Submitted:
25 February 2026
Posted:
27 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results
3.1. Definition and Pathophysiology of Erythroderma
3.2. Consequences of Erythroderma
3.3. Causes of Erythroderma
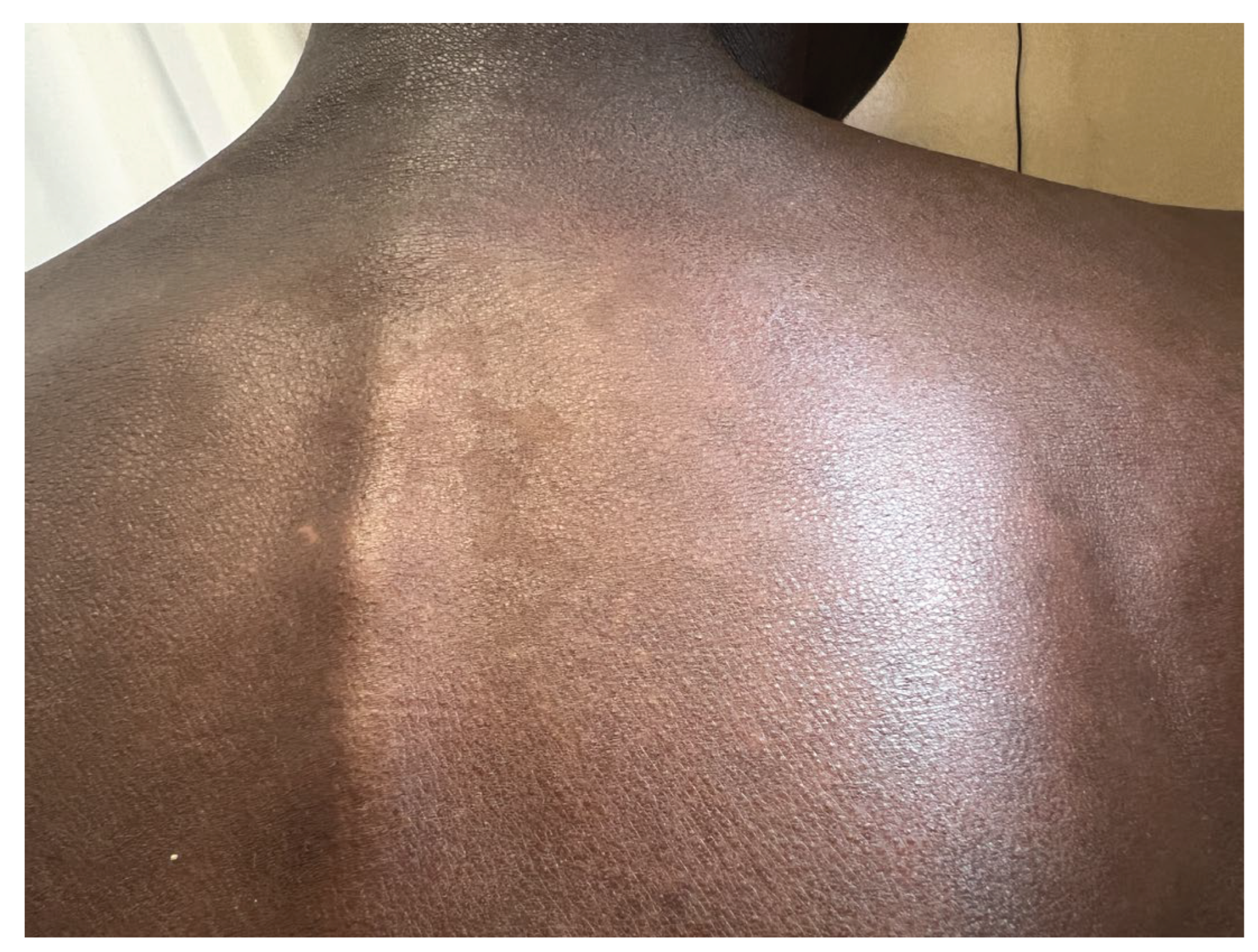
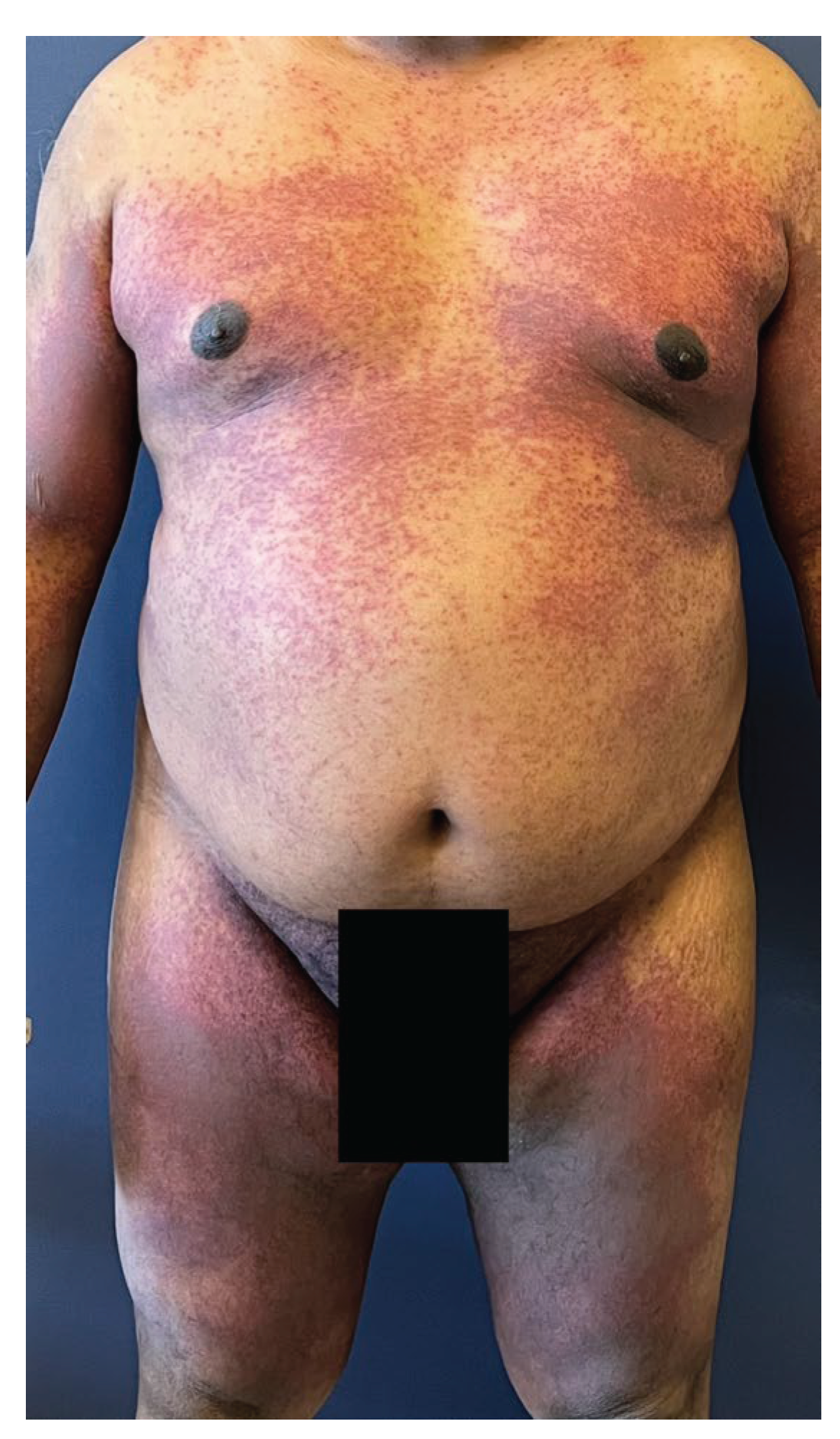
3.4. Clinical Presentation
- Yellowish thickened palmoplantar skin
- “Islands of sparing” (patches of uninvolved skin within diffuse erythema)
- Nail abnormalities
- Character and morphology of scale (fine, lamellar, greasy, or bran-like)
3.5. Initial Evaluation and Investigations
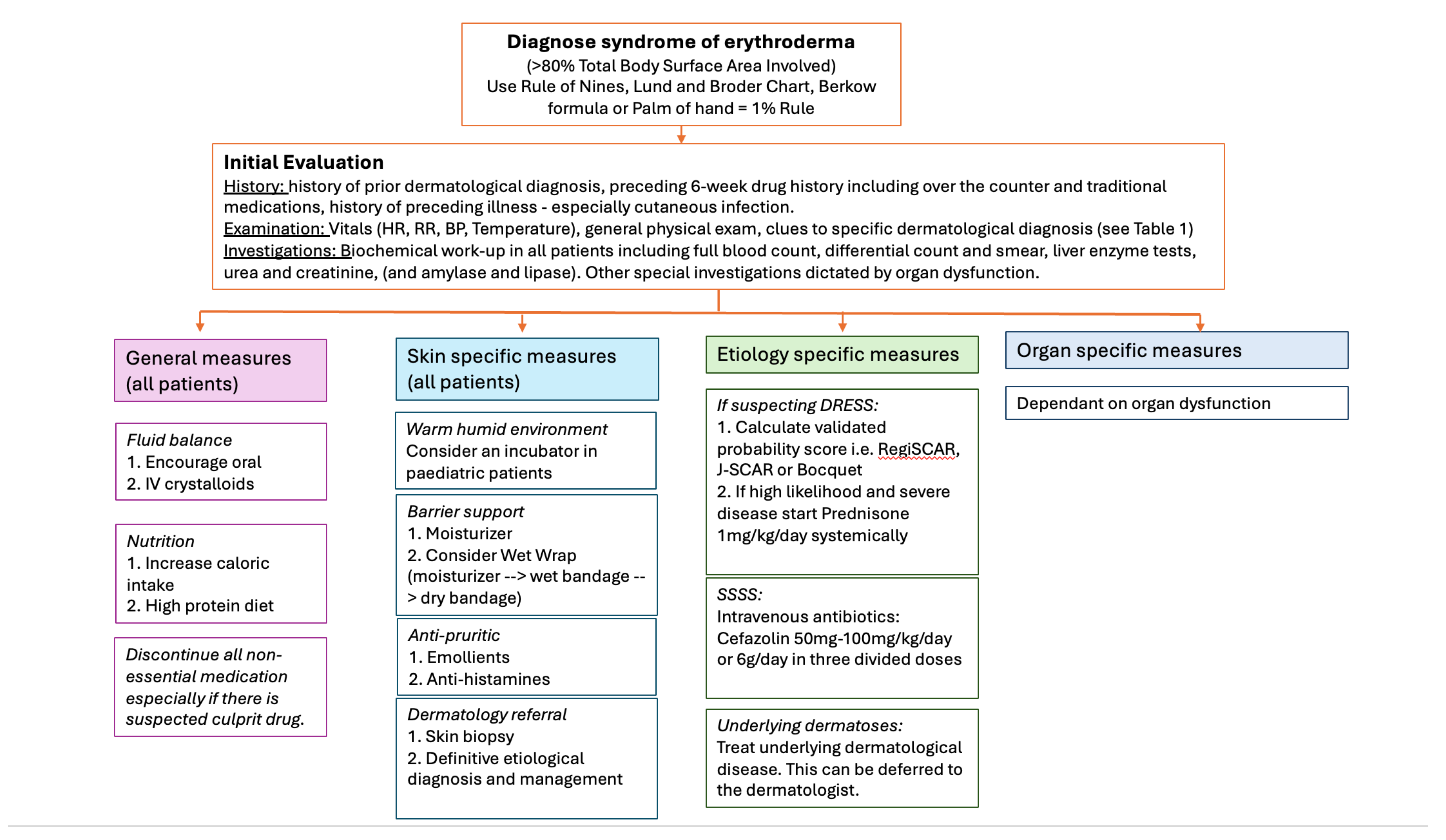
3.6. Management of Erythroderma in the Emergency Department
3.6.1. General Measures
Fluid Management
Nutritional Support
Discontinuation of Non-Essential Medications
3.6.2. Skin Specific Measures
Environmental Modification
Partial Restoration of Skin Barrier Function
Anti-Pruritic Measures
Management of Secondary infection
3.6.3. Definitive Diagnosis and Referral
3.6.4. Organ Supportive Measures
3.6.5. Etiological Causes That Require Specific Management in the Emergency Department
Drug Reactions
Staphylococcal Scalded Skin Syndrome (SSSS)
Crusted Scabies
Stevens–Johnson Syndrome/Toxic Epidermal Necrolysis (SJS/TEN)
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ED | Emergency Department |
| SSSS | Staphylococcal scalded skin syndrome |
| DRESS | Drug reaction with eosinophilia and systemic symptoms |
| TEN | Toxic Epidermal Necrolysis |
| SJS | Steven Johnson Syndrome |
| AGEP | Acute Generalized Exanthomatous Pustulosis |
| AD | Atopic Dermatitis |
References
- Kilic, D.; Yigit, O.; Kilic, T.; Buyurgan, C.S.; Dicle, O. Epidemiologic Characteristics of Patients Admitted to Emergency Department with Dermatological Complaints; a Retrospective Cross sectional Study. Arch Acad Emerg Med 2019, 7, e47.
- Agarwal, A.; Verma, H.; Pathak, N.; Gulati, N. An update on dermatological trends among emergency department visits: a cross-sectional analysis, 2017–2021. Arch. Dermatol. Res. 2025, 317, 1–3. [CrossRef]
- Alegre-Sánchez, A.; de Perosanz-Lobo, D.; Pascual-Sánchez, A.; Pindado-Ortega, C.; Fonda-Pascual, P.; Moreno-Arrones, Ó.; Jaén-Olasolo, P. Impact on Quality of Life in Dermatology Patients Attending an Emergency Department. Actas Dermo-Sifiliogr. 2017, 108, 918–923. [CrossRef]
- Baibergenova, A.; Shear, N.H. Skin Conditions That Bring Patients to Emergency Departments. Arch. Dermatol. 2011, 147, 118–120. [CrossRef]
- Nadkarni, A.; Domeisen, N.; Hill, D.; Feldman, S.R. The most common dermatology diagnoses in the emergency department. J. Am. Acad. Dermatol. 2016, 75, 1261–1262. [CrossRef]
- Grillo, E.; Vañó-Galván, S.; Jiménez-Gómez, N.; Ballester, A.; Muñoz-Zato, E.; Jaén, P. Dermatologic Emergencies: Descriptive Analysis of 861 Patients in a Tertiary Care Teaching Hospital. Actas Dermo-Sifiliograficas 2013, 104, 316–324. [CrossRef]
- Okezie PU. Dermatologic Emergency And Rates Of Admission To The Hospital GlobalRPH. Global PxPh The Clinician’s Ultimate Reference 2024. https://globalrph.com/2024/08/dermatologic-emergency-admission-to-hospital/ (accessed January 30, 2026).
- Tso, S.; Satchwell, F.; Moiz, H.; Tushar, H.; Dhariwal, S.; Barlow, R.; Forbat, E.; Randeva, H.; Tan, Y.T.; Ilchyshyn, A.; et al. Erythroderma (exfoliative dermatitis). Part 1: underlying causes, clinical presentation and pathogenesis. Clin. Exp. Dermatol. 2021, 46, 1001–1010. [CrossRef]
- Harper-Kirksey, K. Erythroderma. Life-Threatening Rashes 2018:265. [CrossRef]
- Hulmani, M.; NandaKishore, B.; Bhat, M.; Sukumar, D.; Martis, J.; Kamath, G.; Srinath, M. Clinico-etiological study of 30 erythroderma cases from tertiary center in South India. Indian Dermatol. Online J. 2014, 5, 25–9. [CrossRef]
- Mistry, N.; Gupta, A.; Alavi, A.; Sibbald, R.G. A Review of the Diagnosis and Management of Erythroderma (Generalized Red Skin). Adv. Ski. Wound Care 2015, 28, 228–236. [CrossRef]
- Miyashiro, D.; Sanches, J.A. Erythroderma: a prospective study of 309 patients followed for 12 years in a tertiary center. Sci. Rep. 2020, 10, 1–13. [CrossRef]
- Egeberg, A.; Thyssen, J.; Gislason, G.; Skov, L. Prognosis after Hospitalization for Erythroderma. Acta Dermato-Venereologica 2016, 96, 959–962. [CrossRef]
- Baethge, C.; Goldbeck-Wood, S.; Mertens, S. SANRA—a scale for the quality assessment of narrative review articles. Res. Integr. Peer Rev. 2019, 26, 5. [CrossRef]
- Whittaker S. Erythroderma. In: Bolognia JL, Schaffer J V, Cerroni L, editors. Dermatology , vol. 5, Elsevier ; 2024.
- Cuellar-Barboza, A.; Ocampo-Candiani, J.; Herz-Ruelas, M. A Practical Approach to the Diagnosis and Treatment of Adult Erythroderma. Actas Dermo-Sifiliograficas 2018, 109, 777–790. [CrossRef]
- Kanthraj, G.R.; Srinivas, C.R.; Devi, P.U.; Ganasoundari, A.; Shenoi, S.D.; Deshmukh, R.P.; Suresh, B.J.; Pai, S.B.; .; Pgdca Quantitative estimation and recommendations for supplementation of protein lost through scaling in exfoliative dermatitis. Int. J. Dermatol. 1999, 38, 91–95. [CrossRef]
- Sehgal, V.N.; Srivastava, G.; Sardana, K. Erythroderma/exfoliative dermatitis: a synopsis. Int. J. Dermatol. 2003, 43, 39–47. [CrossRef]
- Kurokawa, R.; Hagiwara, A.; Niijima, Y.; Kojima, K. Computed tomography imaging findings in erythrodermic psoriasis treated with infliximab: A case report. Radiol. Case Rep. 2018, 13, 460–463. [CrossRef]
- Shuster, S. HIGH-OUTPUT CARDIAC FAILURE FROM SKIN DISEASE. Lancet 1963, 281, 1338–1340. [CrossRef]
- Lu, L.-Y.; Yu, Y.-Z.; Wang, S.-H.; Zhang, J.; Fan, X.; Qi, Y.; Lin, B.-J. A Clinicopathological, Etiological Analysis of 136 Cases of Erythroderma: A Single-Center Retrospective Study. Int. J. Dermatol. Venereol. 2023, 7, 137–143. [CrossRef]
- Iqbal, M.; Tan, C.-H. Erythroderma and acute skin failure. Medicine 2025, 53, 512–516. [CrossRef]
- Holm, J.; Ren, Z.; McCarthy, M.; Pang, Y.; Nguyen, W.; Guitart, J.; Zhou, X. 64894 Retrospective analysis of demographic and clinical characteristics of erythroderma patients reveals potential risk factors. J. Am. Acad. Dermatol. 2025, 93, AB290. [CrossRef]
- Surekha, A.; Harikrishna, J.; Kushma, T. Aetiology and clinical profile of patients presenting with erythroderma. J. Clin. Sci. Res. 2025. [CrossRef]
- Vasconcellos C, Domingues PP, Aoki V, Miyake RK, Sauaia N, Martins EC. Erythroderma: analysis of 247 cases n.d.
- Avandi B, Ghahartar M, Kashkooli NM, Ahramiyanpour N, Parvizi MM. Epidemiological and clinical features of hospitalized erythroderma patients: a cross-sectional study. Iranian Journal of Dermatology 2022;25:60–5. [CrossRef]
- Sehgal, V.N.; Srivastava, G. Erythroderma/generalized exfoliative dermatitis in pediatric practice: An overview. Int. J. Dermatol. 2006, 45, 831–839. [CrossRef]
- Dhalimi, A.; A., M.A. Neonatal and infantile erythroderma: A clinical and follow-up study of 42 cases. J. Dermatol. 2007, 34, 302–307. [CrossRef]
- Kalsy, J.; Puri, K.J.P.S. Erythroderma in children: Clinico-etiological study from Punjab. Indian J. Paediatr. Dermatol. 2013, 14, 9. [CrossRef]
- Sarkar, R.; Garg, V.K. Erythroderma in children. Indian J. Dermatol. Venereol. Leprol. 2010, 76, 341–7. [CrossRef]
- Pruszkowski, A.; Bodemer, C.; Fraitag, S.; Teillac-Hamel, D.; Amoric, J.-C.; de Prost, Y. Neonatal and Infantile Erythrodermas. Arch. Dermatol. 2000, 136, 875–880. [CrossRef]
- Michalek IMaria, Loring Belinda, John SMalte. Global report on psoriasis 2016:44.
- Emanuel P, Cheng H. Eczema pathology. DermNet 2013:1. https://dermnetnz.org/topics/eczema-pathology (accessed February 7, 2026).
- Jeskey, J.; Kurien, C.; Blunk, H.; Sehmi, K.; Areti, S.; Nguyen, D.; Hostoffer, R. Atopic Dermatitis: A Review of Diagnosis and Treatment. J. Pediatr. Pharmacol. Ther. 2024, 29, 587–603. [CrossRef]
- Guttman-Yassky E, Renert-Yuval Y, Brunner PM. Atopic dermatitis. The Lancet 2025;405:583–96. [CrossRef]
- Joshi, T.P.; Duvic, M. Pityriasis Rubra Pilaris: An Updated Review of Clinical Presentation, Etiopathogenesis, and Treatment Options. Am. J. Clin. Dermatol. 2023, 25, 243–259. [CrossRef]
- Yuan, X.-Y.; Guo, J.-Y.; Dang, Y.-P.; Qiao, L.; Liu, W. Erythroderma: A clinical-etiological study of 82 cases. Eur. J. Dermatol. 2010, 20, 373–377. [CrossRef]
- Dummer, R.; Vermeer, M.H.; Scarisbrick, J.J.; Kim, Y.H.; Stonesifer, C.; Tensen, C.P.; Geskin, L.J.; Quaglino, P.; Ramelyte, E. Cutaneous T cell lymphoma. Nat. Rev. Dis. Prim. 2021, 7, 1–22. [CrossRef]
- Gutiérrez-Cerrajero, C.; Sprecher, E.; Paller, A.S.; Akiyama, M.; Mazereeuw-Hautier, J.; Hernández-Martín, A.; González-Sarmiento, R. Ichthyosis. Nat. Rev. Dis. Prim. 2023, 9, 1–23. [CrossRef]
- Vaillant AAJ, Qurie A. Immunodeficiency. StatPearls 2023.
- Qian, Q.; Shen, J.; Vinturache, A.; Ding, G. Staphylococcal scalded skin syndrome in a child. Lancet Infect. Dis. 2023, 23, 634–634. [CrossRef]
- Mishra, A.K.; Yadav, P.; Mishra, A. A Systemic Review on Staphylococcal Scalded Skin Syndrome (SSSS): A Rare and Critical Disease of Neonates. Open Microbiol. J. 2016, 10, 150–159. [CrossRef]
- Liy-Wong, C.; Pope, E.; Weinstein, M.; Lara-Corrales, I. Staphylococcal scalded skin syndrome: An epidemiological and clinical review of 84 cases. Pediatr. Dermatol. 2020, 38, 149–153. [CrossRef]
- Wang, X.-D.; Shen, H.; Liu, Z.-H. Contagious Erythroderma. J. Emerg. Med. 2016, 51, 180–181. [CrossRef]
- Devi, G.C.; Hazarika, N. Erythroderma secondary to crusted scabies. BMJ Case Rep. 2021, 14, e248000. [CrossRef]
- Talty, R.; Micevic, G.; Damsky, W.; King, B.A. Erythrodermic scabies in an immunocompetent patient. JAAD Case Rep. 2022, 29, 112–115. [CrossRef]
- Cox, S.; Kriho, K.; De Klerk, S.; van Dijk, M.; Rode, H. Total body and hand surface area: Measurements, calculations, and comparisons in ethnically diverse children in South Africa. Burns 2017, 43, 1567–1574. [CrossRef]
- Brüggen, M.-C.; Walsh, S.; Ameri, M.M.; Anasiewicz, N.; Maverakis, E.; French, L.E.; Ingen-Housz-Oro, S.; DRESS Delphi consensus group; Abe, R.; Ardern-Jones, M.; et al. Management of Adult Patients with Drug Reaction with Eosinophilia and Systemic Symptoms. JAMA Dermatol. 2024, 160, 37–44. [CrossRef]
- Wei, B.M.; Fox, L.P.; Kaffenberger, B.H.; Korman, A.M.; Micheletti, R.G.; Mostaghimi, A.; Noe, M.H.; Rosenbach, M.; Shinkai, K.; Kwah, J.H.; et al. Drug-induced hypersensitivity syndrome/drug reaction with eosinophilia and systemic symptoms. Part II diagnosis and management. J. Am. Acad. Dermatol. 2023, 90, 911–926. [CrossRef]
- Lee, E.Y.; Peter, J. Diagnosing and Managing Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) Amidst Remaining Uncertainty. J. Allergy Clin. Immunol. Pr. 2025, 13, 979–988. [CrossRef]
- Mohapatra, L.; Singh, B.S.T.P.; Satapathy, S.; Samal, K. Pediatric erythroderma – A clinical and therapeutic review. Indian J. Ski. Allergy 2025, 4, 7–18. [CrossRef]
- Mistry, N.; Gupta, A.; Alavi, A.; Sibbald, R.G. A Review of the Diagnosis and Management of Erythroderma (Generalized Red Skin). Adv. Ski. Wound Care 2015, 28, 228–236. [CrossRef]
- González-López, G.; Ceballos-Rodríguez, R.; González-López, J.; Rodríguez, M.F.; Herranz-Pinto, P. Efficacy and safety of wet wrap therapy for patients with atopic dermatitis: a systematic review and meta-analysis. Br. J. Dermatol. 2016, 177, 688–695. [CrossRef]
- Andersen, R.; Thyssen, J.; Maibach, H. The Role of Wet Wrap Therapy in Skin Disorders – A Literature Review. Acta Dermato-Venereologica 2015, 95, 933–939. [CrossRef]
- Kim, D.-H.; Koh, Y.-I. Comparison of Diagnostic Criteria and Determination of Prognostic Factors for Drug Reaction with Eosinophilia and Systemic Symptoms Syndrome. Allergy, Asthma Immunol. Res. 2014, 6, 216–21. [CrossRef]
- Brazel, M.; Desai, A.; Are, A.; Motaparthi, K. Staphylococcal Scalded Skin Syndrome and Bullous Impetigo. Medicina 2021, 57, 1157. [CrossRef]
- Gray, L.; Hansen, A.M.; Cipriano, S.D. Pediatric Staphylococcal Scalded Skin Syndrome: A Systematic Review of the Literature to Inform Work-Up and Management. Pediatr. Dermatol. 2025, 42, 978–984. [CrossRef]
| Etiology |
Adults (%) * [9,12,21,23,24,25,26] |
Children (%) *[27,28,29,30,31] |
Disease information |
| A. Exacerbation of diagnosed underlying dermatoses | 23 to 36.3 | 20 | In cases of exacerbation of a pre-existing dermatosis, a prior history of the underlying condition may be elicited. However, some patients may initially present with erythroderma as the first manifestation of their disease. |
| 1. Psoriasis | 14.5 to 45 | 18 | A multisystem immune-mediated inflammatory disease that classically presents with erythematous plaques and silvery scale, often with nail and joint involvement [32]. Nail findings such as irregular pitting, onycholysis, and “oil drop” discoloration are important diagnostic clues. |
| 2. Eczematous dermatoses | 8 to 33.9 | 14.3 to 20 | Eczema is a clinical term describing cutaneous inflammation that histologically manifests as intercellular oedema of the epidermis (spongiosis) [33]. Like erythroderma, it represents a syndrome with multiple etiologies. Common subtypes are outlined below. |
| 2.1 Atopic dermatitis (AD) | 8.7 to 21.8 | 12 | A chronic relapsing pruritic inflammatory dermatosis characterised by scaly patches on a background of xerosis. AD is part of the atopic spectrum, which includes allergic rhinitis, conjunctivitis, asthma, and food allergy [34,35]Fine scaling commonly accompanies erythrodermic transformation. |
| 2.2 Seborrheic dermatitis | Rare | 5 | Associated with overgrowth of Malassezia furfur, predominantly affecting sebaceous gland–rich areas. In immunocompromised individuals, including patients with HIV, the disease may generalise. The scale is typically greasy and yellowish with a bran-like appearance. |
| 3. Pityriasis Rubra Pilaris (PRP) | 1 | 2.4 | A rare papulosquamous disorder in which a trigger activates the IL-23/Th17 inflammatory pathway, leading to follicular hyperkeratosis and coalescence into widespread erythema [36]. PRP is subdivided into six clinical types, with types I, III, and VI potentially presenting with erythroderma. “Islands of sparing” and waxy palmoplantar keratoderma are characteristic clinical clues. |
| B. Drug reactions | 7.3 to 67 | 29 to 42.8 | Drug reactions may be immunologically or non-immunologically mediated. Temporal association with medication exposure is a key diagnostic clue as demonstrated in table 2. Acute generalized exanthematous pustulosis (AGEP), toxic epidermal necrolysis (TEN), and drug reaction with eosinophilia and systemic symptoms (DRESS) may present with erythroderma [21,24]. Rapid onset of widespread erythema should raise suspicion for a drug-induced cause. A comprehensive medication history must include prescription drugs, over-the-counter preparations, and traditional remedies. In a Chinese cohort, traditional herbal medications were the most frequently implicated agents [37]. Among hospitalized erythroderma patients, anticonvulsants are commonly reported culprit drugs [21,24,26]. |
| C. Cutaneous T Cell Lymphoma (CTCL) | 4 to 21.8 | Rare | A group of non-Hodgkin lymphomas characterized by monoclonal T-cell infiltration of the skin without extracutaneous involvement at initial diagnosis [38]. Progressive pruritus, lymphadenopathy, and alopecia may be clinical clues. |
| D. Idiopathic | 4.4 to 30 | 30 | No identifiable underlying cause despite appropriate investigation. |
| E. Primary blistering diseases | Rare | Autoimmune or genetic disorders affecting adhesion molecules that maintain structural integrity between keratinocytes and between the epidermis and dermis. Autoantibodies or genetic mutations may disrupt these adhesion proteins, leading to widespread blistering and secondary erythroderma. | |
| F. Genodermatoses | Rare | 18 to 70 ^ | Genetic syndromes with prominent cutaneous manifestations involving skin, hair, and nails. |
| 1. Icthysiform disorders | Rare | 25 to 31.5 | Inherited disorders of keratinization characterized by abnormal epidermal differentiation and barrier dysfunction [39]. |
| 2. Immunodeficiency syndromes | Rare | 4.8 to 30 | Primary or secondary immune system defects that predispose patients to recurrent infections and inflammatory skin disease [40]. |
| G. Infections | 12 | 40 | |
| 1. Staphylococcal scalded skin syndrome | Rare | 7.4 to 18 | Caused by exfoliative toxins produced by Staphylococcus aureus, leading to superficial epidermal splitting [41,42,43]. Skin denudation typically begins on the central face and flexural areas (neck, axillae, groin). Radial fissuring around the mouth and eyes is a useful clinical clue. |
| 2. Crusted scabies | Rare | Occurs predominantly in immunocompromised or debilitated individuals with impaired sensory response [44,45,46]. In this hyper infestation state, millions of Sarcoptes scabiei var. hominis mites inhabit hyperkeratotic scale [46]. Prolonged duration and thick yellow crusted plaques are characteristic. Bedside scraping with potassium hydroxide microscopy is usually diagnostic. Importantly, crusted scabies is highly contagious [44]. | |
| Day | 0 | D4-7 | D7 | D14 | D21 | D28 | D35 | D40 | |
| Urticaria Anaphylaxis |
AGEP | SJS/TEN | DRESS | ||||||
| Fixed drug reactions | |||||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




