Submitted:
25 February 2026
Posted:
27 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
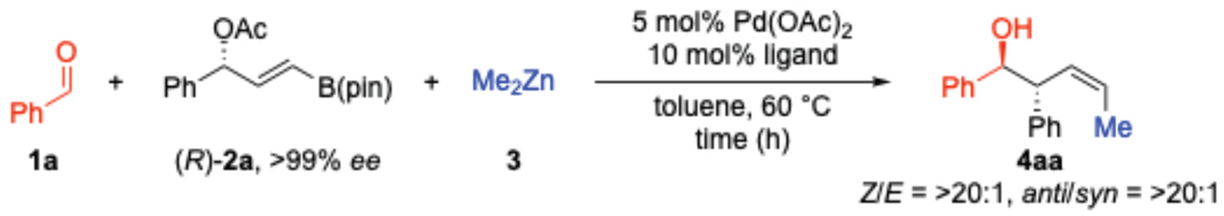
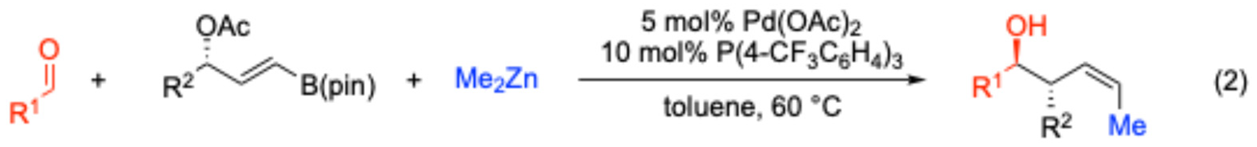
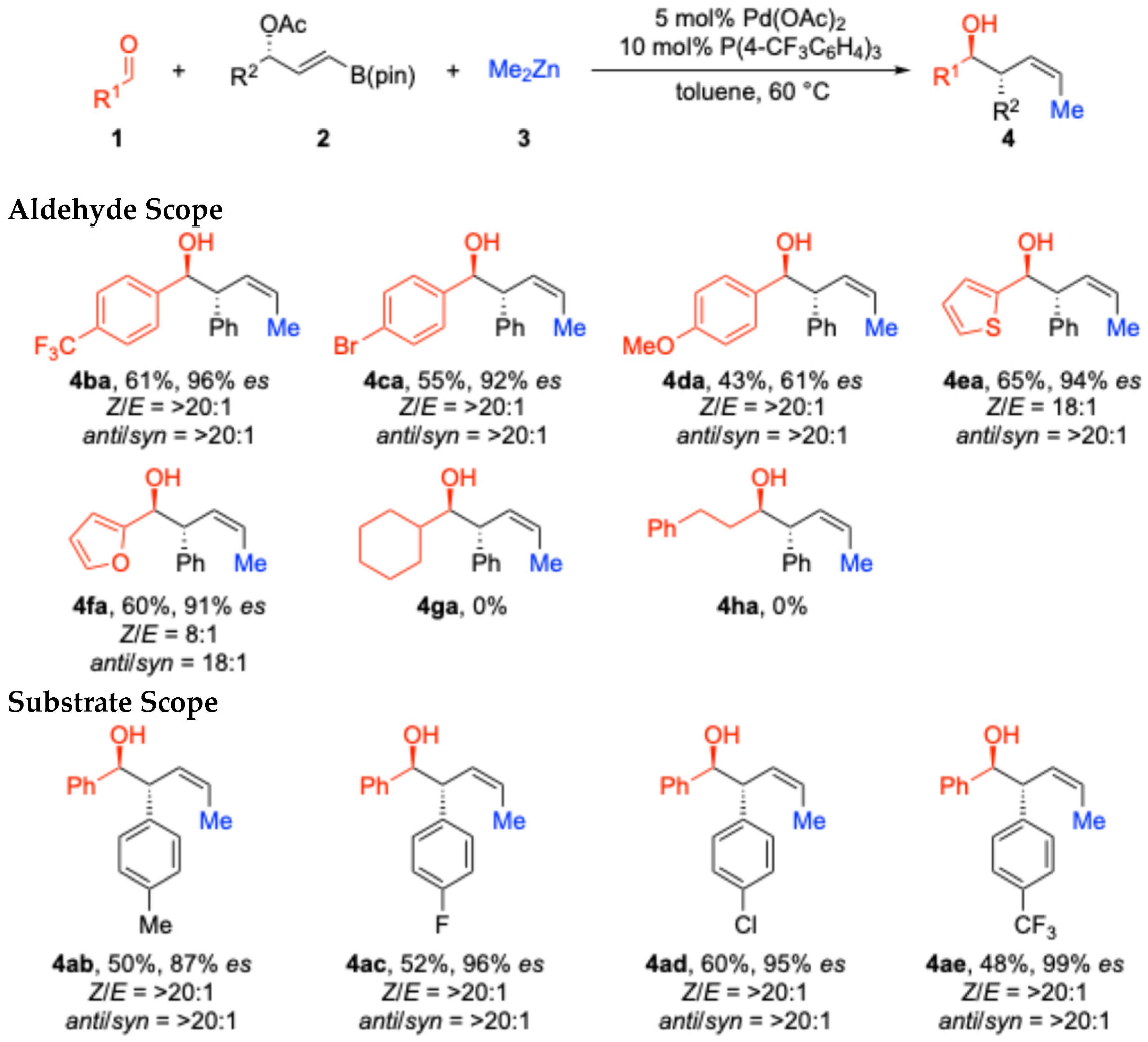
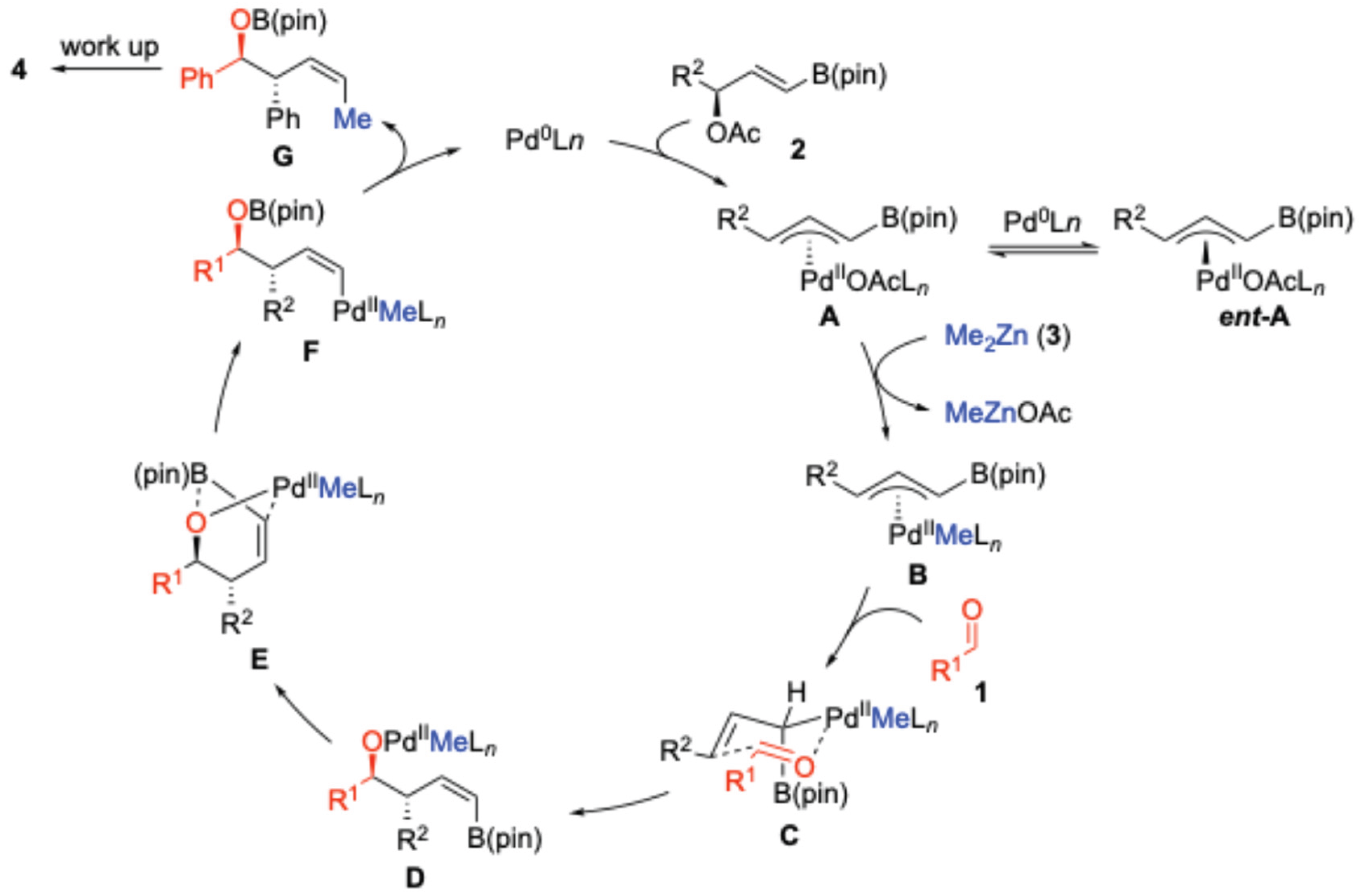
2. Results and Discussion
3. Materials and Methods
3.1. General Method for Arylation Process
Characterization Data
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yeung, K.; Paterson, I. Advances in the Total Synthesis of Biologically Important Marine Macrolides; 2005; pp. 4237–4313. [Google Scholar] [CrossRef]
- Florence, G.J.; Gardner, N.M.; Paterson, I. Development of Practical Syntheses of the Marine Anticancer Agents Discodermolide and Dictyostatin. Nat. Prod. Rep. 2008, 25, 342–375. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Verboom, K.L.; Krische, M.J. Catalytic Enantioselective C–C Coupling of Alcohols for Polyketide Total Synthesis beyond Chiral Auxiliaries and Premetalated Reagents. Chem. Rev. 2024, 124, 13715–13735. [Google Scholar] [CrossRef] [PubMed]
- Denmark, S.E.; Fu, J. Catalytic Enantioselective Addition of Allylic Organometallic Reagents to Aldehydes and Ketones. Chem. Rev. 2003, 103, 2763–2794. [Google Scholar] [CrossRef] [PubMed]
- Yus, M.; Gonz, C.; Foubelo, F. Catalytic Enantioselective Allylation of Carbonyl Compounds and Imines; 2011; pp. 7774–7854. [Google Scholar] [CrossRef]
- Yus, M.; González-Gómez, J.C.; Foubelo, F. Catalytic Enantioselective Allylation of Carbonyl Compounds and Imines. Chem. Rev. 2011, 111, 7774–7854. [Google Scholar] [CrossRef] [PubMed]
- Sedgwick, D.; Grayson, M.; Fustero, S.; Barrio, P. Recent Developments and Applications of the Chiral Brønsted Acid Catalyzed Allylboration of Carbonyl Compounds. Synthesis 2018, 50, 1935–1957. [Google Scholar] [CrossRef]
- Spielmann, K.; Niel, G.; de Figueiredo, R.M.; Campagne, J.-M. Catalytic Nucleophilic 'Umpoled' π-Allyl Reagents. Chem. Soc. Rev. 2018, 47, 1159–1173. [Google Scholar] [CrossRef] [PubMed]
- Holmes, M.; Schwartz, L.A.; Krische, M.J. Intermolecular Metal-Catalyzed Reductive Coupling of Dienes, Allenes, and Enynes with Carbonyl Compounds and Imines. Chem. Rev. 2018, 118, 6026–6052. [Google Scholar] [CrossRef] [PubMed]
- Lu, W.; Chong, Q.; Meng, F. Cu-Catalyzed Enantioselective Reductive Coupling of 1,3-Dienes and Carboxylic Acid Anhydrides. Asian J. Org. Chem. 2023, 12, e202300436. [Google Scholar] [CrossRef]
- Mitsunuma, H.; Kanai, M. Catalytic Asymmetric Allylation of Aldehydes with Alkenes through Allylic C(sp3)–H Functionalization Mediated by Organophotoredox and Chiral Chromium Hybrid Catalysis. Chem. Sci. 2019, 10, 3459–3465. [Google Scholar] [CrossRef] [PubMed]
- Xie, H.; Breit, B. Organophotoredox/Ni-Cocatalyzed Allylation of Allenes: Regio- and Diastereoselective Access to Homoallylic Alcohols. ACS Catal. 2022, 12, 3249–3255. [Google Scholar] [CrossRef]
- Hesse, M.J.; Essafi, S.; Watson, C.G.; Harvey, J.N.; Hirst, D.; Willis, C.L.; Aggarwal, V.K. Highly Selective Allylborations of Aldehydes Using α,α-Disubstituted Allylic Pinacol Boronic Esters. Angew. Chem. Int. Ed. 2014, 53, 6145–6149. [Google Scholar] [CrossRef] [PubMed]
- Tao, Z.L.; Adili, A.; Shen, H.C.; Han, Z.Y.; Gong, L.Z. Catalytic Enantioselective Assembly of Homoallylic Alcohols from Dienes, Aryldiazonium Salts, and Aldehydes. Angew. Chem. Int. Ed. 2016, 55, 4322–4326. [Google Scholar] [CrossRef] [PubMed]
- Shen, H.C.; Wang, P.S.; Tao, Z.L.; Han, Z.Y.; Gong, L.Z. An Enantioselective Multicomponent Carbonyl Allylation of Aldehydes with Dienes and Alkynyl Bromides Enabled by Chiral Palladium Phosphate. Adv. Synth. Catal. 2017, 359, 2383–2389. [Google Scholar] [CrossRef]
- Lv, Y.-F.; Shi, Z.; Liu, G.; Shen, H.; Wang, Z. Stereoselective Synthesis of Chiral Homoallylic Alcohols with Functionalized Alkenes via Radical-Involved Triple Catalysis. ACS Catal. 2025, 15, 20157–20169. [Google Scholar] [CrossRef]
- Horino, Y.; Aimono, A.; Abe, H. Pd-Catalyzed Three-Component Reaction of 3-(Pinacolatoboryl)Ally Acetates, Aldehydes, and Organoboranes: A New Entry to Stereoselective Synthesis of (Z)-anti-Homoallylic Alcohols. Org. Lett. 2015, 17, 2824–2827. [Google Scholar] [CrossRef] [PubMed]
- Schmidtmann, E.S.; Oestreich, M. Enantiospecific Synthesis and Allylation of All-Carbon-Substituted α-Chiral Allylic Stannanes. Angew. Chem. Int. Ed. 2009, 48, 4634–4638. [Google Scholar] [CrossRef] [PubMed]
- Katsuki, T.; Sharpless, K.B. The First Practical Method for Asymmetric Epoxidation. J. Am. Chem. Soc. 1980, 102, 5974–5976. [Google Scholar] [CrossRef]
- Horino, Y.; Ishibashi, M.; Sakamoto, J.; Murakami, M.; Korenaga, T. Palladium-Catalyzed Diastereoselective Synthesis of (Z)-Conjugated Enynyl Homoallylic Alcohols. Adv. Synth. Catal. 2021, 363, 3592–3599. [Google Scholar] [CrossRef]
- Oono, M.; Yamada, A.; Kimura, M.; Kanomata, K.; Akai, S. Lipase-Palladium Co-Catalyzed Dynamic Kinetic Resolution of Racemic Allylic Esters. Chem. Eur. J. 2025, 2, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Cooze, C.; Dada, R.; Lundgren, R.J. Direct Formic Acid Mediated Z-Selective Reductive Coupling of Dienes and Aldehydes. Angew. Chem. Int. Ed. 2019, 58, 12246–12251. [Google Scholar] [CrossRef] [PubMed]
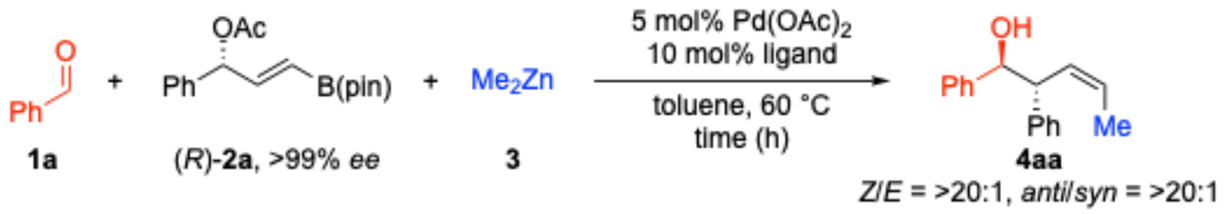 | |||||
| Entry | Ligand | Time (h) | 4aa (%) | ee (%)2 | es (%)3 |
| 1 | PPh3 | 4 | 44 | 88 | 88 |
| 2 | P(4-MeOC6H4)3 | 6 | 43 | 84 | 84 |
| 3 | P(4-CF3C6H4)3 | overnight | 44 | 88 | 88 |
| 44 | PPh3 | 4 | 56 | 93 | 93 |
| 54 | P(4-MeOC6H4)3 | 4 | 57 | 93 | 93 |
| 64 | P(4-CF3C6H4)3 | overnight | 60 | 93 | 93 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.





