Submitted:
17 February 2026
Posted:
27 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
2.1. Literature Search Strategy
2.2. Study Selection and Data Synthesis
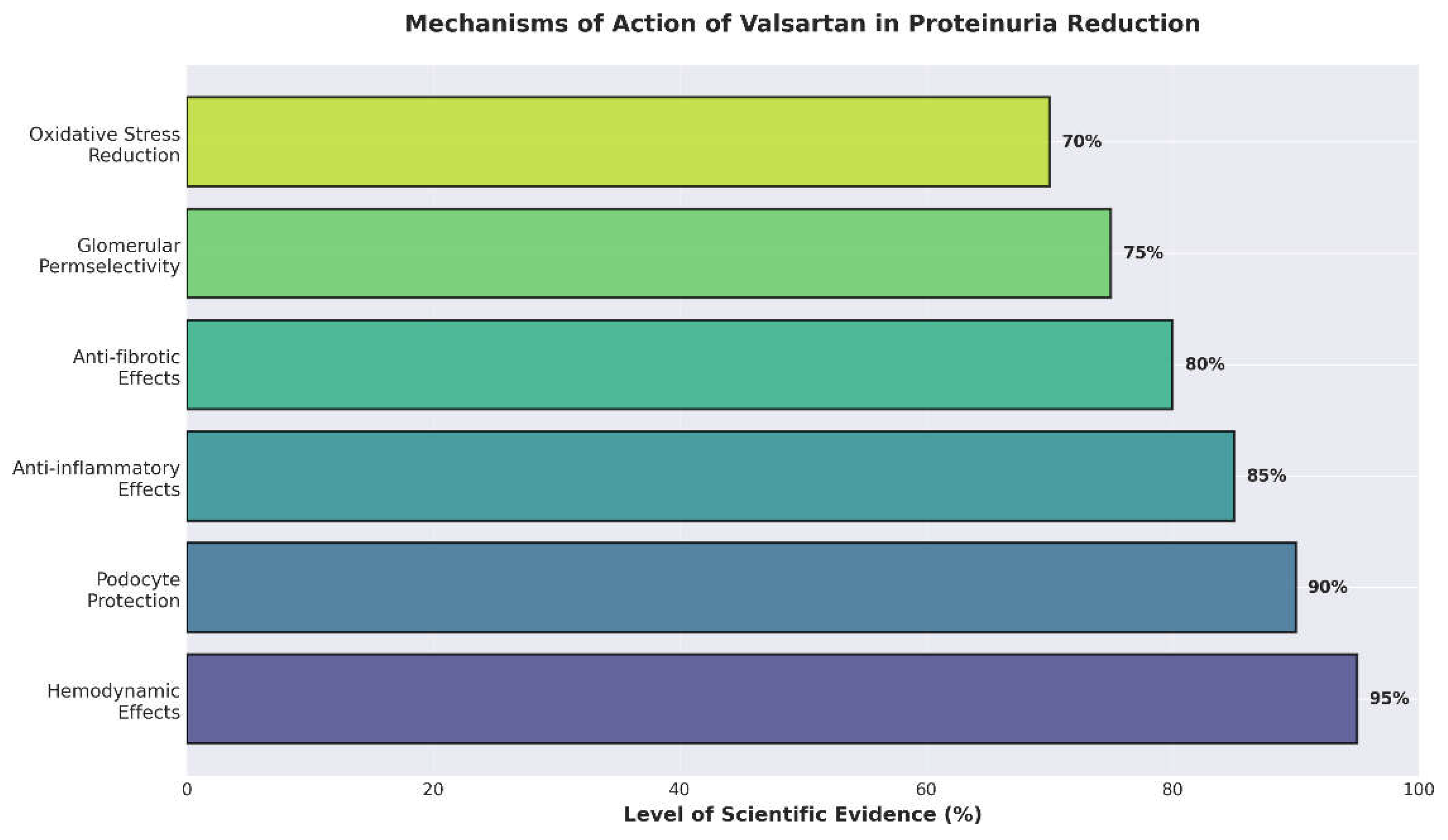
2.3. Mechanisms of Action
2.4. Hemodynamic Effects
2.5. Podocyte Protection
2.6. Anti-inflammatory and Anti-fibrotic Effects
2.7. Improvement of Glomerular Permselectivity
2.8. Oxidative Stress Reduction
3. Clinical Efficacy
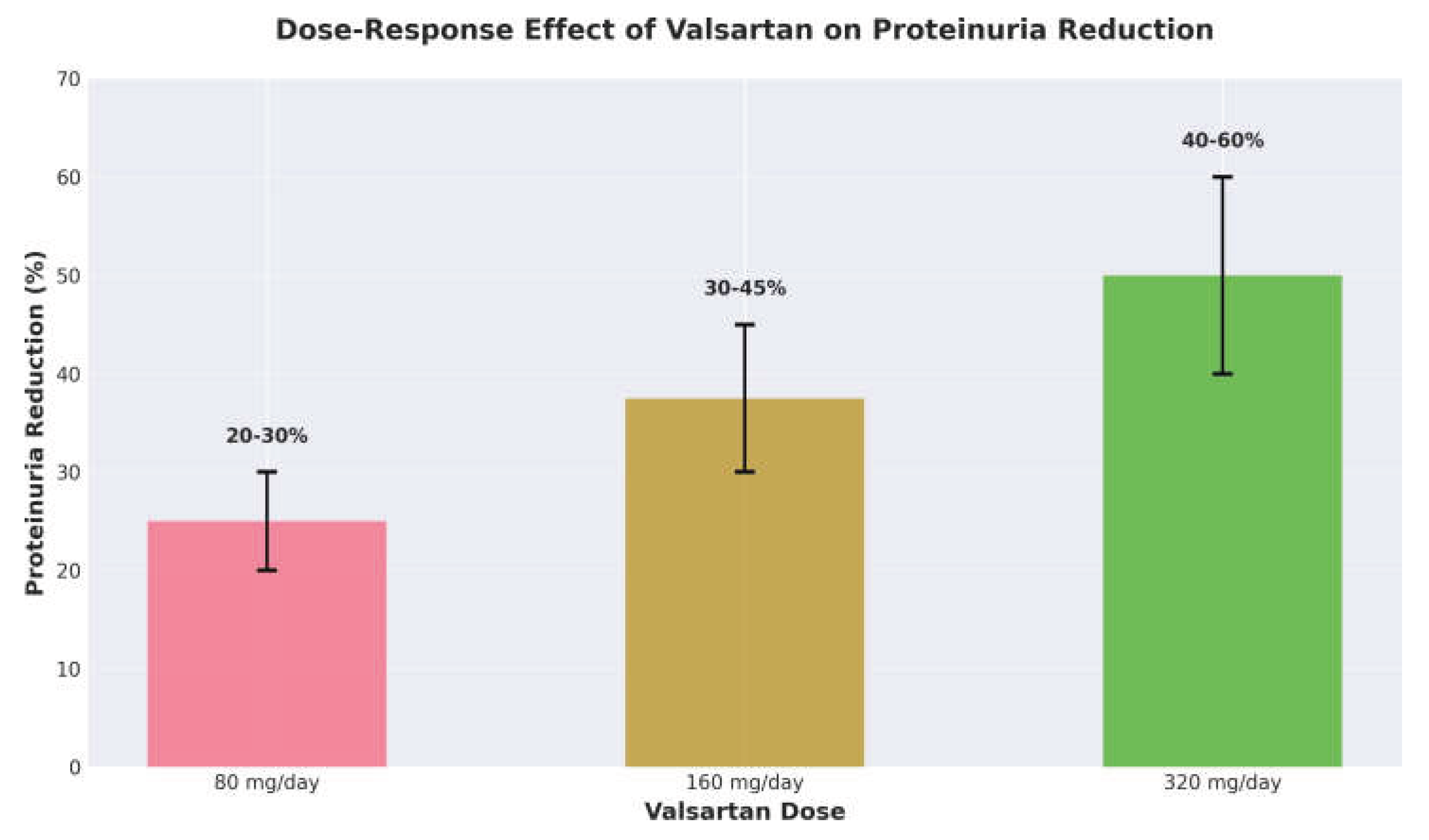
3.1. Dose–Response Relationship
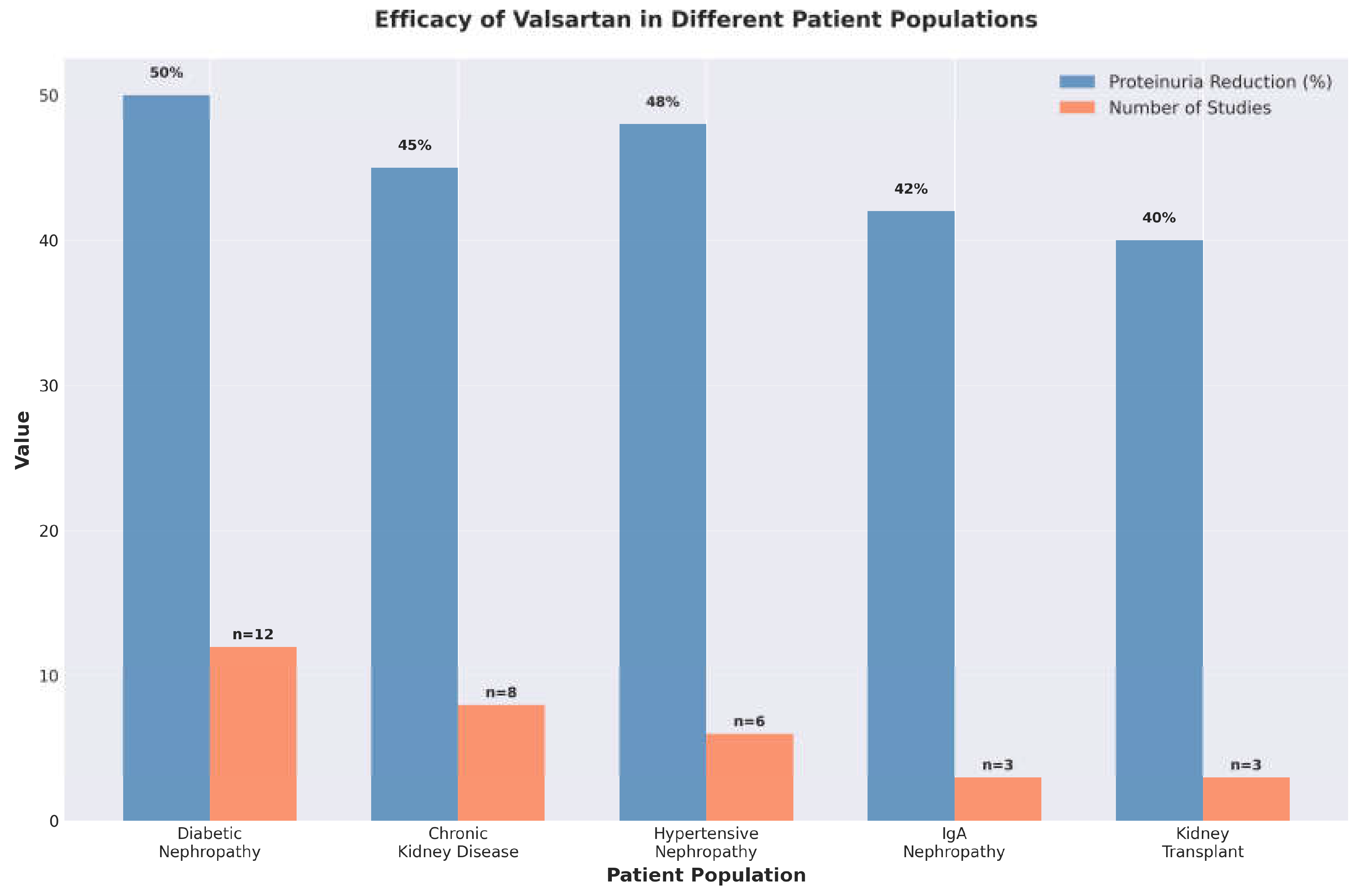
3.2. Efficacy Across Patient Populations
3.2.1. Diabetic Nephropathy
3.2.2. Chronic Kidney Disease
3.2.3. Hypertensive Nephropathy
3.2.4. IgA Nephropathy
3.2.5. Kidney Transplant Recipients
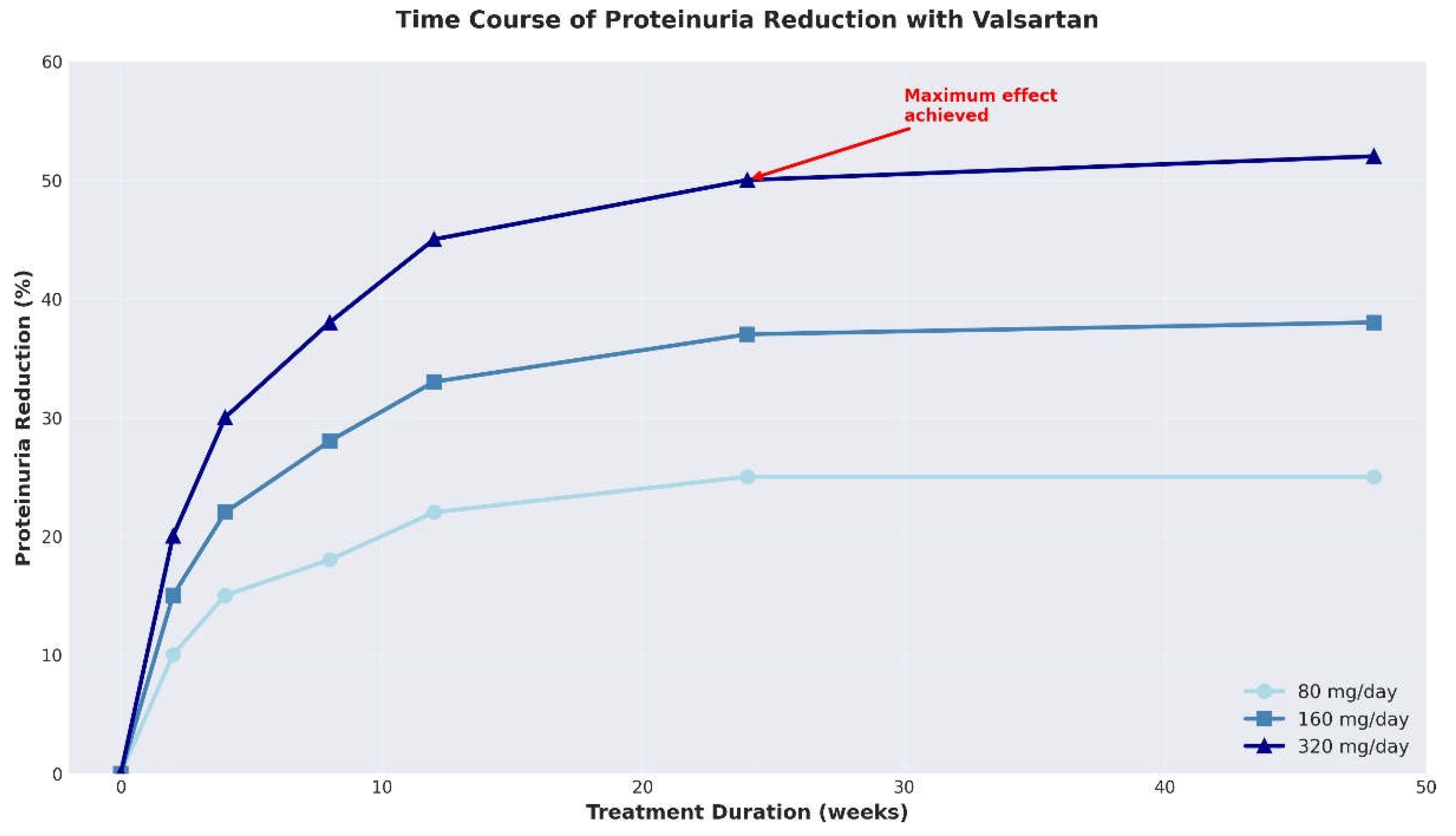
3.3. Time Course of Effects
4. Comparative Effectiveness
5. Safety and Tolerability
5.1. Common Adverse Effects
5.2. Contraindications
5.3. Monitoring Requirements
5.4. Clinical Recommendations
- Initiation: Valsartan should be considered first-line therapy for patients with proteinuria exceeding 300 mg/day or an albumin-to-creatinine ratio exceeding 30 mg/g, unless contraindicated.
- Dosing: An initial dose of 80 mg once daily is recommended, with gradual titration every 2–4 weeks based on tolerability and response. A target dose of 160–320 mg/day should be pursued for maximal renoprotection in patients who tolerate higher doses.
- Monitoring: Serum potassium and creatinine should be checked at baseline, 2 weeks after initiation or dose change, and every 3–6 months during maintenance. Proteinuria should be monitored every 3–6 months to assess therapeutic response.
- Combination Therapy: Combination with ACE inhibitors may be considered in carefully selected patients with refractory proteinuria, but requires intensive monitoring for adverse effects.
- Special Populations: In diabetic nephropathy, hypertensive nephropathy, and CKD, valsartan represents first-line therapy. In transplant recipients and advanced CKD (stages 4–5), increased monitoring and potential dose adjustment are warranted.
5.5. Future Directions
5.6. Personalized Medicine
5.7. Novel Combination Strategies
5.8. Long-term Outcomes
5.9. Mechanistic Studies
6. Conclusions
References
- Ruggenenti, P.; et al. Preventing microalbuminuria in type 2 diabetes. New England Journal of Medicine 2012, 351(19), 1941–1951. [Google Scholar] [CrossRef] [PubMed]
- Brenner, B. M.; et al. Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. New England Journal of Medicine 2001, 345(12), 861–869. [Google Scholar] [CrossRef] [PubMed]
- Remuzzi, G.; et al. Pathophysiology of progressive nephropathies. New England Journal of Medicine 2006, 339(20), 1448–1456. [Google Scholar] [CrossRef] [PubMed]
- Viberti, G.; Wheeldon, N. M. Microalbuminuria reduction with valsartan in patients with type 2 diabetes mellitus: a blood pressure–independent effect. Circulation 2002, 106(6), 672–678. [Google Scholar] [CrossRef] [PubMed]
- Plum, J.; et al. Effects of the angiotensin II antagonist valsartan on blood pressure, proteinuria, and renal hemodynamics in patients with chronic renal failure and hypertension. Journal of the American Society of Nephrology 1998, 9(12), 2223–2234. [Google Scholar] [CrossRef] [PubMed]
- Rossing, K.; et al. Impact of arterial blood pressure and albuminuria on the progression of diabetic nephropathy in IDDM patients. Diabetes 2005, 42(5), 715–719. [Google Scholar] [CrossRef] [PubMed]
- Kirmizis, D.; et al. IgA Nephropathy: Insights into Genetic Basis and Treatment Options; 2011. [Google Scholar] [CrossRef]
- Katayama, S.; et al. Is renoprotection by angiotensin receptor blocker dependent on blood pressure?: the Saitama Medical School, Albuminuria Reduction in Diabetics with Valsartan (STAR) study. Hypertension Research 2007, 30(6), 529–533. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; et al. Valsartan slows the progression of diabetic nephropathy in db/db mice via a reduction in podocyte injury, and renal oxidative stress and inflammation. Clinical Science 2014, 126(9), 707–720. [Google Scholar] [CrossRef] [PubMed]
- Lee, H. W.; et al. Renoprotective efficacy of valsartan in chronic non-diabetic proteinuric nephropathies with renin–angiotensin system gene polymorphisms. Nephrology 2011, 16(4), 416–423. [Google Scholar] [CrossRef] [PubMed]
- Bilić, A.; et al. Effects of ramipril and valsartan on proteinuria and renal function in patients with nondiabetic proteinuria. Collegium Antropologicum 2011, 35(4), 1123–1129. [Google Scholar]
- Hu, Z.; et al. A Randomized Non-Controlled and Multi-Center Clinical Research to Evaluate the Safety and Effect of Valsartan in Chronic Renal Disease. 2003. [Google Scholar] [CrossRef]
- Zhang, Y.; et al. Effects of Benazepril and Valsartan Alone or in Combination on Proteinuria in Chronic Glomerular Disease. 2007. [Google Scholar] [CrossRef]
- Ohishi, M.; et al. Renal protective effect in hypertensive patients: the high doses of angiotensin II receptor blocker (HARB) study. Hypertension Research 2007, 30(12), 1187–1194. [Google Scholar] [CrossRef] [PubMed]
- Bilić, A., et al. (2011). Effects of ramipril and valsartan on proteinuria and renal function in patients with nondiabetic proteinuria [Učinci ramiprila i valsartana na proteinuriju i bubrežnu funkciju u bolesnika s nedijabetičkom proteinurijom].
- Plum, J.; et al. Effects of the angiotensin II antagonist valsartan on blood pressure, proteinuria, and renal hemodynamics in patients with chronic renal failure and hypertension. Journal of the American Society of Nephrology 1998, 9(12), 2223–2234. [Google Scholar] [CrossRef] [PubMed]
- Saikawa, S.; et al. Is the reno-protective effect of valsartan dose dependent? A comparative study of 80 and 160 mg day(−1). Hypertension Research 2010, 33(8), 834–838. [Google Scholar] [CrossRef] [PubMed]
- Chao, L.; et al. Effect of High Dose of Valsartan, Single or Combined with Lotensin in Chronic Kidney Disease. 2006. [Google Scholar] [CrossRef]
- Tang, X.; et al. Effects of Angiotensin II Receptor Antagonists—Valsartan on the Level of Endothelin-1 (ET-1) and Transforming Growth Factor beta-1 of Diabetic Nephropathy. 2010. [Google Scholar] [CrossRef]
- He, Y.; et al. Clinical Observation of the Therapeutic Effect of Valsartan for Renal Hypertension in 64 Cases. 2003. [Google Scholar] [CrossRef]
- Wang, L.; et al. Protective effects of aliskiren and valsartan in mice with diabetic nephropathy. Journal of the Renin–Angiotensin–Aldosterone System 2014, 15(4), 384–395. [Google Scholar] [CrossRef] [PubMed]
- Sun, N.; et al. Effects of valsartan combined with alpha-lipoic acid on renal function in patients with diabetic nephropathy: a systematic review and meta-analysis. BMC Endocrine Disorders 2021, 21(1), 175. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



