Submitted:
25 February 2026
Posted:
26 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
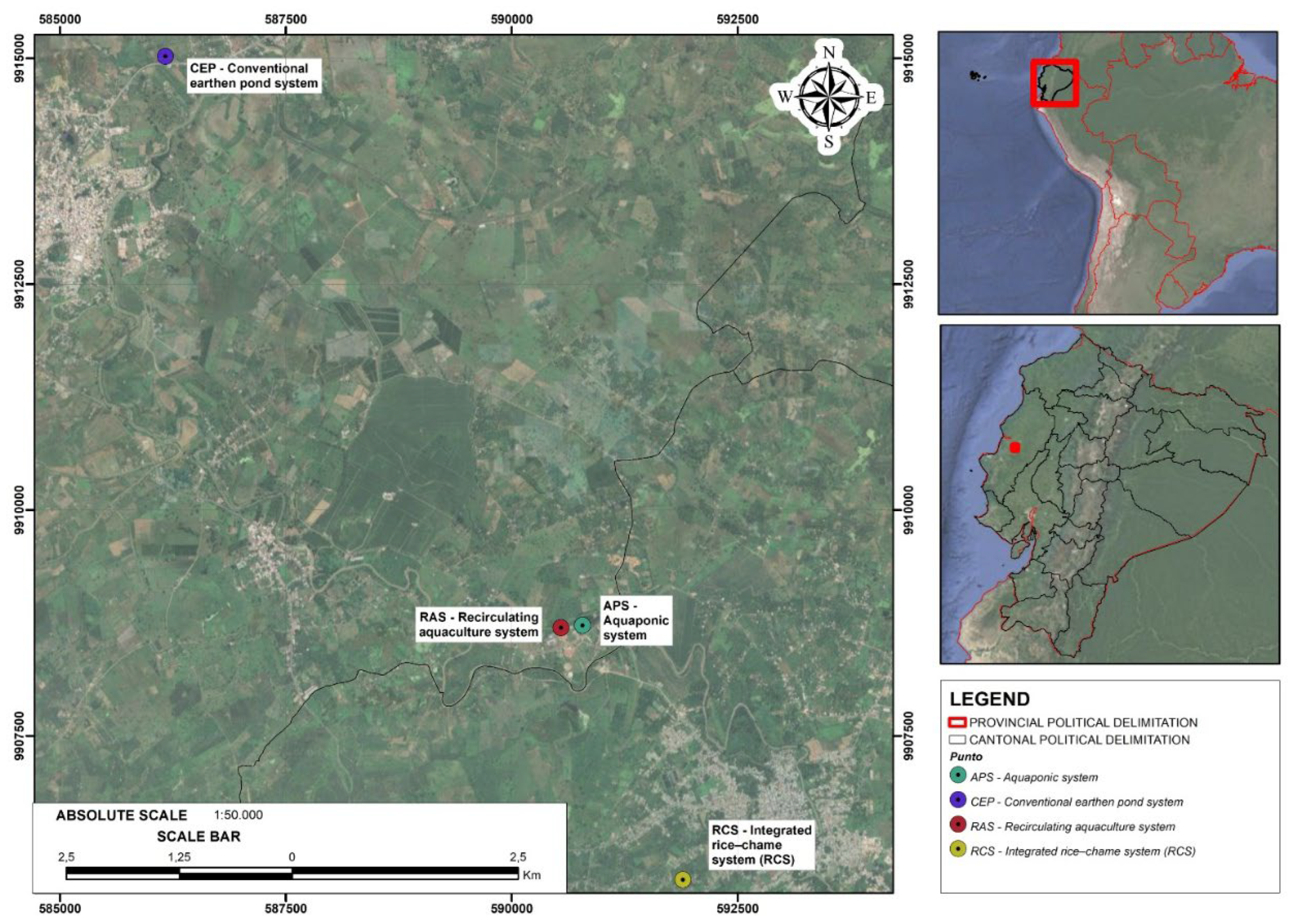
2.1. Study Area and Experimental Design
2.2. Production Systems and Animal Management
2.3. Water Sampling and Quality Parameters
2.4. Statistical Analysis
3. Results
3.1. Water Quality Patterns
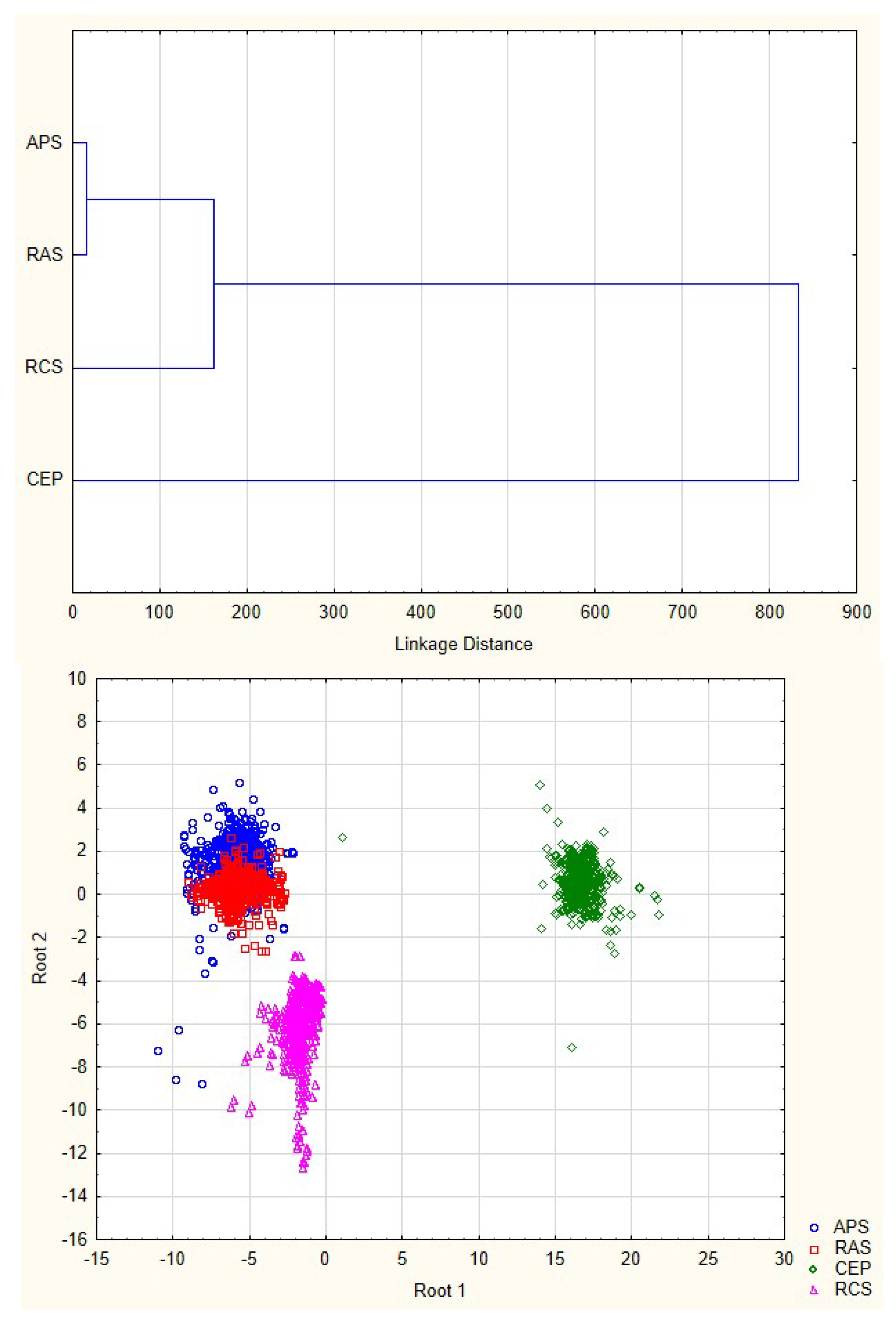
3.2. Discriminant Analysis of Water Quality Parameters
4. Discussion
4.1. Water Quality Patterns and Intensification Gradient in Tropical Pond-Based Systems
4.2. Sustainability Implications, Management Strategies, and Study Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bostock, J.; McAndrew, B.; Richards, R.; Jauncey, K.; Telfer, T.; Lorenzen, K.; Little, D.; Ross, L.; Handisyde, N.; Gatward, I.; Corner, R. Aquaculture: Global status and trends. Philos. Trans. R. Soc. B 2010, 365, 2897–2912. [Google Scholar] [CrossRef]
- FAO. (2022). El estado mundial de la pesca y la acuicultura 2022. In El estado mundial de la pesca y la acuicultura 2022. Roma. 288 p. Available online: https://doi.org/10.4060/cc0461es (accessed on 10 February 2026).
- Partelow, S.; Nagel, B.; Gentry, R.; Gephart, J.; Rocha, J.C. Archetypes of aquaculture development across 150 countries. Aquaculture 2025, 595, 741484. [Google Scholar] [CrossRef]
- Troell, M.; Naylor, R.L.; Metian, M.; Beveridge, M.; Tyedmers, P.H.; Folke, C.; Arrow, K.J.; Barrett, S.; Crépin, A.-S.; Ehrlich, P.R.; et al. Does aquaculture add resilience to the global food system? Proc. Natl. Acad. Sci. USA 2014, 111, 13257–13263. [Google Scholar] [CrossRef]
- Troell, M.; Costa-Pierce, B.; Stead, S.; Cottrell, R.S.; Brugere, C.; Farmery, A.K.; Little, D.C.; Strand, Å.; Pullin, R.; Soto, D.; et al. Perspectives on aquaculture’s contribution to the Sustainable Development Goals. J. World Aquac. Soc. 2023, 54, 882–905. [Google Scholar] [CrossRef]
- Ahmed, N.; Turchini, G.M. The evolution of the blue-green revolution of rice-fish cultivation for sustainable food production. Sustain. Sci. 2021, 16, 1375–1390. [Google Scholar] [CrossRef]
- Garlock, T.M.; Asche, F.; Anderson, J.L.; Eggert, H.; Anderson, T.M.; Che, B.; Chávez, C.A.; Chu, J.; Chukwuone, N.; Dey, M.M.; et al. Environmental, economic, and social sustainability in aquaculture: The aquaculture performance indicators. Nat. Commun. 2024, 15, 5274. [Google Scholar] [CrossRef]
- Boyd, C. E.; Tucker, C. S. Pond aquaculture water quality management. Springer. 1998. [Google Scholar] [CrossRef]
- Páez-Osuna, F. The environmental impact of shrimp aquaculture: Causes, effects, and mitigating alternatives. Environ. Manag. 2017, 60, 954–970. [Google Scholar] [CrossRef]
- Hargreaves, J.A. Nitrogen biogeochemistry of aquaculture ponds. Aquaculture 1998, 166, 181–212. [Google Scholar] [CrossRef]
- Yang, P.; Zhao, G.; Tong, C.; Tang, K.W.; Lai, D.Y.F.; Li, L.; Tang, C. Assessing nutrient budgets and environmental impacts of coastal land-based aquaculture systems in southeastern China. Agric. Ecosyst. Environ. 2021, 322, 107662. [Google Scholar] [CrossRef]
- Boyd, C.E. Water Quality: An Introduction; Springer: Cham, Switzerland, 2020. [Google Scholar]
- Wetzel, R.G. Limnology: Lake and River Ecosystems, 3rd ed.; Academic Press: San Diego, CA, USA, 2001. [Google Scholar]
- Oberle, M.; Salomon, S.; Ehrmaier, B.; Richter, P.; Lebert, M.; Strauch, S.M. Diurnal stratification of oxygen in shallow aquaculture ponds in central Europe and recommendations for optimal aeration. Aquaculture 2019, 501, 482–487. [Google Scholar] [CrossRef]
- Duan, J.; Zhang, Y.; Li, X.; Liu, H.; Chen, Y. Multivariate assessment of water quality dynamics in pond aquaculture systems under different management regimes. Aquaculture 2024, 585, 740771. [Google Scholar] [CrossRef]
- Hossain, M.B.; Lipi, J.A.; Pingki, F.H.; Sarker, M.M.; Nur, A.-A.-U.; Islam, M.M.; Albeshr, M.F.; Arai, T. Traditional fish farming based on indigenous knowledge in homestead ponds can uplift socioeconomic status and sustainability. Sustainability 2023, 15, 13583. [Google Scholar] [CrossRef]
- Haque, A.B.M.M.; Khan, M.A.; Hossain, M.M.; Hossain, M.E.; Nahiduzzaman, M.; Islam, M.S. Improved aquaculture management practices and their impact on small-scale rural aquaculture farmers in Bangladesh. Aquaculture 2025, 594, 741459. [Google Scholar] [CrossRef]
- Dixon, W.; Chiswell, B. Review of aquatic monitoring program design. Water Res. 1996, 30, 1935–1948. [Google Scholar] [CrossRef]
- Wenning, R.J.; Erickson, G.A. Interpretation and analysis of complex environmental data using chemometric methods. TrAC Trends Anal. Chem. 1994, 13, 446–457. [Google Scholar] [CrossRef]
- Legendre, P.; Legendre, L. Numerical Ecology, 3rd ed.; Elsevier: Amsterdam, The Netherlands, 2012. [Google Scholar]
- Johnson, R.A.; Wichern, D.W. Applied Multivariate Statistical Analysis, 6th ed.; Pearson: Upper Saddle River, NJ, USA, 2007. [Google Scholar]
- Henriksson, P.J.G.; Tran, N.; Mohan, C.V.; Chan, C.Y.; Rodriguez, U.P. Indonesian aquaculture futures—Evaluating environmental and socioeconomic potentials and constraints. J. World Aquac. Soc. 2018, 49, 857–876. [Google Scholar] [CrossRef]
- Dompreh, E.B.; Wang, Q.; Su, J.; Dam Lam, R.; Barman, B.K.; Rossignoli, C.M.; Gasparatos, A. Differentiated characteristics, sustainability performance and preferences among small-scale aquaculture producers: Implications for sustainable intensification. Sustain. Sci. 2025. [Google Scholar] [CrossRef]
- Diana, J.S. Aquaculture production and biodiversity conservation. BioScience 2009, 59, 27–38. [Google Scholar] [CrossRef]
- Aguirre, W.E.; Alvarez-Mieles, G.; Anaguano-Yancha, F.; Burgos Morán, R.; Cucalón, R.V.; Escobar-Camacho, D.; Jácome-Negrete, I.; Jiménez-Prado, P.; Laaz, E.; Miranda-Troya, K.; et al. Conservation threats and future prospects for the freshwater fishes of Ecuador: A hotspot of Neotropical fish diversity. J. Fish Biol. 2021, 99, 192–210. [Google Scholar] [CrossRef]
- Aréchiga-Palomera, M.A.; Nieves-Rodríguez, K.; Chong-Carrillo, O.; Nolasco-Soria, H.; Peña-Marín, E.; Álvarez-González, C.; Martínez-García, R.; López-Ramírez, S.; Ortiz-Galindo, J.L.; Vega-Villasante, F. Dormitator latifrons (Richardson, 1844), a Pacific fat sleeper: Review of published research, commercial importance and aquaculture potential. Lat. Am. J. Aquat. Res. 2022, 50, 451–460. [Google Scholar] [CrossRef]
- González, M.A.; Rodríguez, J.M.; Angón, E.; Martínez, A.; García, A.; Peña, F. Characterization of morphological and meristic traits and their variations between two populations (wild and cultured) of Cichlasoma festae. Arch. Tierz. 2016, 59, 435–444. [Google Scholar]
- Hernán, M.A.; Robins, J.M. Causal Inference: What If; Chapman & Hall/CRC: Boca Raton, FL, USA, 2020. [Google Scholar]
- Duan, N.; Yu, X.; Jiang, Z.; Chen, H.; Feng, H.; Kang, Y.; Ma, H.; Zhu, H. Impacts of an intensive traditional mariculture model on offshore environments as evidenced by dissolved organic matter and bacterial communities. Water Res. 2024, 267, 122530. [Google Scholar] [CrossRef]
- Gonzalez-Martinez, A.; Lopez, M.; Molero, H.M.; Rodriguez, J.; González, M.; Barba, C.; García, A. Morphometric and meristic characterization of native chame fish (Dormitator latifrons) in Ecuador using multivariate analysis. Animals 2020, 10, 1805. [Google Scholar] [CrossRef]
- Rodríguez, J.; Angón, E.; González, M.; Perea, J.; Barba, C.; García, A. Allometric relationship and growth models of juveniles of Cichlasoma festae (Perciformes: Cichlidae), a freshwater species native to Ecuador. Rev. Biol. Trop. 2017, 65, 1185–1193. [Google Scholar] [CrossRef]
- Tacon, A.G.J.; Metian, M. Fish matters: Importance of aquatic foods in human nutrition and global food supply. Rev. Fish. Sci. 2013, 21, 22–38. [Google Scholar] [CrossRef]
- Goddek, S.; Delaide, B.; Mankasingh, U.; Ragnarsdottir, K.V.; Jijakli, M.H.; Thorarinsdottir, R. Challenges of sustainable and commercial aquaponics. Aquac. Int. 2015, 23, 1–20. [Google Scholar] [CrossRef]
- Goddek, S.; Joyce, A.; Kotzen, B.; Burnell, G.M. (Eds.) Aquaponics Food Production Systems; Springer: Cham, Switzerland, 2019. [Google Scholar] [CrossRef]
- Wongkiew, S.; Hu, Z.; Chandran, K.; Lee, J.W.; Khanal, S.K. Nitrogen transformations in aquaponic systems: A review. Aquac. Eng. 2017, 76, 9–19. [Google Scholar] [CrossRef]
- Timmons, M.B.; Ebeling, J.M. Recirculating Aquaculture; Cayuga Aqua Ventures: Ithaca, NY, USA, 2010. [Google Scholar]
- Summerfelt, S.T.; Penne, C.R. Design and Management of Conventional Recirculating Aquaculture Systems; Wiley-Blackwell: Hoboken, NJ, USA, 2018. [Google Scholar]
- Frei, M.; Becker, K. Integrated rice–fish farming: A new paradigm for sustainable agriculture. Agric. Ecosyst. Environ. 2005, 37, 187–206. [Google Scholar] [CrossRef]
- López Vera, M.R. Caracterización del Chame (Dormitator latifrons) de la Subcuenca del Carrizal, Manabí, Ecuador. Master’s Thesis, University of Córdoba, Córdoba, Spain, 2017. [Google Scholar]
- APHA. Standard Methods for the Examination of Water and Wastewater, 23rd ed.; American Public Health Association: Washington, DC, USA, 2017. [Google Scholar]
- Mera Vélez, A.G.; Vélez Espinoza, J.J.; Delgado Villafuerte, C.R. Evaluación de la calidad de agua en sistemas acuapónicos para la producción de chame (Dormitator latifrons) en el área Agropecuaria, ESPAM-MFL, 2025. (Unpublished data).
- Eding, E.H.; Kamstra, A.; Verreth, J.A.J.; Huisman, E.A.; Klapwijk, A. Design and operation of nitrifying trickling filters in recirculating aquaculture: A review. Aquac. Eng. 2006, 34, 234–260. [Google Scholar] [CrossRef]
- Martins, C.I.M.; Eding, E.H.; Verdegem, M.C.J.; Heinsbroek, L.T.N.; Schneider, O.; Blancheton, J.-P.; Roque d’Orbcastel, E.; Verreth, J.A.J. New developments in recirculating aquaculture systems in Europe: A perspective on environmental sustainability. Aquac. Eng. 2010, 43, 83–93. [Google Scholar] [CrossRef]
- Badiola, M.; Mendiola, D.; Bostock, J. Recirculating aquaculture systems (RAS) analysis: Main issues on management and future challenges. Aquac. Eng. 2012, 51, 26–35. [Google Scholar] [CrossRef]
- Chen, S.; Ling, J.; Blancheton, J.-P. Nitrification kinetics of biofilm as affected by water quality factors. Aquac. Eng. 2006, 34, 179–197. [Google Scholar] [CrossRef]
- Estim, A.; Saufie, S.; Mustafa, S. Water quality remediation using aquaponics sub-systems as biological and mechanical filters in aquaculture. J. Water Process Eng. 2019, 30, 100566. [Google Scholar] [CrossRef]
- Sa’adah, W.; Hanani, N.; Muhaimin, A.W. Linking Livelihood Assets to Technical Efficiency: Empirical Evidence from Pond-Based Grouper Aquaculture in the Coastal Area of Lamongan, Indonesia. Sustainability 2026, 18, 1738. [Google Scholar] [CrossRef]

| Parameter | Analytical Method 1 |
| pH | Potentiometry |
| Turbidity (NTU) | Turbidimetry |
| Temperature (°C) | Temperature probe |
| Salinity (mg L⁻¹) | Conductimetry |
| Alkalinity (mg L⁻¹) | Volumetric titration |
| Dissolved oxygen (mg L⁻¹) | Oximetry |
| Color (UPC) | Spectrophotometry |
| Nitrites (mg L⁻¹) | Spectrophotometry |
| Phosphates (mg L⁻¹) | Spectrophotometry |
| Total dissolved solids (mg L⁻¹) | Conductimetry |
| Total hardness (mg L⁻¹) | Volumetric titration |
| Electrical conductivity (mS cm⁻¹) | Conductimetry |
| Parameters | Aquaculture Systems 1 | P_value | ||||
|
All (n = 3,544) |
APS (n = 1152) |
RAS (n = 1152) |
CEP (n = 840) |
RCS (n = 400) |
||
| pH | 7.94±0.01 (4.21) | 8.02±0.01 (2.33) b | 8.14±0.01 (2.39) a | 7.55±0.01 (4.52) d | 7.97±0.01 (3.58) c | < 0.001 |
| Total dissolved solids, mg/l | 890.06±9.97 (66.67) | 522.42±2.54 (16.50) c | 529.01±2.44 (15.63) c | 1.928.42±4.74 (7.12) a | 808.13±2.78 (6.87) b | < 0.001 |
| Temperature, °C | 27.40±0.03 (5.53) | 27.07±0.02 (2.44) c | 26.35±0.02 (3.12) d | 28.99±0.06 (5.61) a | 28.08±0.08 (5.41) b | < 0.001 |
| Dissolved oxygen, mg/l | 4.87±0.03 (40.63) | 3.88±0.01 (12.02) d | 4.06±0.02 (12.81) c | 5.79±0.09 (44.18) b | 8.12±0.08 (19.25) a | < 0.001 |
| Salinity, ppm | 1094.40±12.42 (67.57) | 644.25±3.40 (17.91) c | 638.07±3.35 (17.81) c | 2.391.06±4.65 (5.64) a | 982.11±2.81 (5.73) b | < 0.001 |
| Electrical conductivity, mS/cm | 1.70±0.02 (67.99) | 1.00±0.01 (17.91) c | 0.99±0.01 (17.79) c | 3.74±0.01 (5.71) a | 1.52±0.00 (5.73) b | < 0.001 |
| Turbidity, NTU | 8.72±0.14 (98.51) | 10.89±0.26 (82.46) b | 4.22±0.09 (70.42) d | 6.38±0.11 (51.79) c | 20.34±0.62 (61.19) a | < 0.001 |
| Chlorides, mg/l | 40.78±0.64 (92.76) | 39.94±0.95 (80.71) c | 48.25±0.62 (43.55) b | n.m. | 107.33±0.91 (17.05) a | < 0.001 |
| Alkalinity, mg/l | 176.03±4.98 (168.36) | 65.71±0.48 (24.69) c | 79.96±3.11 (132.21) c | 163.37±4.57 (81.05) b | 797.00±25.56 (64.14) a | < 0.001 |
| Colour, Pt/Co | 121.51±1.55 (76.06) | 202.54±2.83 (47.48) a | 83.80±1.70 (68.72) b | 85.86±1.57 (52.85) b | 71.65±4.20 (117.10) c | < 0.001 |
| Nitrate, mg/l | 0.61±0.04 (397.16) | 1.15±0.04 (130.15) a | 0.13±0.00 (54.66) b | 0.82±0.16 (560.40) b | 0.03±0.00 (31.41) c | < 0.001 |
| Phosphates, mg/l | 7.15±0.10 (80.57) | 12.56±0.14 (38.05) a | 8.73±0.02 (9.69) b | n.m. | 2.00±0.04 (44.90) c | < 0.001 |
| Ammonium, mg/l | 1.61±0.03 (101.34) | 2.08±0.06 (94.24) b | 0.59±0.01 (45.75) d | 2.49±0.06 (70.77) a | 1.32±0.03 (40.70) c | < 0.001 |
| Total hardness, mg/l | 764.93±9.24 (71.92) | 521.37±3.26 (21.22) b | 488.43±4.40 (30.58) c | 1.663.39±13.31 (23.18) a | 375.89±8.95 (47.63) d | < 0.001 |
| Parameters | Wilks’ Lambda | Partial Lambda | F-remove | p-level | Toler | 1-Toler |
| Electrical conductivity | 0.00 | 0.99 | 9.99 | < 0.001 | 0.01 | 0.99 |
| Turbidity | 0.00 | 0.77 | 354.19 | < 0.001 | 0.87 | 0.13 |
| Phosphates | 0.00 | 0.83 | 241.20 | < 0.001 | 0.92 | 0.08 |
| Chlorides | 0.00 | 0.78 | 328.78 | < 0.001 | 0.90 | 0.10 |
| Colour | 0.00 | 0.76 | 369.63 | < 0.001 | 0.74 | 0.26 |
| Total dissolved solids | 0.00 | 0.68 | 564.08 | < 0.001 | 0.48 | 0.52 |
| Dissolved oxygen | 0.00 | 0.87 | 179.33 | < 0.001 | 0.79 | 0.21 |
| Nitrates | 0.00 | 0.82 | 253.04 | < 0.001 | 0.52 | 0.48 |
| Turbidity | 0.00 | 0.85 | 199.39 | < 0.001 | 0.78 | 0.22 |
| Temperature | 0.00 | 0.82 | 254.61 | < 0.001 | 0.87 | 0.13 |
| pH | 0.00 | 0.88 | 159.13 | < 0.001 | 0.89 | 0.11 |
| Ammonium | 0.00 | 0.94 | 78.73 | < 0.001 | 0.85 | 0.15 |
| Total hardness | 0.00 | 0.97 | 36.62 | < 0.001 | 0.80 | 0.20 |
| Salinity | 0.00 | 0.99 | 6.67 | < 0.001 | 0.01 | 0.99 |
| System 1 | Percent | APS | RAS | CEP | RCS |
| APS | 84.81 | 977 | 171 | 0 | 4 |
| RAS | 98.78 | 14 | 1138 | 0 | 0 |
| CEP | 99.88 | 1 | 0 | 838 | 0 |
| RCS | 100.00 | 0 | 0 | 0 | 400 |
| Total | 94.64 | 992 | 1309 | 838 | 404 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




