Submitted:
23 February 2026
Posted:
26 February 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Main
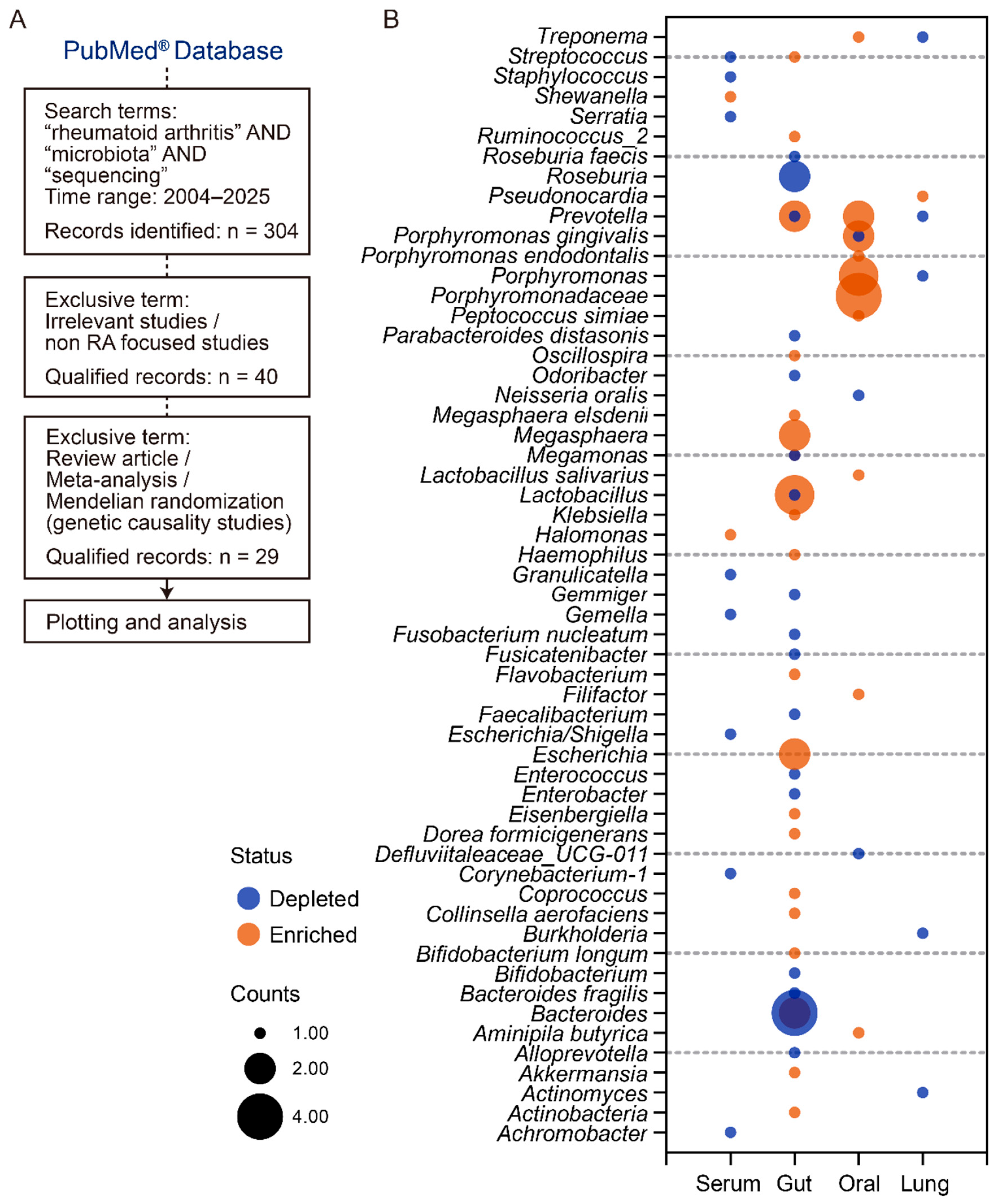
- Mucosa Dysbiosis Contributes to RA Pathogenesis
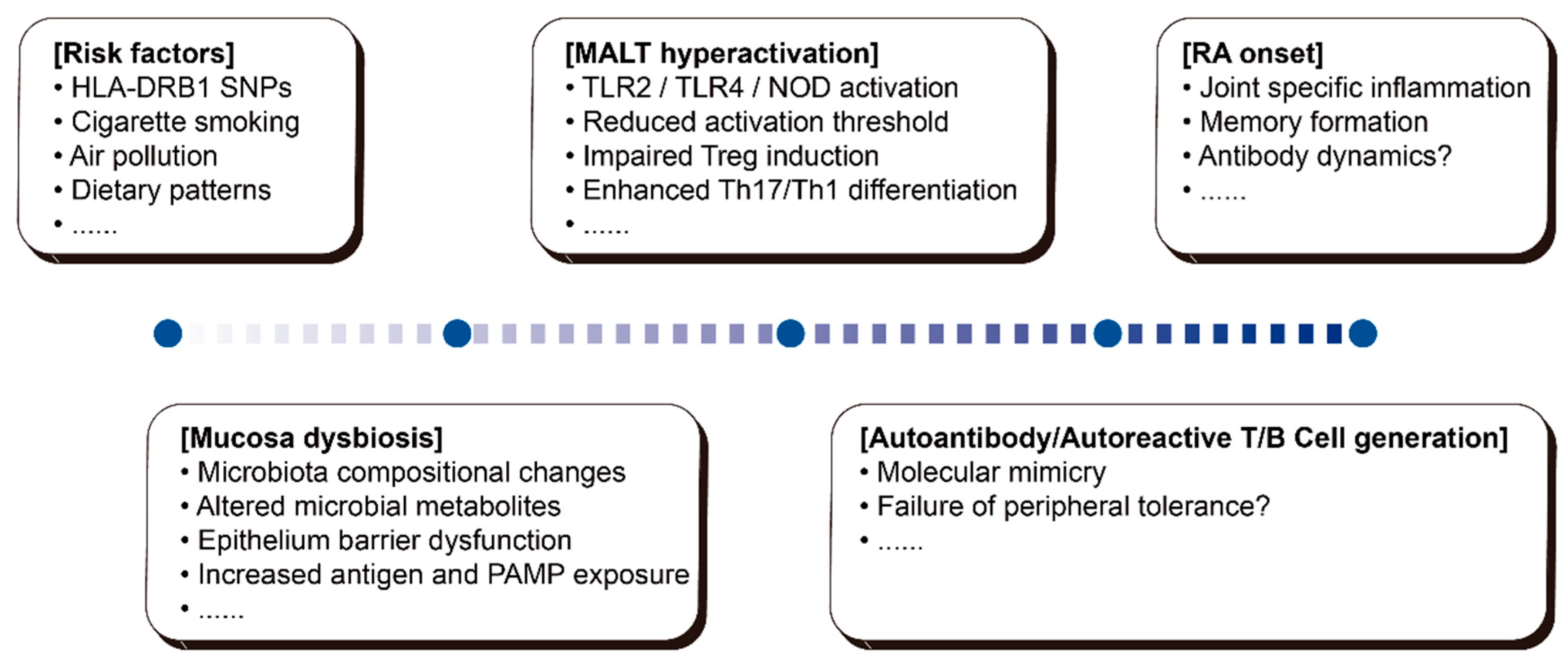
- Integrated Mucosal Mechanisms in RA Initiation
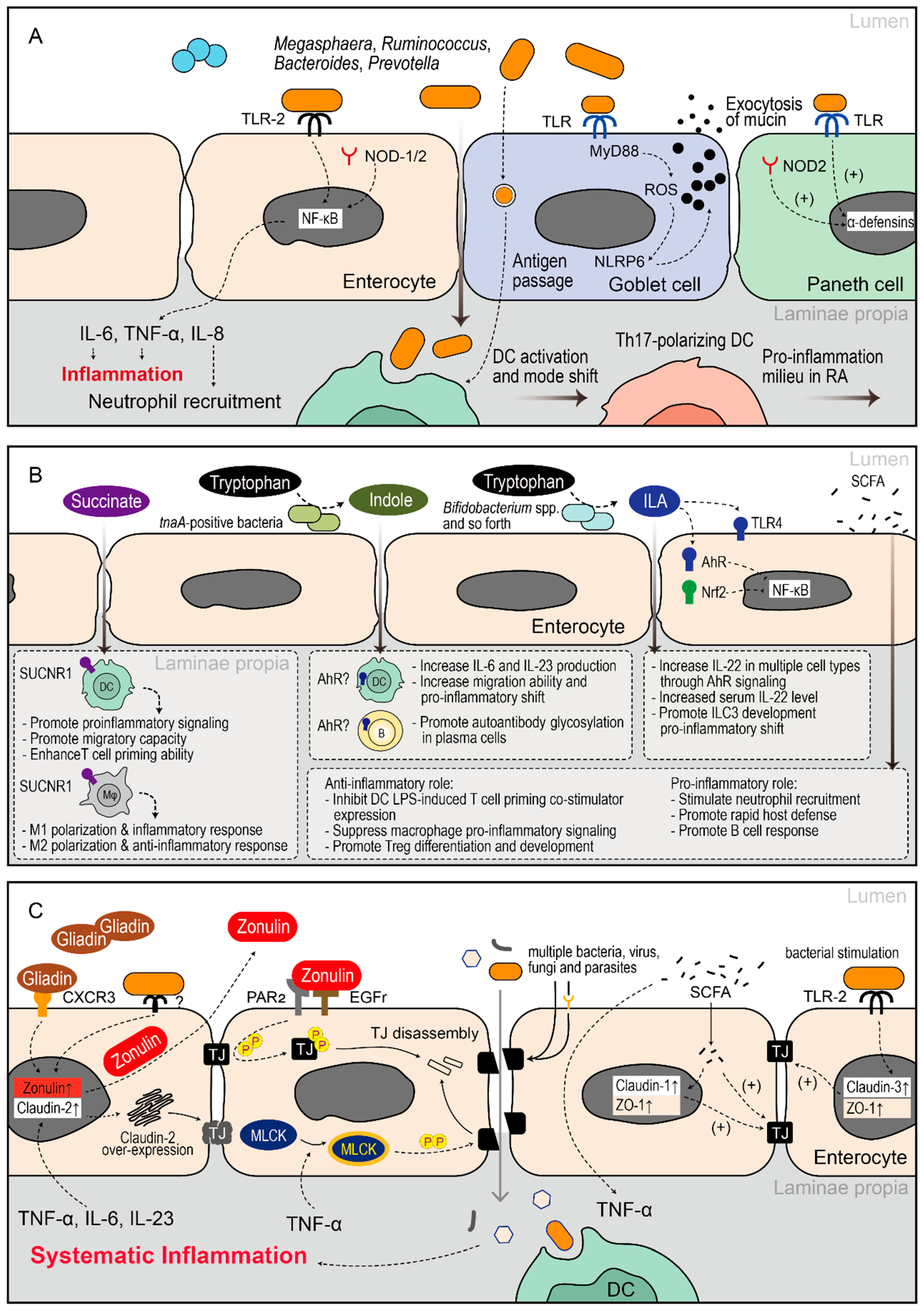
- Microbe Initiated Pro-Inflammatory Skewing Milieu
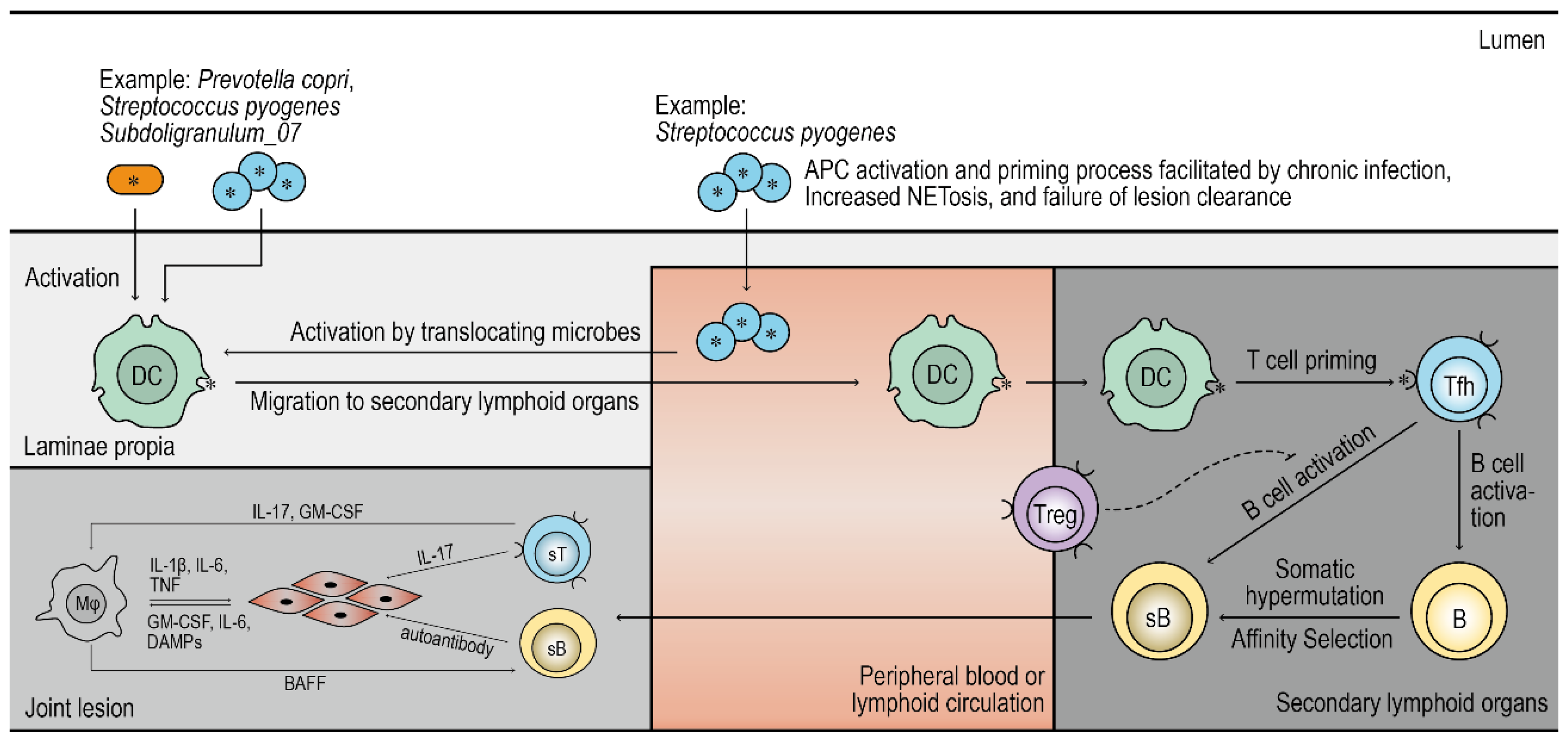
- Molecular Mimicry: Microbial Antigens Prime Cross-Reactive Adaptive Responses
- Parallel Joint Inflammation Contributing to Mechanisms
Conclusion
Supplementary Materials
References
- Abrass, C.K. (1984) ’Evaluation of the presence of circulating immune complexes and their relationship to glomerular IgG deposits in streptozotocin-induced diabetic rats.,’ PubMed, 57(1), pp. 17–24. https://pubmed.ncbi.nlm.nih.gov/6378460.
- Adami, G. et al. (2021) ’Association between environmental air pollution and rheumatoid arthritis flares,’ Lara D. Veeken, 60(10), pp. 4591–4597. [CrossRef]
- Alivernini, S., Firestein, G.S. and McInnes, I.B. (2022) ’The pathogenesis of rheumatoid arthritis,’ Immunity, 55(12), pp. 2255–2270. [CrossRef]
- Arslan, L, Melief, M-J, Tak, PP, Hazenberg, MPH & Laman, J 2000, ’Antigen-presenting cells containing bacterial peptidoglycan in synovial tissues of rheumatoid arthritis patients coexpress costimulatory molecules and cytokines’, Arthritis & Rheumatism, vol. 43, no. 10, pp. 2160-2168. [CrossRef]
- Atarashi, K. et al. (2010) ’Induction of colonic regulatory T cells by indigenous clostridium species,’ Science, 331(6015), pp. 337–341. [CrossRef]
- Badillo-Soto, M.A. et al. (2016) ’Potential protein targets of the peptidylarginine deiminase 2 and peptidylarginine deiminase 4 enzymes in rheumatoid synovial tissue and its possible meaning,’ European Journal of Rheumatology, 3(2), pp. 44–49. [CrossRef]
- Bingham, C.O. and Moni, M. (2013) ’Periodontal disease and rheumatoid arthritis,’ Current Opinion in Rheumatology, 25(3), pp. 345–353. [CrossRef]
- Block, K.E. et al. (2016) ’Gut Microbiota Regulates K/BxN Autoimmune Arthritis through Follicular Helper T but Not Th17 Cells,’ The Journal of Immunology, 196(4), pp. 1550–1557. [CrossRef]
- Boya, B.R. et al. (2021) ’Diversity of the tryptophanase gene and its evolutionary implications in living organisms,’ Microorganisms, 9(10), p. 2156. [CrossRef]
- Brewer, R.C. et al. (2023) ’Oral mucosal breaks trigger anti-citrullinated bacterial and human protein antibody responses in rheumatoid arthritis,’ Science Translational Medicine, 15(684), p. eabq8476. [CrossRef]
- Capaldo, C.T., Powell, D.N. and Kalman, D. (2017) ’Layered defense: how mucus and tight junctions seal the intestinal barrier,’ Journal of Molecular Medicine, 95(9), pp. 927–934. [CrossRef]
- Chang, P.V. et al. (2014) ’The microbial metabolite butyrate regulates intestinal macrophage function via histone deacetylase inhibition,’ Proceedings of the National Academy of Sciences, 111(6), pp. 2247–2252. [CrossRef]
- Chen, J. et al. (2016) ’An expansion of rare lineage intestinal microbes characterizes rheumatoid arthritis,’ Genome Medicine, 8(1), p. 43. [CrossRef]
- Chriswell, M.E. et al. (2022) ’Clonal IgA and IgG autoantibodies from individuals at risk for rheumatoid arthritis identify an arthritogenic strain of Subdoligranulum,’ Science Translational Medicine, 14(668), p. eabn5166. [CrossRef]
- Ciesielski, O. et al. (2022) ’Citrullination in the pathology of inflammatory and autoimmune disorders: recent advances and future perspectives,’ Cellular and Molecular Life Sciences, 79(2), p. 94. [CrossRef]
- Connors, J., Dawe, N. and Van Limbergen, J. (2018) ’The role of succinate in the regulation of intestinal inflammation,’ Nutrients, 11(1), p. 25. [CrossRef]
- Di Sante, G. et al. (2021) ’Haemophilus parasuis (Glaesserella parasuis) as a Potential Driver of Molecular Mimicry and Inflammation in Rheumatoid Arthritis,’ Frontiers in Medicine, 8, p. 671018. [CrossRef]
- Drago, L. (2019) ’Prevotella Copri and microbiota in rheumatoid arthritis: fully convincing evidence?,’ Journal of Clinical Medicine, 8(11), p. 1837. [CrossRef]
- Esmail, H. et al. (2018) ’Complement pathway gene activation and rising circulating immune complexes characterize early disease in HIV-associated tuberculosis,’ Proceedings of the National Academy of Sciences, 115(5), pp. E964–E973. [CrossRef]
- Fasano, A. (2020) ’All disease begins in the (leaky) gut: role of zonulin-mediated gut permeability in the pathogenesis of some chronic inflammatory diseases,’ F1000Research, 9, p. 69. [CrossRef]
- Fernández-Veledo, S. and Vendrell, J. (2019) ’Gut microbiota-derived succinate: Friend or foe in human metabolic diseases?,’ Reviews in Endocrine and Metabolic Disorders, 20(4), pp. 439–447. [CrossRef]
- Foulquier, C. et al. (2007) ’Peptidyl arginine deiminase type 2 (PAD-2) and PAD-4 but not PAD-1, PAD-3, and PAD-6 are expressed in rheumatoid arthritis synovium in close association with tissue inflammation,’ Arthritis & Rheumatism, 56(11), pp. 3541–3553. [CrossRef]
- Fusco, W. et al. (2023) ’Short-Chain Fatty-Acid-Producing bacteria: key components of the human gut microbiota,’ Nutrients, 15(9), p. 2211. [CrossRef]
- González-Febles, J. et al. (2020) ’Association between periodontitis and anti-citrullinated protein antibodies in rheumatoid arthritis patients: a cross-sectional study,’ Arthritis Research & Therapy, 22(1), p. 27. [CrossRef]
- Guo, Q. et al. (2018) ’Rheumatoid arthritis: pathological mechanisms and modern pharmacologic therapies,’ Bone Research, 6(1), p. 15. [CrossRef]
- Hammad, D.B.M., Liyanapathirana, V. and Tonge, D.P. (2019) ’Molecular characterisation of the synovial fluid microbiome in rheumatoid arthritis patients and healthy control subjects,’ PLoS ONE, 14(11), p. e0225110. [CrossRef]
- Harber, K.J. et al. (2020) ’Succinate is an Inflammation-Induced immunoregulatory metabolite in macrophages,’ Metabolites, 10(9), p. 372. [CrossRef]
- Hickey, F.B. and Martin, F. (2013) ’Diabetic kidney disease and immune modulation,’ Current Opinion in Pharmacology, 13(4), pp. 602–612. [CrossRef]
- Holers, V.M. et al. (2018) ’Rheumatoid arthritis and the mucosal origins hypothesis: protection turns to destruction,’ Nature Reviews Rheumatology, 14(9), pp. 542–557. [CrossRef]
- Holers, V.M. et al. (2024) ’Distinct mucosal endotypes as initiators and drivers of rheumatoid arthritis,’ Nature Reviews Rheumatology, 20(10), pp. 601–613. [CrossRef]
- Huang, H. et al. (2024) ’Cellular succinate metabolism and signaling in inflammation: implications for therapeutic intervention,’ Frontiers in Immunology, 15, p. 1404441. [CrossRef]
- Huang, Y. et al. (2020) ’Prevotella induces the production of TH17 cells in the colon of mice,’ Journal of Immunology Research, 2020(1), p. 9607328. [CrossRef]
- Iljazovic, A. et al. (2020) ’Perturbation of the gut microbiome by Prevotella spp. enhances host susceptibility to mucosal inflammation,’ Mucosal Immunology, 14(1), pp. 113–124. [CrossRef]
- Ishikawa, Y. and Terao, C. (2020) ’The Impact of cigarette smoking on risk of rheumatoid arthritis: A Narrative review,’ Cells, 9(2), p. 475. [CrossRef]
- Javed, I. and Crowson, C.S. (2025) ’The apprehension of seronegative rheumatoid arthritis,’ Nature Reviews Rheumatology, 21(10), pp. 575–576. [CrossRef]
- Kasiraja, V., Bakar, N.A.A. and Suliman, N.A. (2025) ’Rheumatoid arthritis unmasked: the immune complex as a key driver of disease progression,’ Exploration of Immunology, 5. [CrossRef]
- Kim, M.H. et al. (2013) ’Short-Chain fatty acids activate GPR41 and GPR43 on intestinal epithelial cells to promote inflammatory responses in mice,’ Gastroenterology, 145(2), pp. 396-406.e10. [CrossRef]
- Kinloch, A. et al. (2008) ’Synovial fluid is a site of citrullination of autoantigens in inflammatory arthritis,’ Arthritis & Rheumatism, 58(8), pp. 2287–2295. [CrossRef]
- Kuhn, K.A. et al. (2025) ’Bacterial catabolism of dietary tryptophan into indole enhances DC-mediated pro-inflammatory responses in autoimmunity 3709,’ The Journal of Immunology, 214(Supplement_1). [CrossRef]
- Kumar, V., Abbas, A.K. and Aster, J.C. (2012) Robbins Basic Pathology E-Book. Elsevier Health Sciences.
- Laurent, L. et al. (2014) ’IgM rheumatoid factor amplifies the inflammatory response of macrophages induced by the rheumatoid arthritis-specific immune complexes containing anticitrullinated protein antibodies,’ Annals of the Rheumatic Diseases, 74(7), pp. 1425–1431. [CrossRef]
- Lee, J.Y. et al. (2015) ’Association between anti-Porphyromonas gingivalis or anti-α-enolase antibody and severity of periodontitis or rheumatoid arthritis (RA) disease activity in RA,’ BMC Musculoskeletal Disorders, 16(1), p. 190. [CrossRef]
- Li, J. and Kuhn, K.A. (2025) ’Microbial threads in the tapestry of rheumatoid arthritis,’ Journal of Clinical Investigation, 135(18). [CrossRef]
- Li, J. et al. (2020) ’Targeted Combination of Antioxidative and Anti-Inflammatory Therapy of Rheumatoid Arthritis using Multifunctional Dendrimer-Entrapped Gold Nanoparticles as a Platform,’ Small, 16(49), p. e2005661. [CrossRef]
- Lu, J. et al. (2025) ’Linking microbial communities to rheumatoid arthritis: focus on gut, oral microbiome and their extracellular vesicles,’ Frontiers in Immunology, 16, p. 1503474. [CrossRef]
- Lucchino, B. et al. (2019) ’Mucosa–Environment interactions in the pathogenesis of rheumatoid arthritis,’ Cells, 8(7), p. 700. [CrossRef]
- Maeda, Y. and Takeda, K. (2019) ’Host–microbiota interactions in rheumatoid arthritis,’ Experimental & Molecular Medicine, 51(12), pp. 1–6. [CrossRef]
- Maeda, Y. et al. (2016) ’Dysbiosis contributes to arthritis development via activation of autoreactive T cells in the intestine,’ Arthritis & Rheumatology, 68(11), pp. 2646–2661. [CrossRef]
- Mansouri, A., Akthar, I. and Miyamoto, A. (2025) ’TLR2 and TLR4 bridge physiological and pathological inflammation in the reproductive system,’ Communications Biology, 8(1), p. 1008. [CrossRef]
- Maslowski, K.M. et al. (2009) ’Regulation of inflammatory responses by gut microbiota and chemoattractant receptor GPR43,’ Nature, 461(7268), pp. 1282–1286. [CrossRef]
- Matei, D.E. et al. (2021) ’Intestinal barrier dysfunction plays an integral role in arthritis pathology and can be targeted to ameliorate disease,’ Med, 2(7), pp. 864-883.e9. [CrossRef]
- Menofy, N.G.E. et al. (2022) ’Bacterial Compositional Shifts of Gut Microbiomes in Patients with Rheumatoid Arthritis in Association with Disease Activity,’ Microorganisms, 10(9), p. 1820. [CrossRef]
- Mohsenzadeh, A. et al. (2025) ’The gut microbiota and cardiovascular disease: Exploring the role of microbial dysbiosis and metabolites in pathogenesis and therapeutics,’ Life Sciences, 381, p. 123981. [CrossRef]
- Moten, D. et al. (2022) ’Molecular Mimicry of the Rheumatoid Arthritis-Related Immunodominant T-Cell Epitope within Type II Collagen (CII260-270) by the Bacterial L-Asparaginase,’ International Journal of Molecular Sciences, 23(16), p. 9149. [CrossRef]
- Nastasi, C. et al. (2015) ’The effect of short-chain fatty acids on human monocyte-derived dendritic cells,’ Scientific Reports, 5(1), p. 16148. [CrossRef]
- Nonaka, S., Kadowaki, T. and Nakanishi, H. (2022) ’Secreted gingipains from Porphyromonas gingivalis increase permeability in human cerebral microvascular endothelial cells through intracellular degradation of tight junction proteins,’ Neurochemistry International, 154, p. 105282. [CrossRef]
- Ordoñez-Rodriguez, A. et al. (2023) ’Changes in gut microbiota and multiple sclerosis: A Systematic review,’ International Journal of Environmental Research and Public Health, 20(5), p. 4624. [CrossRef]
- Ossipova, E. et al. (2014) ’Affinity purified anti-citrullinated protein/peptide antibodies target antigens expressed in the rheumatoid joint,’ Arthritis Research & Therapy, 16(4), p. R167. [CrossRef]
- Paradis, T. et al. (2021) ’Tight junctions as a key for pathogens invasion in intestinal epithelial cells,’ International Journal of Molecular Sciences, 22(5), p. 2506. [CrossRef]
- Paroli, M. and Sirinian, M.I. (2023) ’When autoantibodies are missing: The challenge of seronegative rheumatoid arthritis,’ Antibodies, 12(4), p. 69. [CrossRef]
- Pianta, A. et al. (2016) ’Evidence of the Immune Relevance of Prevotella copri, a Gut Microbe, in Patients With Rheumatoid Arthritis,’ Arthritis & Rheumatology, 69(5), pp. 964–975. [CrossRef]
- Pianta, A. et al. (2017) ’Two rheumatoid arthritis–specific autoantigens correlate microbial immunity with autoimmune responses in joints,’ Journal of Clinical Investigation, 127(8), pp. 2946–2956. [CrossRef]
- Pianta, A. et al. (2021) ’Identification of Novel, Immunogenic HLA–DR-Presented Prevotella copri Peptides in Patients With Rheumatoid Arthritis,’ Arthritis & Rheumatology, 73(12), pp. 2200–2205. [CrossRef]
- Pitter, M.R. and Zou, W. (2025) ’Citrullination in tumor immunity and therapy,’ Journal of Clinical Investigation, 135(20). [CrossRef]
- Pratt, A.G. and Isaacs, J.D. (2014) ’Seronegative rheumatoid arthritis: Pathogenetic and therapeutic aspects,’ Best Practice & Research Clinical Rheumatology, 28(4), pp. 651–659. [CrossRef]
- Van der Heijden, I M et al. “Presence of bacterial DNA and bacterial peptidoglycans in joints of patients with rheumatoid arthritis and other arthritides.” Arthritis and rheumatism vol. 43,3 (2000): 593-8. [CrossRef]
- Regueiro, C. et al. (2020) ’A predominant involvement of the triple seropositive patients and others with rheumatoid factor in the association of smoking with rheumatoid arthritis,’ Scientific Reports, 10(1), p. 3355. [CrossRef]
- Reichardt, N. et al. (2014) ’Phylogenetic distribution of three pathways for propionate production within the human gut microbiota,’ The ISME Journal, 8(6), pp. 1323–1335. [CrossRef]
- Sakurai, T., Odamaki, T. and Xiao, J.-Z. (2019) ’Production of Indole-3-Lactic acid by bifidobacterium strains isolated from Human Infants,’ Microorganisms, 7(9), p. 340. [CrossRef]
- Santana, P.T. et al. (2022) ’Dysbiosis in inflammatory bowel Disease: Pathogenic role and potential therapeutic targets,’ International Journal of Molecular Sciences, 23(7), p. 3464. [CrossRef]
- Saraiva, A.L. et al. (2018) ’Succinate receptor deficiency attenuates arthritis by reducing dendritic cell traffic and expansion of T h 17 cells in the lymph nodes,’ The FASEB Journal, 32(12), pp. 6550–6558. [CrossRef]
- Scher, J.U. et al. (2013) ’Expansion of intestinal Prevotella copri correlates with enhanced susceptibility to arthritis,’ eLife, 2, p. e01202. [CrossRef]
- Serek, P. and Oleksy-Wawrzyniak, M. (2021) ’The effect of bacterial infections, probiotics and zonulin on intestinal barrier integrity,’ International Journal of Molecular Sciences, 22(21), p. 11359. [CrossRef]
- Seymour, B.J. et al. (2023) ’Microbiota-dependent indole production stimulates the development of collagen-induced arthritis in mice,’ Journal of Clinical Investigation, 134(4). [CrossRef]
- Seymour, B.J., Allen, B.E. and Kuhn, K.A. (2024) ’Microbial mechanisms of rheumatoid arthritis pathogenesis,’ Current Rheumatology Reports, 26(4), pp. 124–132. [CrossRef]
- Smith, P.M. et al. (2013) ’The Microbial Metabolites, Short-Chain Fatty Acids, Regulate Colonic T reg Cell Homeostasis,’ Science, 341(6145), pp. 569–573. [CrossRef]
- Smolen, J.S. et al. (2018) ’Rheumatoid arthritis,’ Nature Reviews Disease Primers, 4(1), p. 18001. [CrossRef]
- Spindler, M.P. et al. (2022) ’Human gut microbiota stimulate defined innate immune responses that vary from phylum to strain,’ Cell Host & Microbe, 30(10), pp. 1481-1498.e5. [CrossRef]
- Sun, M. et al. (2016) ’Microbiota metabolite short chain fatty acids, GPCR, and inflammatory bowel diseases,’ Journal of Gastroenterology, 52(1), pp. 1–8. [CrossRef]
- Tajik, N. et al. (2020) ’Targeting zonulin and intestinal epithelial barrier function to prevent onset of arthritis,’ Nature Communications, 11(1), p. 1995. [CrossRef]
- Taylor, P.C. (2020) ’Update on the diagnosis and management of early rheumatoid arthritis,’ Clinical Medicine, 20(6), pp. 561–564. [CrossRef]
- Tedeschi, S.K. et al. (2017) ’Diet and rheumatoid arthritis symptoms: Survey results from a rheumatoid arthritis registry,’ Arthritis Care & Research, 69(12), pp. 1920–1925. [CrossRef]
- Torres, J.S.S. and Annamaraju, P. (2025) Type III hypersensitivity reaction. https://www.ncbi.nlm.nih.gov/books/NBK559122/.
- Trauelsen, M. et al. (2017) ’Receptor structure-based discovery of non-metabolite agonists for the succinate receptor GPR91,’ Molecular Metabolism, 6(12), pp. 1585–1596. [CrossRef]
- Trauelsen, M. et al. (2021) ’Extracellular succinate hyperpolarizes M2 macrophages through SUCNR1/GPR91-mediated Gq signaling,’ Cell Reports, 35(11), p. 109246. [CrossRef]
- Velikova, T. et al. (2024) ’Mucosal immunity and trained innate immunity of the gut,’ Gastroenterology Insights, 15(3), pp. 661–675. [CrossRef]
- Venetsanopoulou, A.I. et al. (2023) ’Epidemiology and risk factors for rheumatoid arthritis development,’ Mediterranean Journal of Rheumatology, 34(4), p. 404. [CrossRef]
- Vinolo, M. a. R. et al. (2008) ’Effects of short chain fatty acids on effector mechanisms of neutrophils,’ Cell Biochemistry and Function, 27(1), pp. 48–55. [CrossRef]
- Vinolo, M. a. R. et al. (2009) ’Short-chain fatty acids stimulate the migration of neutrophils to inflammatory sites,’ Clinical Science, 117(9), pp. 331–338. [CrossRef]
- Watanabe, S. et al. (1987) ’Detection of immunoglobulins and/or complement in kidney tissues from non-obese diabetic (NOD) mice.,’ PubMed, 12(3), pp. 201–8. https://pubmed.ncbi.nlm.nih.gov/3454080.
- Wei, Y.-H. et al. (2023) ’Succinate metabolism and its regulation of host-microbe interactions,’ Gut Microbes, 15(1), p. 2190300. [CrossRef]
- Wu, C.-Y., Yang, H.-Y. and Lai, J.-H. (2020) ’Anti-Citrullinated Protein Antibodies in Patients with Rheumatoid Arthritis: Biological Effects and Mechanisms of Immunopathogenesis,’ International Journal of Molecular Sciences, 21(11), p. 4015. [CrossRef]
- Wu, H.-J. et al. (2010) ’Gut-Residing segmented filamentous bacteria drive autoimmune arthritis via T Helper 17 cells,’ Immunity, 32(6), pp. 815–827. [CrossRef]
- Wu, J. et al. (2025) ’Investigating correlation between gut microbiota and rheumatoid arthritis subtypes by Mendelian randomization,’ Pathogens, 14(4), p. 385. [CrossRef]
- Wu, R. et al. (2024) ’Impaired immune tolerance mediated by reduced Tfr cells in rheumatoid arthritis linked to gut microbiota dysbiosis and altered metabolites,’ Arthritis Research & Therapy, 26(1), p. 21. [CrossRef]
- Xia, Y. et al. (2023) ’Lactobacillus-derived indole-3-lactic acid ameliorates colitis in cesarean-born offspring via activation of aryl hydrocarbon receptor,’ iScience, 26(11), p. 108279. [CrossRef]
- Xiao, X. et al. (2009) ’Cellular and humoral immune responses in the early stages of diabetic nephropathy in NOD mice,’ Journal of Autoimmunity, 32(2), pp. 85–93. [CrossRef]
- Yang, M.-L. et al. (2021) ’Citrullination and PAD enzyme biology in Type 1 diabetes – regulators of inflammation, autoimmunity, and pathology,’ Frontiers in Immunology, 12, p. 678953. [CrossRef]
- Yang, W. et al. (2019) ’Microbiota Metabolite Short-Chain Fatty Acids Facilitate Mucosal Adjuvant Activity of Cholera Toxin through GPR43,’ The Journal of Immunology, 203(1), pp. 282–292. [CrossRef]
- Zhao, Y. et al. (2018) ’Detection and characterization of bacterial nucleic acids in culture-negative synovial tissue and fluid samples from rheumatoid arthritis or osteoarthritis patients,’ Scientific Reports, 8(1), p. 14305. [CrossRef]
- Zhou, X. et al. (2025b) ’Mucosal immune response in biology, disease prevention and treatment,’ Signal Transduction and Targeted Therapy, 10(1), p. 7. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



