Submitted:
25 February 2026
Posted:
26 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. General Overview of Breast Cancer
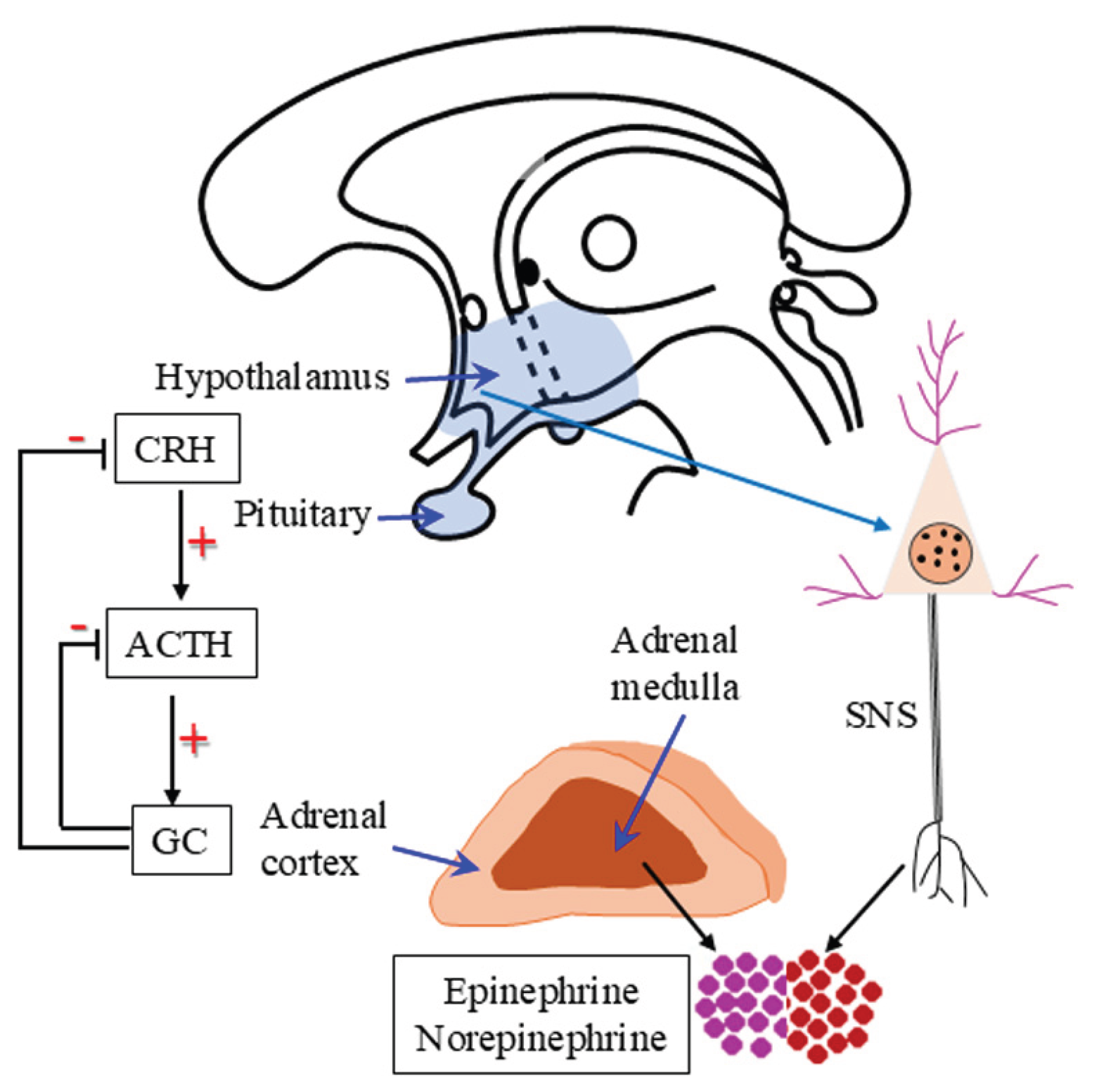
3. Stress and Stress Responses
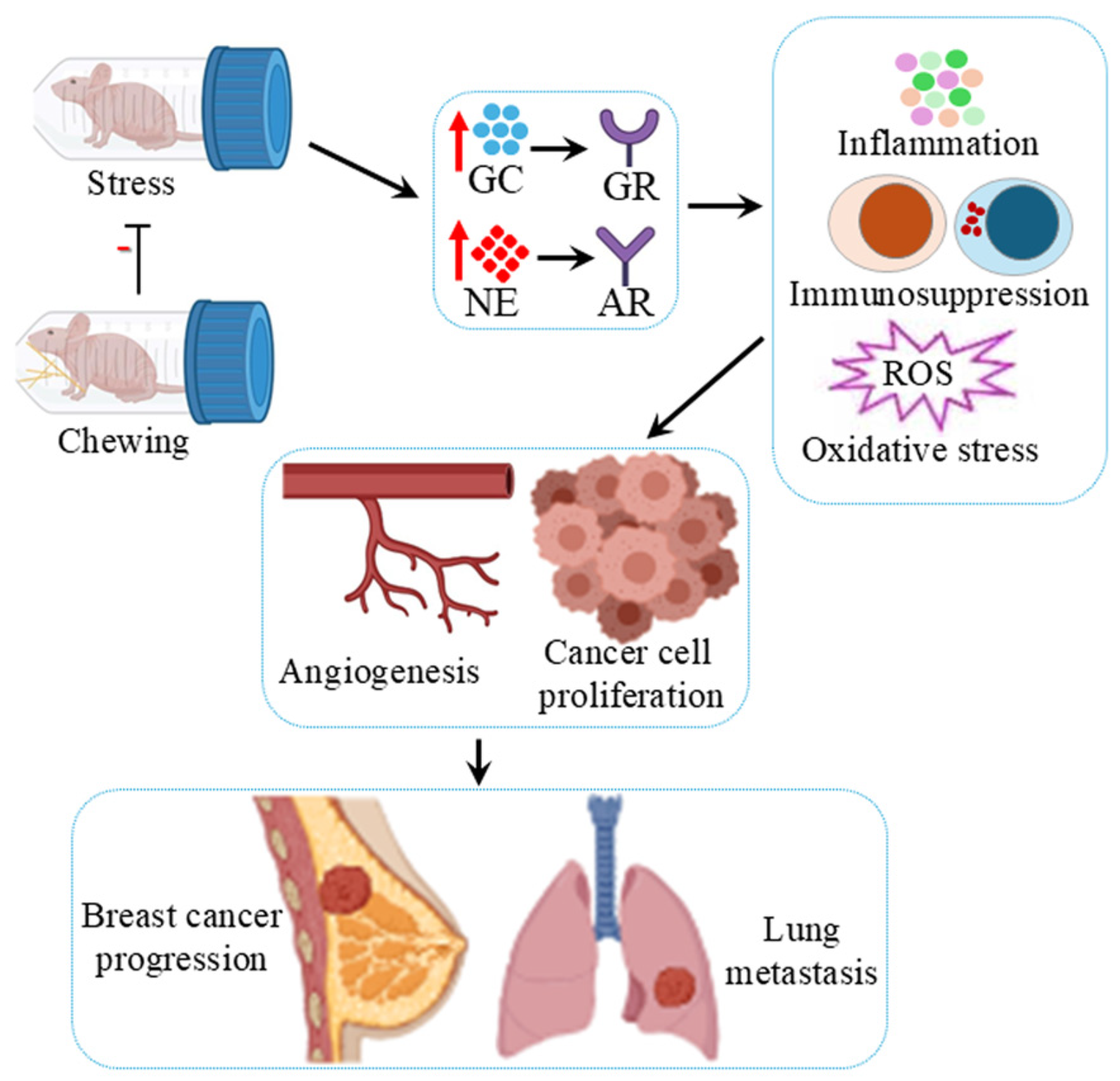
4. Chronic Stress Acts as a Risk Factor for Breast Cancer
5. Chewing Attenuates Promotion of Breast Cancer by Chronic Stress
6. Conclusions and Future Perspectives
Author Contributions
Data Availability Statement
Conflicts of Interest
Abbreviations
| ACTH β-AR BLA CeA CRH EMT GC GR HPA |
Adrenocorticotropic hormone β-adrenergic receptor Basolateral amygdala Central amygdala Corticotropin-releasing hormone Epithelial-mesenchymal transition Glucocorticoid Glucocorticoid receptor Hypothalamic-pituitary-adrenal |
| 4-HNE iNOS IL LTP MDSC MMP NK NE NET NMDA PAG PMN PVN RNS ROS SNS |
4-hydroxynonenal inducible nitric oxide synthase Interleukin Long-term potentiation Myeloid-derived suppressor cell Matrix metalloproteinase Natural killer Norepinephrine Neutrophil extracellular trap N-methyl-D-aspartate Periaqueductal gray Premetastatic niche Paraventricular nucleus Reactive nitrogen species Reactive oxygen species Sympathetic nervous system |
| SOD2 TAM VEGF |
Superoxide dismutase 2 Tumor-associated macrophage Vascular endothelial growth factor |
References
- Freihat, O.; Sipos, D.; Arpad Kovacs, A. Global burden and projections of breast cancer incidence and mortality to 2050: a comprehensive analysis of GLOBOCAN data. Front Public Health 2025, 13, 1622954. [Google Scholar] [CrossRef]
- Łukasiewicz, S.; Czeczelewski, M.; Forma, A.; Baj, J.; Robert Sitarz, R.; Stanisławek, A. Breast Cancer—Epidemiology, Risk Factors, Classification, Prognostic Markers, and Current Treatment Strategies—An Updated Review. Cancers 2021, 13, 4287. [Google Scholar] [CrossRef] [PubMed]
- Liu, H-M.; Ma, L.; Chunyu Li, C.; Cao, B.; Jiang, Y.; Han, L.; Xu, R.; Lin, J.; Zhang, D. The molecular mechanism of chronic stress affecting the occurrence and development of breast cancer and potential drug therapy. Transl. Oncol. 2022, 15, 101281. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Wang, M.; Yu, K.; Xu, S.; Qiu, P.; Lyu, Z.; Zhang, X.; Xu, Y. Chronic stress-induced immune dysregulation in breast cancer: Implications of psychosocial factors. J. Transl. Int. Med. 2022, 11, 226–233. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Wang, N.; Wang, S.; Zhang, J.; Yang, B.; Wang, Z. Chronic psychological stress promotes breast cancer pre-metastatic niche formation by mobilizing splenic MDSCs via TAM/CXCL1 signaling. J. Exp. Clin. Cancer Res. 2023, 42, 129. [Google Scholar] [CrossRef]
- Mravec, B.; Tibensky, M.; Horvathova, L. Stress and cancer. Part I: Mechanisms mediating the effect of stressors on cancer. J. Neuroimmunol. 2020, 346, 577311. [Google Scholar] [CrossRef]
- Chen, H.; Liu, D.; Guo, L.; Cheng, X.; Guo, N.; Shi, M. Chronic psychological stress promotes lung metastatic colonization of circulating breast cancer cells by decorating a pre-metastatic niche through activating β-adrenergic signaling. J. Pathol. 2018, 244, 49–60. [Google Scholar] [CrossRef]
- Bahri, N.; Najafi, T.F.; Shandiz, F.H.; Tohidinik, H.R.; Khajavi, A. The relation between stressful life events and breast cancer: a systematic review and meta-analysis of cohort studies. Breast Cancer Res. Treat. 2019, 176, 53–61. [Google Scholar] [CrossRef]
- Khan, A.; Song, M.; Dong, Z. Chronic stress: a fourth etiology in tumorigenesis? Mol. Cancer 2025, 24, 196. [Google Scholar] [CrossRef]
- Zhang, Z.; Wang, Y.; Li, Q. Mechanisms underlying the effects of stress on tumorigenesis and metastasis (Review). Int. J. Oncol. 2018, 53, 2332–2342. [Google Scholar] [CrossRef]
- Russell, G.; Lightman, S. The human stress response. Nat. Rev. Endocrinol. 2019, 15, 525–534. [Google Scholar] [CrossRef] [PubMed]
- Glaser, R.; Kiecolt-Glaser, J.K. Stress-induced immune dysfunction: implications for health. Nat. Rev. Immunol. 2005, 5, 243–251. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Le, M.; Bai, Y. Chronic Stress Mediates Inflammatory Cytokines Alterations and Its Role in Tumorigenesis. J. Inflamm. Res. 2025, 18, 1067–1090. [Google Scholar] [CrossRef] [PubMed]
- Caparica, R.; Bruzzone, M.; Agostinetto, E.; Angelis, C.D.; Fêde, Â.; Ceppi, M.; de Azambuja, E. Beta-blockers in early-stage breast cancer: a systematic review and meta-analysis. ESMO Open 2021, 6, 100066. [Google Scholar] [CrossRef]
- Antoni, M.H.; Dhabhar, F.S. The impact of psychosocial stress and stress management on immune responses in patients with cancer. Cancer 2019, 125, 1417–1431. [Google Scholar] [CrossRef]
- Kubo, K-Y.; Iinuma, M.; Chen, H. Mastication as a Stress-Coping Behavior. Biomed. Res. Int. 2015, 2015, 876409. [Google Scholar] [CrossRef]
- Azuma, K.; Zhou, Q.; Niwa, M.; Kubo, K-Y. Association between Mastication, the Hippocampus, and the HPA Axis: A Comprehensive Review. Int. J. Mol. Sci. 2017, 18(8), 1687. [Google Scholar] [CrossRef]
- Yaman-Sözbir, Ş.; Ayaz-Alkaya, S.; Bayrak-Kahraman, B. Effect of chewing gum on stress, anxiety, depression, self-focused attention, and academic success: A randomized controlled study. Stress Health 2019, 35, 441–446. [Google Scholar] [CrossRef]
- Siegel, R.L.; Giaquinto, A.N.; Jemal, A. Cancer statistics, 2024. CA Cancer J. Clin. 2024, 74, 12–49. [Google Scholar] [CrossRef]
- Xiong, X.; Zheng, L-W.; Ding, Y.; Chen, Y-F.; Cai, Y-W.; Wang, L-P.; Huang, L.; Liu, C-C.; Shao, Z-M.; Yu, K-D. Breast cancer: Pathogenesis and treatments. Signal Transduct. Target Ther. 2025, 10, 49. [Google Scholar] [CrossRef]
- Anderson, K.N.; Schwab, R.B.; Martinez, M.E. Reproductive risk factors and breast cancer subtypes: a review of the literature. Breast Cancer Res. Treat. 2014, 144, 1–10. [Google Scholar] [CrossRef]
- Namazi, N.; Irandoost, P.; Heshmati, J.; Larijani, B; Azadbakht, L. The association between fat mass and the risk of breast cancer: A systematic review and metaanalysis. Clin. Nutr. 2019, 38, 1496–1503. [Google Scholar] [CrossRef]
- Bagnardi, V.; Rota, M.; Botteri, E.; Tramacere, I.; Islami, F.; Fedirko, V.; Scotti, L.; Jenab, M.; Turati, F.; Pasquali, E.; Pelucchi, C.; Galeone, C.; Bellocco, R.; Negri, E.; Corrao, G.; Boffetta, P.; La Vecchiaet, C. Alcohol consumption and site-specific cancer risk: a comprehensive dose-response meta-analysis. Br. J. Cancer 2015, 112, 580–593. [Google Scholar] [CrossRef]
- Gaudet, M.M.; Gapstur, S.M.; Sun, J.; Diver, W.R.; Hannan, L.M.; Thun, M.J. Active smoking and breast cancer risk: original cohort data and meta-analysis. J. Natl. Cancer Inst. 2013, 105, 515–525. [Google Scholar] [CrossRef]
- Koo, E.; Henderson, M.A.; Dwyer, M.; Skandarajah, A.R. Management and Prevention of Breast Cancer After Radiation to the Chest for Childhood, Adolescent, and Young Adulthood Malignancy. Ann. Surg. Oncol. 2015, 22, S545–S551. [Google Scholar] [CrossRef]
- Selye, H. A Syndrome produced by Diverse Nocuous Agents. Nature 1936, 138, 32. [Google Scholar] [CrossRef]
- McCarty, R.; Horwatt, K.; Konarska, M. Chronic stress and sympathetic-adrenal medullary responsiveness. Soc. Sci. Med. 1998, 26, 333–341. [Google Scholar] [CrossRef] [PubMed]
- Vasconcelos, M.; Stein, D.J.; Gallas-Lopes, M.; Landau, L.; de Almeida, R.M.M. Corticotropin-releasing factor receptor signaling and modulation: implications for stress response and resilience. Trends Psychiatry Psychother. 2020, 42, 195–206. [Google Scholar] [CrossRef] [PubMed]
- Hostinar, C.E.; Sullivan, R.M.; Gunnar, M.R. Psychobiological mechanisms underlying the social buffering of the hypothalamic-pituitary-adrenocortical axis: a review of animal models and human studies across development. Psychol. Bull. 2014, 140, 256–282. [Google Scholar] [CrossRef]
- Besedovsky, H.; del Rey, A. A Glucocorticoid-Mediated Immunoregulatory Circuit Integrated at Brain Levels: Our Early Studies and a Present View. Neuroimmunomodulation 2024, 31, 230–245. [Google Scholar] [CrossRef]
- Akalestou, E.; Genser, L.; Rutter, G.A. Glucocorticoid Metabolism in Obesity and Following Weight Loss. Front Endocrinol. 2020, 11, 59. [Google Scholar] [CrossRef] [PubMed]
- Reichard, S.D.; Agathe Amouret, A.; Chiara Muzzi, C.; Sabine Vettorazzi, S.; Tuckermann, J.P.; Lühder, F.; Reichardt, H.M. The Role of Glucocorticoids in Inflammatory Diseases. Cells 2021, 10, 2921. [Google Scholar] [CrossRef] [PubMed]
- Selye, H. Forty years of stress research: principal remaining problems and misconceptions. Can. Med. Assoc. J. 1976, 115, 53–56. [Google Scholar] [PubMed]
- Zhou, Q.; Katano, M.; Zhang, J-H.; Liu, X.; Wang, K-Y.; Iinuma, M.; Kubo, K-Y.; Azuma, K. Chewing Behavior Attenuates the Tumor Progression-Enhancing Effects of Psychological Stress in a Breast Cancer Model Mouse. Brain Sci. 2021, 11, 479. [Google Scholar] [CrossRef]
- Zhang, J.H.; Wang, K-Y.; Kubo, K-Y.; Azuma, K. Chewing Behavior Attenuates Lung-Metastasis-Promoting Effects of Chronic Stress in Breast-Cancer Lung-Metastasis Model Mice. Cancers 2022, 14, 5950. [Google Scholar] [CrossRef]
- Sanders, V.M.; Kohm, A.P. Sympathetic nervous system interaction with the immune system. Int. Rev. Neurobiol. 2002, 52, 17–41. [Google Scholar]
- Lei, Y.; Liao, F.; Tian, Y.; Wang, Y.; Xia, F.; Wang, J. Investigating the crosstalk between chronic stress and immune cells: implications for enhanced cancer therapy. Front Neurosci. 2023, 17, 1321176. [Google Scholar] [CrossRef]
- Zefferino, R.; Di Gioia, S.; Conese, M. Molecular links between endocrine, nervous and immune system during chronic stress. Brain Behav. 2020, 11, e01960. [Google Scholar] [CrossRef]
- Flaherty, R.L.; Owen, M.; Fagan-Murphy, A.; Intabli, H.; Healy, D.; Patel, B.A.; Allen, M.C.; Patel, B.A.; Flint, M.S. Glucocorticoids induce production of reactive oxygen species/reactive nitrogen species and DNA damage through an iNOS mediated pathway in breast cancer. Breast Cancer Res. 2017, 19, 35. [Google Scholar] [CrossRef]
- Flaherty, R.L.; Intabli, H.; Falcinelli, M.; Bucca, G.; Hesketh, A.; Patel, B.A.; Allen, M.C.; Smith, C.P.; Flint, M.S. Stress hormone-mediated acceleration of breast cancer metastasis is halted by inhibition of nitric oxide synthase. Cancer Lett. 2019, 459, 59–71. [Google Scholar] [CrossRef]
- Silva, D.; Quintas, C.; Gonçalves, J.; Fresco, P. b2-Adrenoceptor Activation Favor Acquisition of Tumorigenic Properties in Non-Tumorigenic MCF-10A Breast Epithelial Cells. Cells 2024, 13, 262. [Google Scholar] [CrossRef]
- Sun, C.; Shen, Y.; Wang, F.; Lu, T.; Zhang, J. Sympathetic nervous system in tumor progression and metabolic regulation: mechanisms and clinical potential. J. Translational. Med. 2025, 23, 836. [Google Scholar] [CrossRef] [PubMed]
- Peixoto, R.; de Lourdes Pereira, M.; Oliveira, M. Beta-Blockers and Cancer: Where Are We? Pharmaceuticals. 2020, 13, 105. [Google Scholar] [CrossRef] [PubMed]
- Alotiby, A. Immunology of Stress: A Review Article. J. Clin. Med. 2024, 13, 6394. [Google Scholar] [CrossRef] [PubMed]
- Slavich, G.M.; Irwin, M.R. From stress to inflammation and major depressive disorder: a social signal transduction theory of depression. Psychol. Bull. 2014, 140, 774–815. [Google Scholar] [CrossRef]
- Kuebler, U.; Zuccarella-Hackl, C.; Arpagaus, A.; Wolf, J.M.; Farahmand, F.; von Känel, R.; Ehlert, U.; Wirtz, P.H. Stress-induced modulation of NF-κB activation, inflammation-associated gene expression, and cytokine levels in blood of healthy men. Brain Behav. Immun. 2015, 46, 87–95. [Google Scholar] [CrossRef]
- Vilasco, M.; Communal, L.; Mourra, N.; Courtin, A.; Forgez, P.; Gompel, A. Glucocorticoid receptor and breast cancer. Breast Cancer Res. Treat. 2011, 130, 1–10. [Google Scholar] [CrossRef]
- Krolick, K.N.; Shi, H. Estrogenic Action in Stress-Induced Neuroendocrine Regulation of Energy Homeostasis. Cells 2022, 11, 879. [Google Scholar] [CrossRef]
- Zhang, X.; Ma, N.; Jin, C.; Cao, X. Role of gut microbiota and immune response in breast cancer progression. Biomolecules and Biomedicine 2026, 26, 100–114. [Google Scholar] [CrossRef]
- Zhang, L.; Pan, J.; Wang, M.; Yang, J.; Zhu, S.; Li, L.; Hu, X.; Wang, Z.; Pang, L.; Li, P.; Jia, Fang; Ren, G.; Zhang, Y.; Xu, D.; Qiu, F.; Huang, J. Chronic Stress-Induced and Tumor Derived SP1+ Exosomes Polarizing IL-1β+ Neutrophils to Increase Lung Metastasis of Breast Cancer. Adv. Sci. 2025, 12, 2310266. [Google Scholar] [CrossRef]
- Momčilović, S.; Milošević, M.; Kočović, D.M.; Marković, D.; Zdravković, D.; Petrinović, S.V. Macrophages at the Crossroads of Chronic Stress and Cancer. Int. J. Mol. Sci. 2025, 26, 6838. [Google Scholar] [CrossRef]
- He, X-Y.; Gao, Y.; Ng, D.; Michalopoulou, E.; George, S.; Adrover, J.M.; Sun, L.; Albrengues, J.; Daßler-Plenker, J.; Han, X.; Wan, L.; Wu, X.S.; Shui, L.S.; Huang, Y-H.; Liu, B.; Su, C.; Spector, D.L.; Vakoc, C.R.; Aelst, L.V.; Egeblad, M. Chronic stress increases metastasis via neutrophil-mediated changes to the microenvironment. Cancer Cell 2024, 42, 474–486.e12. [Google Scholar] [CrossRef] [PubMed]
- Li, K.; Deng, Z.; Lei, C.; Ding, X.; Li, J.; Wang, C. The Role of Oxidative Stress in Tumorigenesis and Progression. Cells 2024, 13, 441. [Google Scholar] [CrossRef] [PubMed]
- Li, Y-P.; Tian, F-G.; Shi, P-C.; Guo, L-Y.; Wu, H-M.; Chen, R-Q.; Xue, J-M. 4-Hydroxynonenal promotes growth and angiogenesis of breast cancer cells through HIF-1α stabilization. Asian Pac. J. Cancer Prev. 2014, 15, 10151–10156. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.S.; Vallur, P.G.; Phaëton, R.; Mythreye, K.; Hempel, N. Insights into the Dichotomous Regulation of SOD2 in Cancer. Antioxidants 2017, 6, 86. [Google Scholar] [CrossRef]
- Takai, N.; Yamaguchi, M.; Aragaki, T.; Eto, K.; Uchihashi, K.; Nishikawa, Y. Effect of psychological stress on the salivary cortisol and amylase levels in healthy young adults. Arch. Oral. Biol. 2004, 49, 9633–968. [Google Scholar] [CrossRef]
- Hingorjo, M.R.; Owais, M.; Siddiqui, S.U.; Nazar, S.; Ali, Y.S. The impact of psychological stress on salivary cortisol levels in periodontitis patients: a case-control study. BMC Oral Health 2025, 21, 25:276. [Google Scholar] [CrossRef]
- Lee, Y-H.; Suk, C.; Shin, S-I.; Hong, J-Y. Salivary cortisol, dehydroepiandrosterone, and chromogranin A levels in patients with gingivitis and periodontitis and a novel biomarker for psychological stress. Front Endocrinol. 2023, 14, 1147739. [Google Scholar] [CrossRef]
- Dinapoli, L.; Colloca, G.; Di Capua, B.; Valentini, V. Psychological Aspects to Consider in Breast Cancer Diagnosis and Treatment. Curr. Oncol. Rep. 2021, 23, 38. [Google Scholar] [CrossRef]
- Cormanique, T.F.; de Almeida, L.E.; Rech, C.A.; Rech, D.; Herrera, A.C.; Panis, C. Chronic psychological stress and its impact on the development of aggressive breast cancer. Einstein 2015, 13, 352–356. [Google Scholar] [CrossRef]
- Gold, S.M.; Zakowski, S.G.; Valdimarsdottir, H.B.; Bovbjerg, D.H. Stronger endocrine responses after brief psychological stress in women at familial risk of breast cancer. Psychoneuroendocrinology 2003, 28, 584–593. [Google Scholar] [CrossRef] [PubMed]
- Kubo, K-Y.; Yamada, Y.; Iinuma, M.; Iwaku, F.; Tamura, Y.; Watanabe, K.; Nakamura, H.; Onozuka, M. Occlusal disharmony induces spatial memory impairment and hippocampal neuron degeneration via stress in SAMP8 mice. Neurosci. Lett. 2007, 414, 188–191. [Google Scholar] [CrossRef] [PubMed]
- Miyake, H.; Mori, D.; Katayama, T.; Fujiwara, S.; Sato, Y.; Azuma, K.; Kubo, K-Y. Novel stress increases hypothalamic-pituitary-adrenal activity in mice with a raised bite. Arch. Oral Biol. 2016, 68, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Kubo, K-Y.; Kojo, A.; Yamamoto, T.; Onozuka, M. The bite-raised condition in aged SAMP8 mice induces dendritic spine changes in the hippocampal region. Neurosci. Lett. 2008, 441, 141–144. [Google Scholar] [CrossRef]
- Vincent, G.P.; Paré, W.P.; Prenatt, J.E.; Glavin, G.B. Aggression, body temperature, and stress ulcer. Physiol. Behav. 1984, 32, 265–268. [Google Scholar] [CrossRef]
- Kubo, K-Y.; Sasaguri, K.; Ono, Y.; Yamamoto, T.; Takahashi, T.; Watanabe, K.; Karasawa, N.; Onozuka, M. Chewing under restraint stress inhibits the stress-induced suppression of cell birth in the dentate gyrus of aged SAMP8 mice. Neurosci. Lett. 2009, 466, 109–113. [Google Scholar] [CrossRef]
- Miyake, S.; Yoshikawa, G.; Yamada, K.; Sasaguri, K-I.; Yamamoto, T.; Onozuka, M.; Sato, S. Chewing ameliorates stress-induced suppression of spatial memory by increasing glucocorticoid receptor expression in the hippocampus. Brain Res. 2012, 1446, 34–39. [Google Scholar] [CrossRef]
- Ono, Y.; Kataoka, T.; Miyake, S.; Sasaguri, K.; Sato, S.; Onozuka, M. Chewing rescues stress-suppressed hippocampal long-term potentiation via activation of histamine H1 receptor. Neurosci. Res. 2009, 64, 385–390. [Google Scholar] [CrossRef]
- Azuma, K.; Adachi, Y.; Hayashi, H.; Kubo, K-Y. Chronic Psychological Stress as a Risk Factor of Osteoporosis. J. UOEH 2015, 37, 245–253. [Google Scholar] [CrossRef]
- Furuzawa, M.; Chen, H.; Fujiwara, S.; Yamada, K.; Kubo, K-Y. Chewing ameliorates chronic mild stress-induced bone loss in senescence-accelerated mouse (SAMP8), a murine model of senile osteoporosis. Exp. Gerontol. 2014, 55, 12–18. [Google Scholar] [CrossRef]
- Ono, Y.; Lin, S-C.; Tzen, K-Y.; Chen, H-H.; Yang, P-F.; Lai, W-S.; Chen, J-H.; Onozuka, M.; Yen, C-T. Active coping with stress suppresses glucose metabolism in the rat hypothalamus. Stress 2012, 15, 207–217. [Google Scholar] [CrossRef] [PubMed]
- Okada, S.; Hori, N.; Kimoto, K.; Onozuka, M.; Sato, S.; Sasaguri, K. Effects of biting on elevation of blood pressure and other physiological responses to stress in rats: biting may reduce allostatic load. Brain Res. 2007, 1185, 189–194. [Google Scholar] [CrossRef] [PubMed]
- Koizumi, S.; Minamisawa, S.; Sasaguri, K.; Onozuka, M.; Sato, S.; Ono, Y. Chewing reduces sympathetic nervous response to stress and prevents poststress arrhythmias in rats. Am. J. Physiol. 2011, 301, H1551–H1558. [Google Scholar] [CrossRef] [PubMed]
- Pessoa, L. A Network Model of the Emotional Brain. Trends Cogn. Sci. 2017, 21, 357–371. [Google Scholar] [CrossRef]
- McEwen, B.S. The neurobiology of stress: from serendipity to clinical relevance. Brain Res. 2000, 886, 172–189. [Google Scholar] [CrossRef]
- Binder, E.B; Nemeroff, C.B. The CRF system, stress, depression and anxiety-insights from human genetic studies. Mol. Psychiatry 2010, 15, 574–588. [Google Scholar] [CrossRef]
- Sasaguri, K.; Kikuchi, M.; Hori, N.; Yuyama, N.; Onozuka, M.; Sato, S. Suppression of stress immobilization-induced phosphorylation of ERK 1/2 by biting in the rat hypothalamic paraventricular nucleus. Neurosci. Lett. 2005, 383, 160–164. [Google Scholar] [CrossRef]
- Van de Kar, L.D.; Piechowski, R.A.; Rittenhouse, P.A.; Gray, T.S. Amygdaloid lesions: differential effect on conditioned stress and immobilization-induced increases in corticosterone and renin secretion. Neuroendocrinology 1991, 54, 89–95. [Google Scholar] [CrossRef]
- Chen, H.; Iinuma, M.; Onozuka, M.; Kubo, K-Y. Chewing Maintains Hippocampus-Dependent Cognitive Function. Int. J. Med, Sci. 2015, 12, 502–509. [Google Scholar] [CrossRef]
- Prewitt, C.M.; Herman, J.P. Hypothalamo-Pituitary-Adrenocortical Regulation Following Lesions of the Central Nucleus of the Amygdala. Stress 1997, 1, 263–280. [Google Scholar] [CrossRef]
- Reznikov, L.R.; Reagan, L.P.; Fadel, J.R. Effects of acute and repeated restraint stress on GABA efflux in the rat basolateral and central amygdala. Brain Res. 2009, 1256, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.J.; Koo, J.W.; Lee, H.J.; Han, J.S. Amygdalar inactivation blocks stress-induced impairments in hippocampal long-term potentiation and spatial memory. J. Neurosci. 2005, 25, 1532–1539. [Google Scholar] [CrossRef] [PubMed]
- Yamada, K.; Narimatsu, Y.; Ono, Y.; Sasaguri, K-I.; Onozuka, M.; Kawata, T.; Yamamoto, T. Chewing suppresses the stress-induced increase in the number of pERK-immunoreactive cells in the periaqueductal grey. Neurosci. Lett. 2015, 599, 43–48. [Google Scholar] [CrossRef] [PubMed]
- de Abreu, A.R.; Abreu, A.R.; Santos, L.T.; de Souza, A.A.; da Silva, L.G., Jr.; Chianca, D.A., Jr.; de Menezes, C.R. Amygdalar neuronal activity mediates the cardiovascular responses evoked from the dorsolateral periaqueductal gray in conscious rats. Neuroscience 2015, 284, 737–750. [Google Scholar] [CrossRef]
- Tasaka, A.; Takeuchi, K.; Sasaki, H.; Yoshii, T.; Soeda, R.; Ueda, T.; Sakurai, K. Influence of chewing time on salivary stress markers. J. Prosthodont. Res. 2014, 58, 48–54. [Google Scholar] [CrossRef]
- Tahara, Y.; Sakurai, K.; Ando, T. Influence of chewing and clenching on salivary cortisol levels as an indicator of stress. J. Prosthodont. 2007, 16, 129–135. [Google Scholar] [CrossRef]
- Tasaka, A.; Kikuchi, M.; Nakanishi, K.; Ueda, T.; Yamashita, S.; Sakurai, K. Psychological stress-relieving effects of chewing - Relationship between masticatory function-related factors and stress-relieving effects. J. Prosthodont. 2018, 62, 50–55. [Google Scholar] [CrossRef]
- Onozuka, M.; Fujita, M.; Watanabe, K.; Hirano, Y.; Niwa, M.; Nishiyama, K.; Saito, S. Mapping brain region activity during chewing: a functional magnetic resonance imaging study. J. Dent. Res. 2002, 81, 743–746. [Google Scholar] [CrossRef]
- Hirano, Y.; Onozuka, M. Chewing and attention: a positive effect on sustained attention. Biomed. Res. Int. 2015, 2015, 367026. [Google Scholar] [CrossRef]
- Hirano, Y.; Obata, T.; Kashikura, K.; Nonaka, H.; Tachibana, A.; Ikehira, H.; Onozuka, M. Effects of chewing in working memory processing. Neurosci. Lett. 2008, 436, 189–192. [Google Scholar] [CrossRef]
- Yu, H.; Chen, X.; Liu, J.; Zhou, X. Gum chewing inhibits the sensory processing and the propagation of stress-related information in a brain network. PLoS ONE 2013, 8, e57111. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



