Background
Single-ventricle congenital heart disease encompasses a heterogeneous group of malformations in which biventricular repair is not feasible, necessitating staged palliation culminating in a cavo-pulmonary connection. The bidirectional Glenn procedure represents a critical intermediate step in this pathway, typically performed after completion of stage I palliation, such as the Norwood operation, pulmonary artery banding, or placement of a systemic-to-pulmonary artery shunt. Optimal timing and patient selection for the Glenn procedure are essential, as adverse hemodynamics at this stage may compromise pulmonary vascular development and negatively impact candidacy for subsequent Fontan completion [
1,
2].
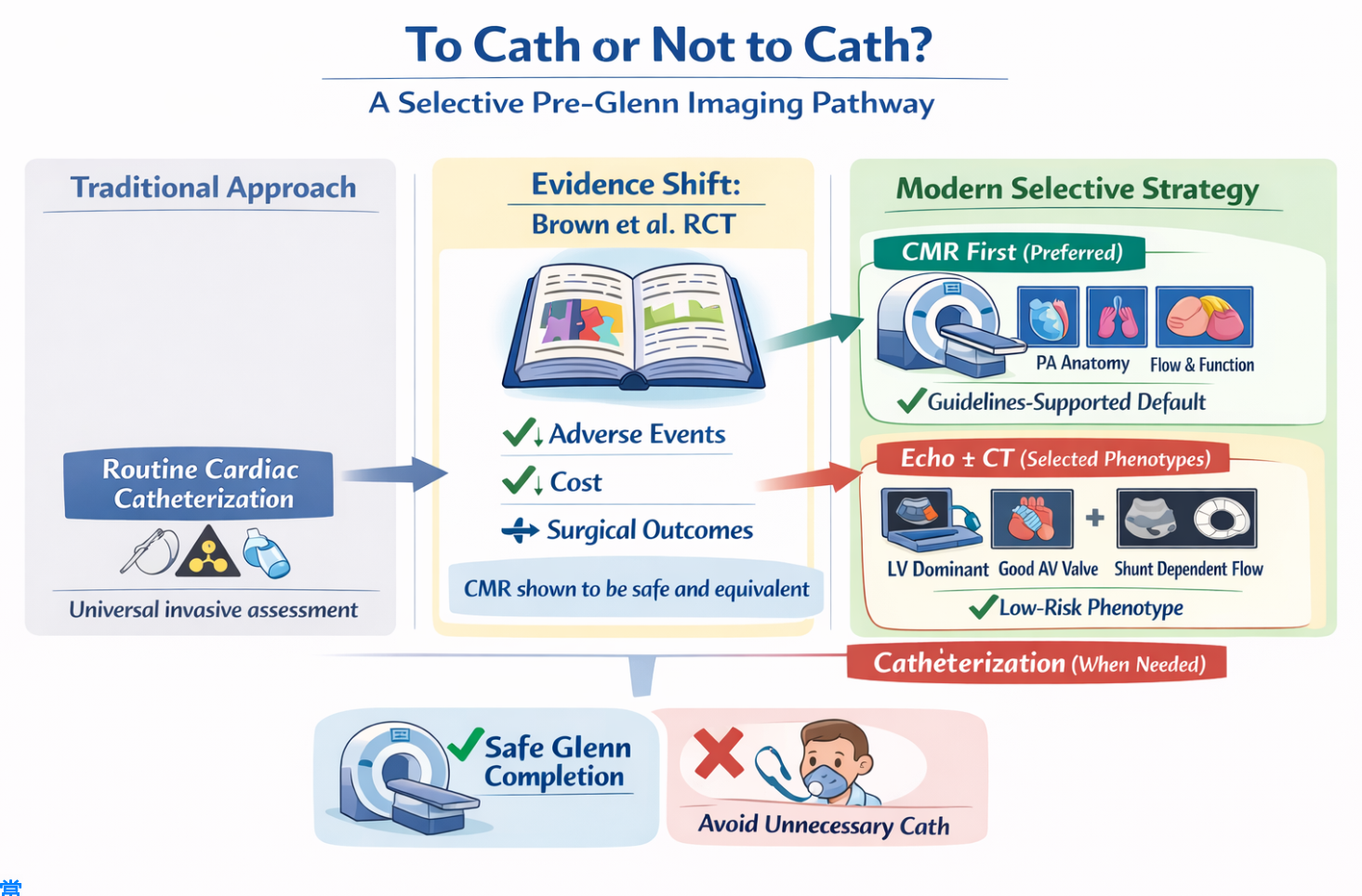
Traditionally, cardiac catheterization has been considered a cornerstone of the pre-Glenn evaluation. Following stage I palliation, patients are believed to require invasive hemodynamic assessment to ensure suitability for cavopulmonary circulation. This standard approach is grounded in the need to comprehensively evaluate pulmonary artery (PA) anatomy and growth, measure pulmonary artery pressures, and assess single-ventricle end-diastolic pressure (EDP). Adequate pulmonary artery size and low pulmonary vascular resistance are prerequisites for successful Glenn physiology, as the pulmonary circulation becomes passively driven by venous pressure rather than ventricular output. Elevated PA (Pulmonary Artery) pressures or distorted pulmonary artery anatomy may lead to suboptimal Glenn performance, superior vena cava syndrome, or early failure of the cavopulmonary circuit [
3]
In addition, assessment of ventricular filling pressures has traditionally been emphasized. Elevated single-ventricle EDP (end-diastolic pressure) reflects impaired diastolic compliance and has been associated with worse outcomes following cavopulmonary connections. Cardiac catheterization allows direct measurement of ventricular pressures and calculation of transpulmonary gradients, providing data that are not always reliably inferred from non-invasive imaging alone. Furthermore, catheterization enables identification of residual lesions such as aortic arch obstruction, systemic-to-pulmonary shunt stenosis, pulmonary artery branch distortion, or venovenous and aortopulmonary collateral vessels, which may require transcatheter or surgical intervention prior to Glenn completion [
4].
Historically, therefore, pre-Glenn catheterization has been viewed not only as a diagnostic tool but also as a therapeutic opportunity, allowing hemodynamic optimization before progression to the next surgical stage. This paradigm has been reinforced by earlier eras of limited echocardiographic resolution and reduced availability of advanced cross-sectional imaging. As a result, routine catheterization prior to Glenn has been widely adopted in many centers as a default strategy, regardless of clinical stability or non-invasive imaging findings [
5].
However, cardiac catheterization is not without risk, particularly in infants with fragile physiology. Complications such as vascular injury, arrhythmias, contrast-induced nephropathy, radiation exposure, and hemodynamic instability remain important considerations. With advancements in echocardiography, including improved assessment of pulmonary artery anatomy and ventricular diastolic function, as well as increasing use of cardiac magnetic resonance imaging and computed tomography, the necessity of routine invasive assessment in all patients prior to Glenn has been increasingly questioned [
2].
Against this backdrop, re-examining the role of routine pre-Glenn catheterization in the contemporary era is timely and clinically relevant, particularly in carefully selected patients with favorable non-invasive findings.
Main Body
Transition Toward a Non-Invasive Pre-Glenn Evaluation Strategy
Over the past two decades, significant advances in non-invasive cardiovascular imaging have challenged the paradigm of routine invasive assessment prior to the bidirectional Glenn procedure. Improvements in cardiac magnetic resonance (CMR), in particular, have enabled comprehensive evaluation of pulmonary artery anatomy, ventricular volumes and function, flow distribution, and extracardiac vasculature without the risks inherent to cardiac catheterization. These technological developments have prompted critical reappraisal of whether routine pre-Glenn catheterization is necessary in all patients, particularly those with favorable clinical status and reassuring echocardiographic findings [
6].
A pivotal milestone in this transition was the prospective randomized trial by
Brown et al., which directly compared routine cardiac catheterization with cardiac magnetic resonance in infants with functional single-ventricle physiology undergoing evaluation for bidirectional Glenn anastomosis. In this landmark study, cardiac magnetic resonance was shown to provide all clinically relevant anatomical and functional information required for surgical decision-making, with
significantly fewer adverse events, shorter hospital stays, and substantially lower costs compared with catheterization. Importantly, early postoperative outcomes, surgical success rates, and short-term clinical status were comparable between the two groups, despite a higher rate of catheter-based interventions in the invasive arm [
7]. These findings demonstrated that, in carefully selected patients, routine catheterization rarely altered surgical candidacy or outcomes and that cardiac magnetic resonance could safely serve as a primary diagnostic modality before Glenn. Subsequent observational studies and institutional experiences further reinforced this shift. Large single-center series documented successful transitions to predominantly non-invasive imaging pathways, with catheterization reserved for patients with suspected elevated pulmonary vascular resistance, significant ventricular dysfunction, atrioventricular valve regurgitation, pulmonary venous obstruction, or when a transcatheter intervention was anticipated. These real-world data highlighted not only the diagnostic reliability of cardiac magnetic resonance but also its favorable safety profile and cumulative reduction in radiation exposure and procedural burden, particularly relevant in infants with lifelong surveillance needs [
8].
This evolving evidence base was ultimately reflected in international guidelines and consensus documents. The
AEPC (Guidelines for the Management of Hypoplastic Left Heart Syndrome from the Association for European Pediatric Cardiology) acknowledged the growing role of cross-sectional imaging and emphasized that pre-Glenn catheterization should not be considered mandatory in all patients. Instead, invasive assessment was recommended selectively, particularly when non-invasive imaging suggested unfavorable hemodynamics or when interventional procedures were anticipated [
9].
More recently, the
2022–2023 multi-society cardiac magnetic resonance guidelines, endorsed by major cardiovascular and imaging societies, explicitly positioned cardiac magnetic resonance as a central modality in the evaluation of patients with single-ventricle physiology. These documents describe cardiac magnetic resonance as a “one-stop” diagnostic tool capable of delivering high-fidelity anatomical, functional, and flow-related data, supporting its preferential use in stable patients prior to staged cavopulmonary connections [
10]
Collectively, these trials, institutional data, and consensus statements mark a clear transition from routine invasive assessment toward a selective, imaging-driven approach, with cardiac magnetic resonance favored as the primary modality before Glenn in appropriately selected patients. This shift underscores a broader movement in congenital heart disease care toward precision-based, patient-tailored evaluation strategies that minimize risk while preserving diagnostic accuracy.
Is Echocardiography +/- CT Angiography Sufficient in Some Selected Phenotypes?
As mentioned earlier in the article, the pre-Glenn evaluation of patients with single-ventricle physiology has traditionally relied on routine cardiac catheterization to assess pulmonary artery anatomy and hemodynamics, particularly pulmonary artery pressure and pulmonary vascular resistance. While this approach has been considered the gold standard, it is inherently invasive and carries non-negligible risks, including vascular complications, radiation exposure, and the need for general anesthesia in a fragile population. More recently, cardiovascular magnetic resonance (CMR) has emerged as a less invasive alternative capable of providing detailed anatomic assessment and surrogate hemodynamic data through flow quantification. However, cardiac magnetic resonance is resource-intensive, time-consuming, frequently requires anesthesia in infants, and may not be universally available. Importantly, both catheterization and cardiac magnetic resonance are often applied uniformly across heterogeneous single-ventricle phenotypes, despite substantial physiological differences that influence the likelihood of abnormal pre-Glenn hemodynamics [
7].
We propose a third, phenotype-guided approach for a selected subgroup of single-ventricle patients, specifically those with pulmonary atresia and a favorable ventricular–valvular profile. This subgroup is typically characterized by a dominant left ventricular morphology, a dominant mitral atrioventricular valve with a low risk of significant regurgitation, preserved ventricular systolic function, and a ductal- or Blalock–Taussig shunt-dependent pulmonary circulation. From a physiological standpoint, these features are associated with a low probability of pulmonary venous hypertension, ventricular dysfunction, or elevated pulmonary vascular resistance prior to the Glenn procedure. In this context, comprehensive targeted echocardiography can reliably assess the key determinants of Glenn suitability, including ventricular performance, atrioventricular valve competence, atrial pressures through Doppler surrogates, and proximal pulmonary artery anatomy. In patients where echocardiographic visualization of branch pulmonary arteries or distal pulmonary arborization is suboptimal, a focused, non-lengthy computed tomography angiography can be added to provide high-resolution anatomic detail without the prolonged acquisition time or physiologic perturbations associated with cardiac magnetic resonance, and with lower procedural risk compared to diagnostic catheterization [
3,
11,
12,
13]
This proposed strategy emphasizes proportionality and risk stratification rather than universal testing. In the described phenotype, the pre-test probability of discovering prohibitive pulmonary hemodynamics at catheterization is low, while the cumulative procedural burden of invasive or lengthy non-invasive studies remains substantial. By reserving cardiac catheterization or cardiac magnetic resonance for patients with echocardiographic red flags, discordant clinical findings, or borderline physiology, this approach maintains patient safety while reducing unnecessary invasiveness, anesthesia exposure, and healthcare resource utilization. Importantly, this is not an argument for abandoning catheterization or cardiac magnetic resonance, but rather for integrating them more selectively within a phenotype-driven algorithm. Such a strategy aligns with contemporary principles of precision medicine and may represent a pragmatic evolution in pre-Glenn assessment for carefully selected single-ventricle patients. Prospective validation of this approach could further clarify its role in optimizing outcomes while minimizing procedural risk [
6,
8,
14,
15,
16,
17,
18]
Conclusion
Routine cardiac catheterization before the bidirectional Glenn procedure has historically been considered essential for assessing pulmonary artery anatomy and hemodynamic suitability. However, contemporary evidence demonstrates that this universal invasive approach is no longer necessary for all patients. Advances in non-invasive imaging—particularly cardiac magnetic resonance—now allow comprehensive anatomical and functional assessment with lower procedural risk, reduced cost, and elimination of ionizing radiation. Randomized and observational data consistently show that selective non-invasive strategies do not compromise early surgical outcomes when applied to appropriately selected patients.
Recent international guidelines have therefore endorsed a shift toward individualized pre-Glenn evaluation, reserving catheterization for patients with suspected unfavorable physiology or when transcatheter intervention is anticipated. Furthermore, a phenotype-guided strategy may safely identify a subset of low-risk patients in whom targeted echocardiography, with adjunctive computed tomography when needed, is sufficient. This evolving paradigm reflects a precision-based approach that balances diagnostic accuracy with proportionality of risk, optimizing patient safety and resource utilization in contemporary single-ventricle care.
Author Contributions
Conceptualization, AFA; Methodology, AFA, JED, HAT, AAA, NA, MA, SEB, JS, FEG; software, AFA, JED, HAT, AAA, NA, MA, SEB, JS, FEG; investigation AFA, JED, HAT, AAA, NA, MA, SEB, JS, FEG; resources, AFA, JED, HAT, AAA, NA, MA, SEB, JS, FEG, data curation, AFA, JED, HAT, AAA, NA, MA, SEB, JS, FEG; writing—original draft preparation, AFA, JED, HAT, AAA, NA, MA, SEB, JS, FEG; writing—review and editing, AFA, JED, HAT, AAA, NA, MA, SEB, JS, FEG; supervision, AFA; project administration, AFA; funding acquisition, (non-applicable). All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
not applicable as this study is a hypothesis/Review article.
Informed Consent Statement
not applicable as this study is a viewpoint/editorial.
Data Availability Statement
All data is made available within the manuscript and are owned by the authors.
Acknowledgments
To our families, who see beyond our limits—and by believing in us, give us the courage to reach the skies above.
Conflicts of Interest
The authors declare no conflict of interest. The manuscript is submitted under Creative Commons Licensing CC-BY-NC-ND. A large language model has been used to proofread the article.
References
- Javadi, E.; et al. Predicting Hemodynamic Performance of Fontan Operation for Glenn Physiology using Computational Fluid Dynamics: Ten Patient-specific Cases. J. Clin. images Med. case reports 2022, 3. [Google Scholar] [CrossRef] [PubMed]
- Schranz, D. Hemodynamics under General Anesthesia in Glenn/Fontan Circulation? Pediatr. Cardiol. 2021, 42, 465–466. [Google Scholar] [CrossRef] [PubMed]
- AbdelMassih, A.; et al. Predictors of protein losing enteropathy after Fontan completion: An 8-year retrospective study at Sheikh Khalifa Medical City. Glob. Cardiol. Sci. Pract. 2023. [Google Scholar] [CrossRef] [PubMed]
- Nichay, N. R.; et al. Risk Factors For Unfavorable Outcomes After Bidirectional Cavopulmonary Anastomosis. World J. Pediatr. Congenit. Hear. Surg. 2017, 8, 575–583. [Google Scholar] [CrossRef] [PubMed]
- Nakanishi, T. Cardiac Catheterization Is Necessary Before Bidirectional Glenn and Fontan Procedures in Single Ventricle Physiology. Pediatr. Cardiol. 2005, 26, 159–161. [Google Scholar] [CrossRef] [PubMed]
- Abdula, G.; et al. Non-invasive estimation of mean pulmonary artery pressure by cardiovascular magnetic resonance in under 2 min scan time. Eur. Hear. J. - Imaging Methods Pract. 2023, 1. [Google Scholar] [CrossRef] [PubMed]
- Brown, D. W.; et al. Cardiac magnetic resonance versus routine cardiac catheterization before bidirectional Glenn anastomosis in infants with functional single ventricle: A prospective randomized trial. Circulation 2007, 116, 2718–2725. [Google Scholar] [CrossRef] [PubMed]
- Yanovskiy, A.; et al. Institutional transition from invasive to non-invasive imaging in children with univentricular heart defects: safety and cost savings. Cardiol. Young 2023, 33, 970–976. [Google Scholar] [CrossRef] [PubMed]
- Alphonso, N.; et al. Guidelines for the management of neonates and infants with hypoplastic left heart syndrome: The European Association for Cardio-Thoracic Surgery (EACTS) and the Association for European Paediatric and Congenital Cardiology (AEPC) Hypoplastic Left Heart Syndrome Guidelines Task Force. Eur. J. Cardio-thoracic Surg. 2020, 58, 416–499. [Google Scholar]
- Fogel, M. A.; et al. Society for Cardiovascular Magnetic Resonance/European Society of Cardiovascular Imaging/American Society of Echocardiography/Society for Pediatric Radiology/North American Society for Cardiovascular Imaging Guidelines for the use of cardiovascular magnet. J. Cardiovasc. Magn. Reson. 2022, 24, 1–78. [Google Scholar] [CrossRef] [PubMed]
- Alsoufi, B.; et al. The Impact of Dominant Ventricle Morphology on Palliation Outcomes of Single Ventricle Anomalies. Ann. Thorac. Surg. 2016, 102, 593–601. [Google Scholar] [CrossRef] [PubMed]
- Kido, T.; et al. Impacts of stage 1 palliation and pre-Glenn pulmonary artery pressure on long-term outcomes after Fontan operation. Eur. J. Cardio-Thoracic Surg. 2021, 60, 369–376. [Google Scholar] [CrossRef] [PubMed]
- Krupickova, S.; et al. Echocardiographic arterial measurements in complex congenital diseases before bidirectional Glenn: comparison with cardiovascular magnetic resonance imaging. Eur. Hear. J. – Cardiovasc. Imaging 2016, jew069. [Google Scholar] [CrossRef] [PubMed]
- Stern, K. W. D.; McElhinney, D. B.; Gauvreau, K.; Geva, T.; Brown, D. W. Echocardiographic evaluation before bidirectional glenn operation in functional single-ventricle heart disease comparison to catheter angiography. Circ. Cardiovasc. Imaging 2011, 4, 498–505. [Google Scholar] [CrossRef] [PubMed]
- Barfuss, S. B.; et al. Pre-Glenn CT: how I do it. Pediatr. Radiol. 2025, 55, 2314–2324. [Google Scholar] [CrossRef] [PubMed]
- Alsaied, T.; et al. Reaching Consensus for Unified Medical Language in Fontan Care. ESC Hear. Fail. 2021, 8, 3894–3905. [Google Scholar] [CrossRef]
- Stern, K. W. D.; McElhinney, D. B.; Gauvreau, K.; Geva, T.; Brown, D. W. Echocardiographic Evaluation Before Bidirectional Glenn Operation in Functional Single-Ventricle Heart Disease. Circ. Cardiovasc. Imaging 2011, 4, 498–505. [Google Scholar] [CrossRef] [PubMed]
- Azhar, A.; et al. Outcomes of bidirectional Glenn surgery done without prior cardiac catheterization. Egypt. Hear. J. 2022, 74. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).