Submitted:
23 February 2026
Posted:
26 February 2026
You are already at the latest version
Abstract
Keywords:
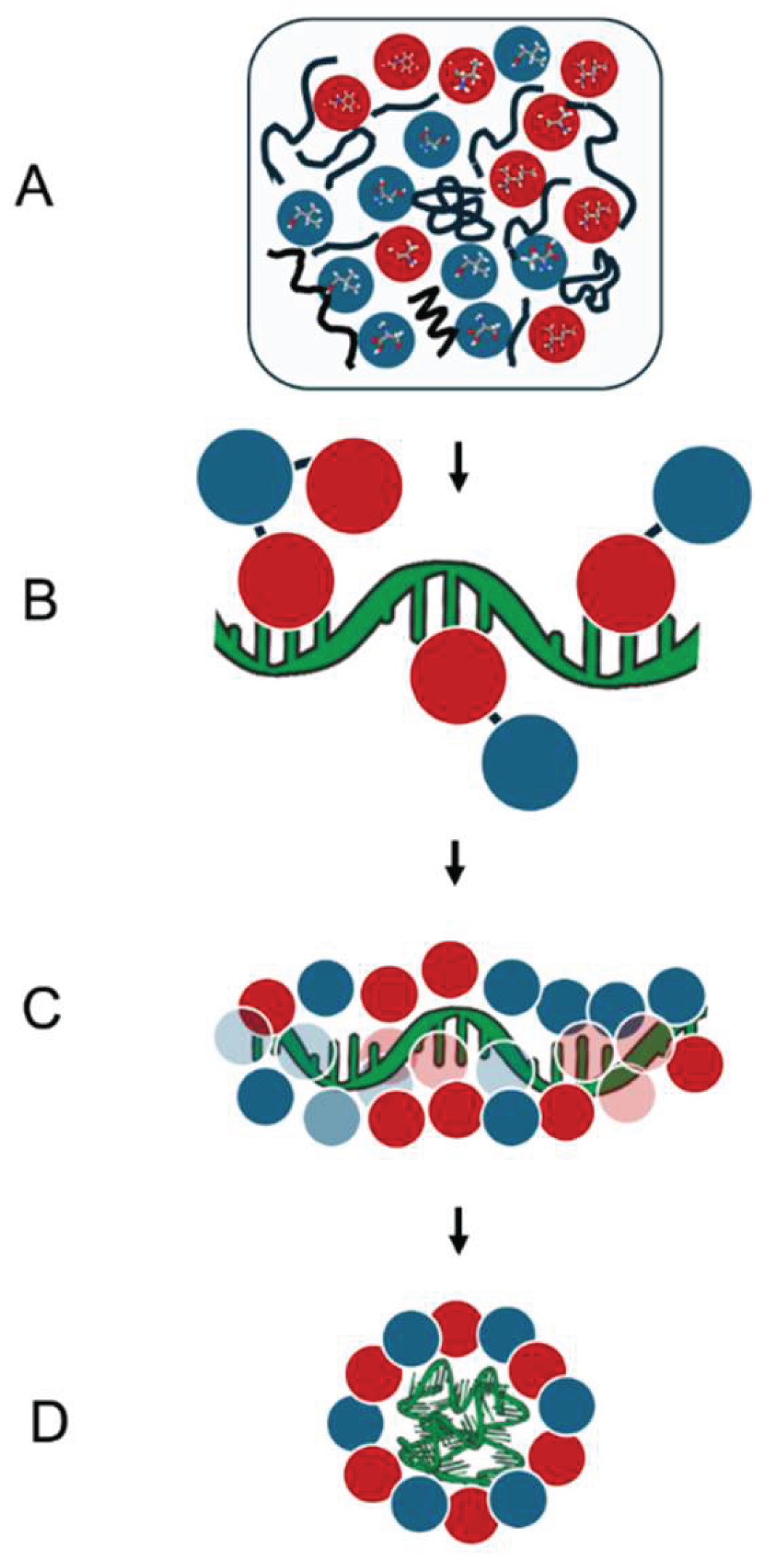
1. Introduction
2. Main Text
3. Conclusions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Declaration on the Use of Artificial Intelligence
References
- Alva V, Soding J, Lupas AN. 2015. A vocabulary of ancient peptides at the origin of folded proteins. eLife 4: e09410. [CrossRef]
- Bada JL. 2013. New insights into prebiotic chemistry from Stanley Miller’s spark discharge experiments. Chemical Society Reviews 42: 2186–96. [CrossRef]
- Blanco C, Bayas M, Yan F, Chen IA. 2018. Analysis of evolutionarily independent protein-RNA complexes yields a criterion to evaluate the relevance of prebiotic scenarios. Curr Biol 28: 526–537.e5. [CrossRef]
- Bushman FD. 2025. Virolithopanspermia: Might viruses be transported in rocks through space? PLoS Pathog 21: e1012955. [CrossRef]
- Cadena-Nava RD, Comas-Garcia M, Garmann RF, Rao ALN, Knobler CM, Gelbart WM. 2012. Self-assembly of viral capsid protein and RNA molecules of different sizes: requirement for a specific high protein/rna mass ratio. J Virol 86: 3318–3326. [CrossRef]
- Callahan MP, Smith KE, Cleaves HJ, Dworkin JP. 2011. Carbonaceous meteorites contain a wide range of extraterrestrial nucleobases. Proc Natl Acad Sci 108: 13995–13998. [CrossRef]
- Claverie JM. 2006. Viruses take center stage in cellular evolution. Genome Biology 7: 110. [CrossRef]
- Cold Spring Harbor Symposia on Quantitative Biology. 1966. The Genetic Code. Cold Spring Harb Symp Quant Biol 31.
- Crick FHC, Orgel LE. 1973. Directed Panspermia. Icarus 19: 341–346. [CrossRef]
- Eigen M, Schuster P. 1977. A principle of natural self-organization. Naturwissenschaften 64: 541–565.
- Forterre P. 2006. The origin of viruses and their possible roles in major evolutionary transitions. Virus Res 117: 5–6. [CrossRef]
- Gamow G. 1954. Possible relation between deoxyribonucleic acid and protein structures. Nature 173: 318. [CrossRef]
- Gilbert W. 1986. Origin of life: the RNA world. Nature 319: 618. [CrossRef]
- Giulio D. 1998. Reflections on the origin of the genetic code: a hypothesis. J Theor Biol 191: 191–196.
- Haldane JBS. 1929. The origin of life. Ration Annu 148: 3–10.
- Higgs PG. 2009. A four-column theory for the origin of the genetic code: tracing the evolutionary pathways that gave rise to an optimized code. Biol Direct 4: 16. [CrossRef]
- Higgs PG, Pudtritz RE. 2009. A Thermodynamic basis for prebiotic amino acid synthesis and the nature of the first genetic code. Astrobiology 9: 483–490.
- Hopfield JJ. 1978. Origin of the genetic code: a testable hypothesis based on tRNA structure, sequence, and kinetic proofreading. Proc Natl Acad Sci 75: 4334–4338. [CrossRef]
- Koonin EV, Dolja VV. 2014. Virus world as an evolutionary network of viruses and capsidless selfish elements. Microbiology and Molecular Biology Reviews 78: 278–303. [CrossRef]
- Koonin EV. 2014. The origins of cellular life. Antonie van Leeuwenhoek 106: 27–41. [CrossRef]
- Krupovic M, Koonin EV. 2017. Multiple origins of viral capsid proteins from cellular ancestors. Proc Natl Acad Sci 114: E2401–E2410. [CrossRef]
- Krupovic M, Dolja VV, Koonin EV. 2019. Origin of viruses: primordial replicators recruiting capsids from hosts. Nature Reviews Microbiology 17: 449–458.
- Lei L, Burton ZF. 2021. Evolution of the genetic code. Transcription 12: 28–53.
- Longo LM, Despotović D, Weil-Ktorza O, Walker MJ, Jabłońska J, Fridmann-Sirkis Y, Varani G, Metanis N, Tawfik DS. 2020. Primordial emergence of a nucleic acid-binding protein via phase separation and statistical ornithine-to-arginine conversion. Proc Natl Acad Sci 117: 15731–15739. [CrossRef]
- McLachlan AD, Bloomer AC, Butler PJG. 1980. Structural Repeats and Evolution of Tobacco Mosaic Virus Coat Protein and RNA. J Mol Biol 136: 203–224. [CrossRef]
- Miller SL. 1953. Production of amino acids under possible primitive Earth conditions. Science 117: 528–529. [CrossRef]
- Noller FH. 2012. Evolution of protein synthesis from an RNA world. Cold Spring Harb Perspect Biol 4: a003681.
- Frenkel-Pinter M, Haynes JW, Mohyeldin AM, Martin C, Sargon AB, Petrov AS, Krishnamurthy R, Hud NV, Williams LD, Leman LJ. 2020. Mutually stabilizing interactions between proto-peptides and RNA. Nat Commun 11: 3137. [CrossRef]
- Oparin AI. 1924. The origin of life. Izd Moskovskii Rabochii (In Russian).
- Oparin AI. 1936. The origin of life. Macmillan New York, NY.
- Poole AM, Jeffares DC, Penny D. 1998. The path from the RNA world. J Mol Evol 46: 1–17. [CrossRef]
- Raoult D, Forterre P. 2008. Redefining viruses: lessons from Mimivirus. Nat Rev Microbiol 6: 315–319. [CrossRef]
- Richardson ES. 1981. The Anatomy and Taxonomy of Protein Structure. Advances in Protein Chemistry 34: 167–339.
- Robertson MP, Joyce GF. 2012. The Origins of the RNA World. Cold Spring Harb Perspect Biol 4: a003608. [CrossRef]
- Root-Bernstein R, Kim Y, Sanjay A, Burton ZF. 2016. tRNA evolution from the proto-tRNA minihelix world. Transcription 7: 153–163. [CrossRef]
- Sonneborn TM. 1965. Degeneracy of the Genetic Code: Extent, Nature, and Genetic Implications. Evolving Genes and Proteins 1: 377–397.
- Szathmáry E. 1999. The origin of the genetic code: amino acids as cofactors in an RNA world. Trends in Genetics 15: 223–229. [CrossRef]
- Furubayashi T, Ueda K, Bansho Y, Motooka D, Nakamura S, Mizuuchi R, Ichihanshi N. 2020. Emergence and diversification of a host-parasite RNA ecosystem through Darwinian evolution. eLife 9: e56038. [CrossRef]
- van der Gulik PTS, Speijer D. 2015. How Amino acids and peptides shaped the RNA World. Life (Basel) 5: 230–246.
- Wehbi S, Wheeler A, Morel B, Manepalli N, Minh BQ, Lauretta DS, Masel J. 2024. Order of amino acid recruitment into the genetic code resolved by last universal common ancestor’s protein domains. Proc Natl Acad Sci U S A 121: e2412152121. [CrossRef]
- Woese CR. 1965. On the evolution of the genetic code. Proc Natl Acad Sci 54: 1546–1552.
- Woese CR. 1968. The fundamental nature of the genetic code: prebiotic interactions between polynucleotides and polyamino acids or their derivatives. Proc Natl Acad Sci 59: 110–117. [CrossRef]
- Wolf YI, Koonin EV. 2007. On the origin of the translation system and the genetic code in the RNA world by means of natural selection, exaptation, and subfunctionalization. Biology Direct 2: 14. [CrossRef]
- Wong JTF. 1975. A co-evolution theory of the genetic code. PNAS 72: 1909–1912. [CrossRef]
- Yagi S, Tagami S. 2024. An ancestral fold reveals the evolutionary link between RNA polymerase and ribosomal proteins. Nature Communications 15: 5938. [CrossRef]
- Yagi S, Padhi AK, Vucinic J, Barbe S, Schiex T, Nakagawa R, Simoncini D, Zhang KYJ, Tagami S. 2021. Seven amino acid types suffice to create the core fold of RNA polymerase. J Am Chem Soc 143: 15998–16006. [CrossRef]
- Yarus M, Caporaso JG, Knight R. 2005. Origins of the genetic code: the escaped triplet theory. Annual Review of Biochemistry 74: 179–198. [CrossRef]
- Ye L, Ambi UB, Olguin-Nava M, Gribling-Burrer AS, Ahmad S, Bohn P, Weber MM, Smyth RP. 2021. RNA Structures and Their Role in Selective Genome Packaging. Viruses 13: 1788. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



