Submitted:
25 February 2026
Posted:
26 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
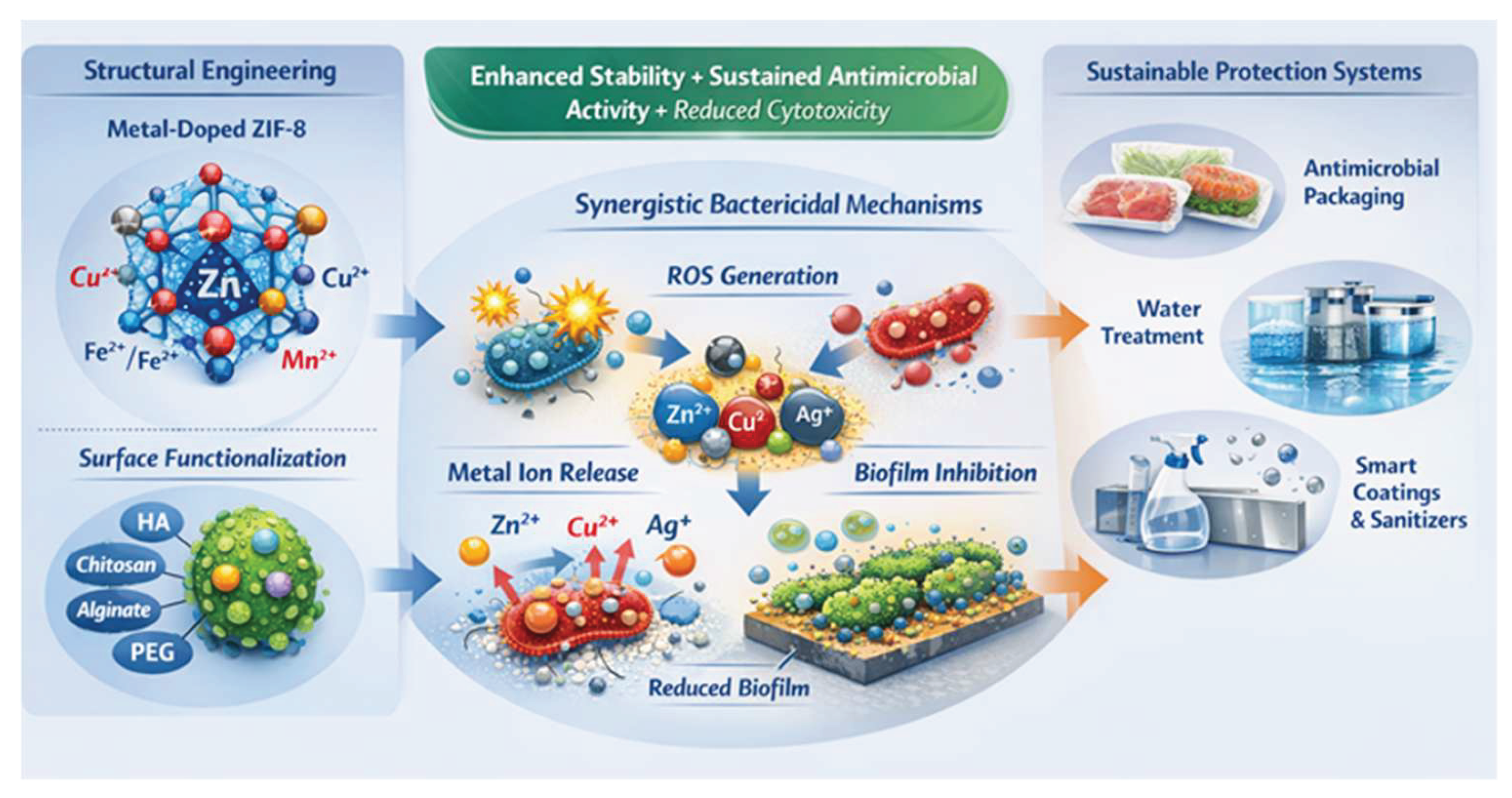
2. Synthesis Strategies and Structural Characteristics of Metal-Doped ZIF-8
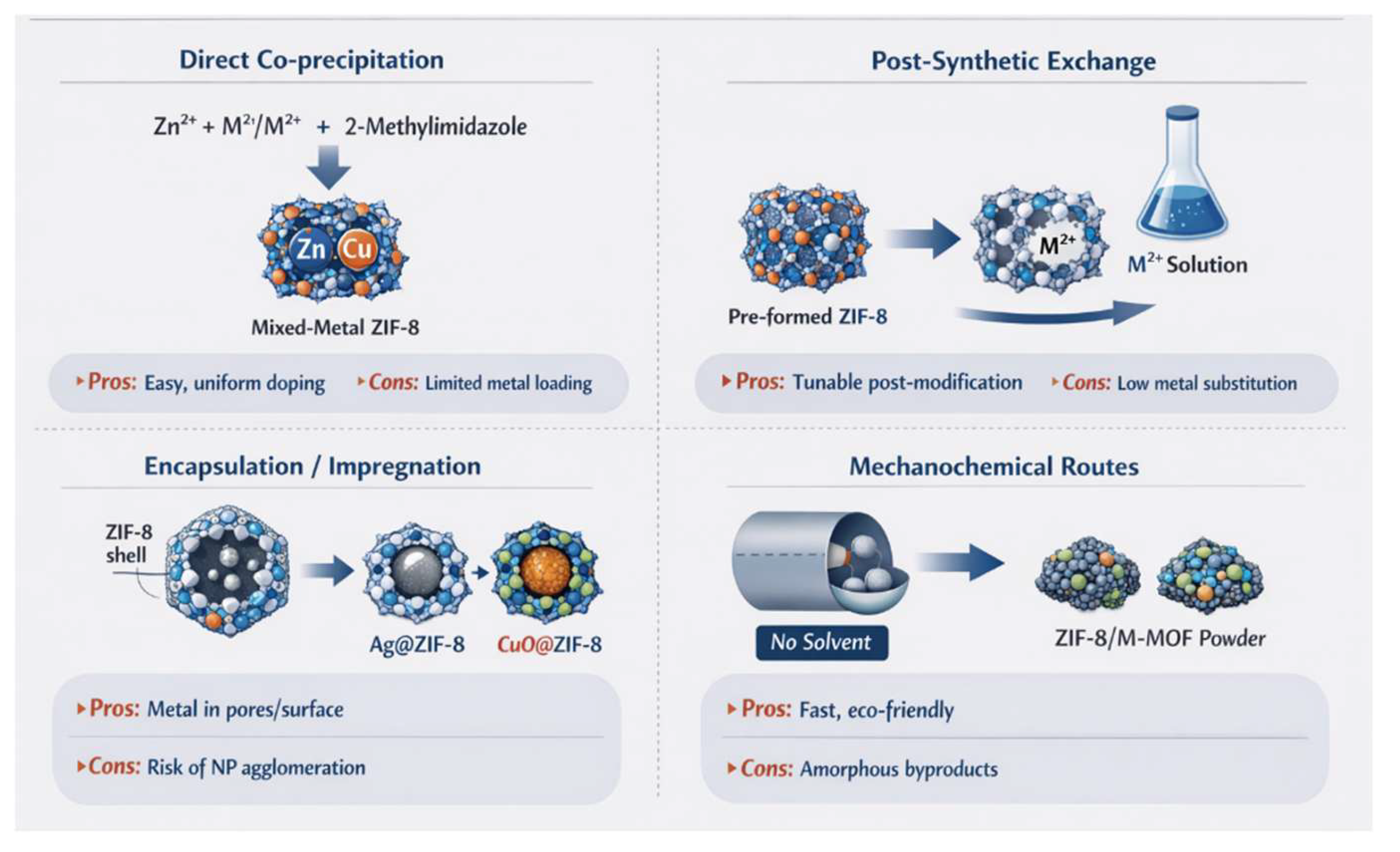
2.1. One-Pot Synthesis of Bimetallic ZIF-8 (In-Situ Doping)
2.2. Post-Synthetic Metal Incorporation and Composite Doping
2.3. Structural Characteristics and Stability of Metal-Doped ZIF-8
2.3.1. Crystallinity and Phase
2.3.2. Particle Morphology
2.3.3. Porosity and Surface Area
2.3.4. Chemical and Thermal Stability
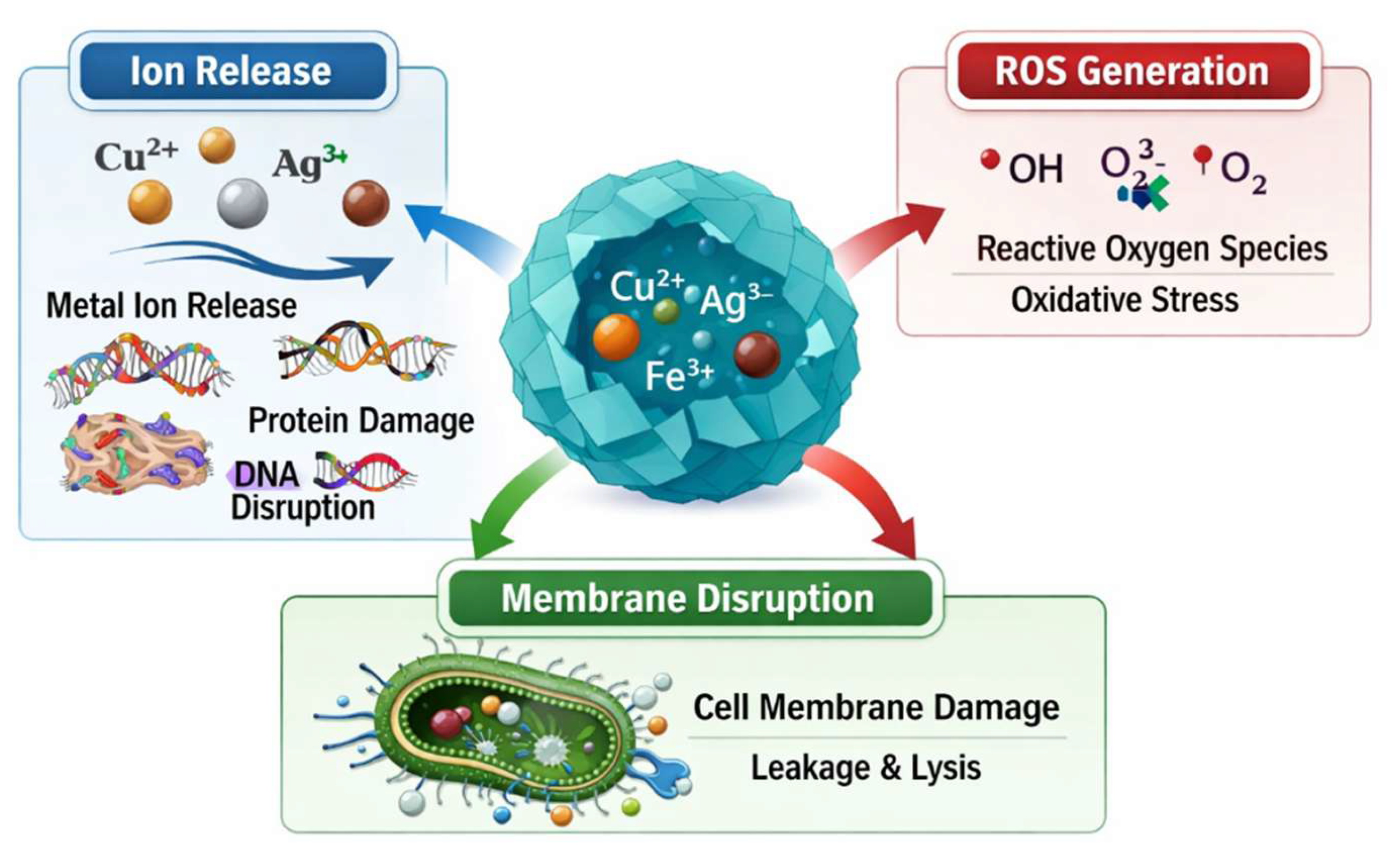
3. Antibacterial Mechanisms of Doped and Functionalized ZIF-8
3.1. Metal Ion Release and Membrane Interaction
3.2. Membrane Disruption and Direct Contact Killing
3.3. Reactive Oxygen Species (ROS) Generation and Photocatalytic Effects
3.4. Synergistic and Multi-Modal Mechanisms with Functional Additives
4. Surface Functionalization of ZIF-8: Polymers, Biomolecules, and Hybrids
4.1. Synergistic and Multi-Modal Mechanisms with Functional Additives
4.2. Biomolecule and Ligand Functionalization
4.3. Hybrid and Composite Nanoplatforms
4.3.1. ZIF-8 on Graphene Oxide (GO)
4.3.2. Magnetic Hybrids (ZIF-8@Fe3O4)
4.3.3. ZIF-8 Derived Hybrids
4.3.4. Layered Hybrids
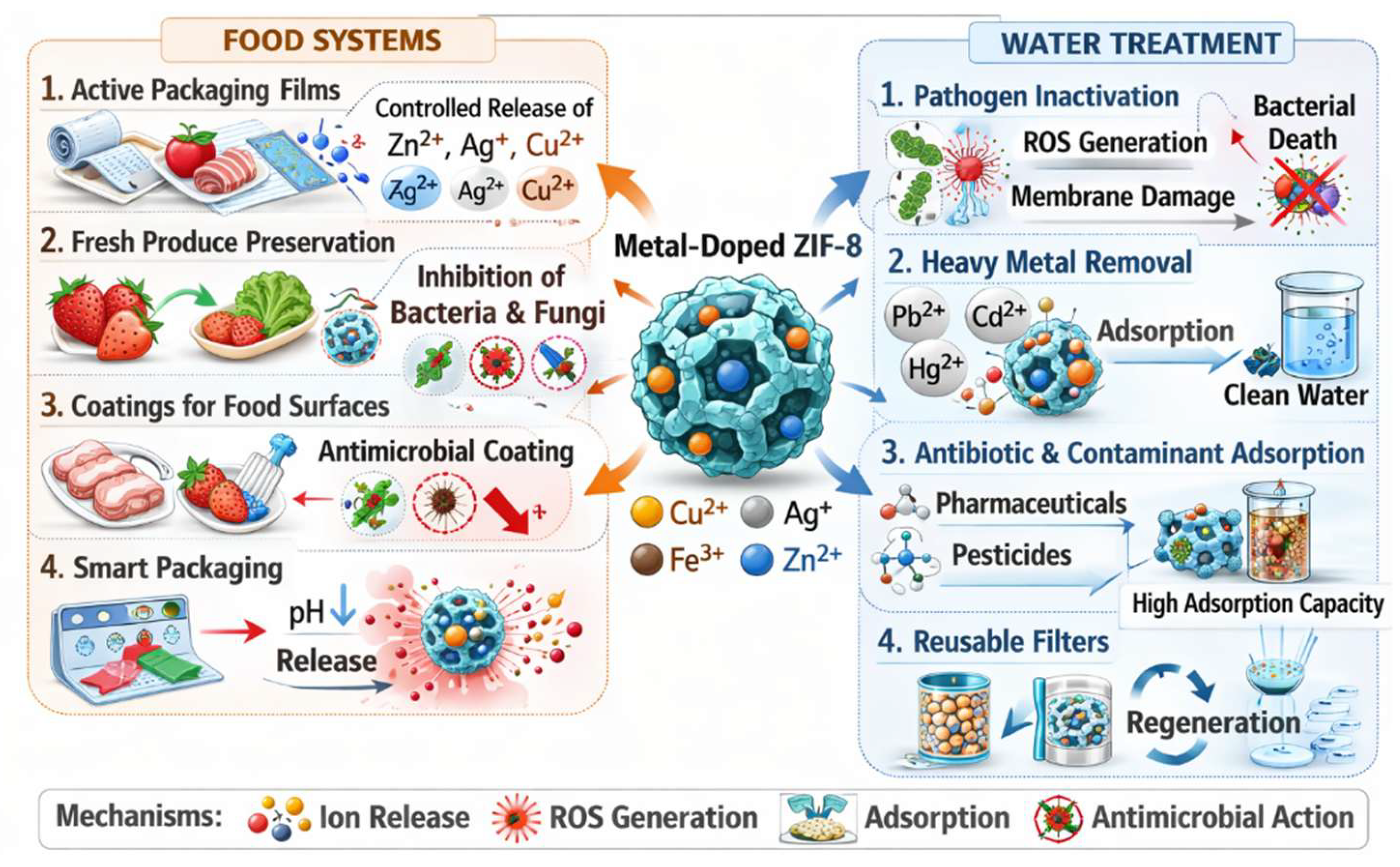
5. Applications in Food Systems: Packaging, Surface Coatings, and Fresh Produce
5.1. Antimicrobial Food Packaging Films
5.1.1. Biopolymer-Based Films
5.1.2. Gelatin/Chitosan Composite Films
5.1.3. Plastic Films
5.2. Antimicrobial Surface Coatings for Food Processing
5.3. Antimicrobial Surface Coatings for Food Processing
6. Applications in Water Treatment with Pathogen Removal and Biofilm Control
6.1. Photocatalytic and Metal-Ion Water Disinfection
6.2. Antibiofilm Strategies and Biofouling Control
6.3. Other Potential Applications and Current Limitations
7. Comparative Analysis of ZIF-8 and Other MOFs in Antimicrobial Applications
7.1. Structural Differences, Porosity, and Stability
7.2. Antimicrobial Mechanisms and Efficacy in Food and Water Systems
7.3. Toxicity and Safety Profile
8. Conclusions
Funding
Informed Consent Statement
Conflicts of Interest
Abbreviations
| MBC | Minimum bactericidal concentration |
| MIC | Minimum inhibitory concentration |
| MOF | Metal-organic framework |
| ZIF | Zeolitic imidazolate framework |
References
- Havelaar, A.H.; Kirk, M.D.; Torgerson, P.R.; Gibb, H.J.; Hald, T.; Lake, R.J.; Praet, N.; Bellinger, D.C.; Silva, M.R.; Gargouri, N.; Speybroeck, N.; Cawthorne, A.; Mather, C.; Stein, C.; Angulo, F.J.; Devleesschauwer, B. World Health Organization Global Estimates and Regional Comparisons of the Burden of Foodborne Disease in 2010. PLoS Med CrossRef. 2015, 12(12), e1001923. [Google Scholar] [CrossRef] [PubMed]
- Prüss-Ustün, A.; Wolf, J.; Bartram, J.; Clasen, T.; Cumming, O.; Freeman, M.C.; Gordon, B.; Hunter, P.R.; Medlicott, K.; Johnston, R. Burden of disease from inadequate water, sanitation and hygiene for selected adverse health outcomes: An updated analysis with a focus on low- and middle-income countries. International Journal of Hygiene and Environmental Health CrossRef. 2019, 222(5), 765–777. [Google Scholar] [CrossRef] [PubMed]
- Shen, M.; Forghani, F.; Kong, X.; Liu, D.; Ye, X.; Chen, S.; Ding, T. Antibacterial application of metal-organic framework and their composites. Comprehensive Review in Food Science and Food Safety CrossRef. 2020, 19(4), 1397–1419. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Z.; Cui, J.; Wu, S.; Cao, Z.; Cao, P. Engineering Metal-Organic Frameworks for Enhanced Antimicrobial Efficacy: Synthesis Methodologies, Mechanistic Perspectives, and Versatile Applications. Journal of Functional Biomaterials CrossRef. 2025, 16(9), 353. [Google Scholar] [CrossRef]
- Shen, B.; Wang, Y.; Wang, X.; Amal, F.E.; Zhu, L.; Jiang, L. A Cruciform Petal-like (ZIF-8) with Bactericidal Activity against Foodborne Gram-Positive Bacteria for Antibacterial Food Packaging. International Journal of Molecular Sciences CrossRef. 2022, 23(14), 7510. [Google Scholar] [CrossRef]
- Tian, Y.; Wang, B.; Zhang, Z.; Kai, T.; Wu, P.; Ding, P. Recent advances in metal-organic frameworks for antibacterial applications: mechanisms and emerging strategies. RSC Advances CrossRef. 2025, 15(33), 26710–26727. [Google Scholar] [CrossRef]
- Abdelkareem, M.A.; Abbas, Q.; Mouselly, M.; Alawadhi, H.; Olabi, A.G. High-performance effective metal-organic frameworks for electrochemical applications. Journal of Science: Advanced Materials and Devices CrossRef. 2022, 7(3), 100465. [Google Scholar] [CrossRef]
- Pettinari, C.; Pettinari, R.; Di Nicola, C.; Tombesi, A.; Scuri, S.; Marchetti, F. Antimicrobial MOFs. Coordination Chemistry Reviews CrossRef. 2021, 446, 214121. [Google Scholar] [CrossRef]
- Sava Gallis, D.F.; Butler, K.S.; Agola, J.O.; Pearce, C.J.; McBride, A.A. Antibacterial Countermeasures via Metal-Organic Framework-Supported Sustained Therapeutic Release. ACS Appl Mater Interfaces CrossRef. 2019, 11(8), 7782–7791. [Google Scholar] [CrossRef]
- Kermanshahi, P.K.; Akhbari, K. The antibacterial activity of three zeolitic-imidazolate frameworks and zinc oxide nanoparticles derived from them. RSC Advances CrossRef. 2024, 14, 5601–5608. [Google Scholar] [CrossRef]
- Subhadarshini, A.; Nanda, B. Zeolitic imidazolium framework (ZIF-8) and their derivative-based material for antibacterial study: a comprehensive review. Discov Mater CrossRef. 2025, 5, 50. [Google Scholar] [CrossRef]
- Yan, D.; Yang, M.; Cheng, F.; Sun, Y. Novel antimicrobial agents prepared on the basis of metal-organic frameworks (MOFs) and their applications in the food field. Trends in Food Science & Technology CrossRef. 2025, 161, 105070. [Google Scholar] [CrossRef]
- Liang, Z.; Wang, H.; Zhang, K.; Ma, G.; Zhu, L.; Zhou, L.; Yan, B. Oxygen-defective MnO2/ZIF-8 nanorods with enhanced antibacterial activity under solar light. Chemical Engineering Journal CrossRef. 2022, 131349. [Google Scholar] [CrossRef]
- Bhakare, M.A.; Likhite, V.V.; Bhosale, K.L.; Lokhande, K.D.; Bondarde, M.P.; Dhumal, P.S.; Degani, M.; Some, S. Synthesis of a dual-functional fluorescent zinc-based metal organic framework for antibacterial and anti-counterfeit ink applications. Journal of the Indian Chemical Society CrossRef. 2025, 101898. [Google Scholar] [CrossRef]
- Afsharpour, M.; Imani, S. Preventive protection of paper works by using nanocomposite coating of zinc oxide. Journal of Cultural Heritage CrossRef. 2017, 142–148. [Google Scholar] [CrossRef]
- Moharramnejad, M.; Ehsani, A.; Salmani, S.; Shahi, M. Zinc-based metal-organic frameworks: synthesis and recent progress in biomedical application. Journal of Inorganic and Organometallic Polymers and Materials CrossRef. 2022, 32(5). [Google Scholar] [CrossRef]
- Hoop, M.; Walde, C.F.; Riccò, R.; Mushtaq, F.; Terzopoulou, A.; Chen, X.Z.; J. DeMello, A.; Doonan, C.J.; Falcaro, P.; Nelson, B.J.; Puigmartí-Luis, J.; Pané, S. Biocompatibility characteristics of the metal organic framework ZIF-8 for therapeutical applications. Applied Materials Today CrossRef. 2018, 13–21. [Google Scholar] [CrossRef]
- Krishnamoorthy, R.; Athinarayanan, J.; Periyasamy, V.S.; Alshuniaber, M.A.; Alshammari, G.; Hakeem, M.J.; Ahmed, M.A.; Alshatwi, A.A. Antibacterial Mechanisms of Zinc Oxide Nanoparticle against Bacterial Food Pathogens Resistant to Beta-Lactam Antibiotics. Molecules CrossRef. 2022, 27(8), 2489. [Google Scholar] [CrossRef]
- Taheri, M.; Ashak, D.; Sen, T.; Enge, T.G.; Verma, N.K.; Tricoli, A.; Lowe, A.; Nisbet, D.R.; Tsuzuki, T. Stability of ZIF-8 nanopowders in bacterial culture media and its implication for antibacterial properties. Chemical Engineering Journal CrossRef. 2021, 127511. [Google Scholar] [CrossRef]
- Wang, D.; Zhu, Y.; Wan, Z.; Zhang, X.; Zhang, J. Colloidal semiconductor nanocrystals for biological photodynamic therapy applications: Recent progress and perspectives. Progress in Natural Science: Materials International CrossRef. 2020, 443–455. [Google Scholar] [CrossRef]
- Wang, Y.; Malkmes, M.J.; Jiang, C.; Wang, P.; Zhu, L.; Zhang, H.; Zhang, Y.; Huang, H.; Jiang, L. Antibacterial mechanism and transcriptome analysis of ultra-small gold nanoclusters as an alternative of harmful antibiotics against Gram-negative bacteria. Journal of Hazardous Materials CrossRef. 2021, 126236. [Google Scholar] [CrossRef] [PubMed]
- Saif, M.S.; Hasan, M.; Zafar, A.; Ahmed, M.M.; Tariq, T.; Waqas, M.; Hussain, R.; Zafar, A.; Xue, H.; Shu, X. Advancing Nanoscale Science: Synthesis and Bioprinting of Zeolitic Imidazole Framework-8 for Enhanced Anti-Infectious Therapeutic Efficacies. Biomedicines CrossRef. 2023, 11(10), 2832. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Sun, Y.; Li, S.; Zhang, P.; Yao, Q. Synthesis and modification of ZIF-8 and its application in drug delivery and tumor therapy. RSC Advances CrossRef. 2020, 10(62), 37600–37620. [Google Scholar] [CrossRef] [PubMed]
- Dring, J.; Kaczynski, M.; Zureikat, R.M.; Kaczynski, M.; Forma, A.; Baj, J. Review of Applications of Zeolites in Dermatology: Molecular Perspectives and Translational Potentials. International Journal of Molecular Sciences CrossRef. 2025, 26(14), 6821. [Google Scholar] [CrossRef]
- Nguyen, H.L.; Moreira, R.G.; Castell-Perez, M.E. Antibacterial Effectiveness of Zeolitic Imidazolate Framework-8 (ZIF-8) Nanoparticle Solutions and Its Derivatives Against Salmonella Typhimurium ATCC13311 on Loose-Leaf Lettuce (Lactuca sativa). Journal of Food Safety CrossRef. 2025, 45(4), e70031. [Google Scholar] [CrossRef]
- Guo, Y.; Fang, W.; Ru, J.; Wu, Y.; Zheng, J.; Gao, G.; Chen, C.; Yan, R.; Huang, S.; Wang, C. Facile synthesis of Ag@ZIF-8 core-shell heterostructure nanowires for improved antibacterial activities. Applied Surface Science CrossRef. 2018, 149–155. [Google Scholar] [CrossRef]
- Martín-Perales, A.I.; Peña-Ortiz, M.; Resende, J.A.; Malpartida, I.; Luque, R.; Balu, A.B. Continuous-flow mechanochemical preparation of Ag-Cu@ZIF-8 bimetallic system for antimicrobial applications. Results in Engineering CrossRef. 2025, 103581. [Google Scholar] [CrossRef]
- Abishad, P.M.; Yayashankar, M.; Namratha, K.; Srinath, B.S.; Kurkure, N.V.; Barbuddhe, S.B.; Rawool, D.B.; Vergis, J.; Byrappa, K. Synthesis of ZIF-8(Fe) Functionalized with Citral as Potent Antimicrobial Candidate against Multi-Drug Resistant Enteroaggregative Escherichia coli and Non-Typhoidal Salmonella Spp. Russian Journal of Bioorganic Chemistry CrossRef. 2023, 49, 360–366. [Google Scholar] [CrossRef]
- Fu, D.; Ding, Y.; Guo, R.; Zhang, J.; Wang, H.; Niu, B.; Yan, H. Polylactic acid/polyvinyl alcohol–quaternary ammonium chitosan double-layer films doped with novel antimicrobial agent CuO@ZIF-8 nanoparticles for fruit preservation. International Journal of Biological Macromolecules CrossRef. 2022, 195, 538–546. [Google Scholar] [CrossRef]
- Nguyen, H.L.; Moreira, R.G.; Castell-Perez, M.E. Multifunctional OEO-ZIF-8-HA Nanoparticles for Antibacterial Control on Latex Surfaces and Baby Arugula (Eruca Sativa) Leaves. Journal of Food Science CrossRef. 2026, 91(2), e70896. [Google Scholar] [CrossRef]
- Tran, T.V.; Dang, H.H.; Nguyen, H.; Nguyen, N.T.T.; Nguyen, D.H.; Nguyen, T.T.T. Synthesis methods, structure, and recent trends of ZIF-8-based materials in the biomedical field. Nanoscale Adv. CrossRef. 2025, 7(13), 3941–3960. [Google Scholar] [CrossRef] [PubMed]
- Vatani, P.; Aliannezhadi, M.; Tehrani, F.S. ZIFs for wastewater treatment from copper ions and dye. Scientific Reports CrossRef. 2024, 14, 15434. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Chen, T.; Lu, J.; Hu, H.; Zheng, H.; Zhu, P.; Pan, X. Metal-organic frameworks doped with metal ions for efficient sterilization: Enhanced photocatalytic activity and photothermal effect. Water Res. CrossRef. 2023, 229, 119366. [Google Scholar] [CrossRef] [PubMed]
- Usman, M.; Suliman, M.H. Silver-Doped Zeolitic Imidazolate Framework (Ag@ZIF-8): An Efficient Electrocatalyst for CO2 Conversion to Syngas. Catalysts CrossRef. 2023, 13(5), 867. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, X.; Song, J.; Jin, L.; Wang, X.; Quan, C. Ag/H-ZIF-8 Nanocomposite as an Effective Antibacterial Agent Against Pathogenic Bacteria. Nanomaterials CrossRef. 2019, 9(11), 1579. [Google Scholar] [CrossRef]
- Mechoor, A.; Berchmans, S.; Venkatachalam, G. Bimetallic Cu-Zn Zeolitic Imidazolate Framework as Peroxidase Mimics for the Detection of Hydrogen Peroxide: Electrochemical and Spectrophotometric Evaluation. ACS Omega CrossRef. 2023, 8(42), 39636–39650. [Google Scholar] [CrossRef]
- Ren, H.; Gao, C.; Huang, S.; Du, L.; Liu, S.; Cao, X.; Lv, Y. Photocatalytic Antibacterial Mechanism and Biotoxicity Trade-Off of Metal-Doped M ZIF-8 (M=Co, Cu): Progress and Challenges. Inorganics CrossRef. 2026, 14(2), 43. [Google Scholar] [CrossRef]
- Awadallah-F, A.; Hillman, F.; Al-Muhtaseb, S.A. Influence of doped metal center on morphology and pore structure of ZIF-8. MRS Communication CrossRef. 2019, 9, 288–291. [Google Scholar] [CrossRef]
- Thanh, M.T.; Thien, T.V.; Chau, V.T.T.; Du, P.D.; Hung, N.P.; Khieu, D.Q. Synthesis of Iron Doped Zeolite Imidazolate Framework-8 and Its Remazol Deep Black RGB Dye Adsorption Ability. Journal of Chemistry CrossRef. 2017, 1, 5045973. [Google Scholar] [CrossRef]
- Gohr, M.S.; Ali, H.; Gad, S.; Soliman, H.M.; Giba, A.E. Preparation and characterization of holmium doped ZIF-8 nanocrystals for white light emitting phosphors. Sci Rep CrossRef. 2026, 16, 4116. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, X.; Song, J.; Jin, L.; Wang, X.; Quan, C. Ag/H-ZIF-8 Nanocomposite as an Effective Antibacterial Agent Against Pathogenic Bacteria. Nanomaterials CrossRef. 2019, 9(11), 1579. [Google Scholar] [CrossRef] [PubMed]
- Ngidi, N.P.D.; Ollengo, M.A.; Nyamori, V.O. Effect of Doping Temperatures and Nitrogen Precursors on the Physicochemical, Optical, and Electrical Conductivity Properties of Nitrogen-Doped Reduced Graphene Oxide. Materials (Basel) CrossRef. 2019, 12(20), 3376. [Google Scholar] [CrossRef] [PubMed]
- De Jesus Velazquez-Garcia, J.; Frenzke, S.; de los Santos Valladares, L.; Barnes, C.H.W.; Copeman, C.; Singh, J.; Kulkarni, S.; Keller, T.F.; Cornejo, H.S.; Huanaco-Quispe, D.; Anwary, M.; Elorche, R.; Asprilla-Herrera, L.M.; Lukaszczyk, W.; Eroglu, N.; Eifler, D.; Techert, S. Size matters: limitations of the ZIF-8 monolith and its Ni-, Co-, and Cu-doped variants for the adsorption of rhodamine B. Material Advances CrossRef. 2025, 6, 7800–7811. [Google Scholar] [CrossRef]
- Hongyu, W.; Henghui, Y.; Guoqing, W.; Lei, H.; Jia, Y.; Xianjie, L.; Yuanke, M.; Mingjie, W.; Yuanlin, Z.; Jianxin, Y.; Hongguo, Z. Co/Fe co-doped ZIF-8 derived hierarchically porous composites as high-performance electrode materials for Cu2+ ions capacitive deionization. Chemical Engineering Journal CrossRef. 2023, 460, 141621. [Google Scholar]
- Han, L.; Huang, Z.; Zhu, M.; Zhu, Y.; Li, H. Drug-loaded zeolite imidazole framework-8-functionalized bioglass scaffolds with antibacterial activity for bone repair. Ceramics International CrossRef. 2022, 48(5), 6890–6898. [Google Scholar] [CrossRef]
- Su, Y.; Wang, J.; Wang, Y.; Han, X.; Liu, Y.; Gong, H.; Zhang, M.; Ma, S.; Zhao, J. Zeolitic Imidazolate Framework-8 as a pH-Responsive Smart Targeted Release Nanoplatform for Antibacterial Therapy and Wound Regeneration. Chemistry An Asian Journal CrossRef. 2025, 20(19), e00682. [Google Scholar] [CrossRef]
- He, J.; Peng, L.; Zhang, Z.; Wu, Y.; Qu, W. Antibacterial hydrogel filters via in situ growth of Ag-doped ZIF-8 on cellulose nanofibers: A novel strategy for biofouling control in water treatment. Carbohydrate Polymers CrossRef. 2025, 368(2), 124174. [Google Scholar] [CrossRef]
- Oteiza, P.; Clegg, M.; Zago, M.P.; Keen, C.L. Zinc deficiency induces oxidative stress and AP-1 activation in 3T3 cells. Carbohydrate Polymers CrossRef. 2000, 28(7), 1091–1099. [Google Scholar] [CrossRef]
- Cortese-Krott, M.M.; Münchow, M.; Pirev, E.; Heβner, F.; Bozkurt, A.; Uciechowski, P.; Pallua, N.; Kröncke, K.D.; Suschek, C.V. Silver ions induce oxidative stress and intracellular zinc release in human skin fibroblasts. Free Radical Biology and Medicine CrossRef. 2009, 47(11), 1570–1577. [Google Scholar] [CrossRef]
- Tien Vo, T.T.; Peng, T.Y.; Nguyen, T.H.; Huyen Bui, T.N.; Wang, C.; Lee, W.; Chen, Y.; Lee, I. The crosstalk between copper-induced oxidative stress and cuproptosis: a novel potential anticancer paradigm. Cell Communication and Signaling CrossRef. 2024, 22, 353. [Google Scholar] [CrossRef]
- Abdelkhalek, M.M.; Mohamed, A.M.; Abdallah, R.Z.; Khedr, G.E.; Siam, R.; Allam, N.K. Zeolitic imidazolate framework-8 encapsulated with Mo-based polyoxometalates as surfaces with antibacterial activity against Escherichia coli†. Nanoscale Advances CrossRef. 2024, 6(13), 3355–3366. [Google Scholar] [CrossRef] [PubMed]
- Shen, D.; Wu, W.; Li, Y.; Abate, A.; Wei, M. 2-Methylimidazole as an interlayer for the enhancement of the open-circuit voltage in perovskite solar cells. Journal of Power Sources CrossRef. 2020, 450, 227714. [Google Scholar] [CrossRef]
- Wu, X.; Gao, J.; Wang, J.; Zhang, H. A ZIF-8@Apt nanocomposite enables synergistic antibacterial action via membrane disruption and transcriptional inhibition. Chemical Engineering Journal CrossRef. 2026, 531, 174204. [Google Scholar] [CrossRef]
- Xia, A.; Song, X.; Li, Y.; Hou, W.; Ly, H.; Li, F.; Li, Y.; Liu, J.; Li, X. Antibacterial and anti-inflammatory ZIF-8@Rutin nanocomposite as an efficient agent for accelerating infected wound healing. Frontiers in Bioengineering and Biotechnology CrossRef. 2022, 10, 1026743. [Google Scholar] [CrossRef]
- Poznanski, P.; Hameed, A.; Orczyk. Chitosan and Chitosan Nanoparticles: Parameters Enhancing Antifungal Activity. Molecules CrossRef. 2023, 28(7), 2996. [Google Scholar] [CrossRef]
- Tu, Y.; Lei, C.; Deng, F.; Chen, Y.; Wang, Y.; Zhang, Z. Core–shell ZIF-8@polydopamine nanoparticles obtained by mitigating the polydopamine coating induced self-etching of MOFs: prototypical metal ion reservoirs for sticking to and killing bacteria. New Journal of Chemistry CrossRef. 2021, 19(45), 8701–8713. [Google Scholar] [CrossRef]
- Wu, Z.; Nie, R.; Wang, Y.; Wang, W.; Li, X.; Liu, Y. Precise antibacterial therapeutics based on stimuli-responsive nanomaterials. Frontiers in Bioengineering and Biotechnology 2023, 11. [Google Scholar] [CrossRef]
- Radan, N.; Nejad, Z.G.; Ghahnaz, S.; Yaghmaei, S. Boosting antibacterial efficiency of gelatin/chitosan composite films through synergistic interaction of ag nanoparticles and ZIF-8 metal-organic frameworks for food packaging. International Journal of Biological Macromolecules CrossRef. 2025, 305(2), 141175. [Google Scholar] [CrossRef]
- Lushchak, I.V. Free radicals, reactive oxygen species, oxidative stress and its classification. Chemico-Biological Interactions CrossRef. 2014, 224, 164–175. [Google Scholar] [CrossRef]
- Juan, C.A.; Pérez de la Lastra, J.M.; Lou, F.; Pérez-Lebeña, E. The Chemistry of Reactive Oxygen Species (ROS) Revisited: Outlining Their Role in Biological Macromolecules (DNA, Lipids and Proteins) and Induced Pathologies. International Journal of Molecular Sciences CrossRef. 2021, 22(9), 4642. [Google Scholar] [CrossRef]
- Gebretsadik, A.; Reddy, S.G.; Gonfa, B.A.; Abede, B. Ag-decorated Cu-doped ZnO nanomaterial for enhanced antibacterial application. Scientific Reports CrossRef. 2026, 16, 5552. [Google Scholar] [CrossRef] [PubMed]
- Lv, H.; Macharia, D.; Liu, Z.; Zhang, L.; Yu, C.; Lu, C.; Liu, H.; Zhang, y.; Chen, Z. Au-loaded ZIF-8 derived porous carbon with improved photothermal catalysis ability from interfacial heating instead of hot-electrons. Chemical Engineering Journal CrossRef. 2024, 482, 148963. [Google Scholar] [CrossRef]
- Wang, J.; Liang, H.; Zhou, H.; Zhang, M.; Sun, X.; Zhang, S.; Wang, P.; Gao, X. Cobalt-doped ZIF-8 nanozyme with bifunctional catalytic activity for integrated monitoring and remediation of sulfadiazine contaminants. Elsevier iScience CrossRef. 2025, 28(12), 113678. [Google Scholar] [CrossRef] [PubMed]
- Taheri, M.; Enge, T.G.; Tsuzuki, T. Water stability of cobalt doped ZIF-8: a quantitative study using optical analyses. Materials Today Chemistry CrossRef. 2020, 16, 100231. [Google Scholar] [CrossRef]
- Niu, X.; Liu, B.; Hu, P.; Zhu, H.; Wang, M. Nanozymes with Multiple Activities: Prospects in Analytical Sensing. Biosensors CrossRef. 2022, 12(4), 251. [Google Scholar] [CrossRef]
- Weng, Y.; Li, J.; Xie, D.; Peng, S.; Wen, Y.; Wan, J.; Ma, D.; Li, G; Zhang, W. ZIF-8-Based Composites for Enhanced Biofilm Penetration and Synergistic Antibacterial Therapy against Periodontal Pathogens. ACS Applied Materials & Interfaces CrossRef. 2025, 17(32), 45413–45428. [Google Scholar] [CrossRef]
- Lan, J.; Zou, J.; Xin, H.; Sun, J.; Han, T.; Sun, M.; Niu, M. Nanomedicines as disruptors or inhibitors of biofilms: Opportunities in addressing antimicrobial resistance. Journal of Controlled Release CrossRef. 2025, 381, 113589. [Google Scholar] [CrossRef]
- Xiao, Y.; Xu, M.; Lv, N.; Cheng, C.; Huang, P.; Li, J.; Hu, Y.; Sun, M. Dual stimuli-responsive metal-organic framework-based nanosystem for synergistic photothermal/pharmacological antibacterial therapy. Acta Biomaterialia CrossRef. 2021, 1, 122:2910305. [Google Scholar] [CrossRef]
- Hara, Y.; Castell-Perez, M.E.; Moreira, R.G. Antimicrobial properties of poly (vinyl alcohol) films with zeolitic imidazolate framework (ZIF-8) nanoparticles for food packaging. Journal of Food Sciences CrossRef. 2023, 88(6), 2512–2522. [Google Scholar] [CrossRef]
- Ding, Y.; Yuan, J.; Mo, F.; Wu, S.; Ma, Y.; Li, R.; Li, M. A pH-Responsive Essential Oil Delivery System Based on Metal-organic Framework (ZIF-8) for Preventing Fungal Disease. Journal of Agricultural and Food Chemistry CrossRef. 2023, 71)47, 18312–18322. [Google Scholar] [CrossRef]
- Kalhor, H.; Piraman, Z.; Fathali, Y. Hen egg white lysozyme encapsulated in ZIF-8 for performing promiscuous enzymatic Mannich reaction. iScience CrossRef. 2023, 26(10), 107807. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.; Sum, T.; Zeng, Y.; Zheng, M.; Zhang, F.; Wang, Y.; Lin, Z.; Lin, R. Hierarchical Micro- and Mesoporous Zeolitic Imidazolate Framework-8-Based Enzyme Hybrid for Combination Antimicrobial by Lysozyme and Lactoferrin. Inorganic Chemistry CrossRef. 2024, 63(26), 12377–12384. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Yao, H.; Yang, J.; Chen, C.; Shi, J. Construction of a two-dimensional artificial antioxidase for nanocatalytic rheumatoid arthritis treatment. Nature Communications CrossRef. 2022, 13. [Google Scholar] [CrossRef] [PubMed]
- Jahani, S.; Shakiba, A.; Jahani, L. The Antimicrobial Effect of Lactoferrin on Gram-Negative and Gram-Positive Bacteria. International Journal of Infection CrossRef. 2015, 2(3), e27954. [Google Scholar] [CrossRef]
- Xia, X.; Song, X.; Li, Y.; Hou, W.; Lv, H.; Li, F.; Li, Y.; Liu, J.; Li, X. Antibacterial and anti-inflammatory ZIF-8@Rutin nanocomposite as an efficient agent for accelerating infected wound healing. Frontiers in Bioengineering and Biotechnology CrossRef. 2022, 10, 1026743. [Google Scholar] [CrossRef]
- Chen, X.; Argandona, S.M.; Melle, F.; Rampal, N.; Fairen-Jimenez. Advances in surface functionalization of next-generation metal-organic frameworks for biomedical applications: Design, strategies, and prospects. Chem CrossRef. 2024, 10(2), 504–543. [Google Scholar] [CrossRef]
- Chen, Q.; Han, D.; Qu, X.; Zhang, W.; Peng, Y.; Li, S.; Qin, K.; Ren, S.; Wang, Y.; Zhou, H.; Zhao, P.; Wu, Z.; Gao, Z. Self-enhanced multifunctional composite membranes based on metal-organic frameworks embedded with thymol nanoparticles for post-harvest preservation of fruits. Food Hydrocolloids CrossRef. 2025, 164, 111208. [Google Scholar] [CrossRef]
- Channab, B.; El Idrissi, A.; Ammar, A.; Akil, A.; White, J.C.; Zahouily, M. ZIF-8 metal organic framework, carboxymethylcellulose and polyvinyl alcohol bio-nanocomposite controlled-release phosphorus fertilizer: Improved P management and tomato growth. Chemical Engineering Journal CrossRef. 2024, 495, 153610. [Google Scholar] [CrossRef]
- Makhetha, T.A.; Ray, S.C.; Moutloali, R.M. Zeolitic Imidazolate Framework-8-Encapsulated Nanoparticle of Ag/Cu Composites Supported on Graphene Oxide: Synthesis and Antibacterial Activity. ACS Omega CrossRef. 2020, 5(17), 9626–9640. [Google Scholar] [CrossRef]
- Jamiri, F.; Fasaei, B.N.; Joghataei, S.M.; Yahyaraeyat, R.; Mazloom-Jalali, A. Synergistic antibacterial activity of chitosan-polyethylene glycol nanocomposites films containing ZIF-8 and doxycycline. BMC Biotechnology CrossRef. 2025, 25:19. [Google Scholar] [CrossRef]
- Huang, T.; Yang, L.; Wang, S.; Lin, C.; Wu, X. Enhanced performance of ZIF-8 nanocrystals hybrid monolithic composites via an in-situ growth strategy for efficient capillary microextraction of perfluoroalkyl phosphonic acids. Talanta CrossRef. 2023, 259, 124452. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Tao, Y.; Chen, Z.; Wang, Z.; Yang, J.; Wu, S.; Pang, Y.; Chen, H. Synergistic Zn2+ nucleation control via zincophilic 3D confinement enables dendrite-free Zn anodes. Electrochimica Acta CrossRef. 2026, 550, 148132. [Google Scholar] [CrossRef]
- Kong, W.; Serdechnova, M.; Kasneryk, V.; Gao, D.; Wang, H.; Xie, X.; Blawert, C.; Zheludkevich, M.L.; Zhang, Y. ZIF-8 based bifunctional coatings with anticorrosive and antibacterial properties: A new design strategy for more efficiency. Surface and Coatings Technology CrossRef. 2024, 482, 130812. [Google Scholar] [CrossRef]
- Rahman, M.; Kabir, M.; Li, K.; Li, Y.; Chen, S.; Wu, S. Electrospun zeolitic imidazole framework-8 loaded silk fibroin/polycaprolactone nanofibrous scaffolds for biomedical application. Journal of the Mechanical Behavior of Biochemical Materials CrossRef. 2024, 160, 106769. [Google Scholar] [CrossRef]
- Ki, H.; Yanilmaz, M.; Kim, J. Antibacterial dual-drug delivery fiber system via a multi-layered porous Poly(caprolactone)/poly lactic acid/ZIF-8 composites. International Journal of Pharmaceutics CrossRef. 2025, 685, 126282. [Google Scholar]
- Huan, Y.; Kong, Q.; Mou, H.; Yi, H. Antimicrobial peptides: Classification, Design, Application and Research Progress in Multiple Fields. Front Microbiol CrossRef. 2020, 11, 582779. [Google Scholar] [CrossRef]
- Saha, S.; Mishra, A. Protein-directed synthesis of ZIF-8 functionalized with a polymer as core-shell drug coatings with antibacterial and anti-inflammatory properties. Biomaterial Science CrossRef. 2023, 11, 481–488. [Google Scholar] [CrossRef]
- Shen, X.; Wang, J.; Cao, B.; Wang, M.; Yang, H.; Fu, Q.; Han, P.; Lin, H.; Zhang, X.; Yin, C.; Lan, J.; Sang, S.; Huang, Y. Antimicrobial peptide-ZIF8 embedded silk protein-lysozyme composite films: A promising multifunctional solution for infected bone regeneration. Int J Biol Macromol CrossRef. 2025, 311(1), 143553. [Google Scholar] [CrossRef]
- Behera, D.; Priyadarshini, P.; Parida, K. ZIF-8 metal-organic frameworks and their hybrid materials: emerging photocatalysts for energy and environmental applications. Dalton Trans CrossRef. 2025, 54, 2681–2708. [Google Scholar] [CrossRef]
- Perdikaki, A.; Galeou, A.; Pilatos, G.; Karatasios, I.; Kanellopoulos, N.K.; Prombona, A.; Karanikolos, G.N. Ag and Cu monometallic and Ag/Cu bimetallic nanoparticle–graphene composites with enhanced antibacterial performance. ACS Appl. Mater. Interfaces CrossRef. 2016, 8, 27498–27510. [Google Scholar] [CrossRef]
- Wu, X.; Zhang, H.; Yin, Z.; Yang, Y.; Wang, Z. ZIF-8/GO sandwich composite membranes through a precursor conversion strategy for H2/CO2 separation. Journal of Membrane Science CrossRef. 2022, 647, 120291. [Google Scholar] [CrossRef]
- Phatai, P.; Pholtue, C.; Sihanatang, N.; Bantao, N.; Prasitnok, K.; Khemthong, P.; Youngjan, S.; Butburee, T.; Khunphonoi, R.; Kamonwannasit, S.; Pinitsoontorn, S.; Prasitnok, O. Plant Extract-Coated Fe3O4/ZIF-8 and Fe3O4/ZIF-67 Nanocomposites: Efficient Antibacterial Activity and Magnetic Recovery. ChemistrySelect CrossRef. 2025, 10(44), e05890. [Google Scholar] [CrossRef]
- Kim, E.B.; Akhtar, M.S.; Kong, I.; Ameen, S. Carbonized porous zeolitic imidazolate framework as promising electrode for electrochemical supercapacitors. Electrochimica Acta CrossRef. 2024, 507, 145110. [Google Scholar] [CrossRef]
- Sadiq, S.; Khan, I.; Humayun, M.; Wu, P.; Khan, A.; Khan, S.; Khan, A.; Khan, S.; Alanazi, A.F.; Bououdina, M. Synthesis of Metal–Organic Framework-Based ZIF-8@ZIF-67 Nanocomposites for Antibiotic Decomposition and Antibacterial Activities. ACS Omega 2023, 8(51), 49244–49258. [Google Scholar] [CrossRef]
- Fonseca, J.; Broto-Ribas, A.; Jiao, L.; Pei, X. Pickering emulsions stabilized by metal-organic framework nanoparticles. Advances in Colloid and Interface Science CrossRef. 2025, 342, 103552. [Google Scholar] [CrossRef]
- Li, H.; Shu, C.; Zhou, Y.; Zhang, K.; Wang, Y. pH-triggered starch/ordered macroporous ZIF-8 smart films: synergistic antioxidant–antibacterial activity via clove oil encapsulation for sustainable food packaging. Food Quality and Safety CrossRef. 2026, 10, fyaf076. [Google Scholar] [CrossRef]
- Liu, Z.; Yan, A.; Lin, J.; Liu, B.; Wang, Y.; Wen, L.; Jin, B.; Wu, S.; Wang, L. Nanocellulose/ZIF-8/Calcium Hybrid Films for Sustainable Food Preservation. ACS Applied Nano Materials CrossRef. 2026. [Google Scholar] [CrossRef]
- Zhou, Q.; Yu, T.; Yang, R.; Guo, X.; Chen, Y. ZIF-8 and Its Derivative Adsorbents for Heavy Metal Removal in Water: A Review. ACS Omega CrossRef. 2025, 10(41), 47790–47801. [Google Scholar] [CrossRef]
- Li, H.; Wang, S.; Chen, J.; Sun, M.; Tang, H. Preparation and characterization of thymol-loaded ZIF-8/κ-carrangeenan/Zein composite film as active food packaging with enhanced functional properties for blueberry preservation. Carbohydrate Polymers CrossRef. 2025, 356, 123406. [Google Scholar] [CrossRef]
- Liu, J.; Feng, J.; Xu, Z.; Zhang, J.; Wang, H. Intelligent Food Packaging Films Based on pH-Responsive Eugenol@ZIF-8/PVA-HACC with Enhanced Antimicrobial Activity. Molecules CrossRef. 2026, 31(4), 669. [Google Scholar] [CrossRef]
- Hall, A.G.; King, J.C. Zinc Fortification: Current Trends and Strategies. Nutrients CrossRef. 2022, 14(19), 3895. [Google Scholar] [CrossRef]
- Sukhareva, K.; Chernetsov, V.; Burmistrov, I. A Review of Antimicrobial Polymer Coatings on Steel for the Food Processing Industry. Polymers CrossRef. 2024, 16(6), 809. [Google Scholar] [CrossRef]
- Yamane, S.; Bin Yusri, A.H.; Chen, P.; van der Vlies, A.J.; Ben Mabrouk, A.; Fetzer, I.; Hasegawa, U. Surface Coating of ZIF-8 Nanoparticles with Polyacrylic Acid: A Facile Approach to Enhance Chemical Stability for Biomedical Applications. Macromol Biosci CrossRef. 2024, 25(2), 2400382. [Google Scholar] [CrossRef]
- Xiong, L.; Wu, M.; Lio, J.; Gu, Z.; Fu, Z.; Fan, X.; Zhu, M. In-situ encapsulation of organic and inorganic inhibitors into 2D zeolitic imidazolate framework assisted by sodium gluconate for enhanced anticorrosion protection of AA2024. Corrosion Science CrossRef. 2025, 256, 113226. [Google Scholar] [CrossRef]
- Schumann-Muck, F.M.; Hillig, N.; Braun, P.G.; Griebel, J.; Koethe, M. Impact of nanoscale coating of stainless steel on Salmonella Enteritidis and Escherichia coli. Journal of Food Safety CrossRef. 2023, 43(5), e13075. [Google Scholar] [CrossRef]
- Abdelhamid, H.N.; Mathew, A.P. Cellulose-zeolitic imidazolate frameworks (CelloZIFs) for multifunctional environmental remediation: Adsorption and catalytic degradation. Chemical Engineering Journal CrossRef. 2021, 426, 131733. [Google Scholar] [CrossRef]
- Li, P.; Li, J.; Feng, X.; Li, J.; Hao, Y.; Zhang, J.; Wang, H.; Yin, A.; Zhou, J.; Ma, X.; Wang, B. Metal-organic frameworks with photocatalytic bactericidal activity for integrated air cleaning. Nature Communications CrossRef. 2019, 10, 2177. [Google Scholar] [CrossRef]
- Benitez, J.; Aryal, J.; Lituma, I.; Moreira, J.; Adhikari, A. Evaluation of the Effectiveness of Aeration and Chlorination during Washing to Reduce E. coli O157:H7, Salmonella enterica, and L. innocua on Cucumbers and Bell Peppers. Foods CrossRef. 2023, 13(1), 146. [Google Scholar] [CrossRef]
- Chang-qing, X.; Yi, G.; Fan-hao, Z. Stability of aqueous nano-ceramic coatings with two different dispersants. Journal of Central South University of Technology CrossRef. 2003, 10, 87–90. [Google Scholar] [CrossRef]
- Wang, R.; Mi, K.; Yuan, X.; Chen, J.; Pu, J.; Shi, X.; Yang, Y.; Zhang, H.; Zhang, H. Zinc Oxide Nanoparticles Foliar Application Effectively Enhanced Zinc and Aroma Content in Rice (Oryza sativa L.) Grains. Rice (NY) CrossRef. 2023, 16:36. [Google Scholar] [CrossRef]
- Plati, F.; Rapi, R.; Paraskevopoulou, A. Characterization of Oregano Essential Oil (Origanum vulgare L. subsp. hirtum) Particles Produced by the Novel Nano Spray Drying Technique. Foods CrossRef. 2021, 10(12), 2923. [Google Scholar] [CrossRef] [PubMed]
- Ravinayagam, V.; Rahman, S. Zeolitic imidazolate framework-8 (ZIF-8) doped TiZSM-5 and Mesoporous carbon for antibacterial characterization. Saudi Journal of Biological Sciences CrossRef. 2020, 27(7), 1726–1736. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Wang, H.; Yue, C.; He, L.; Li, H.; Zhang, H.; Yang, S.; Ma, T. Photocatalytic degradation by TiO2-conjugated/coordination polymer heterojunction: Preparation, mechanisms, and prospects. Applied Catalysis B: Environment and Energy CrossRef. 2024, 344, 123605. [Google Scholar] [CrossRef]
- Thomas, N.; Dionysiou, D.D.; Pillai, S.C. Heterogeneous Fenton catalysts: A review of recent advances. Journal of Hazard Materials CrossRef. 2020, 404, 124082. [Google Scholar] [CrossRef]
- Mishra, N.O.; Quon, A.S.; Nguyen, A.; Papazyan, E.K.; Hao, Y.; Liu, Y. Constructing Physiological Defense Systems against Infectious Disease with Metal–Organic Frameworks: A Review. ACS Applied Bio Materials CrossRef. 2023, 6(8), 3052–3065. [Google Scholar] [CrossRef]
- Wei, T; Yu, Q; Chen, H. Responsive and synergistic antibacterial coatings: fighting against bacteria in a smart and effective way. Advanced Healthcare Materials CrossRef. 2019, 8(3), e1801381. [Google Scholar] [CrossRef]
- Di Pippo, F.; Di Gregorio, L.; Congestri, R.; Tandoi, V.; Rossetti, S. Biofilm growth and control in cooling water industrial systems. FEMS Microbiology Ecology CrossRef. 2018, 94(5), fiy044. [Google Scholar] [CrossRef]
- Ugwu, C.N.; Ezeibe, E.N.; Emencheta, S.C.; Nwagwu, C.S.; Ogbonna, K.O.; Ejiofor, C.V.; Onugwu, A.L.; Berebon, D.P.; Attama, A.A. Biofilms: structure, resistance mechanism, emerging control strategies, and applications. RSC Pharmaceutics CrossRef. 2025, 2(6), 1376–1407. [Google Scholar] [CrossRef]
- Muthalagu, S.; Natarajan, S. Deciphering the Antimicrobial and Antibiofilm Efficiency of Thyme Essential Oil Encapsulated Zeolitic Imidazole Framework-8 Against Foodborne Pathogens. Current Microbiology CrossRef. 2024, 82(1), 49. [Google Scholar] [CrossRef]
- National Academies of Sciences; Engineering; and Medicine; Health and Medicine Division. Division on Earth and Life Studies; Board on Population Health and Public Health Practice; Board on Life Sciences; Water Science and Technology Board; Committee on Management of Legionella in Water Systems. Management of Legionella in Water Systems Strategies for Legionella Control and Their Application. Washington (DC): National Academies Press (US) CrossRef. 2019, 4. [Google Scholar]
- Jiang, W.; Zhou, X.; Yuan, X.; Zhang, L.; Xiao, X.; Zhu, J.; Cheng, W. Multifunctional Metal–Organic Frameworks for Enhancing Food Safety and Quality: A Comprehensive Review. Foods CrossRef. 14(23), 4111. [CrossRef]
- Wang, L.; Cheng, S.; Qin, K.; Yang, X.; Wang, H.; Man, C.; Zhao, Q.; Jiang, Y. Apigenin@ZIF-8 with pH-responsive sustained release function added to propolis-gelatin films achieved an outstanding antibacterial effect. Food Packaging and Shelf Life CrossRef. 2023, 40, 101191. [Google Scholar] [CrossRef]
- Yang, G.; Wang, J.; Wang, R.; Han, L.; Gong, C.; Chen, J.; Chen, M.; Yuan, Y. Zeolitic Imidazolate Framework-8 (ZIF-8) as a Carrier for Kaempferol Delivery to Protect Against Gamma Radiation-Induced Mortality and Damage. Pharmaceutics CrossRef. 2025, 17(11), 1489. [Google Scholar] [CrossRef] [PubMed]
- Hu, F.; Liu, X.; Chen, C.; Liu, Y.; Xu, X.; Han, L.; He, C.; Wang, M. Biomimetic carboxymethyl β-glucan-ZIF-8 nanocarrier enhances structural stability, gastrointestinal release, and antioxidant efficacy of urolithin A via dectin-1-engaged mitophagy. Food Res Int CrossRef. 2026, 227, 118314. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Trinh, B.T.; Kim, K.H.; Moon, J.; Kang, H.; Jo, K.; Akter, R.; Jeong, J.; Lim, E.K.; Jung, J.; Choi, H.S.; Park, H.G.; Kwon, O.S.; Yoon, I.; Kang, T. Au@ZIF-8 SERS paper for food spoilage detection. Biosensors and Bioelectronics CrossRef. 2021, 179, 113063. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Dong, X.; Cai, N.; Yang, F.; Yao, W.; Huang, L. Application of a Novel Au@ZIF-8 Composite in the Detection of Bisphenol A by Surface-Enhanced Raman Spectroscopy. Foods CrossRef. 2023, 12(4), 813. [Google Scholar] [CrossRef]
- Piryaei, M.; Abolghasemi, M.M.; Shokri, M.; Shokri, Z. Synergistic antimicrobial and antioxidant smart packaging enabled by a bio-MOF/ZIF-8 hybrid nanoplatform. LWT CrossRef. 2026, 242, 118977. [Google Scholar] [CrossRef]
- Elaouni, A.; El Ouardi, M.; Zbair, M.; BaQais, A.; Saadi, M.; Ait Ahsaine, H. ZIF-8 metal organic framework materials as a superb platform for the removal and photocatalytic degradation of organic pollutants: a review. RSC Adv CrossRef. 12(49), 31801–31817. [CrossRef]
- Li, K.; Miwornunyuie, N.; Chen, L.; Jingyu, H.; Amaniampong, P.S.; Koomson, D.A.; Ewusi-Mensah, D.; Xue, W.; Li, G.; Lu, H. Sustainable Application of ZIF-8 for Heavy-Metal Removal in Aqueous Solutions. Sustainability CrossRef. 2021, 13(2), 984. [Google Scholar] [CrossRef]
- Talukder, N.; Wang, Y.; Nunna, B.B.; Tong, X.; Lee, E.S. An investigation on the structural stability of ZIF-8 in water versus water-derived oxidative species in aqueous environment. Microporous and Mesoporous Materials CrossRef. 2024, 366, 112934. [Google Scholar] [CrossRef]
- Ebrahimipour, S.Y.; Shahsavari, M.; Sheikhshoaei, M.; Sheikhshoaie, I. Zeolitic imidazolate frameworks (ZIFs): Versatile materials for sensing and mitigating diverse food contaminants. Journal of Food Composition and Analysis CrossRef. 2025, 146, 107825. [Google Scholar] [CrossRef]
- Zheng, H.; Wu, D.; Wang, Y.; Liu, X.; Gao, P.; Liu, W.; Wen, J.; Rebrov, E.V. One-step synthesis of ZIF-8/ZnO composites based on coordination defect strategy and its derivatives for photocatalysis. Journal of Alloys and Compounds CrossRef. 2020, 838, 155219. [Google Scholar] [CrossRef]
- Wang, D.; Wu, Q.; Ren, X.; Niu, M.; Ren, J.; Meng, X. Tunable Zeolitic Imidazolate Framework-8 Nanoparticles for Biomedical Applications. Small Methods CrossRef. 2024, 8(3), 2301270. [Google Scholar] [CrossRef] [PubMed]
- Lan, H.; Gan, N.; Pan, D.; Hu, F.; Li, T.; Long, N.; Shen, H.; Feng, Y. Development of a novel magnetic molecularly imprinted polymer coating using porous zeolite imidazolate framework-8 coated magnetic iron oxide as carrier for automated solid phase microextraction of estrogens in fish and pork samples. Journal of Chromatography A CrossRef. 2014, 1365, 35–44. [Google Scholar] [CrossRef]
- Bai, Y.; Dou, Y.; Xie, L.H.; Rutledge, W.; Li, J.R.; Zhou, H.C. Zr-based metal–organic frameworks: design, synthesis, structure, and applications. Chemical Society Reviews CrossRef. 2016, 8(45), 2327–2367. [Google Scholar] [CrossRef]
- Dai, X.; Jia, X.; Zhao, P.; Wang, T.; Wang, J.; Huang, P.; He, L.; Hou, X. A combined experimental/computational study on metal-organic framework MIL-101(Cr) as a SPE sorbent for the determination of sulphonamides in environmental water samples coupling with UPLC-MS/MS. Talanta CrossRef. 2016, 154, 581–588. [Google Scholar] [CrossRef]
- Chopra, S.; Dhumal, S.; Abeli, P.; Beaudry, R.; Almenar, E. Metal-organic frameworks have utility in adsorption and release of ethylene and 1-methylcyclopropene in fresh produce packaging. Postharvest Biology and Technology CrossRef. 2017, 130, 48–55. [Google Scholar] [CrossRef]
- Sheng, K.; Zheng, X.; Gao, Y.; Long, J.; Bao, T.; Wang, S. Fabrication of metal-organic frameworks containing Cu2+ coated medical PVC catheters with durable antiplatelet and antibacterial activities. Colloids and Surfaces A: Physicochemical and Engineering Aspects CrossRef. 2024, 700, 134779. [Google Scholar] [CrossRef]
- Zou, M.; Dong, M.; Zhao, T. Advances in Metal-Organic Frameworks MIL-101(Cr). Int J Mol Sci CrossRef. 2022, 23(16), 9396. [Google Scholar] [CrossRef]
- Gangu, K.K.; Maddila, S.; Jonnalagadda, S.B. The pioneering role of metal–organic framework-5 in ever-growing contemporary applications – a review. RSC Adv CrossRef. 2022, 12(22), 14282–14298. [Google Scholar] [CrossRef]
- Schoofs, H.; Schmit, J.; Rink, L. Zinc Toxicity: Understanding the Limits. Molecules CrossRef. 2024, 29(13), 3130. [Google Scholar] [CrossRef]
- Elmehrath, S.; Ahsan, K.; Munawar, N.; Alzamly, A.; Nguyen, H.L.; Greish, Y. Antibacterial efficacy of copper-based metal–organic frameworks against Escherichia coli and Lactobacillus. RSC Adv CrossRef. 2024, 14(22), 15821–15831. [Google Scholar] [CrossRef]
- Chen, G.; Leng, X.; Luo, J.; You, L.; Qu, C.; Dong, X.; Huang, H.; Yin, X.; Ni, J. In Vitro Toxicity Study of a Porous Iron(III) Metal‒Organic Framework. Molecules CrossRef. 2019, 24(7), 1211. [Google Scholar] [CrossRef]

| Metal Dopant | In Situ Doping Method | Max. Doping Achieved | Structural Effects | References |
|---|---|---|---|---|
| Co (Zn/Co ZIF-8) | One-pot room-temp co-precipitation (vary Zn:Co) | ~50% Co in framework (ZIF-67/ZIF-8 mix) | Retained sodalite ZIF structure; increased pore size, +Visible absorption band; slight lattice contraction at high Co | Vatani et al., 2024; Kolsani et al., 2022 (stability study) |
| Cu (Zn/Cu ZIF-8) | One-pot co-precipitation in methanol or water | ~40% Cu (higher leads to amorphous phases) | Retained ZIF-8 structure at ≤30% Cu; slight peak shifts; possibly improved hydrostability at low Cu doping | Nguyen et al., 2025; Cheng et al., 2022 |
| Ni (Zn/Ni ZIF-8) | One-pot (like Co, Cu) | ~100% (Ni-ZIF-8 known) | Isostructural ZIF-8 type possible (Ni2+ with 2-mIm); Ni substitution narrows bandgap like Co | Li et al., 2023 |
| Fe (Zn/Fe ZIF-8) | One-pot, often slow add or room-temp aging | ~20% (higher leads to ZIF-8 + amorphous Fe phases) | Largely ZIF-8 structure with peak broadening; Fe likely partially substitutes Zn and partially forms extra-framework nodes | Wang et al., 2021; Nguyen et al., 2025 |
| Ag (Ag/Zn ZIF-8) | One-pot or rapid mix (AgNO3 + Zn2+) – uncommon (Ag more often post-loaded) | low% as true dopant (Ag+ can coordinate imidazole weakly) | Mostly ZIF-8 phase; Ag+ may occupy surface sites or defects; minimal XRD change, indicating framework intact | Usman et al., 2023 (Ag in ZIF-8 electrocatalyst) |
| Mechanism | Description | Enhanced By | Example System (Reference) |
|---|---|---|---|
| Controlled metal ion release | Gradual liberation of Zn2+ and dopant ions (Ag+, Cu2+, etc.) that bind to bacterial cell components and disrupt physiological processes. It leads to enzyme inhibition, membrane destabilization, and oxidative stress inside cells. | Metal doping (Ag, Cu), acidic triggers, MOF porosity controlling release rate. | Ag/H-ZIF-8 releasing Zn2++Ag+ killed E. coli & S. aureus more effectively than either ion alone; Cu–ZIF-8 on lettuce released Cu2+ causing 3-log kill in 2 min. |
| Membrane contact disruption | Nanoparticles attach to bacterial cell walls, exert mechanical pressure or create nano-damage, leading to loss of membrane integrity and leakage of cell contents. Also facilitates entry of other agents. | Cationic polymer/peptide coating (improves adhesion); rough/hierarchical particle surface. | ZIF-8@chitosan adhered to E. coli cell membranes causing visible roughness and lysis (SEM); PDA-coated ZIF-8 showed strong binding to S. aureus and boosted killing via membrane rupture. |
| ROS generation (photocatalysis) | Production of reactive oxygen species (1O2, ·OH, O2−·) that oxidize and damage proteins, DNA, and lipids. Can occur under light (photocatalytic) or via enzyme-mimic reactions in dark. | Doping with photocatalytic metals (Co, Fe, Cu); incorporation of noble metal NPs; presence of H2O2 or light. | 5% Co–ZIF-8 under sunlight yielded 1O2 and O2−·, giving 6.6-log E. coli kill (vs <2-log by ZIF-8). Ag/Cu–ZIF-8@CNF filter generated 2.7× more ·OH under light than undoped, enhancing anti-biofilm efficacy. |
| Synergistic additive effects | Combined action with other antimicrobial agents (antibiotics, essential oils, enzymes). ZIF-8 acts as carrier and co-bactericide, weakening microbes so the additive works better; or providing dual function (e.g., enzyme + metal ion). | Loading of antibiotics or natural antimicrobials into ZIF pores; co-immobilization of enzymes or peptides; stimuli-responsive release triggers. | Cinnamaldehyde-loaded ZIF-8 in PVA film completely inactivated E. coli on spinach via Zn2+ + essential oil release. LYZ-Lactoferrin@ZIF-8 composite eradicated bacteria through combined cell wall hydrolysis, nutrient sequestration, Zn toxicity, and light-activated ROS. |
| Polymer | Integration with ZIF-8 | Benefit for Antimicrobial Use | Example |
|---|---|---|---|
| Chitosan (CHI) | Coating on ZIF-8 nanoparticles; blending ZIF-8 into chitosan films or beads. | CHI’s cationic nature improves bacterial adhesion and contact killing; film-forming for coatings; CHI itself is antimicrobial and edible (food-safe). | ZIF-8@chitosan coating on fruit delayed microbial spoilage, as composite coatings slowed mold/bacteria growth on cherry tomatoes. Gelatin/CHI films with Ag@ZIF-8@CMC achieved 100% bacterial kill and extended fruit shelf-life. |
| Polyvinyl Alcohol (PVA) | Mixing or in situ ZIF-8 growth in PVA solution; casting films. | Improves dispersion of ZIF-8 in aqueous media; PVA is flexible and forms transparent films; can incorporate other additives (glycerol, etc.) for packaging. | PVA-ZIF-8@cinnamaldehyde films showed dose-dependent E. coli inhibition on produce, complete kill at ≥4% loading. PVA/Quaternary ammonium polymer matrix with ZIF-8@eugenol yielded pH-responsive release and ~88% bacterial reduction. |
| Polydopamine (PDA) | Ultrathin coating on ZIF-8 surfaces (via dopamine self-polymerization). | Enhances water stability by chelating Zn; provides bioadhesive catechol groups for sticking to surfaces (and bacteria); introduces slight positive charge; can further conjugate other molecules (via PDA chemistry). | ZIF-8@PDA (core–shell) acted as a Zn2+ reservoir with pH-triggered release; showed superior S. aureus killing due to PDA-mediated cell binding. PDA coating on ZIF also allowed further attachment of hyaluronic acid in an OEO-Ag-ZIF-8-HA system, improving stability and biocompatibility. |
| Cellulose (e.g., Nanocellulose fibers, CNC/CNF) | In situ growth of ZIF-8 on cellulose nanofibers; embedding ZIF-8 in cellulose film or hydrogel. | Cellulose provides biodegradable, mechanically strong support; improves handling of MOF powder; CNF networks can create porous hydrogels for filtration; cellulose can enhance adhesion to hydrophilic surfaces. | Ag-doped ZIF-8 grown on TEMPO-oxidized CNF yielded “necklace” nanofibers that were embedded in a CNF hydrogel, resulting in a flexible antibacterial filter that resisted biofouling (only 14% flux decline). Bacterial cellulose loaded with ZIF-8 was proposed as an antimicrobial wound dressing that releases Zn2+ to prevent infection. |
| Food-grade polymers (PLA, PLGA, etc.) | Mixed-matrix composites; electrospun fibers with MOF; coating MOF with polyesters. | These polyesters are safe for food contact; incorporate MOFs to create active packaging or antimicrobial coatings that are also biodegradable; can tailor release profiles by polymer crystallinity. | PLA films containing ZIF-8@cinnamaldehyde nanoparticles significantly reduced S. enterica on meat surfaces (due to vapor-phase cinnamaldehyde release and Zn2+ action) – demonstrated in a 2022 study. PLA/PCL core–shell fibers with ZIF-8 in core and antibiotic in shell provided multi-stage antimicrobial release for medical textiles. |
| Hybrid Composition | Description | Antimicrobial Advantages | Example |
|---|---|---|---|
| Ag@ZIF-8 core–shell | Ag nanowire or nanoparticle core, ZIF-8 shell grown around it. | Controlled Ag+ release, prevention of Ag aggregation, dual ion (Ag+/Zn2+) synergy. Shell porous to allow ion diffusion. | Ag@ZIF-8 nanowires inhibited E. coli and B. subtilis far more than Ag nanowires alone. ZIF-8 shell prolonged Ag+ release (active over several days). |
| ZIF-8@GO composite | ZIF-8 nanoparticles deposited on graphene oxide sheets (or mixed in GO dispersion). | GO provides support and can wrap bacteria, some inherent antimicrobial via oxidative stress; ZIF-8 adds metal ion release. Composite can be formed into coatings or papers. | ZIF-8@GO achieved complete growth inhibition of E. coli, S. aureus in paper disk diffusion, whereas GO alone was bacteriostatic not bactericidal. GO improved dispersion of ZIF-8 in water and the composite could be easily filtered out. |
| Magnetic ZIF-8 (e.g., Fe3O4/ZIF-8) | Either ZIF-8 grown on Fe3O4 nanoparticles or vice versa. Often also coated with silica or polymer for stability. | Magnetic separability from liquids; Fe3O4 can generate •OH from H2O2 (peroxidase mimic); can heat under AC magnetic field (potentially useful for biofilm disruption via localized heating). | Fe3O4@ZIF-8 modified with plant extract showed strong antibacterial effect and could be magnetically recovered and reused 5× with minimal loss of activity. Magnetically retrievable MOFs avoid nanoparticle residues in treated water. |
| ZIF-8/cellulose hybrid | ZIF-8 grown in situ on cellulose fibers (cotton, nanocellulose, bacterial cellulose). | Robust, flexible substrates; easy shaping into films or filters; cellulose improves biocompatibility; ZIF-8 imparts active functionality that cellulose lacks. | Cotton fabric coated with ZIF-8 (via in situ crystallization) was turned into an antibacterial surface that inactivated >99.9% of E. coli on contact, yet fabric was washable and reusable due to strong MOF anchoring (Chen et al., 2019). Cellulose-ZIF-8 filters in Section 6 show biofouling resistance. |
| MOF-on-MOF or MOF-on-oxide hybrid | Layering or integrating ZIF-8 with another framework or porous oxide. | Can combine different pore sizes (capture diverse targets) or add photocatalytic function (e.g., TiO2, g-C3N4) to ZIF-8’s ion release. Potential for sequential action: one component adsorbs toxins while other kills microbes. | Ag3PO4@ZIF-8 hybrid showed excellent photocatalytic disinfection and pollutant degradation simultaneously. g-C3N4@ZIF-8 composites have been tested for synergistic light-driven antibacterial activity (ZIF adsorbs and concentrates bacteria around g-C3N4 sites). |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




