Submitted:
26 February 2026
Posted:
28 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results
3.1. Wetting and Drying in the Environmental SEM
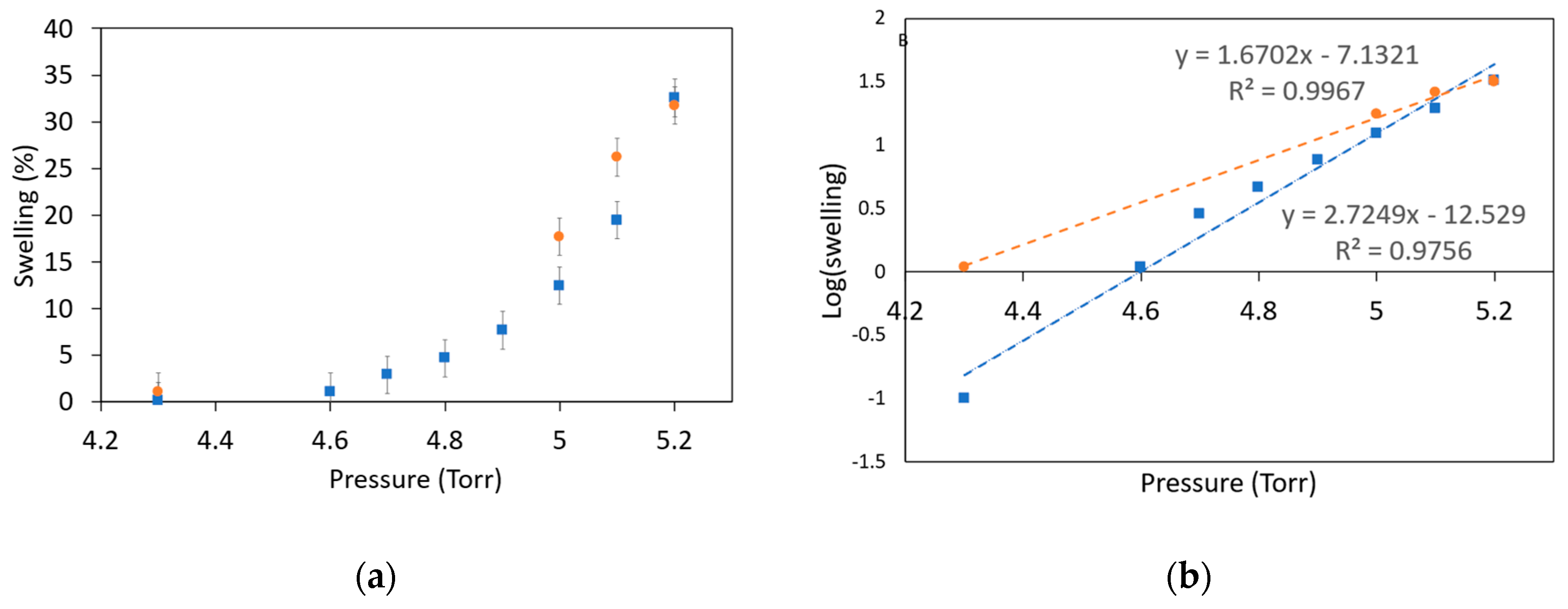
3.2. Swelling and Drying as Function of Relative Humidity Inside XRD
3.3. Water Adsorption Isotherms Based on ESEM Thickness Measurements
4. Conclusions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| SWy-1 | Montmorillonite |
| ESEMJ | Environmental Scanning Electron Microscopy |
| XRD | X-ray Diffraction |
| AOI | Area of interest |
| RH | Relative Humidity |
| Ah | Area of hydrated sample |
| A0 | Initial area of dry sample |
References
- Aylmore, L.A.G.; Quirk, J.P. Swelling of Clay–Water Systems. Nature 1959, 183, 1752-1753. [CrossRef]
- Blackmore, A.V.; Miller, R.D. Tactoid Size and Osmotic Swelling in Calcium Montmorillonite. Soil Science Society of America Journal 1961, 25, 169-173. [CrossRef]
- Diop, S.; Ogawa, Y.; Zhang, M. Effects of Cyclic Wetting and Drying on Physical and Mechanical Properties of Neogene Sandstones and Siltstones from Boso Peninsula, Japan. Jour. Japan Soc. Eng. Geol. 2008, 49, 150-163.
- Gillott, J.E. Chapter 6 - Moisture Interaction with Clays and Clay Minerals. In Developments in Geotechnical Engineering, Gillott, J.E., Ed.; Elsevier: 1987; Volume 41, pp. 167-217.
- Güven, N.; Pollastro, R.M. Clay-water interface and its rheological implications; The Clay Minerals Society: Boulder, 1992; Volume 4, p. 244.
- Newman, A.C.D. The significance of clays in agriculture and soils. Philosophical Transactions Royal Society, London 1984, A 311, 375-389.
- Newman, A.C.D. The interaction of water with clay mineral surfaces. In Chemistry of clays and clay minerals, Newman, A.C.D., Ed.; Longman Scientific & Technical: Harlow, 1987; pp. 237-274.
- Pusch, R.; Nakano, M.; Yong, R.N.; Yang, T. Long-term function of on-ground repositories for hazardous waste - Mechanisms in cyclic drying/wetting of top clay liners. Journal of Earth Sciences and Geotechnical Engineering 2017, 7, 45-62.
- Shahsavani, S.; Vakili, A.H.; Mokhberi, M. The effect of wetting and drying cycles on the swelling-shrinkage behavior of the expansive soils improved by nanosilica and industrial waste. Bulletin of Engineering Geology and the Environment 2020. [CrossRef]
- Taylor, R.K.; Smith, T.J. The engineering geology of clay minerals: swelling, shrinking and mudrock breakdown. Clay Minerals 1986, 21, 235-260. [CrossRef]
- Wang, B.-t.; Zhang, C.-h.; Qiu, X.-l.; Ji, E.-y.; Zhang, W.-h. Research on Wetting-Drying Cycles’ Effect on the Physical and Mechanical Properties of Expansive Soil Improved by OTAC-KCl. Advances in Materials Science and Engineering 2015, 2015, 304276, 304271-304277. [CrossRef]
- Brindley, G.W.; Brown, G. Crystal Structures of Clay Minerals and their X-ray Identification; Mineralogical Society: London, 1984.
- Kuhnel, R.A.; van der Gaast, S.J. Humidity controlled diffractometry and its applications. Advances in X-Ray Analysis 1993, 36, 439-449.
- Van der Gaast, S.J.; Kuhnel, R.A.; Vasterink, J.; Frost, R.L. A new model for the structure of water in the interlayer of Ca-Wyoming montmorillonite. Clays Our Future, Proc. Int. Clay Conf., 11th 1997, 421-427.
- Ferrage, E. Investigation of smectite hydration properties by modeling experimental X-ray diffraction patterns: Part I. Montmorillonite hydration properties. American Mineralogist 2005, 90, 1358-1374. [CrossRef]
- Fu, M.H.; Zhang, Z.Z.; Low, P.F. Changes in the Properties of a Montmorillonite-Water System during the Adsorption and Desorption of Water: Hysteresis1. Clays and Clay Minerals 1990, 38, 485-492. [CrossRef]
- Morida, K.; Fukushi, K.; Sakuma, H.; Tamura, K. Systematic comparison of the hydration and dehydration of Na+-, K+-, and NH4+-saturated montmorillonite, nontronite, hectorite, saponite, and Fe-saponite by in situ X-ray diffraction measurements. Applied Clay Science 2023, 237, 106898. [CrossRef]
- Morodome, S.; Kawamura, K. In situ X-ray diffraction study of the swelling of montmorillonite as affected by exchangeable cations and temperature. Clays and Clay Minerals 2011, 59, 165-175. [CrossRef]
- Tamura, K.; Yamada, H.; Nakazawa, H. Stepwise hydration of high-quality synthetic smectite with various cations. Clays and Clay Minerals 2000, 48, 400-404. [CrossRef]
- Al-Taie, A.; Disfani, M.M.; Evans, R.; Arulrajah, A.; Horpibulsuk, S. Swell-shrink Cycles of Lime Stabilized Expansive Subgrade. Procedia Engineering 2016, 143, 615-622. [CrossRef]
- Vanorio, T. Elastic properties of dry clay mineral aggregates, suspensions and sandstones. Geophysical Journal International 2003, 155, 319-329. [CrossRef]
- Apriyono, A.; Yuliana, Y.; Kamchoom, V.; Leung, A.K.; Jotisankasa, A. The effects of desiccation crack and seasonal variation on hydrological response of compact clay embankment. Canadian Geotechnical Journal 2025, 62, 1-16. [CrossRef]
- Chittoori, B.C.S.; Puppala, A.J.; Pedarla, A. Addressing Clay Mineralogy Effects on Performance of Chemically Stabilized Expansive Soils Subjected to Seasonal Wetting and Drying. Journal of Geotechnical and Geoenvironmental Engineering 2018, 144, 04017097. [CrossRef]
- Kalantari, B. Engineering significant of swelling soils. Research Journal of Applied Sciences, Engineering and Technology 2012, 4, 2874-2878.
- Kumar, A.; Biswas, N.; Puppala, A.J.; Mebarkia, B. Forensic investigation and chemical remediation of a collapsed highway embankment slope in Houston, Texas. Transportation Geotechnics 2025, 51, 101503. [CrossRef]
- Liu, W.; Tan, J.; Wu, J.; Xu, L.; Wan, J. Effect of cyclic wetting on lateritic clay subgrade settlement and train-track dynamic response of high-speed railway. Transportation Geotechnics 2025, 51, 101541. [CrossRef]
- Shah, S.S.A.; Turrakheil, K.S.; Naveed, M. Impact of Wetting and Drying Cycles on the Hydromechanical Properties of Soil and Implications on Slope Stability. Atmosphere 2024, 15, 1368. [CrossRef]
- Walker, C.; Heitor, A.; Clarke, B.G. Impact of drying-wetting cycles on the small strain behaviour of compacted clay. Transportation Geotechnics 2023, 42, 101063. [CrossRef]
- Zemenu, G.; Martine, A.; Roger, C. Analysis of the behaviour of a natural expansive soil under cyclic drying and wetting. Bulletin of Engineering Geology and the Environment 2009, 68, 421-436. [CrossRef]
- Totsche, K.U. Microaggregates in soils. Journal of Plant Nutrition and Soil Science 2017, 180, 15-21. [CrossRef]
- Al-Mahbashi, A.M.; Al-Shamrani, M.A.; Abbas, M.F. Hydromechanical behavior of unsaturated expansive clay under repetitive loading. Journal of Rock Mechanics and Geotechnical Engineering 2021, 13, 1136-1146. [CrossRef]
- Montes-H, G. Swelling–shrinkage measurements of bentonite using coupled environmental scanning electron microscopy and digital image analysis. Journal of Colloid and Interface Science 2005, 284, 271-277. [CrossRef]
- Danilatos, G.D. Foundations of environmental scanning electron microscopy. Advances in Electronics and Electronic Physics 1988, 71, 109-250.
- Danilatos, G.D. Introduction to the ESEM instrument. Microscopy Research and Technique 1993, 25, 354-361.
- Baker, J.C.; Grabowska-Olszewska, B.; Uwins, P.J.R. ESEM study of osmotic swelling of bentonite from Radzionkow (Poland). Applied Clay Science 1995, 9, 465-469.
- Carrier, B.; Wang, L.; Vandamme, M.; Pellenq, R.J.M.; Bornert, M.; Tanguy, A.; Van Damme, H. ESEM Study of the Humidity-Induced Swelling of Clay Film. Langmuir 2013, 29, 12823-12833. [CrossRef]
- Friedrich, F.; Schild, D.; Weidler, P.G.; Schäfer, T.; Schäfer, T.; Dohrmann, R.; Greenwell, H.C. Hydration of Febex Bentonite as Observed by Environmental Scanning Electron Microscopy (ESEM). In Filling the Gaps – from Microscopic Pore Structures to Transport Properties in Shales; Clay Minerals Society: 2016; Volume 21, p. 0.
- Kloprogge, J.T.; Duong, L.V.; Frost, R.L.; Boström, T.L. Use of environmental scanning electron microscopy to study uncoated minerals. Microscopy and Analysis 2005, 19, 17–19.
- Romero, E.; Simms, P.H. Microstructure Investigation in Unsaturated Soils: A Review with Special Attention to Contribution of Mercury Intrusion Porosimetry and Environmental Scanning Electron Microscopy. Geotechnical and Geological Engineering 2008, 26, 705-727. [CrossRef]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nature Methods 2012, 9, 671-675. [CrossRef]
- Chen, L.; Wu, Z.; Xu, L.; Li, Z.; Zhang, Y.; Xia, W. Hydration-Driven Interfacial Behaviors of Nanoconfined Sodium Montmorillonite. Langmuir 2025, 41, 31471-31480. [CrossRef]
- Xie, G.; Xiao, Y.; Deng, M.; Zhang, Q.; Huang, D.; Jiang, L.; Yang, Y.; Luo, P. Quantitative Investigation of the Hydration Behavior of Sodium Montmorillonite by Thermogravimetric Analysis and Low-Field Nuclear Magnetic Resonance. Energy & Fuels 2019, 33, 7483-7492. [CrossRef]
- Tessier, D.; Pedro, G. Electron microscopy study of Na smectite fabric - role of layer charge, salt concentration and suction parameters. In Developments in Sedimentology, van Olphen, H., Veniale, F., Eds.; Elsevier: Amsterdam, 1982; Volume 35, pp. 165-176.
- Tessier, D.L.A.; Petit, J.C. Relation between macroscopic behavior of clays and their microstructural. Applied Geochemistry 1992, Suppl. Issue 1, 151-161.
- Teng, Y.; Chen, C.; Zheng, L.; Whittaker, M.L. Optically Triggered Nucleation Eliminates Hydration Hysteresis in Swelling Minerals. Langmuir 2025, 41, 11307-11313. [CrossRef]
- Teng, F. Optically Triggered Nucleation Eliminates Hydration Hysteresis in Swelling Minerals. Langmuir 2025. [CrossRef]
- Cases, J.M.; Berend, I.; Besson, G.; Francois, M.; Uriot, J.P.; Thomas, F.; Poirier, J.E. Mechanism of adsorption and desorption of water vapor by homoionic montmorillonite. 1. The sodium-exchanged form. Langmuir 1992, 8, 2730-2739. [CrossRef]
- Tompsett, G.A.; Krogh, L.; Griffin, D.W.; Conner, W.C. Hysteresis and Scanning Behavior of Mesoporous Molecular Sieves. Langmuir 2005, 21, 8214-8225. [CrossRef]
- Nair, A.K.N.; Cui, R.; Sun, S. Overview of the Adsorption and Transport Properties of Water, Ions, Carbon Dioxide, and Methane in Swelling Clays. American Chemical Society 2021. [CrossRef]
- Zhang, W.; Chen, S.; Tong, K.; Li, S.; Huang, K.; Dai, Z.; Luo, L. Effects of the Layer Charge Location and Interlayer Cation on Rectorite Swelling Properties: Comparison between Molecular Dynamics Simulations and Experiments. American Chemical Society 2022. [CrossRef]
- Emmerich, K.; Koeniger, F.; Kaden, H.; Thissen, P. Microscopic structure and properties of discrete water layer in Na-exchanged montmorillonite. Journal of Colloid and Interface Science 2015, 448, 24-31. [CrossRef]
- Hetzel, F. The Microstructure of Three NA+ Smectites: The Importance of Particle Geometry on Dehydration and Rehydration. Clays and Clay Minerals 1994, 42, 242-248. [CrossRef]
- Saiyouri, N.; Tessier, D.; Hicher, P.Y. Experimental study of swelling in unsaturated compacted clays. Clay Minerals 2004, 39, 469-479. [CrossRef]
- Sato, T.; Watanabe, T.; Otsuka, R. Effects of layer charge, charge location, and energy change on expansion properties of dioctahedral smectites. Clays and Clay Minerals 1992, 40, 103−113.
- Endo, M.; Sato, H. Swelling Stress of Bentonite: Thermodynamics of Interlayer Water in K-Montmorillonite in Consideration of Alteration. Minerals 2024. [CrossRef]
- Xiong, Q.; Baychev, T.G.; Jivkov, A.P. Review of pore network modelling of porous media: Experimental characterisations, network constructions and applications to reactive transport. Journal of Contaminant Hydrology 2016), 192, 101-117. [CrossRef]
- Prastistho, W.; Apsari, C.N.; Gusnaniar, N.; Budianta, W.; Promentilla, M.; Utami, S.T. Re-evaluating the evidence of aflatoxin B1 intercalation into smectite interlayer: a review based on basal spacing data. Sains Tanah: Journal of Soil Science and Agroclimatology 2025. [CrossRef]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.



