Submitted:
25 February 2026
Posted:
26 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
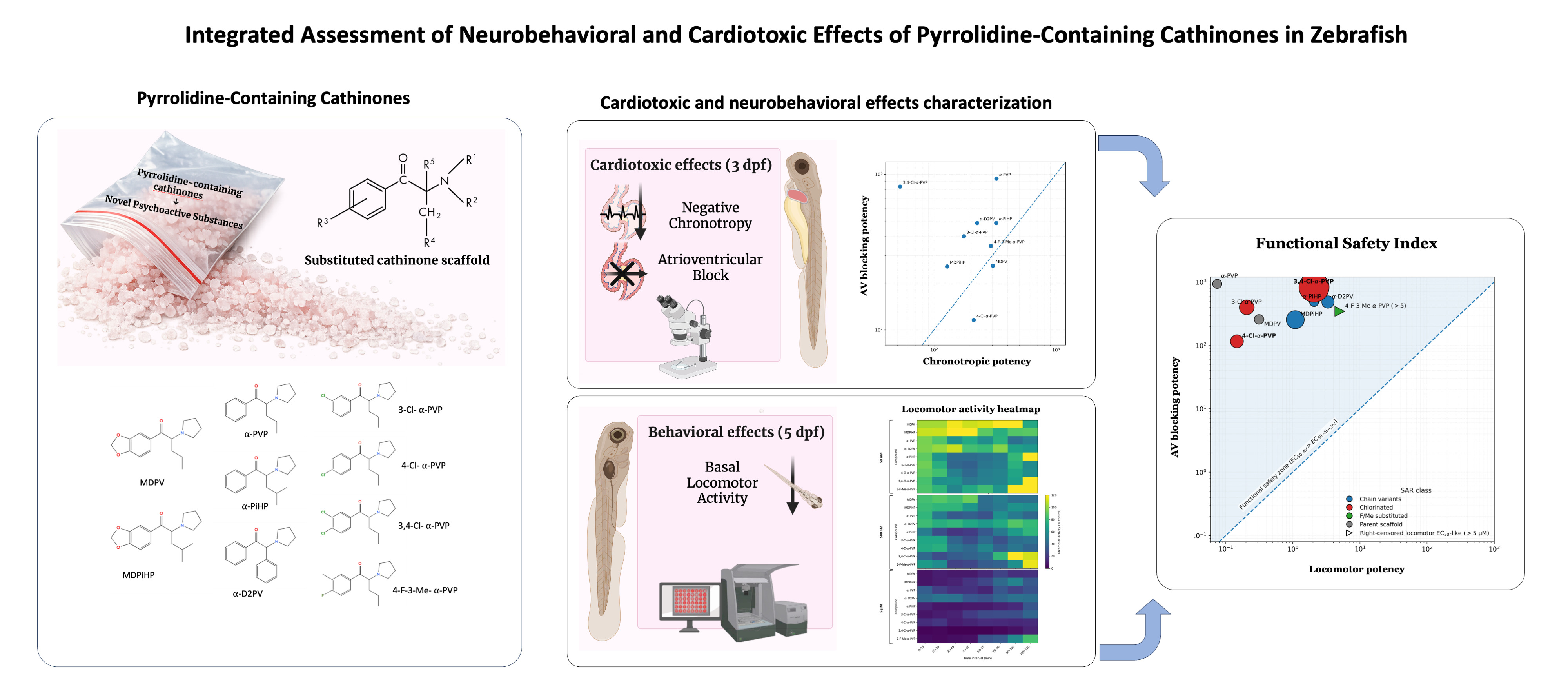
2. Results
2.1. Effects of Pyrrolidinophenone Derivatives on Atrial Chronotropy
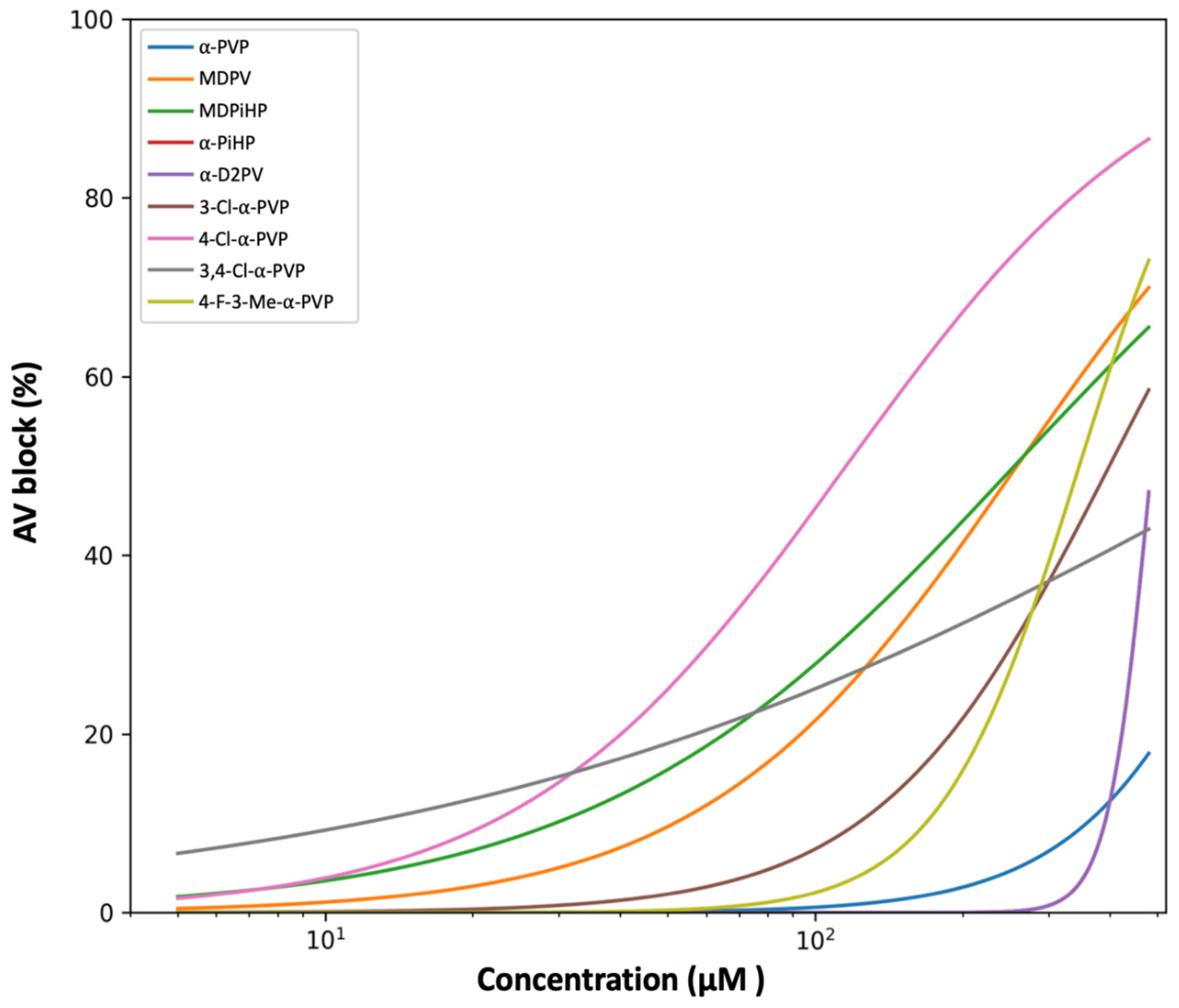
2.2. Effects of Pyrrolidinophenone Derivatives on Atrioventricular Conduction
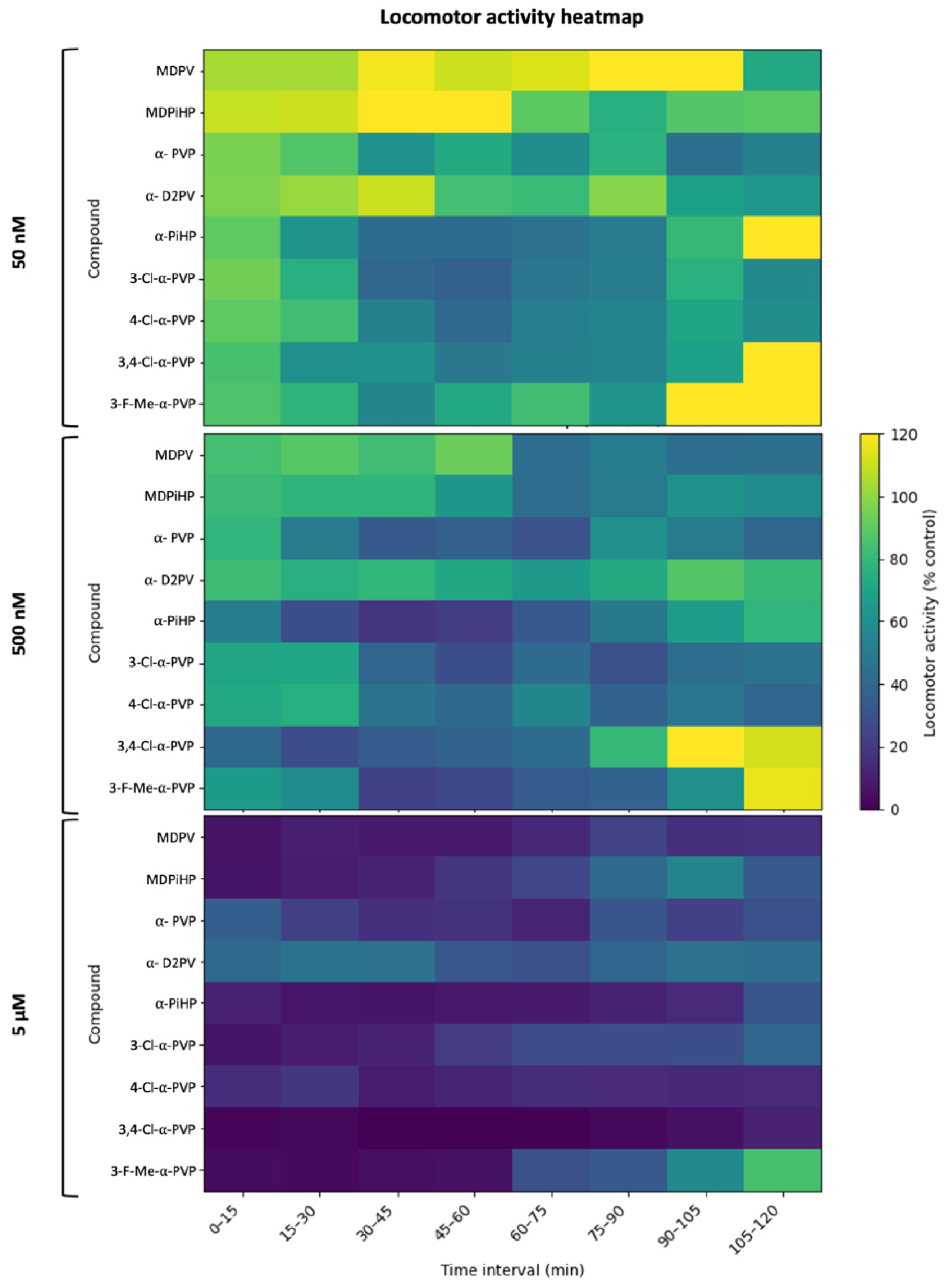
2.3. Effects of Pyrrolidinophenone Derivatives on Locomotor Activity
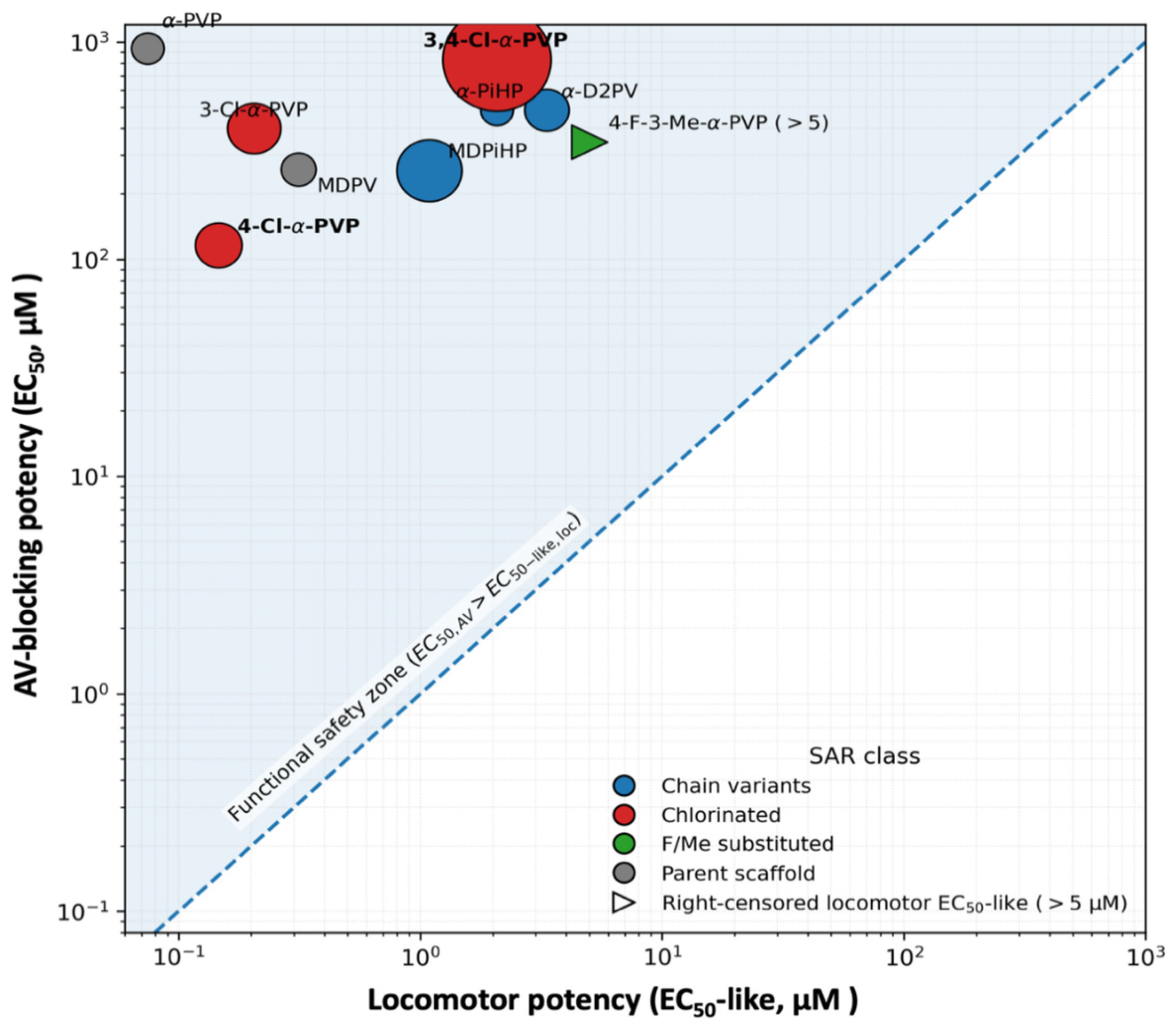
2.4. Relationship Between Atrioventricular Block and Locomotor Inhibition
3. Discussion
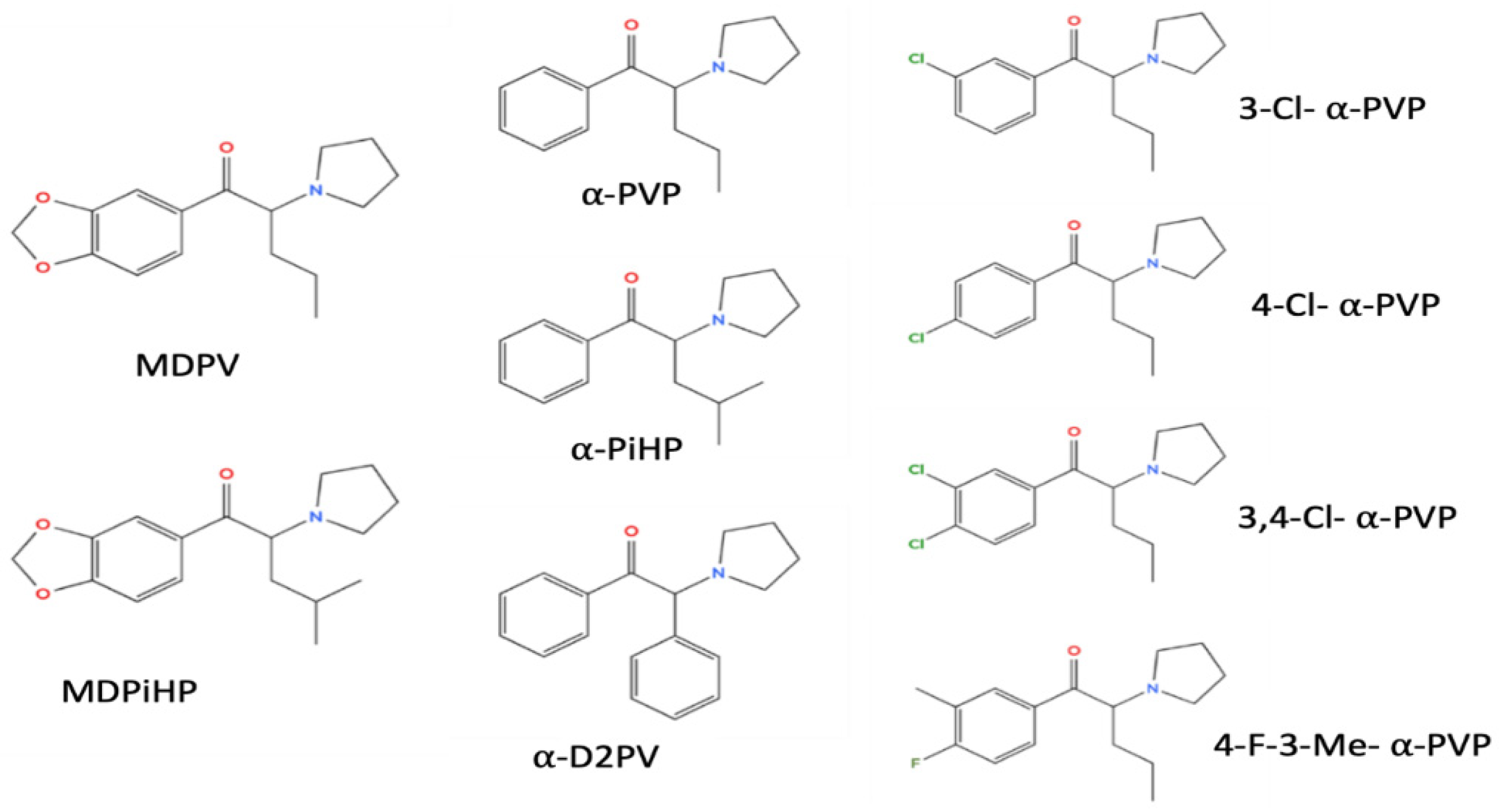
3.1. Structural Determinants of Atrial Chronotropic Effects
3.2. Structural Determinants of AV Conduction Block
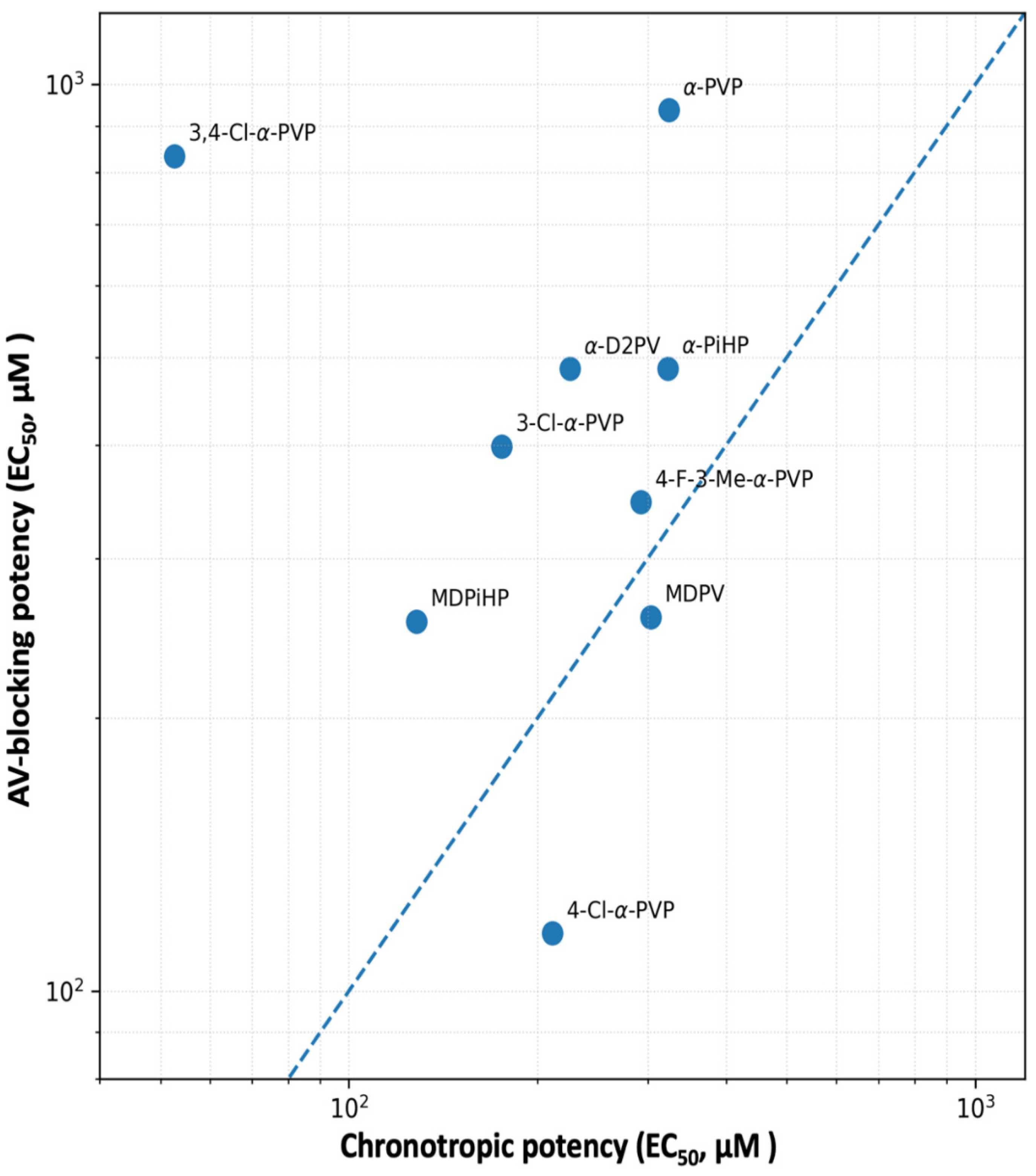
3.3. Functional Dissociation Between Atrial Chronotropy and AV Block
3.4. Implications for Cardiotoxicity and Structure-Based Risk Assessment
3.5. Integration with Locomotor Endpoints: Time-Course, Potency, and Functional Safety Margins
3.6. Pharmacological Alignment: From Molecular DAT Inhibition to In Vivo Locomotor Impairment
3.7. Conclusions
4. Materials and Methods
4.1. Zebrafish Maintenance and Embryo Collection
4.2. Compounds and Exposure Solutions
4.3. Cardiotoxicity Assessment
4.4. Behavioral Assessment
4.5. Safety Index Calculation
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- United Nations Office on Drugs and Crime (UNODC). (n.d.-a). What are NPS? UNODC Early Warning Advisory on New Psychoactive Substances. Accessed 13 February 2026. https://www.unodc.org/LSS/Page/NPS.
- United Nations Office on Drugs and Crime (UNODC). (n.d.-b). Current NPS threats (título exacto de la otra). UNODC Early Warning Advisory on New Psychoactive Substances. Accessed 13 February 2026.
- https://sway.cloud.microsoft/SBdzGtMZGnxpGRRA?ref=Link.
- Oliver, C.F.; Palamar, J.J.; Salomone, A.; Simmons, S.J.; Philogene-Khalid, H.L.; Stokes-McCloskey, N.; Rawls, S.M. Synthetic cathinone adulteration of illegal drugs. Psychopharmacology (Berl). 2019, 236, 869–879. [CrossRef]
- Chen, S.; Zhou, W.; Lai, M. Synthetic Cathinones: Epidemiology, Toxicity, Potential for Abuse, and Current Public Health Perspective. Brain Sci. 2024, 14, 334. [CrossRef]
- Zaami, S.; Giorgetti, R.; Pichini, S.; Pantano, F.; Marinelli, E.; Busardò, F.P. Synthetic cathinones related fatalities: An update. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 268–274. [CrossRef]
- European Union Drugs Agency. New psychoactive substances – the current situation in Europe (European Drug Report 2024);
- European Union Drugs Agency. New psychoactive substances – the current situation in Europe (European Drug Report 2025);
- Nadal-Gratacós, N.; Pazos, M.D.; Pubill, D.; Camarasa, J.; Escubedo, E.; Berzosa, X.; López-Arnau, R. Structure–Activity Relationship of Synthetic Cathinones: An Updated Review. ACS Pharmacol. Transl. Sci. 2024, 7, 2588–2603. [CrossRef]
- Kolaczynska, K.E.; Thomann, J.; Hoener, M.C.; Liechti, M.E. The Pharmacological Profile of Second Generation Pyrovalerone Cathinones and Related Cathinone Derivative. Int. J. Mol. Sci. 2021, 22, 8277. [CrossRef]
- Nadal-Gratacós, N.; Mata, S.; Puigseslloses, P.; De Macedo, M.; Lardeux, V.; Pain, S.; Wang, F.-H.; Källsten, L.; Pubill, D.; Berzosa, X.; et al. Unveiling the potential abuse liability of α-D2PV: A novel α-carbon phenyl-substituted synthetic cathinone. Neuropharmacology 2025, 272, 110425. [CrossRef]
- Nadal-Gratacós, N.; Lleixà, E.; Gibert-Serramià, M.; Estrada-Tejedor, R.; Berzosa, X.; Batllori, X.; Pubill, D.; Camarasa, J.; Escubedo, E.; López-Arnau, R. Neuropsychopharmacology of Emerging Drugs of Abuse: meta- and para-Halogen-Ring-Substituted α-PVP (“flakka”) Derivatives. Int. J. Mol. Sci. 2022, 23, 2226. [CrossRef]
- Kriikku, P.; Ojanperä, I. Findings of synthetic cathinones in post-mortem toxicology. Forensic Sci. Int. 2024, 365, 112297. [CrossRef]
- Pieprzyca, E.; Skowronek, R.; Czekaj, P. Toxicological Analysis of Intoxications with Synthetic Cathinones. J. Anal. Toxicol. 2022, 46, 705–711. [CrossRef]
- Riley, A.L.; Nelson, K.H.; To, P.; López-Arnau, R.; Xu, P.; Wang, D.; Wang, Y.; Shen, H.; Kuhn, D.M.; Angoa-Perez, M.; et al. Abuse potential and toxicity of the synthetic cathinones (i.e., “Bath salts”). Neurosci. Biobehav. Rev. 2020, 110, 150–173. [CrossRef]
- Kolanos, R.; Sakloth, F.; Jain, A.D.; Partilla, J.S.; Baumann, M.H.; Glennon, R.A. Structural Modification of the Designer Stimulant α-Pyrrolidinovalerophenone (α-PVP) Influences Potency at Dopamine Transporters. 2015. [CrossRef]
- Zawilska, J.B.; Wojcieszak, J. Designer cathinones — An emerging class of novel recreational drugs. Forensic Sci. Int. 2013, 231, 42–53. [CrossRef]
- Brueckner, I.; Welter-luedeke, J.; Zangl, A.; Graw, M.; Paul, L.D. α-Pyrrolidinohexanophenone (α-PHP) vs α-pyrrolidinoisohexanophenone (α-PiHP): A toxicological investigation about plasma concentrations and behavior in forensic routine cases. 2024, 299–313.
- Dinis, P.; Franco, J.; Margalho, C. α-Pyrrolidinohexanophenone (α-PHP) and α-Pyrrolidinoisohexanophenone (α-PiHP): A Review. Life 2024, 14, 429. [CrossRef]
- Aljabasini, O.; Tagkalidou, N.; Bedrossiantz, J.; Prats, E.; Arnau, R.L.; Raldua, D. Integrated Assessment of the Cardiotoxic and Neurobehavioral Effects of 3,4-Methylenedioxypyrovalerone (MDPV) in Zebrafish Embryos. Int. J. Mol. Sci. 2025, 27, 59. [CrossRef]
- Stainier, D.Y.R.; Fishman, M.C. The zebrafish as a model system to study cardiovascular development. Trends Cardiovasc. Med. 1994, 4, 207–212. [CrossRef]
- Bakkers, J. Zebrafish as a model to study cardiac development and human cardiac disease. Cardiovasc. Res. 2011, 91, 279–288. [CrossRef]
- Langheinrich, U.; Vacun, G.; Wagner, T. Zebrafish embryos express an orthologue of HERG and are sensitive toward a range of QT-prolonging drugs inducing severe arrhythmia. Toxicol. Appl. Pharmacol. 2003, 193, 370–382. [CrossRef]
- Milan, D.J.; Peterson, T.A.; Ruskin, J.N.; Peterson, R.T.; MacRae, C.A. Drugs that induce repolarization abnormalities cause bradycardia in zebrafish. Circulation 2003, 107, 1355–1358. [CrossRef]
- Teixidó, E.; Riera-Colomer, C.; Raldúa, D.; Pubill, D.; Escubedo, E.; Barenys, M.; López-Arnau, R. First-Generation Synthetic Cathinones Produce Arrhythmia in Zebrafish Eleutheroembryos: A New Approach Methodology for New Psychoactive Substances Cardiotoxicity Evaluation. Int. J. Mol. Sci. 2023, 24. [CrossRef]
- Milan, D.J.; MacRae, C.A. Zebrafish genetic models for arrhythmia. Prog. Biophys. Mol. Biol. 2008, 98, 301–308. [CrossRef]
- Pazos, M.D.; Berzosa, X.; Camarasa, J.; Pubill, D.; Escubedo, E.; Nadal-Gratacós, N.; López-Arnau, R. Psychostimulant and addictive effects of α-PiHP and MDPiHP, two novel second-generation synthetic cathinones. Neurosci. Appl. 2026, 5, 105536. [CrossRef]
- Haverinen, J.; Hassinen, M.; Korajoki, H.; Vornanen, M. Cardiac voltage-gated sodium channel expression and electrophysiological characterization of the sodium current in the zebra fi sh ( Danio rerio ) ventricle. Prog. Biophys. Mol. Biol. 2018, 138, 59–68. [CrossRef]
- Bowley, G.; Kugler, E.; Wilkinson, R.; Lawrie, A.; Eeden, F. Van; Chico, T.J.A.; Evans, P.C.; Noël, E.S.; Serbanovic-canic, J. Zebrafish as a tractable model of human cardiovascular disease. 2022, 900–917. [CrossRef]
- Gauvrit, S.; Bossaer, J.; Lee, J.; Collins, M.M. Modeling Human Cardiac Arrhythmias : Insights from Zebrafish. 2022.
- Vornanen, M.; Badr, A.; Haverinen, J. Cardiac arrhythmias in fish induced by natural and anthropogenic changes in environmental conditions. J. Exp. Biol. 2024, 227. [CrossRef]
- Zakaria, Z.Z.; Benslimane, F.M.; Nasrallah, G.K.; Shurbaji, S.; Younes, N.N.; Mraiche, F.; Da’As, S.I.; Yalcin, H.C. Using Zebrafish for Investigating the Molecular Mechanisms of Drug-Induced Cardiotoxicity. Biomed Res. Int. 2018, 2018. [CrossRef]
- Maciag, M.; Wnorowski, A.; Bednarz, K.; Plazinska, A. Evaluation of β-adrenergic ligands for development of pharmacological heart failure and transparency models in zebrafish. Toxicol. Appl. Pharmacol. 2022, 434, 115812. [CrossRef]
- Fink, M.; Callol-Massot, C.; Chu, A.; Ruiz-Lozano, P.; Belmonte, J.C.I.; Giles, W.; Bodmer, R.; Ocorr, K. A new method for detection and quantification of heartbeat parameters in Drosophila, zebrafish, and embryonic mouse hearts. Biotechniques 2009, 46, 101–113. [CrossRef]
- Nadal-Gratacós, N.; Lleixà, E.; Gibert-Serramià, M.; Estrada-Tejedor, R.; Berzosa, X.; Batllori, X.; Pubill, D.; Camarasa, J.; Escubedo, E.; López-Arnau, R. Neuropsychopharmacology of Emerging Drugs of Abuse: meta-and para-Halogen-Ring-Substituted α-PVP (“flakka”) Derivatives. Int. J. Mol. Sci. 2022, 23, 2226. [CrossRef]
- Glennon, R.A.; Young, R. Neurobiology of 3,4-methylenedioxypyrovalerone (MDPV) and α-pyrrolidinovalerophenone (α-PVP). Brain Res. Bull. 2016, 126, 111–126. [CrossRef]
- Dinis, P. α -Pyrrolidinoisohexanophenone ( α -PiHP ): A Review. 2024.
- Adamowicz, P.; Jurczyk, A.; Gil, D.; Szustowski, S. Case Report A case of intoxication with a new cathinone derivative α -PiHP – A presentation of concentrations in biological specimens. Leg. Med. 2020, 42, 101626. [CrossRef]
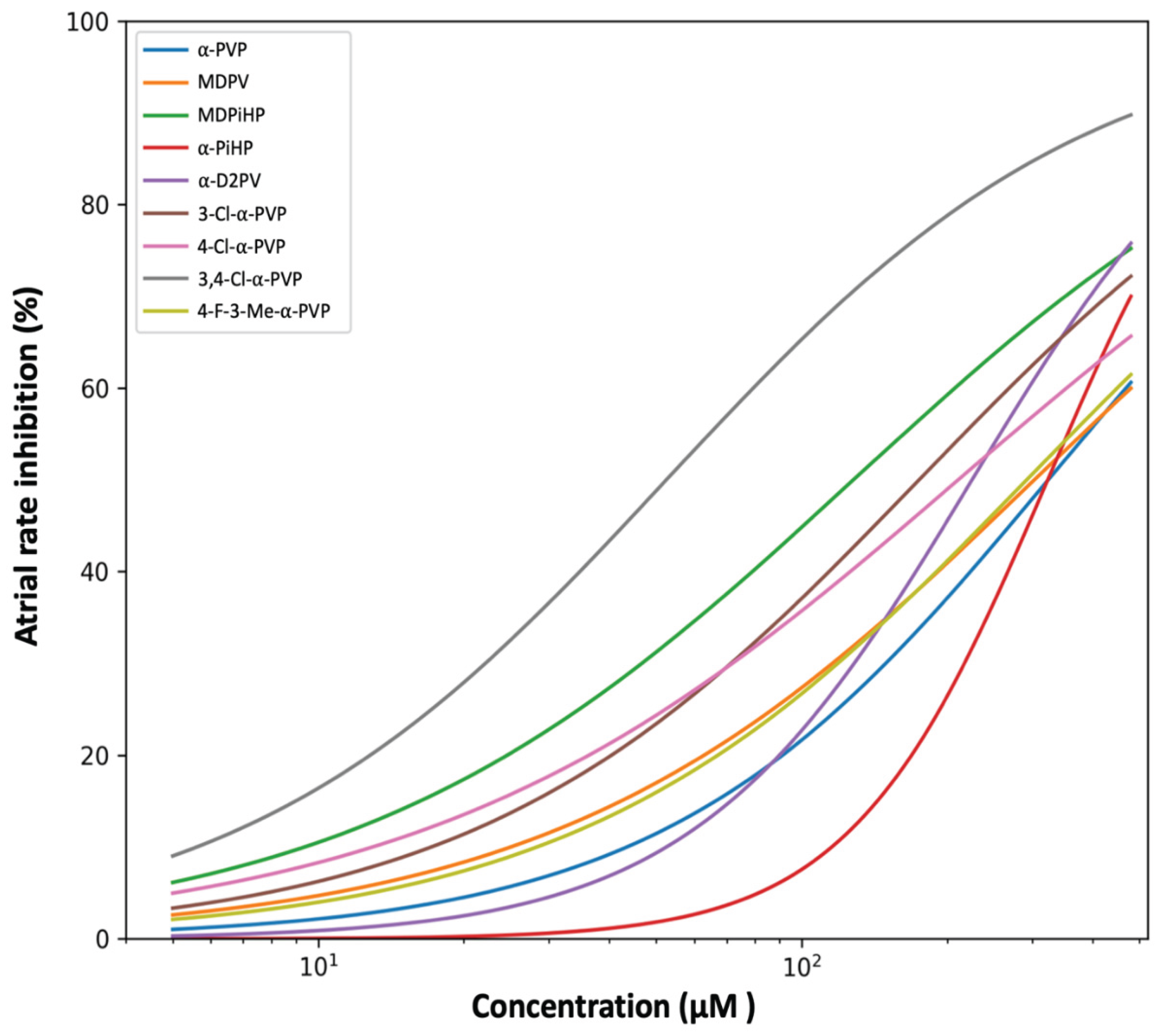
| Compound | EC50(µM) | 95% CI (EC50) | Hill slope | 95% CI (Hill) |
| 3,4-Cl-α-PVP | 52.6 | 46.3–60.4 | 0.98 | 0.89–1.08 |
| MDPiHP | 128.2 | 116.4–140.0 | 0.84 | 0.76–0.94 |
| 3-Cl-α-PVP | 175.2 | 156.4–197.6 | 0.95 | 0.85–1.09 |
| 4-Cl-α-PVP | 211.1 | 183.8–241.6 | 0.79 | 0.70–0.90 |
| α-D2PV | 225.4 | 208.2–244.7 | 1.51 | 1.36–1.68 |
| 4-F-3-Me-α-PVP | 292.5 | 257.1–331.5 | 0.94 | 0.84–1.08 |
| MDPV | 303.4 | 263.9–347.1 | 0.88 | 0.77–1.01 |
| α-PiHP | 323.0 | 302.9–342.4 | 2.14 | 1.84–2.56 |
| α-PVP | 323.9 | 284.3–375.0 | 1.09 | 0.95–1.25 |
| Compound | EC50(µM) | 95% CI (EC50) | Hill slope | 95% CI (Hill) |
| 4-Cl-α-PVP | 115.8 | 94.3–137.7 | 1.31 | 1.00–1.76 |
| MDPiHP | 255.6 | 212.7–311.6 | 1.02 | 0.80–1.30 |
| MDPV | 258.5 | 222.6–307.5 | 1.36 | 1.08–1.76 |
| 4-F-3-Me-α-PVP | 346.1 | 298.4–387.5 | 3.04 | 2.30–4.94 |
| 3-Cl-α-PVP | 398.9 | 327.8–526.3 | 1.86 | 1.39–2.64 |
| α-D2PV | 485.6 | 446.6–566.5 | 10.00 | 9.92–10.00 |
| α-PiHP | 486.0 | 446.6–567.5 | 10.00 | 9.91–10.00 |
| 3,4-Cl-α-PVP | 833.5 | 350.3–6300.2 | 0.52 | 0.31–0.88 |
| α-PVP | 937.0 | 463.0–10727.9 | 2.29 | 0.87–10.00 |
| Compound | EC50 Atrial (µM) | EC50 AV block (µM) | Locomotor EC50-like (µM) | Safety Index |
| MDPV | 303.4 | 258.5 | 0.3125 | 827.11 |
| MDPiHP | 128.2 | 255.6 | 1.088 | 235.02 |
| α-PVP | 323.9 | 937.0 | 0.07449 | 12578.52 |
| α-D2PV | 225.4 | 485.6 | 3.33 | 145.81 |
| α-PiHP | 323.0 | 486.0 | 2.069 | 234.95 |
| 3-Cl-α-PVP | 175.2 | 398.9 | 0.2049 | 1946.86 |
| 4-Cl-α-PVP | 211.1 | 115.8 | 0.1459 | 793.49 |
| 3,4-Cl-α-PVP | 52.6 | 833.5 | 2.068 | 402.98 |
| 4-F-3-Me-α-PVP | 292.5 | 346.1 | >5 | <69.22 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




