Submitted:
25 February 2026
Posted:
26 February 2026
You are already at the latest version
Abstract
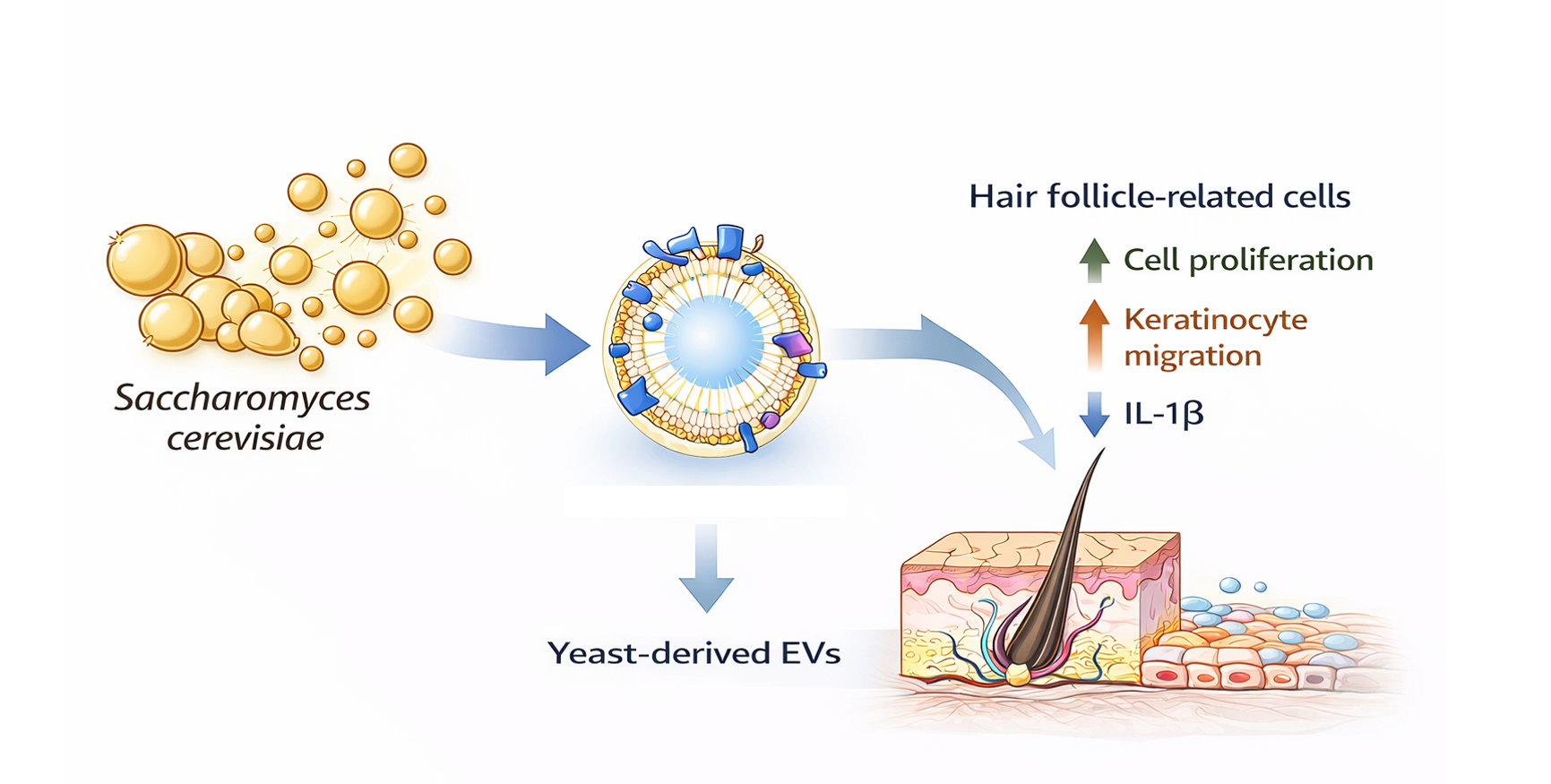
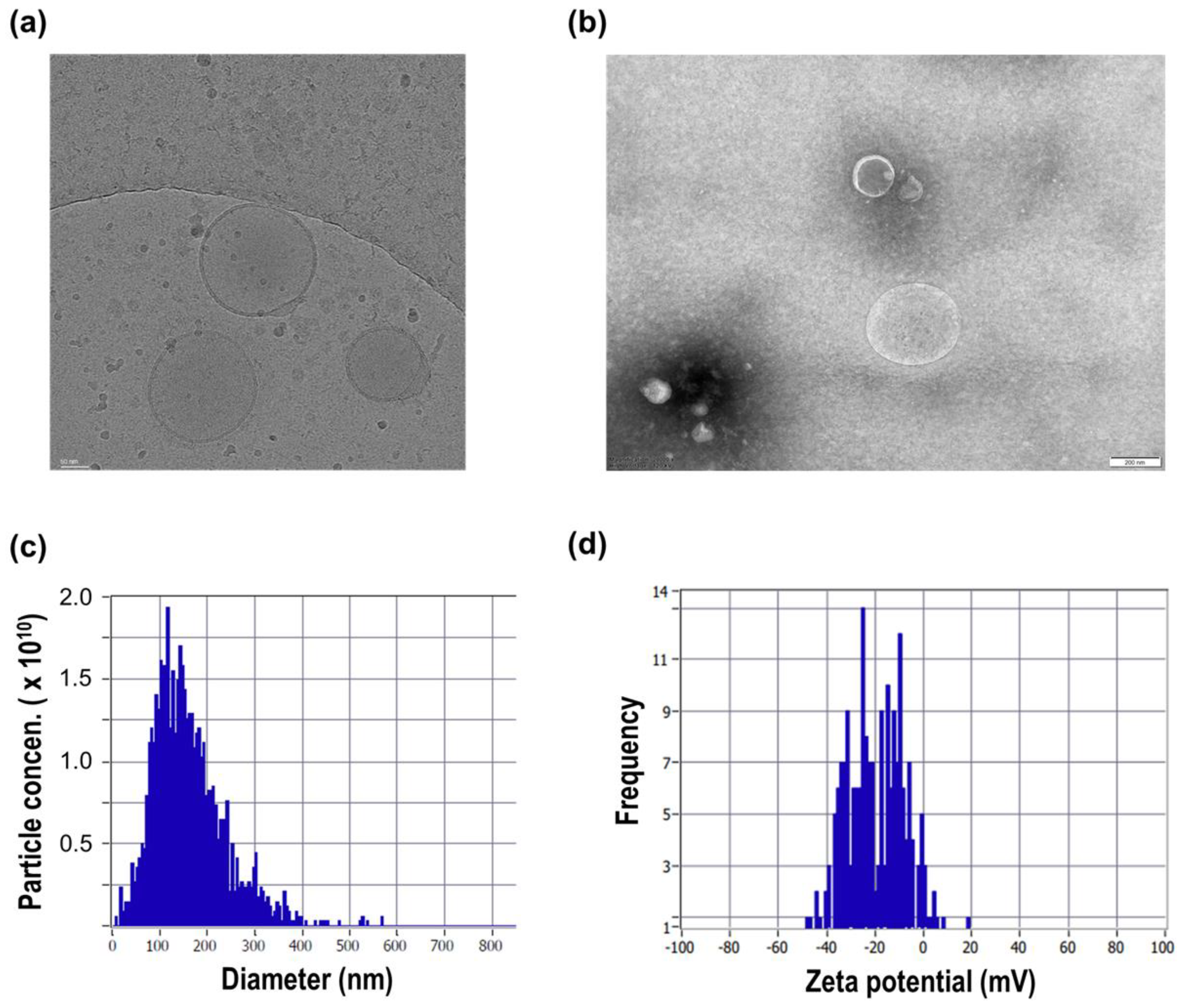
Extracellular vesicles (EVs) derived from microbial sources, including beer yeast (Saccharomyces cerevisiae), have recently attracted increasing attention as bioactive nanostructures with potential biomedical and cosmetic applications. In this study, extracellular vesicles were isolated from Saccharomyces cerevisiae (beer yeast) using an electrokinetic ion-binding filtration system followed by tangential flow filtration (TFF)–based buffer exchange. Their physicochemical characteristics and hair follicle–related biological activities were systematically evaluated. Nanoparticle tracking analysis demonstrated a mean particle size within the typical EV range, and zeta potential analysis confirmed a negatively charged surface. Cryo-transmission electron microscopy further verified the presence of lipid bilayer–enclosed nanovesicles. Biological activity was assessed in human dermal papilla cells, keratinocytes, and dermal fibroblasts, which collectively represent key components of the hair follicle microenvironment. At non-cytotoxic concentrations, yeast-derived EVs enhanced dermal papilla cell proliferation and promoted keratinocyte migration. The EVs attenuated pro-inflammatory cytokine expression under stimulated conditions and upregulated collagen-related gene expression in dermal fibroblasts. In addition, measurable antioxidant activity was observed. Collectively, these findings indicate that S. cerevisiae–derived extracellular vesicles exhibit multifunctional bioactivity relevant to the regulation of hair follicle–associated cellular processes. This study supports the potential of microbial EVs as scalable bioactive platforms for modulating hair follicle microenvironmental homeostasis.
Keywords:
1. Introduction
2. Materials and Methods
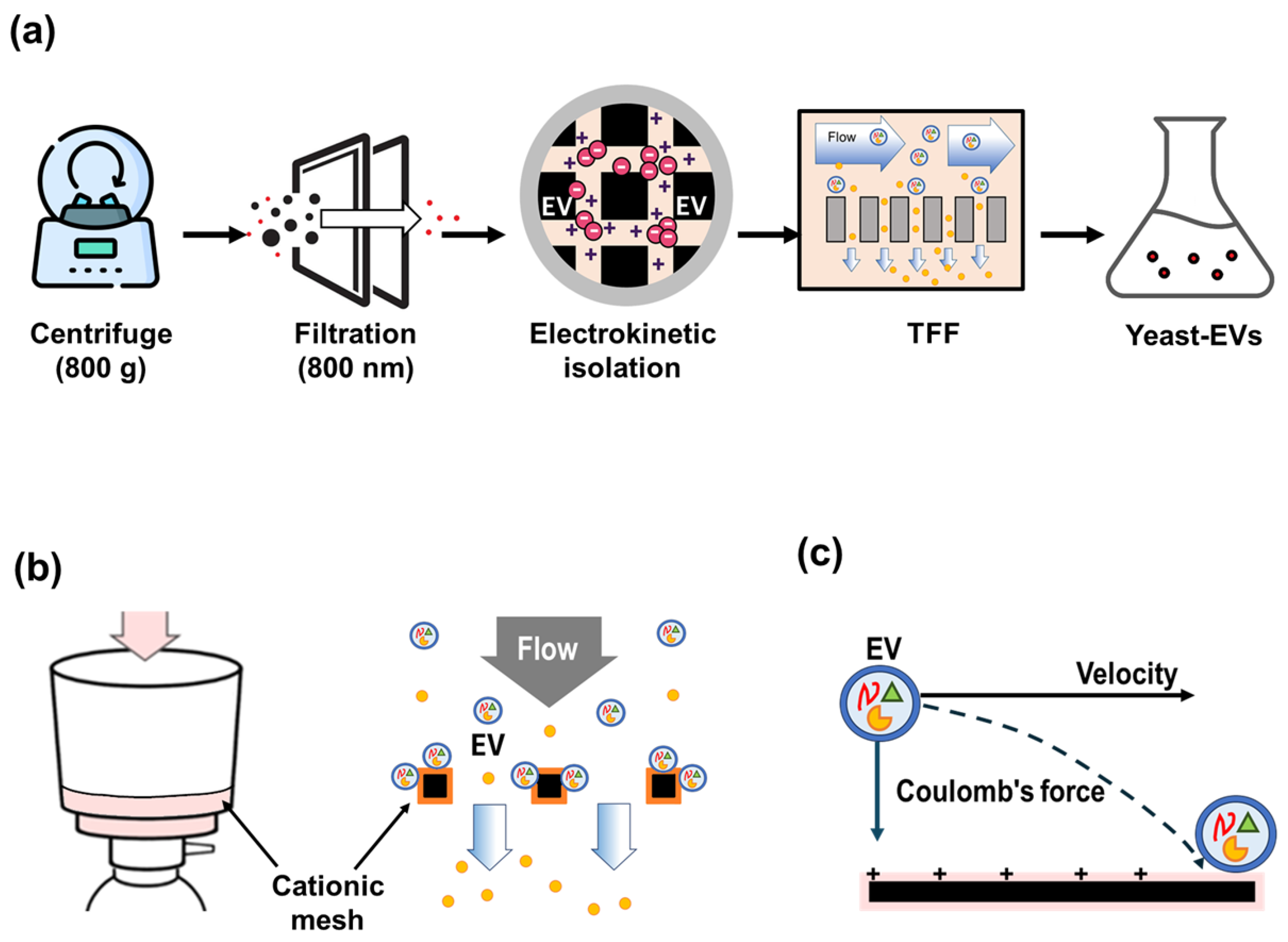
2.1. Isolation Saccharomyces cerevisiae–Derived Extracellular Vesicles
2.2. Electrokinetic EV Isolation Using ExoFilter
2.3. Physicochemical Characterization of EVs
2.4. Cell Culture
2.5. Cell Viability and Proliferation Assay
2.6. Wound Healing Assay
2.7. Inflammatory Cytokine Gene Expression Analysis
2.8. Collagen Gene Expression Analysis
2.9. Antioxidant Activity Assay
2.10. Statistical Analysis
3. Results
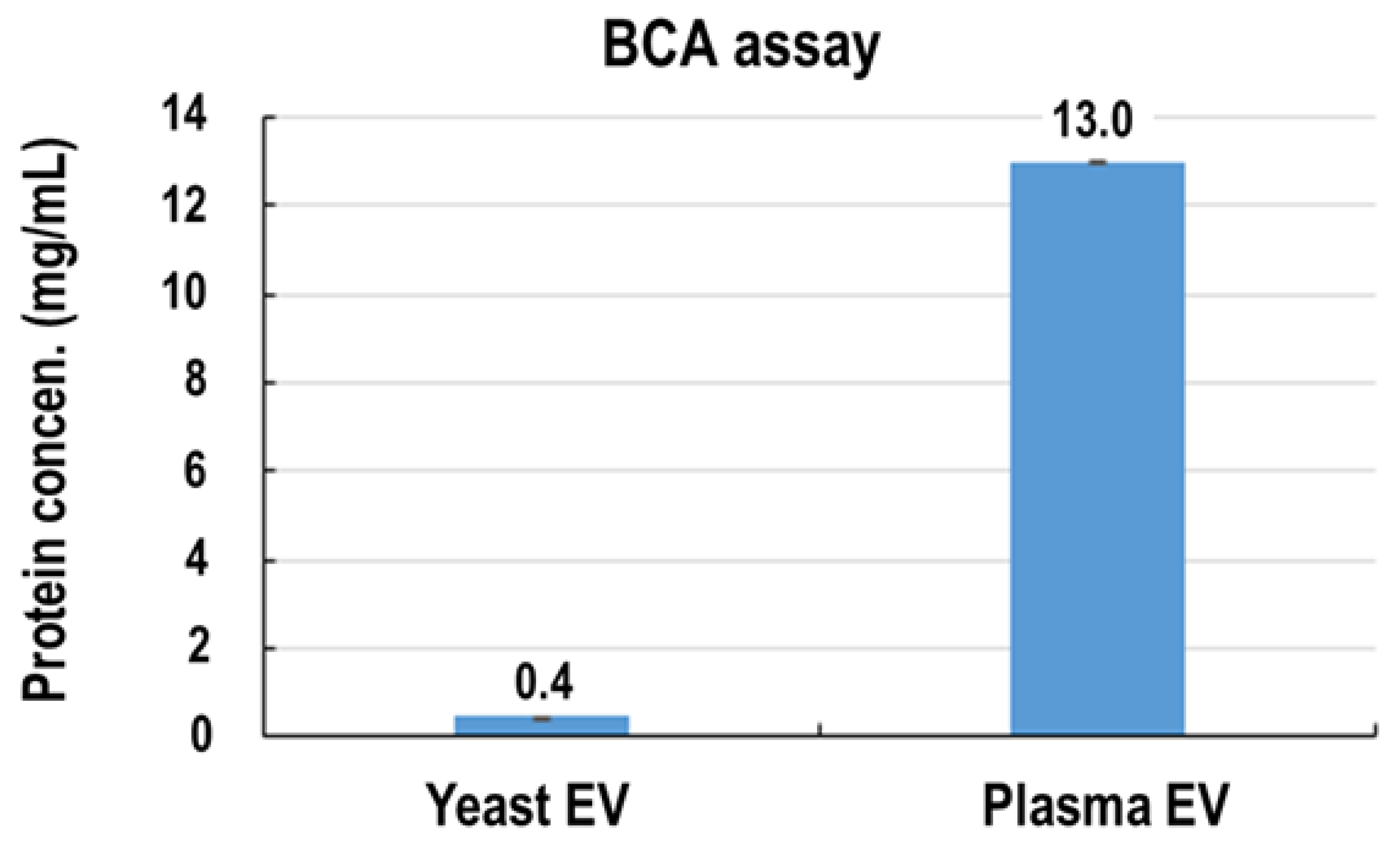
3.1. Isolation and Physicochemical Characterization of Yeast EVs
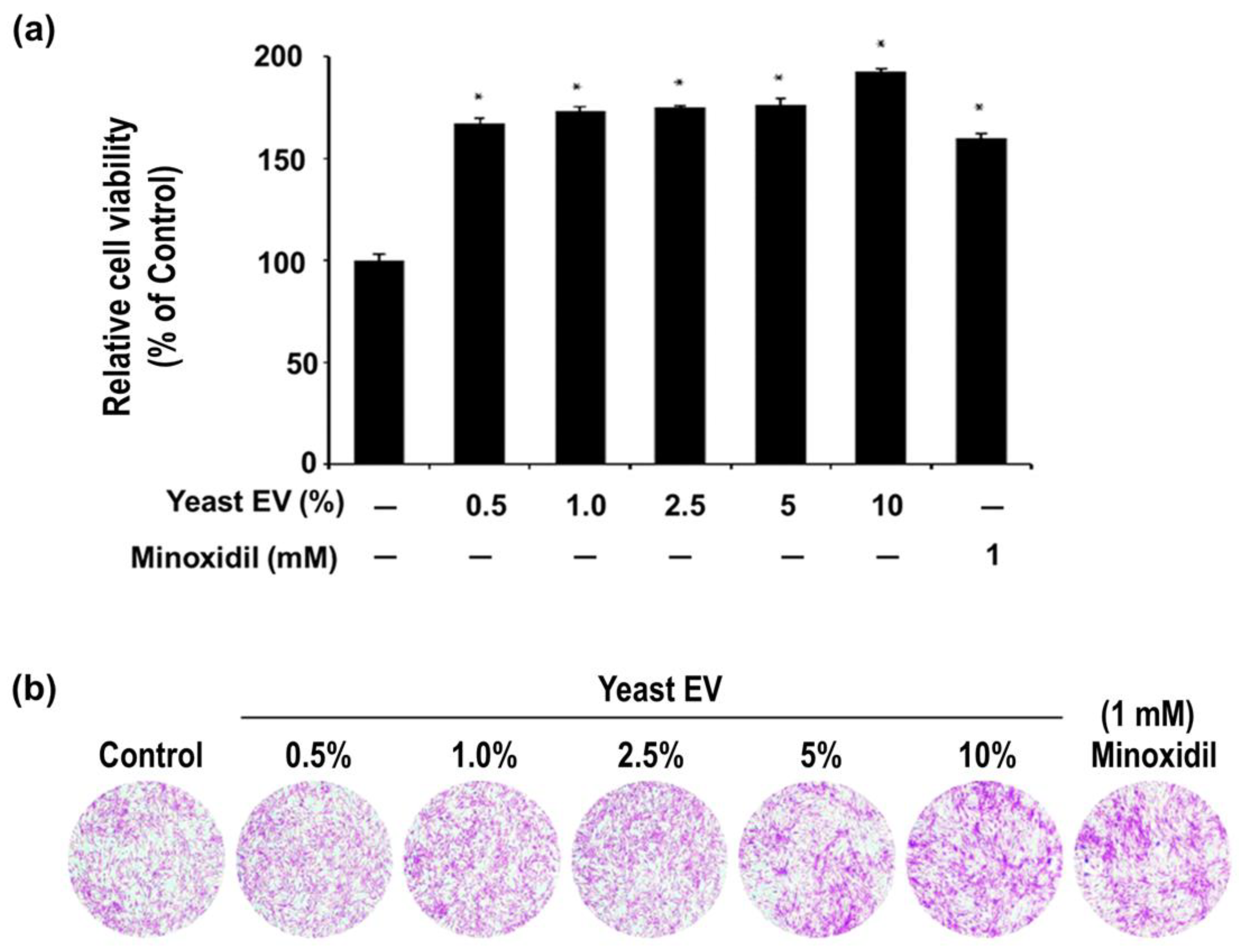
3.2. Effect of Yeast EVs on Dermal Papilla Cell Proliferation
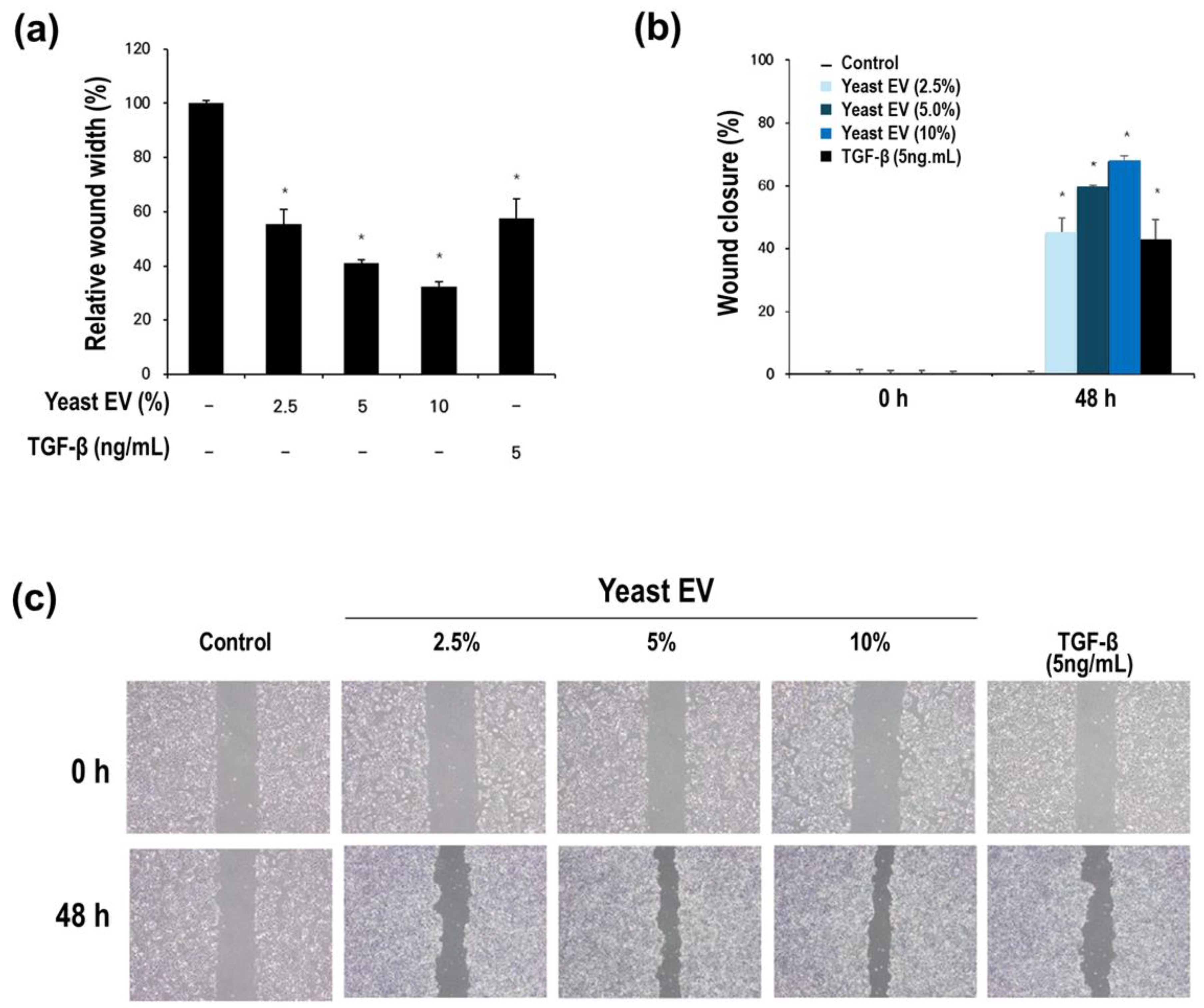
3.3. Effect of Yeast EVs on Keratinocyte Migration and Wound Closure
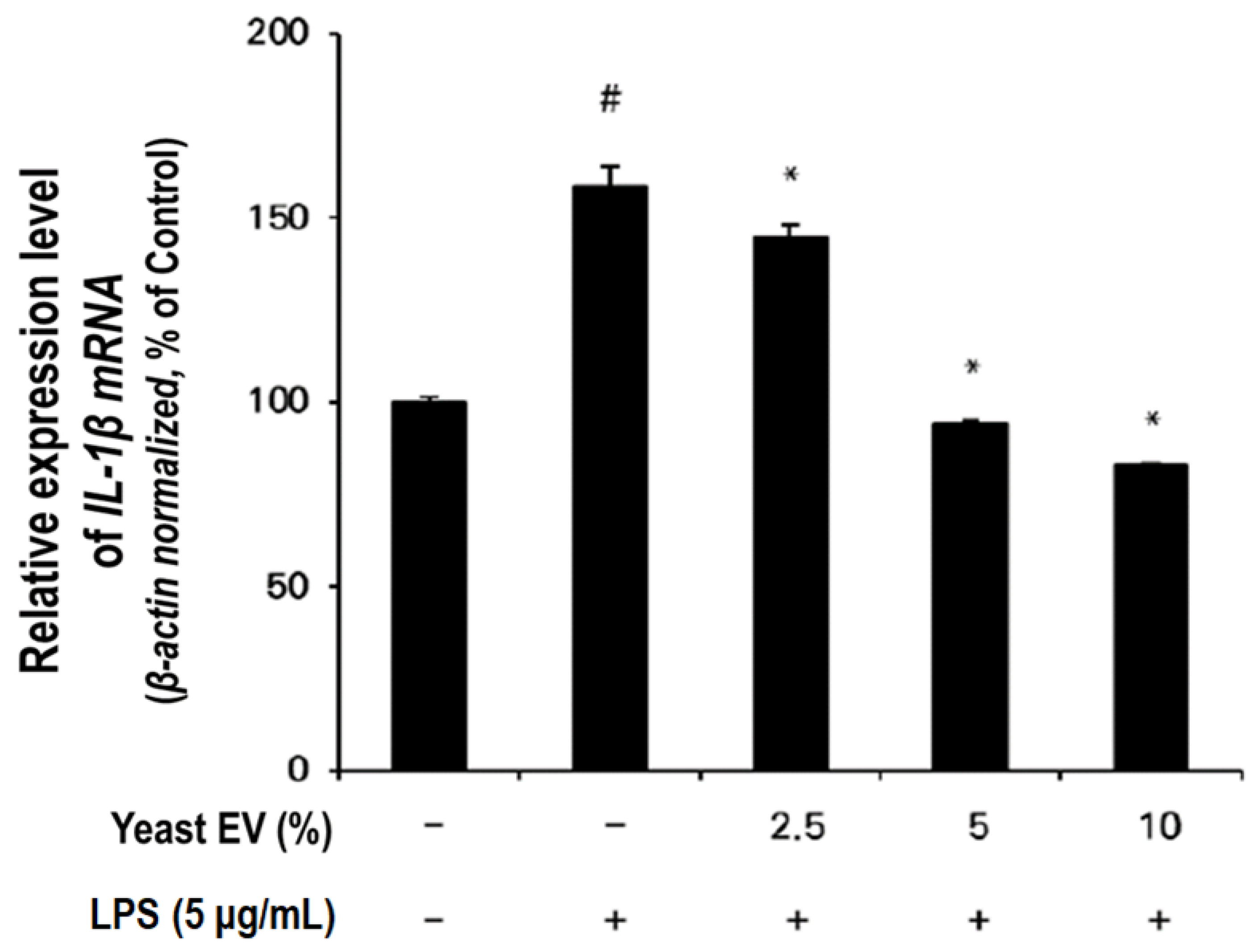
3.4. Anti-Inflammatory Effect of Yeast EVs
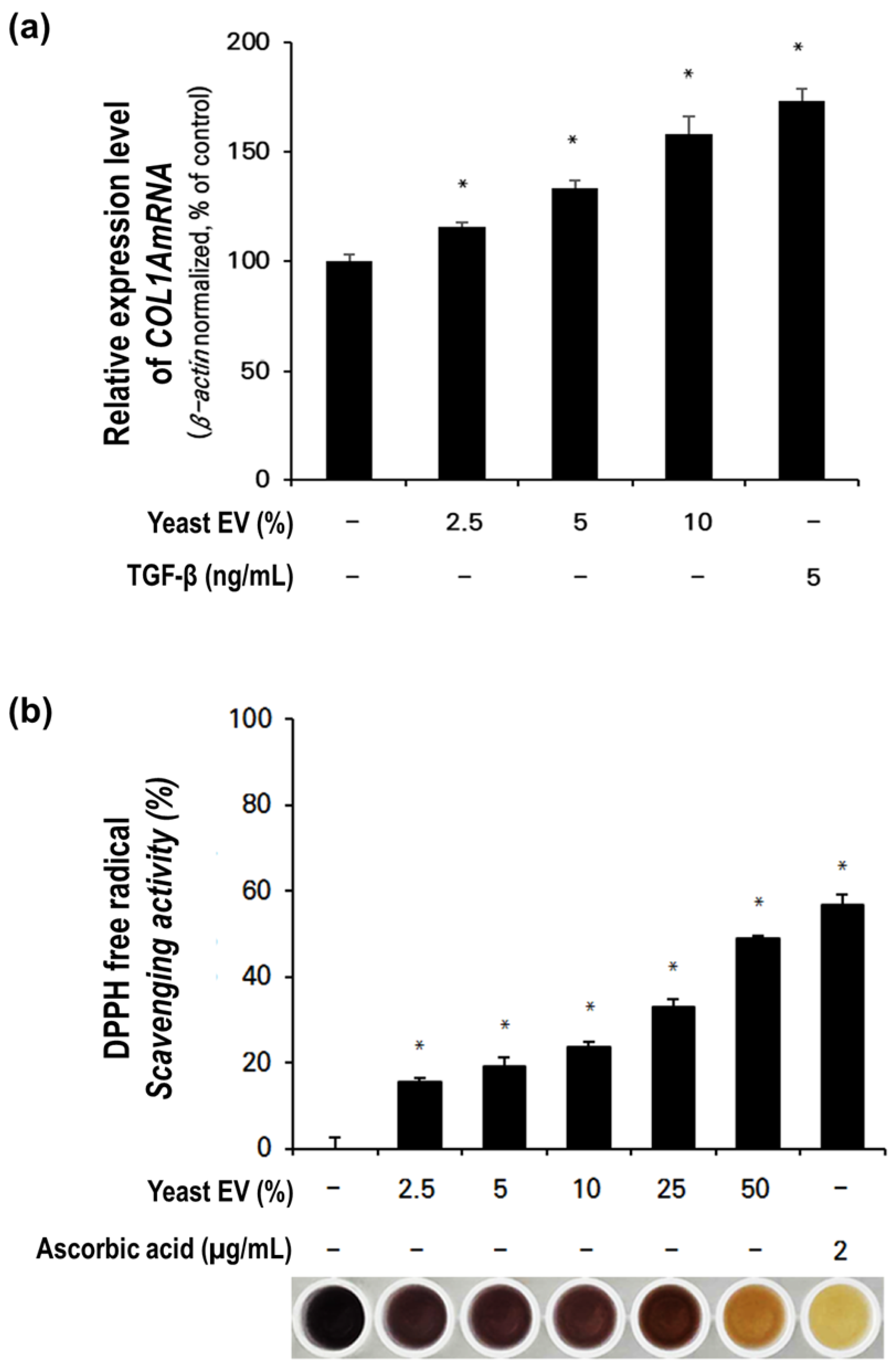
3.5. Effect of Yeast EVs on Collagen Synthesis in Fibroblasts
3.6. Antioxidant Activity of Yeast- EVs
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| EV | Extracellular vesicle |
| SC | Saccharomyces cerevisiae |
| hHFDPCs | Human hair follicle dermal papilla cells |
Appendix A
Appendix A.1
| Gene | Forward Primer (5′→3′) | Reverse Primer (5′→3′) |
| IL-1β | CCACAGACCTTCCAGGAGAATG | GTGCAGTTCAGTGATCGTACAGG |
| β-actin | GGATTCCTATGTGGGCGACGA | CGCTCGGTGAGGATCTTCATG |
| Gene | Forward Primer (5′→3′) | Reverse Primer (5′→3′) |
| COL1A1 | AGGGCCAAGACGAAGACATC | AGATCACGTCATCGCACAACA |
| β-actin | GGATTCCTATGTGGGCGACGA | CGCTCGGTGAGGATCTTCATG |
Appendix A.2
References
- Mathieu M, Martin-Jaular L, Lavieu G, Théry C., Specificities of secretion and uptake of exosomes and other extracellular vesicles for cell-to-cell communication, Nat Cell Biol. 2019;21(1):9-17.
- Théry C, Witwer KW, Aikawa E, Alcaraz MJ, Anderson JD, Andriantsitohaina R, Antoniou A, Arab T, Archer F, Atkin-Smith GK, et al., Minimal information for studies of extracellular vesicles 2018 (MISEV2018): A position statement of the International Society for Extracellular Vesicles, J Extracell Vesicles. 2018;7(1):1535750.
- Rodrigues ML, Casadevall A., A two-way road: novel roles for fungal extracellular vesicles, Nat Rev Microbiol. 2020;18(8):461-472.
- Albuquerque PC, Nakayasu ES, Rodrigues ML, Frases S, Casadevall A, Zancope-Oliveira RM, Almeida IC, Nosanchuk JD., Vesicular transport in Histoplasma capsulatum: An analysis of yeast phase extracellular vesicles, Eukaryot Cell. 2008;7(10):1695-1703.
- Zhao K, Bleackley M, Chisanga D, Gangoda L, Fonseka P, Liem M, Kalra H, Al Saffar H, Keerthikumar S, Ang CS, et al., Extracellular vesicles secreted by Saccharomyces cerevisiae are involved in cell wall remodelling, Cell Rep. 2019;27(12):3682-3695.
- Coelho C, Casadevall A., Answers to naysayers regarding microbial extracellular vesicles, Trends Microbiol. 2019;27(10):873-874.
- Zarnowski R, Sanchez H, Covelli AS, Dominguez E, Jaromin A, Berhardt J, Mitchell AP, Andes DR., Candida albicans biofilm-induced vesicles confer drug resistance through matrix biogenesis, mBio. 2018;9(3):e01376-18.
- Rittié L., Cellular mechanisms of skin repair in humans and other people’s animals, Nat Rev Mol Cell Biol. 2016;17(6):385-396.
- Bai H, Zhang W, Qin X, Yang Y, Wang X, Li Y., Extracellular vesicles as emerging therapeutics in skin wound healing and regeneration, Int J Mol Sci. 2024;25(3):1452.
- Kim HS, Choi DY, Yun SJ, Choi SM, Kang JW, Jung JW, Hwang D, Kim KP, Kim DW., Proteomic analysis of microvesicles derived from human mesenchymal stem cells in hair growth, Int J Mol Sci. 2021;22(6):3024.
- Suchonwanit P, Thammarucha S, Leerunyakul K., Minoxidil and its use in hair disorders: A review, Drug Des Devel Ther. 2019;13:2777-2786.
- Lee, K.; Bae, M.; Kim, Y.; Jeon, S.; Kang, S.; Rhee, W.; Shin, S. Scalable, High-Throughput Isolation of Extracellular Vesicles Using Electrokinetic-Assisted Mesh Filtration: ExoFilter. J. Extracell. Biol. 2025, 4(6), e70054; PMCID: PMC12119903. [CrossRef]
- Kim, YW, Jeon, SY, Lee, KM, Shin, S, Advanced Extracellular Vesicle Isolation: A Hybrid Electrokinetic-Tangential Flow Filtration Approach for Improved Yield, Purity, and Scalability, Anal. Chem. 2025, 97, 31, 16759–16768.
- Schneider, M.R.; Schmidt-Ullrich, R.; Paus, R. The hair follicle as a dynamic miniorgan. Curr. Biol. 2009, 19, R132–R142. [CrossRef]
- Quan, T.; Fisher, G.J. Role of age-associated alterations of the dermal extracellular matrix microenvironment in human skin aging. Gerontology 2015, 61, 427–434. [CrossRef]
- Mahé, Y.F.; Michelet, J.F.; Billoni, N.; Jarrousse, F.; Buan, B.; Commo, S.; Bernard, B.A. Androgenetic alopecia and microinflammation. Int. J. Dermatol. 2000, 39, 576–584. [CrossRef]
- Trüeb, R.M. Oxidative stress in ageing of hair. Int. J. Trichology 2009, 1, 6–14. [CrossRef]
- Yáñez-Mó, M.; Siljander, P.R.M.; Andreu, Z.; Zavec, A.B.; Borràs, F.E.; Buzas, E.I.; Buzás, K.; Casal, E.; Cappello, F.; Carvalho, J.; et al. Biological properties of extracellular vesicles and their physiological functions. J. Extracell. Vesicles 2015, 4, 27066. [CrossRef]
- Konoshenko MY, Lekchnov EA, Vlassov AV, Laktionov PP. Isolation of extracellular vesicles: General methodologies and latest trends. Biomed Res Int. 2018;2018:8545347.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




