamazaki, M.; Murakami, H.; Ohtsu, H. Chronic heat stress induces renal fibrosis and mitochondrial dysfunction in laying hens. J. Anim. Sci. Biotechnol. 2023, 14, 81. [Google Scholar] [CrossRef]
- Sahin, E.; Bagci, R.; Bektur Aykanat, N.E.; Kacar, S.; Sahinturk, V. Silymarin attenuated nonalcoholic fatty liver disease through the regulation of endoplasmic reticulum stress proteins GRP78 and XBP-1 in mice. J. Food Biochem. 2020, 44, e13194. [Google Scholar] [CrossRef] [PubMed]
- Gholamalian, R.; Mahdavi, A.H.; Riasi, A. Hepatic fatty acids profile, oxidative stability and egg quality traits ameliorated by supplementation of alternative lipid sources and milk thistle meal. J. Anim. Physiol. Anim. Nutr. 2022, 106, 860–871. [Google Scholar] [CrossRef] [PubMed]
- Abulikemu, A.; Zhao, X.; Xu, H.; Li, Y.; Ma, R.; Yao, Q.; Wang, J.; Sun, Z.; Li, Y.; Guo, C. Silica nanoparticles aggravated the metabolic associated fatty liver disease through disturbed amino acid and lipid metabolisms-mediated oxidative stress. Redox Biol. 2023, 59, 102569. [Google Scholar] [CrossRef] [PubMed]
- García-Muñoz, A.M.; Victoria-Montesinos, D.; Ballester, P.; Cerdá, B.; Zafrilla, P. A descriptive review of the antioxidant effects and mechanisms of action of berberine and silymarin. Molecules 2024, 29, 4576. [Google Scholar] [CrossRef]
- He, R.; Wei, Y.; Peng, Z.; Yang, J.; Zhou, Z.; Li, A.; Wu, Y.; Wang, M.; Li, X.; Zhao, D.; Liu, Z.; Dong, H.; Leng, X. α-Ketoglutarate alleviates osteoarthritis by inhibiting ferroptosis via the ETV4/SLC7A11/GPX4 signaling pathway. Cell Mol. Biol. Lett. 2024, 29, 88. [Google Scholar] [CrossRef]
- Liu, L.; Zhang, W.; Liu, T.; Tan, Y.; Chen, C.; Zhao, J.; Geng, H.; Ma, C. The physiological metabolite α-ketoglutarate ameliorates osteoarthritis by regulating mitophagy and oxidative stress. Redox Biol. 2023, 62, 102663. [Google Scholar] [CrossRef]
- Cheng, D.; Zhang, M.; Zheng, Y.; Wang, M.; Gao, Y.; Wang, X.; Liu, X.; Lv, W.; Zeng, X.; Belosludtsev, K.N.; Su, J.; Zhao, L.; Liu, J. α-Ketoglutarate prevents hyperlipidemia-induced fatty liver mitochondrial dysfunction and oxidative stress by activating the AMPK-pgc-1α/Nrf2 pathway. Redox Biol. 2024, 74, 103230. [Google Scholar] [CrossRef]
- Branco, R.C.S.; Camargo, R.L.; Batista, T.M.; Vettorazzi, J.F.; Lubaczeuski, C.; Bomfim, L.H.M.; Silveira, L.R.; Boschero, A.C.; Zoppi, C.C.; Carneiro, E.M. Protein malnutrition mitigates the effects of a high-fat diet on glucose homeostasis in mice. J. Cell. Physiol. 2019, 234, 6313–6323. [Google Scholar] [CrossRef]
- Legendre, F.; Tharmalingam, S.; Bley, A.M.; MacLean, A.; Appanna, V.D. Metabolic adaptation and NADPH homeostasis evoked by a sulfur-deficient environment in Pseudomonas fluorescens. Antonie Van Leeuwenhoek 2020, 113, 605–616. [Google Scholar] [CrossRef]
- Silva, A.R.; Ruschel, C.; Helegda, C.; Brusque, A.M.; Wannmacher, C.M.; Wajner, M.; Dustra-Filho, C.S. Inhibition of rat brain lipid synthesis in vitro by 4-hydroxybutyric acid. Metab. Brain Dis. 1999, 14, 157–164. [Google Scholar] [CrossRef]
- Sgaravatti, A.M.; Magnusson, A.S.; Oliveira, A.S.; Mescka, C.P.; Zanin, F.; Sgarbi, M.B.; Pederzolli, C.D.; Wyse, A.T.; Wannmacher, C.M.; Wajner, M.; Dutra-Filho, C.S. Effects of 1,4-butanediol administration on oxidative stress in rat brain: study of the neurotoxicity of gamma-hydroxybutyric acid in vivo. Metab. Brain Dis. 2009, 24, 271–282. [Google Scholar] [CrossRef] [PubMed]
- Sgaravatti, A.M.; Sgarbi, M.B.; Testa, C.G.; Durigon, K.; Pederzolli, C.D.; Prestes, C.C.; Wyse, A.T.; Wannmacher, C.M.; Wajner, M.; Dutra-Filho, C.S. Gamma-hydroxybutyric acid induces oxidative stress in cerebral cortex of young rats. Neurochem. Int. 2007, 50, 564–570. [Google Scholar] [CrossRef] [PubMed]
- Choudhary, R.K.; Choudhary, S.; Verma, R. In vivo response of xanthosine on mammary gene expression of lactating Beetal goat. Mol. Biol. Rep. 2018, 45, 581–590. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Zhou, W.; Fan, Z.; Cheng, Y.; Xiao, Y.; Liu, Y.; Li, X.; Ji, Z.; Fan, Y.; Ma, G. Xanthosine alleviates myocardial ischemia-reperfusion injury through attenuation of cardiomyocyte ferroptosis. Cell Mol. Biol. Lett. 2025, 30, 93. [Google Scholar] [CrossRef]
- Guo, Y.Y.; Liang, N.N.; Zhang, X.Y.; Ren, Y.H.; Wu, W.Z.; Liu, Z.B.; He, Y.Z.; Zhang, Y.H.; Huang, Y.C.; Zhang, T.; et al. Mitochondrial GPX4 acetylation is involved in cadmium-induced renal cell ferroptosis. Redox Biol. 2024, 73, 103179. [Google Scholar] [CrossRef]
- Ahmed, S.A.; Sarma, P.; Barge, S.R.; Swargiary, D.; Devi, G.S.; Borah, J.C. Xanthosine, a purine glycoside mediates hepatic glucose homeostasis through inhibition of gluconeogenesis and activation of glycogenesis via regulating the AMPK/ FoxO1/AKT/GSK3β signaling cascade. Chem. Biol. Interact. 2023, 371, 110347. [Google Scholar] [CrossRef]
- Xie, Z.; Ding, S.Q.; Shen, Y.F. Silibinin activates AMP-activated protein kinase to protect neuronal cells from oxygen and glucose deprivation-re-oxygenation. Biochem. Biophys. Res. Commun. 2014, 454, 313–319. [Google Scholar] [CrossRef]
- Softic, S.; Meyer, J.G.; Wang, G.X.; Gupta, M.K.; Batista, T.M.; Lauritzen, H.P.M.M.; Fujisaka, S.; Serra, D.; Herrero, L.; Willoughby, J.; et al. Dietary sugars alter hepatic fatty acid oxidation via transcriptional and post-translational modifications of mitochondrial proteins. Cell Metab. 2019, 30, 735–753.e4. [Google Scholar] [CrossRef]
- Maréchal, L.; Sicotte, B.; Caron, V.; Brochu, M.; Tremblay, A. Fetal cardiac lipid sensing triggers an early and sex-related metabolic energy switch in intrauterine growth restriction. J. Clin. Endocrinol. Metab. 2021, 106, 3295–3311. [Google Scholar] [CrossRef]
- Hüttl, M.; Markova, I.; Miklankova, D.; Zapletalova, I.; Poruba, M.; Racova, Z.; Vecera, R.; Malinska, H. The beneficial additive effect of silymarin in metformin therapy of liver steatosis in a pre-diabetic model. Pharmaceutics 2021, 14, 45. [Google Scholar] [CrossRef]
- Jiang, X.; Geng, H.; Zhang, C.; Zhu, Y.; Zhu, M.; Feng, D.; Wang, D.; Yao, J.; Deng, L. Circadian rhythm enhances mTORC1/AMPK pathway-mediated milk fat synthesis in dairy cows via the microbial metabolite acetic acid. J. Agric. Food Chem. 2024, 72, 28178–28193. [Google Scholar] [CrossRef]
- Zhou, Y.; Li, Y.; Chen, G.; Guo, X.; Gao, X.; Meng, J.; Xu, Y.; Zhou, N.; Zhang, B.; Zhou, X. ACSL1-mediated fatty acid β-oxidation enhances metastasis and proliferation in endometrial cancer. Front. Biosci. 2024, 29, 1247. [Google Scholar] [CrossRef]
- Jaffar, H.M.; Al-Asmari, F.; Khan, F.A.; Rahim, M.A.; Zongo, E. Silymarin: Unveiling its pharmacological spectrum and therapeutic potential in liver diseases-A comprehensive narrative review. Food Sci. Nutr. 2024, 12, 3097–3111. [Google Scholar] [CrossRef]
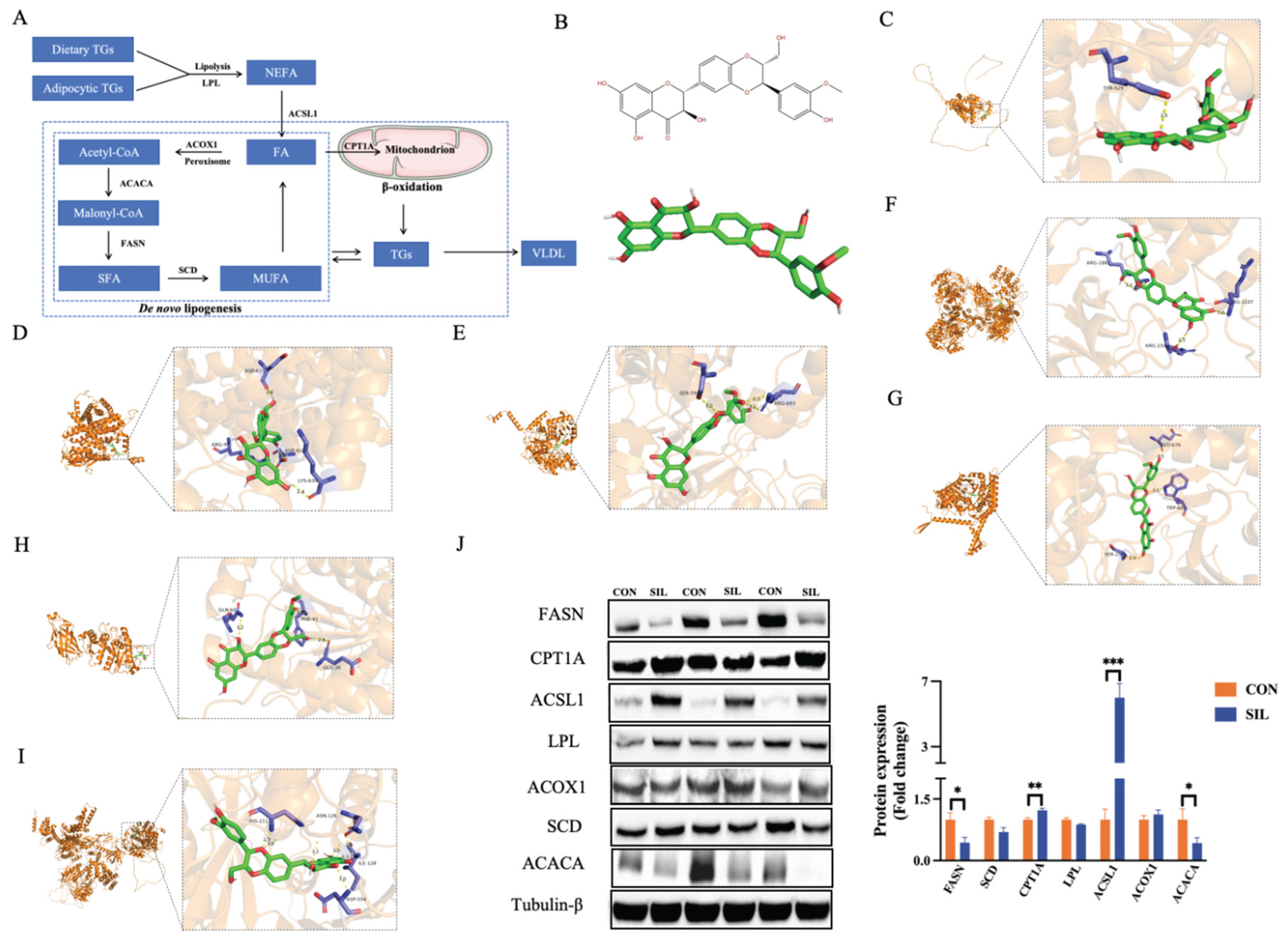
| Protein name |
Binding energy (kcal/mol) |
Residues | Distance(Å) |
| CPT1A | -9.7 | Glu676, Trp682, Ser259 | 2.57 |
| ACSL1 | -8.0 | Ser593, Arg683 | 3.23 |
| ACACA | -8.8 | His211, Asn126, Ile128, Asp554 | 2.96 |
| FASN | -8.7 | Arg1881, Arg1107, Arg1542 | 3.06 |
| SCD | -8.0 | Tyr529 | 3.00 |
| LPL | -8.5 | Gln60, Phe41, Glu38 | 2.90 |
| ACOX1 | -8.4 | Asp615, Arg97, Asp410, Lys639 | 2.72 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




