Submitted:
25 February 2026
Posted:
26 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Aedes aegypti Colonies and Strains
2.2. Chemicals
2.3. Larvicidal Bioassay
2.4. Topical Adulticidal Bioassay
2.5. Mosquito Airborne Repellency Test (MART Assay)
2.6. Statistical Analysis
3. Results
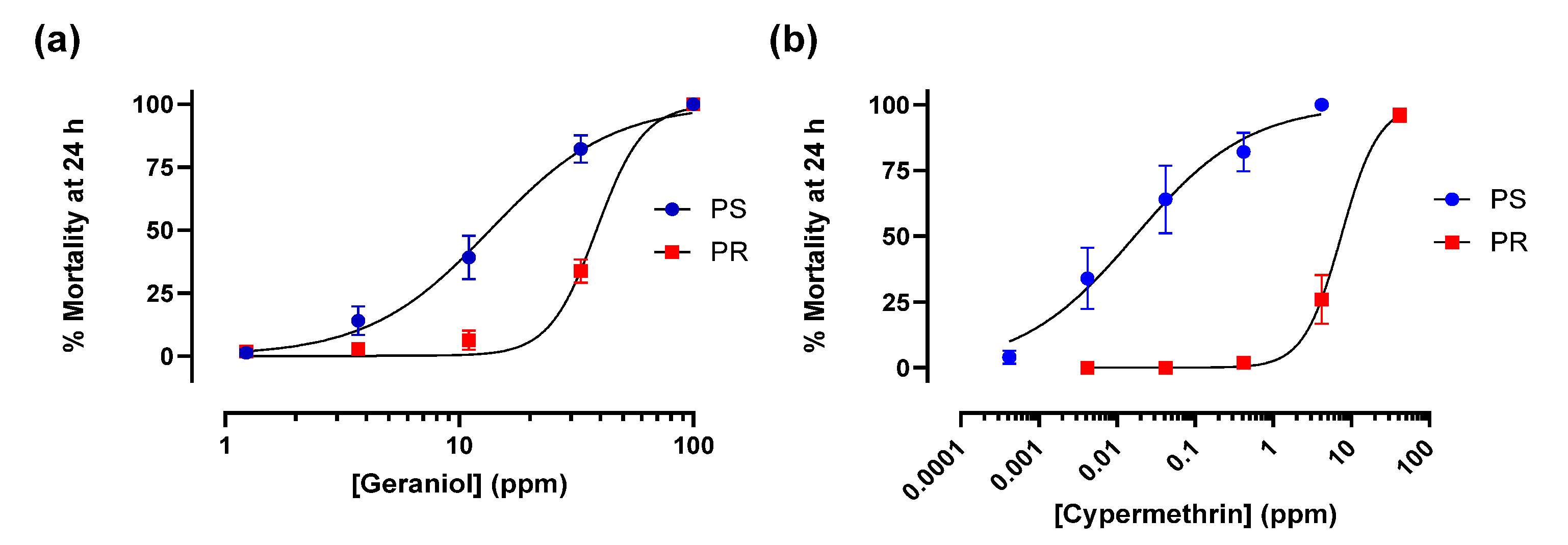
3.1. Geraniol and Cypermethrin Toxicity Against PS and PR Larvae
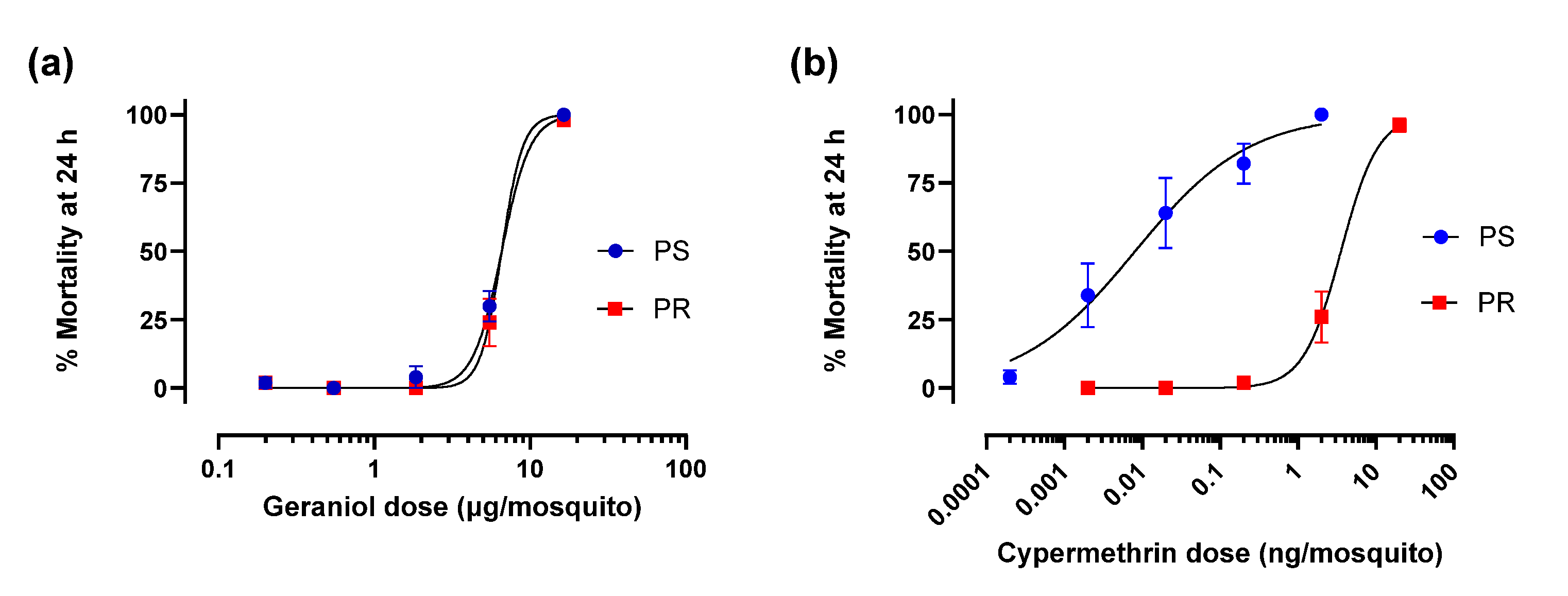
3.2. Geraniol and Cypermethrin Toxicity Against PS and PR Adult Females
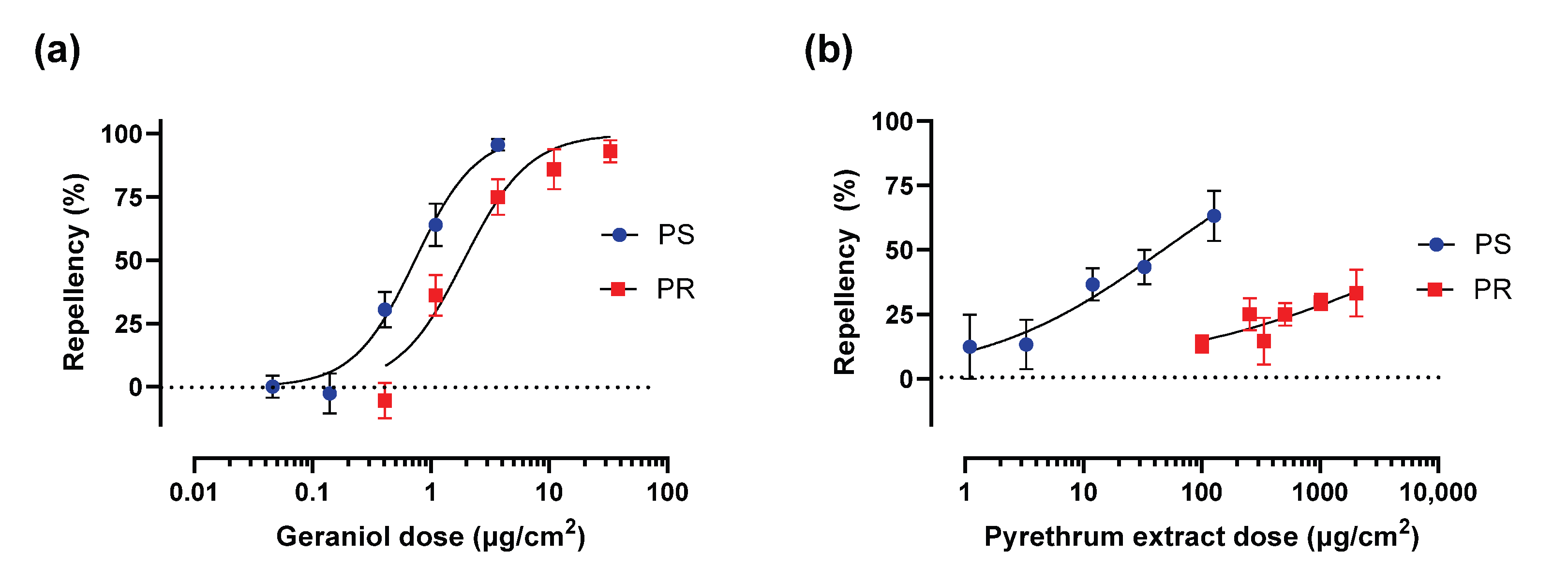
3.3. Geraniol and Pyrethrum Extract Spatial Repellency Against PS and PR Adult Females
4. Discussion
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Benelli, G.; Mehlhorn, H. Declining Malaria, Rising of Dengue and Zika Virus: Insights for Mosquito Vector Control. Parasitol Res 2016, 115, 1747–1754. [Google Scholar] [CrossRef] [PubMed]
- Mulatier, M.; Cohuet, A.; Carrasco, D. Chapter 34: Repellents for Mosquito-Borne Disease Control: Beyond the Repellency Effect. In Sensory ecology of disease vectors; Ignell, R., Lazzari, C.R., Lorenzo, M.G., Hill, S.R., Eds.; Brill | Wageningen Academic, 2022; pp. 879–911. ISBN 978-90-8686-380-8. [Google Scholar]
- Poinsignon, A.; Fournet, F.; Ngowo, H.S.; Franco Martins Barreira, V.; Pinto, J.; Bartumeus, F.; Kaindoa, E.W.; Corbel, V. Advances in Surveillance and Control Methods for Aedes-Borne Diseases and Urban Vectors: Report of the International Conference, August 2024, Tanzania. Parasites Vectors 2025, 18, 212. [Google Scholar] [CrossRef] [PubMed]
- Deletre, E.; Martin, T.; Duménil, C.; Chandre, F. Insecticide Resistance Modifies Mosquito Response to DEET and Natural Repellents. Parasites Vectors 2019, 12, 89. [Google Scholar] [CrossRef]
- Norris, E.J.; Gross, A.D.; Bartholomay, L.C.; Coats, J.R. Plant Essential Oils Synergize Various Pyrethroid Insecticides and Antagonize Malathion in Aedes aegypti. Medical Vet Entomology 2019, 33, 453–466. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, X.; Brown, D.J.; An, M.; Xue, R.-D.; Liu, N. Insecticide Resistance: Status and Potential Mechanisms in Aedes Aegypti. Pesticide Biochemistry and Physiology 2023, 195, 105577. [Google Scholar] [CrossRef]
- Hazarika, H.; Rajan, R.K.; Pegu, P.; Das, P. Insecticide Resistance in Mosquitoes: Molecular Mechanisms, Management, and Alternatives. J Pest Sci 2025. [Google Scholar] [CrossRef]
- Mosquitoes: Biology, Pathogenicity and Management; Omkar, Ed.; Springer Nature Singapore: Singapore, 2024; ISBN 978-981-97-4162-5. [Google Scholar]
- Lin, H.-H.; Li, Z.-T.; Tzeng, H.-Y.; Chang, C.; Dai, S.-M. Correlation between Pyrethroid Knockdown Resistance and Mutation Frequency of Voltage-Gated Sodium Channel and Its Application in Aedes Aegypti Management. Pesticide Biochemistry and Physiology 2024, 198, 105710. [Google Scholar] [CrossRef]
- Mohammadi, H.; Ghassemi-Barghi, N.; Malakshah, O.; Ashari, S. Pyrethroid Exposure and Neurotoxicity: A Mechanistic Approach. Archives of Industrial Hygiene and Toxicology 2019, 70, 74–89. [Google Scholar] [CrossRef]
- Sabarwal, A.; Kumar, K.; Singh, R.P. Hazardous Effects of Chemical Pesticides on Human Health–Cancer and Other Associated Disorders. Environmental Toxicology and Pharmacology 2018, 63, 103–114. [Google Scholar] [CrossRef]
- Erb, M. Plant Defenses against Herbivory: Closing the Fitness Gap. Trends in Plant Science 2018, 23, 187–194. [Google Scholar] [CrossRef]
- Inocente, E.A.; Nguyen, B.; Manwill, P.K.; Benatrehina, A.; Kweka, E.; Wu, S.; Cheng, X.; Rakotondraibe, L.H.; Piermarini, P.M. Insecticidal and Antifeedant Activities of Malagasy Medicinal Plant (Cinnamosma Sp.) Extracts and Drimane-Type Sesquiterpenes against Aedes Aegypti Mosquitoes. Insects 2019, 10, 373. [Google Scholar] [CrossRef]
- Lopez, A.D.; Whyms, S.; Luker, H.A.; Galvan, C.J.; Holguin, F.O.; Hansen, I.A. Repellency of Essential Oils and Plant-Derived Compounds Against Aedes Aegypti Mosquitoes. Insects 2025, 16, 51. [Google Scholar] [CrossRef]
- Mahanta, S.; Khanikor, B. Mosquitocidal Activity of Twenty-Eight Plant Essential Oils and Their Binary Mixtures against Culex Quinquefasciatus, (Diptera: Culicidae). Heliyon 2021, 7, e06128. [Google Scholar] [CrossRef]
- Martin, J.S.; Martin, M.M.; Bernays, E.A. Failure of Tannic Acid to Inhibit Digestion or Reduce Digestibility of Plant Protein in Gut Fluids of Insect Herbivores: Implications for Theories of Plant Defense. J Chem Ecol 1987, 13, 605–621. [Google Scholar] [CrossRef]
- Dobreva, A.; Nedeltcheva-Antonova, D.; Nenov, N.; Getchovska, K.; Antonov, L. Subcritical Extracts from Major Species of Oil-Bearing Roses—A Comparative Chemical Profiling. Molecules 2021, 26, 4991. [Google Scholar] [CrossRef]
- Antonova-Nedeltcheva, D.; Dobreva, A.; Gechovska, K.; Antonov, L. Volatile Compounds Profiling of Fresh R. Alba L. Blossom by Headspace—Solid Phase Microextraction and Gas Chromatography. Molecules 2025, 30, 3102. [Google Scholar] [CrossRef] [PubMed]
- Akçura, S.; Çakmakçi, R.; Ürüşan, Z. Changes in the Essential Oil Content and Composition of Pelargonium Graveolens l’hér with Different Drying Methods. Grasas aceites 2023, 74, e497. [Google Scholar] [CrossRef]
- Itawani; Hayati, R.; Munawar, A. Physicochemical Analysis Using Gas Chromatogrphy Mass Spectrophotometer (GCMS) on the Quality of Citronella Oil (Cymbopogon Nardus L.). IOP Conf. Ser.: Earth Environ. Sci. 2024, 1297, 012011. [Google Scholar] [CrossRef]
- Chuaycharoensuk, T.; Manguin, S.; Duvallet, G.; Chareonviriyaphap, T. Assessment of Geraniol-Incorporated Polymers to Control Aedes Albopictus (Diptera: Culicidae). Parasite 2012, 19, 427–432. [Google Scholar] [CrossRef]
- Huong, L.T.; Dai, D.N.; Thin, D.B.; Hung, N.H.; Thinh, B.B. Essential Oils of Distichochlamys Benenica: Chemical Constituents, Mosquito Larvicidal and Antimicrobial Activities. Natural Product Communications 2023, 18, 1934578X231193541. [Google Scholar] [CrossRef]
- Müller, G.C.; Junnila, A.; Kravchenko, V.D.; Revay, E.E.; Butler, J.; Schlein, Y. Indoor Protection Against Mosquito and Sand Fly Bites: A Comparison Between Citronella, Linalool, and Geraniol Candles. Journal of the American Mosquito Control Association 2008, 24, 150–153. [Google Scholar] [CrossRef] [PubMed]
- Müller, G.C.; Junnila, A.; Butler, J.; Kravchenko, V.D.; Revay, E.E.; Weiss, R.W.; Schlein, Y. Efficacy of the Botanical Repellents Geraniol, Linalool, and Citronella against Mosquitoes. Journal of Vector Ecology 2009, 34, 2–8. [Google Scholar] [CrossRef]
- Liu, X.C.; Dong, H.W.; Zhou, L.; Du, S.S.; Liu, Z.L. Essential Oil Composition and Larvicidal Activity of Toddalia Asiatica Roots against the Mosquito Aedes Albopictus (Diptera: Culicidae). Parasitol Res 2013, 112, 1197–1203. [Google Scholar] [CrossRef]
- Benelli, G.; Pavela, R.; Giordani, C.; Casettari, L.; Curzi, G.; Cappellacci, L.; Petrelli, R.; Maggi, F. Acute and Sub-Lethal Toxicity of Eight Essential Oils of Commercial Interest against the Filariasis Mosquito Culex Quinquefasciatus and the Housefly Musca Domestica. Industrial Crops and Products 2018, 112, 668–680. [Google Scholar] [CrossRef]
- Moungthipmalai, T.; Puwanard, C.; Aungtikun, J.; Sittichok, S.; Soonwera, M. Ovicidal Toxicity of Plant Essential Oils and Their Major Constituents against Two Mosquito Vectors and Their Non-Target Aquatic Predators. Sci Rep 2023, 13, 2119. [Google Scholar] [CrossRef] [PubMed]
- Tabari, M.A.; Youssefi, M.R.; Esfandiari, A.; Benelli, G. Toxicity of β-Citronellol, Geraniol and Linalool from Pelargonium Roseum Essential Oil against the West Nile and Filariasis Vector Culex Pipiens (Diptera: Culicidae). Research in Veterinary Science 2017, 114, 36–40. [Google Scholar] [CrossRef]
- Dehghankar, M.; Maleki-Ravasan, N.; Tahghighi, A.; Karimian, F.; Karami, M. Bioactivities of Rose-Scented Geranium Nanoemulsions against the Larvae of Anopheles Stephensi and Their Gut Bacteria. PLoS ONE 2021, 16, e0246470. [Google Scholar] [CrossRef]
- Elghonemy, M.M.; Ayoob, F.; Abdel-Ghany, H.S.M.; Kassem, A.M.; El-Gendy, A.E.G.; Abdel-Shafy, S.; Abd-ElGawad, A.M.; Imagawa, H.; Elshamy, A.I. Larvicidal Effects of Cymbopogon Commutatus Essential Oil and Its Based Nanoformulations on Culex Pipiens: Chemical Profiling and Physicochemical Characterization. Chemistry & Biodiversity 2025, 22, e202403276. [Google Scholar] [CrossRef]
- Kaufman, P.E.; Mann, R.S.; Butler, J.F. Evaluation of Semiochemical Toxicity to Aedes Aegypti, Ae. Albopictus and Anopheles Quadrimaculatus (Diptera: Culicidae). Pest Management Science 2010, 66, 497–504. [Google Scholar] [CrossRef] [PubMed]
- Reeves, W.K.; Miller, M.M. Aqueous 2% Geraniol as a Mosquito Repellent Failed against Aedes Aegypti on Ponies. Journal of the American Mosquito Control Association 2010, 26, 340–341. [Google Scholar] [CrossRef]
- Setlur, A.S.; K, C.; Pandey, S.; Sarkar, M.; Niranjan, V. Comprehensive Molecular Interaction Studies to Construe the Repellent/Kill Activity of Geraniol During Binding Event Against Aedes Aegypti Proteins. Mol Biotechnol 2023, 65, 726–740. [Google Scholar] [CrossRef]
- Du, Y.; Nomura, Y.; Zhorov, B.; Dong, K. Sodium Channel Mutations and Pyrethroid Resistance in Aedes Aegypti. Insects 2016, 7, 60. [Google Scholar] [CrossRef] [PubMed]
- Naw, H.; Võ, T.C.; Lê, H.G.; Kang, J.-M.; Mya, Y.Y.; Myint, M.K.; Kim, T.-S.; Shin, H.-J.; Na, B.-K. Knockdown Resistance Mutations in the Voltage-Gated Sodium Channel of Aedes Aegypti (Diptera: Culicidae) in Myanmar. Insects 2022, 13, 322. [Google Scholar] [CrossRef]
- Ponce-García, G.; Del Río-Galvan, S.; Barrera, R.; Saavedra-Rodriguez, K.; Villanueva-Segura, K.; Felix, G.; Amador, M.; Flores, A.E. Knockdown Resistance Mutations in Aedes Aegypti (Diptera: Culicidae) From Puerto Rico. J Med Entomol 2016, 53, 1410–1414. [Google Scholar] [CrossRef]
- Martínez Rodríguez, E.J.; Phelan, P.L.; Canas, L.; Acosta, N.; Rakotondraibe, H.L.; Piermarini, P.M. Larvicidal Activity of Hemp Extracts and Cannabidiol against the Yellow Fever Mosquito Aedes Aegypti. Insects 2024, 15, 517. [Google Scholar] [CrossRef] [PubMed]
- Inocente, E.A.; Shaya, M.; Acosta, N.; Rakotondraibe, L.H.; Piermarini, P.M. A Natural Agonist of Mosquito TRPA1 from the Medicinal Plant Cinnamosma Fragrans That Is Toxic, Antifeedant, and Repellent to the Yellow Fever Mosquito Aedes Aegypti. PLoS Negl Trop Dis 2018, 12, e0006265. [Google Scholar] [CrossRef]
- Swale, D.R.; Engers, D.W.; Bollinger, S.R.; Gross, A.; Inocente, E.A.; Days, E.; Kanga, F.; Johnson, R.M.; Yang, L.; Bloomquist, J.R.; et al. An Insecticide Resistance-Breaking Mosquitocide Targeting Inward Rectifier Potassium Channels in Vectors of Zika Virus and Malaria. Sci Rep 2016, 6, 36954. [Google Scholar] [CrossRef]
- Calkins, T.L.; Piermarini, P.M. Pharmacological and Genetic Evidence for Gap Junctions as Potential New Insecticide Targets in the Yellow Fever Mosquito, Aedes Aegypti. PLoS ONE 2015, 10, e0137084. [Google Scholar] [CrossRef]
- Jiang, S.; Yang, L.; Bloomquist, J.R. High-throughput Screening Method for Evaluating Spatial Repellency and Vapour Toxicity to Mosquitoes. Medical Vet Entomology 2019, 33, 388–396. [Google Scholar] [CrossRef]
- Yang, L.; Norris, E.J.; Jiang, S.; Bernier, U.R.; Linthicum, K.J.; Bloomquist, J.R. Reduced Effectiveness of Repellents in a Pyrethroid-resistant Strain of Aedes Aegypti (Diptera: Culicidae) and Its Correlation with Olfactory Sensitivity. Pest Management Science 2020, 76, 118–124. [Google Scholar] [CrossRef] [PubMed]
- Estep, A.S.; Sanscrainte, N.D.; Waits, C.M.; Louton, J.E.; Becnel, J.J. Resistance Status and Resistance Mechanisms in a Strain of Aedes Aegypti (Diptera: Culicidae) From Puerto Rico. Journal of Medical Entomology 2017, 54, 1643–1648. [Google Scholar] [CrossRef] [PubMed]
- López, M.D.; Pascual-Villalobos, M.J. Mode of Inhibition of Acetylcholinesterase by Monoterpenoids and Implications for Pest Control. Industrial Crops and Products 2010, 31, 284–288. [Google Scholar] [CrossRef]
- Jankowska, M.; Rogalska, J.; Wyszkowska, J.; Stankiewicz, M. Molecular Targets for Components of Essential Oils in the Insect Nervous System—A Review. Molecules 2017, 23, 34. [Google Scholar] [CrossRef] [PubMed]
- Liu, N. Insecticide Resistance in Mosquitoes: Impact, Mechanisms, and Research Directions. Annu. Rev. Entomol. 2015, 60, 537–559. [Google Scholar] [CrossRef]
- Bharadwaj, N.; Sharma, R.; Subramanian, M.; Ragini, G.; Nagarajan, S.A.; Rahi, M. Omics Approaches in Understanding Insecticide Resistance in Mosquito Vectors. IJMS 2025, 26, 1854. [Google Scholar] [CrossRef]
- Liu, F.; Wang, Q.; Xu, P.; Andreazza, F.; Valbon, W.R.; Bandason, E.; Chen, M.; Yan, R.; Feng, B.; Smith, L.B.; et al. A Dual-Target Molecular Mechanism of Pyrethrum Repellency against Mosquitoes. Nat Commun 2021, 12, 2553. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



