Submitted:
24 February 2026
Posted:
26 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Statistical Analyses
2.2. Ethics Statement
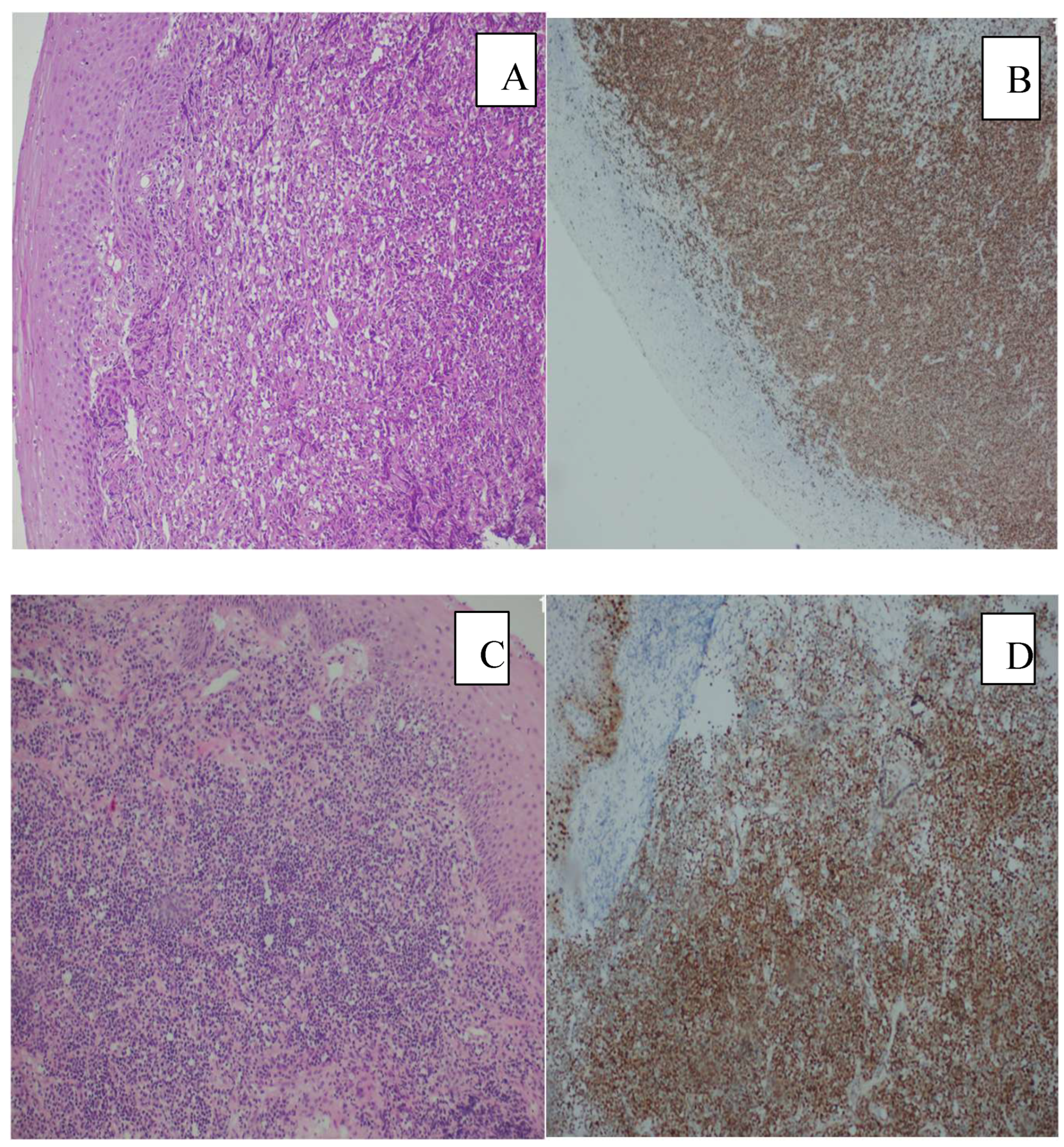
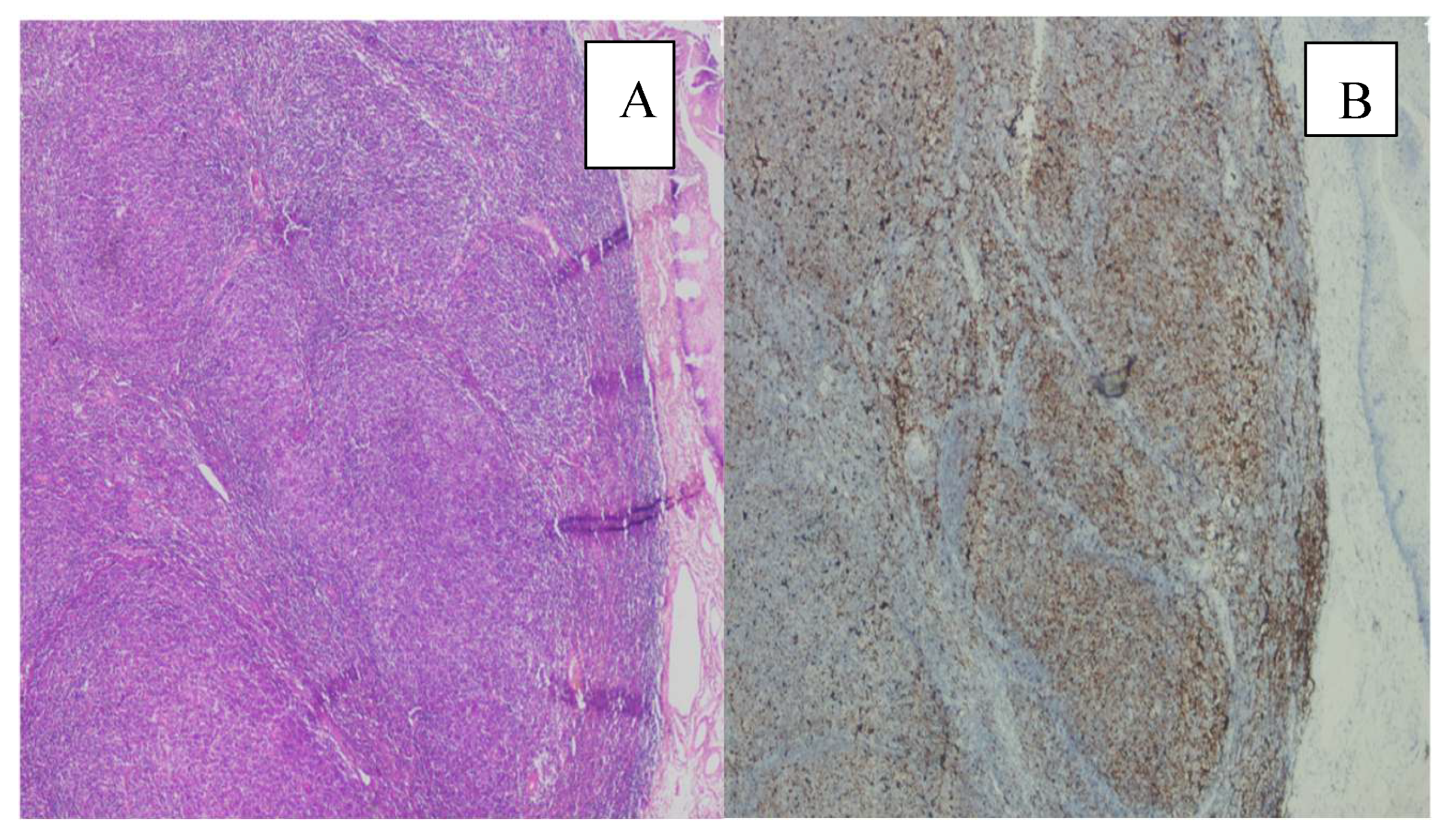
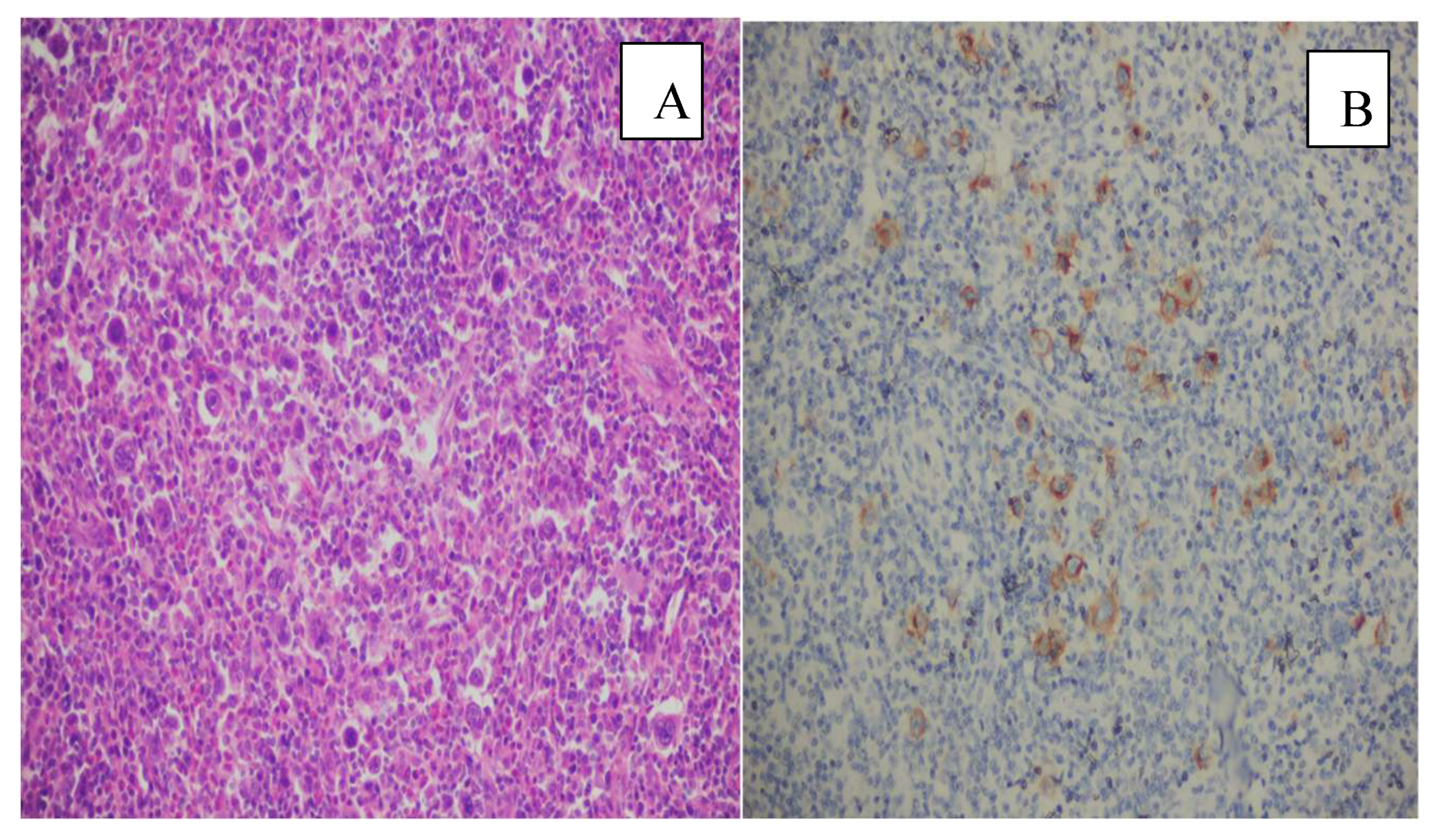
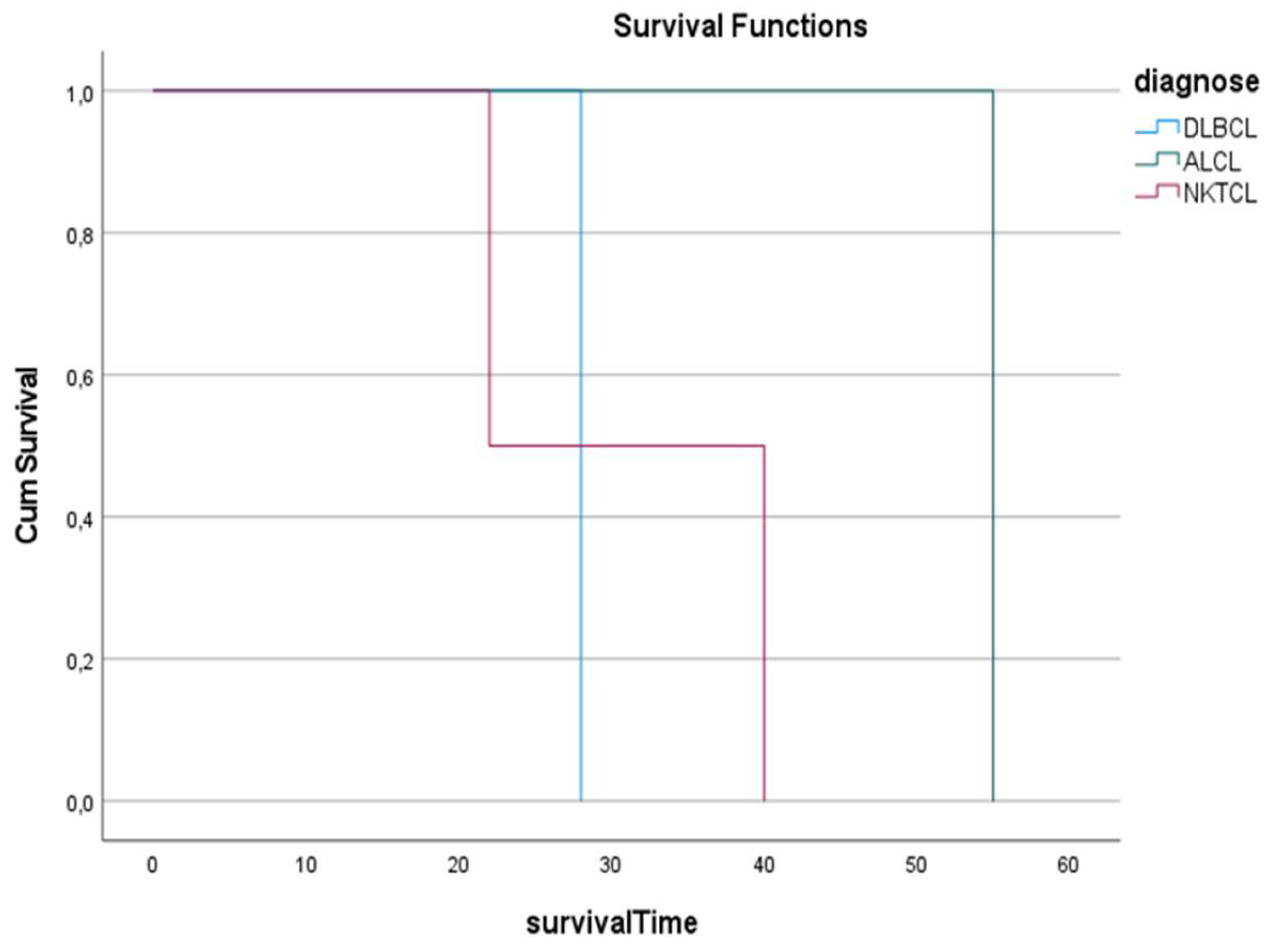
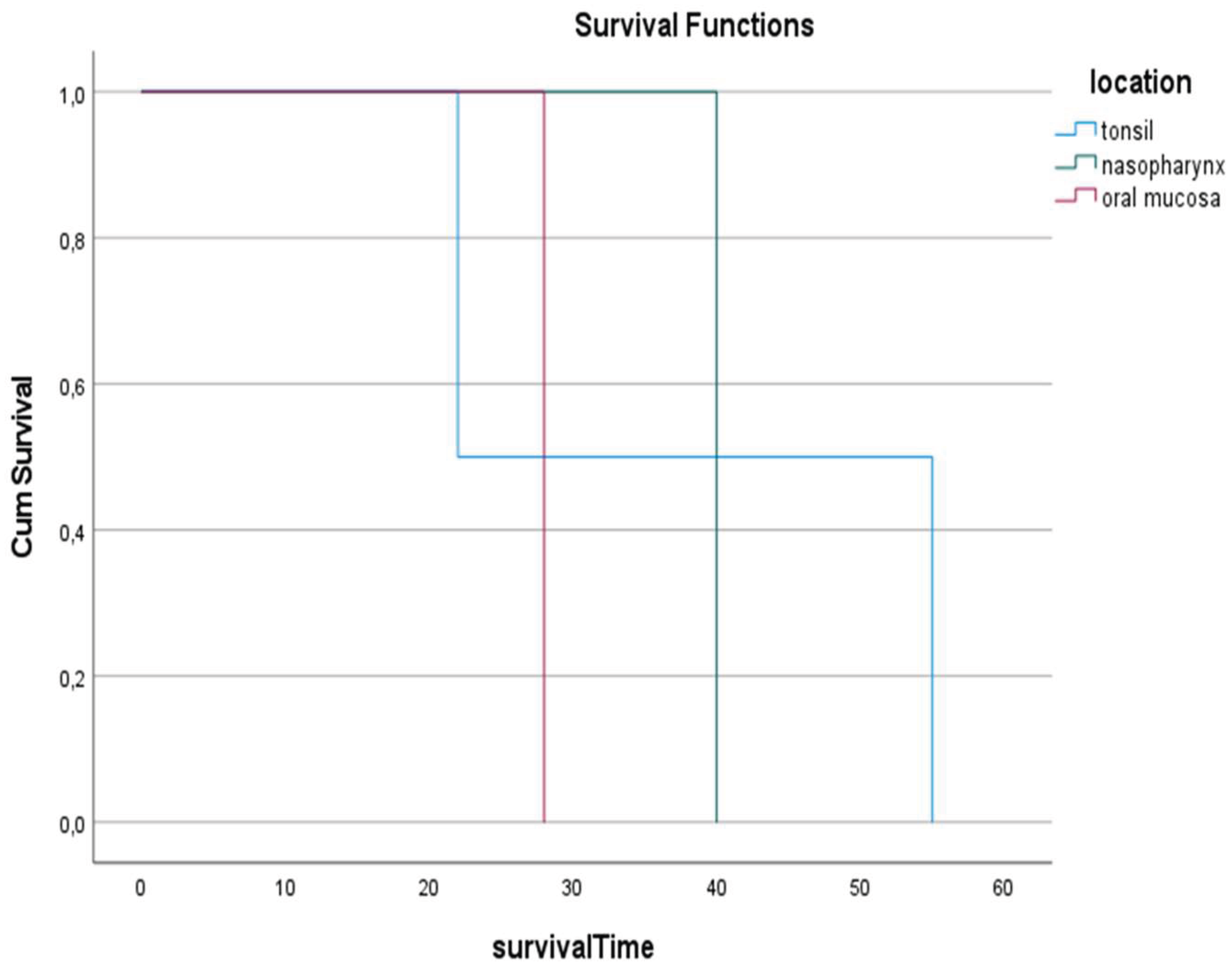
3. Results
4. Discussion
Study Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Vega, F; Lin, Pİ; Medeiros, J. Extra nodal lymhomas of the head and neck. Ann Diagn Pathol. 2005, 9, 340–350. [Google Scholar] [CrossRef] [PubMed]
- Kamiński, B. Lymphomas of the head-and-neck region. J Cancer Res Ther. 2021, 17, 1347–1350. [Google Scholar] [CrossRef] [PubMed]
- Picard, A; Cardinne, C; Denoux, Y; Wagner, I; Chabolle, F; Bach, CA. Extranodal lymphoma of the head and neck: A 67-case series. Eur Ann Otorhinolaryngol Head Neck Dis. 2015, 132, 71–75. [Google Scholar] [CrossRef] [PubMed]
- Mishra, P; Prashar, M; Rehman, N. Primary extranodal lymphomas: Five-year experience from a tertiary care center of North. Indian J Cancer 2024, 61, 16–21. [Google Scholar] [CrossRef] [PubMed]
- Akkari, Y; Alaggio, R; Chan, J. K. C; et al. Who classification of hematolymphoid tumors, Part B, 5th edition; International Agency for Research on Cancer (IARC): Lyon, France, 2024; pp. 295–795. [Google Scholar]
- Akhtar, S; Khafaga, Y; Edesa, W; Al Mubarak, M. Hodgkin lymphoma involving extranodal sites in head and neck: report of twenty-nine cases and review of three-hundred and fifty-seven cases. Hematology 2021, 26, 103–110. [Google Scholar] [CrossRef]
- Bello-Castro, A; Mosquera-Orgueira, A; Gude-Smpedro, F; Varela-Aneiros, I; Seoane-Romero, J; Martin-Biedma, B; Castelo-Baz. Prognostic and survival factors in head and neck extra-nodal non-Hodgkin's lymphoma. Oral Surg Oral Med Oral Pathol Oral Radiol. 2025, 139, 201–210. [Google Scholar] [CrossRef]
- Etemad-Moghadam, S; Tirgary, F; Keshavarz, S; Alaeddini, M. Head and neck non-Hodgkin's lymphoma: A 20-year demographic study of 381 cases Int J Oral Maxillofac Surg. 2010, 39, 869–872. [Google Scholar]
- Wei, MG; Tian, B; Xiong, JK; Feng, J; Wu, ZT; Zhang, X; Zheng, YH. Primary head and neck lymphoid neoplasms in adolescents and young adults: demographics, distribution and survival outcomes. Ann Hematol. 2024, 103, 5871–5880. [Google Scholar] [CrossRef]
- Cabeçadas, J; Martinez, D; Andreasen, S; et al. Lymphomas of the head and neck region: an update. Virchows Arch. 2019, 474, 649–665. [Google Scholar] [CrossRef] [PubMed]
- Brown, NA; Elenitoba-Johnson, KS. Update from the 4th Edition of the World Health Organization Classification of Head and Neck Tumours: Hematolymphoid Tumours. Head Neck Pathol. 2017, 11, 96–109. [Google Scholar] [CrossRef]
- Singh, R; Shaik, S; Negi, BS; Rajguru, JP; Patil, PB; Parihar, AS; Sharma, U. Non-Hodgkin's lymphoma: A review. J Family Med Prim Care 2020, 30, 1834–1840. [Google Scholar]
- Etemad-Moghadam, S; Tirgary, F; Keshavarz, S; Alaeddini, M. Head and neck non-Hodgkin's lymphoma: A 20-year demographic study of 381 cases Int J Oral Maxillofac Surg. 2010, 39, 869–872. [Google Scholar] [PubMed]
- Salplahta, D; Comănescu, MV; Anghelina, F; Ioniţă, E; Mogoantă, CA; Anghelina, L. Non-Hodgkin lymphoma of the Waldeyer’s Ring. Rom J Morphol Embryol. 2012, 53, 1057–1060. [Google Scholar]
- Alli, N; Meer, S. Head and neck lymphomas: A 20-year review in an Oral Pathology Unit, Johannesburg, South Africa, a country with the highest global incidence of HIV/AIDS. Oral Oncol. 2017, 67, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Vega, F; Lin, P; Medeiros, LJ. Extranodal lymphomas of the head and neck. Ann Diagn Pathol. 2005, 9, 340–350. [Google Scholar] [CrossRef]
- Auerbach, A; Aguilera, N. Epstein-Barr virus (EBV)-associated lymphoid lesions of the head and neck. Semin Diagn Pathol. 2015, 32, 12–22. [Google Scholar] [CrossRef] [PubMed]
- Ma, JJ; Zhang, H; Wang, CC; Ji, WL; Zhao, Y; Li, XX. Clinical characteristics and prognostic analysis of three hundred and nineteen cases of primary gastrointestinal diffuse large B-cell lymphoma. World J Gastrointest Oncol. 2025, 17, 113661. [Google Scholar] [CrossRef]
- Meyer, PN; Fu, K; Greiner, TC. Immunohistochemical methods for predicting cell of origin and survival in patients with diffuse large B-cell lymphoma treated with rituximab. Clin Oncol. 2011, 29, 200–207. [Google Scholar] [CrossRef] [PubMed]
- Hans, C P; Weisenburger, D D; Greiner, T. C.; et al. Confirmation of the molecular classification of diffuse large B-cell lym-phoma by immunohistochemistry using a tissue microarray. Blood 2004, 103, 275–282. [Google Scholar] [CrossRef]
- Di Santo, D; Bramati, C; Festa, BM; Pace, GM; Comini, LV; Luparello, P; Cascardi, E; Galizia, D; Galli, A; De Virgilio, A; Giordano, L; Bondi, S. Current evidence on diagnosis and treatment of parotid gland lymphomas: a systematic review. Eur Arch Otorhinolaryngol. 2023, 280, 5219–5227. [Google Scholar] [CrossRef]
- Costes, V. Lymphoid lesions of the head and neck. Ann Pathol. 2009, 29, 323–334. [Google Scholar] [CrossRef]
- Fei, F; Reddy, V; Peker, D; Patel, C; Al Diffalha, S. EBV Positive Mucocutaneous Ulcer (EBVMCU): Single Center Series of Three Cases and Review of Literature. Ann Clin Lab Sci. 2021, 51, 124–130. [Google Scholar]
- Quintanilla-Martinez, L; Swerdlow, SH; Tousseyn, T; Barrionuevo, C; Nakamura, S; Jaffe, ES. New concepts in EBV-associated B, T, and NK cell lymphoproliferative disorders. Virchows Arch. 2023, 482, 227–244. [Google Scholar] [CrossRef]
- Rezkallah, E; Hanna, RS; Elsaify, WM. Thyroid Lymphoma: Case Series Review. Am Surg. 2023, 89, 4811–4816. [Google Scholar] [CrossRef] [PubMed]
- Tzioni, MM; Watanabe, N; Chen, Z; Wu, F; Madej, E; Makker, J; Guo, S; Attygalle, AD; Wotherspoon, A; Sugino, K; Ito, K; Du, MQ. Primary thyroid B-cell lymphoma: molecular insights into its clonal evolution and relapse. J Pathol. 2025, 265, 23–131. [Google Scholar] [CrossRef] [PubMed]
- Shastri, A; Janakiram, M; Mantzaris, I; Yu, Y; Londono, JS; Verma, AK; Barta, SK. Sites of extranodal involvement are prognostic in patients with stage 1 follicular lymphoma. Oncotarget 2017, 8, 78410–78418. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, E. Lymphoma: 2023 update on diagnosis and management. Am J Hematol. 2022, 97, 1638–1651. [Google Scholar] [CrossRef]
- Tashakori, M; Kim, DH; Kanagal-Shamanna, R; Vega, F; Miranda, RN; Jain, P; Wang, M; Medeiros, LJ; Ok, CY. Mantle cell lymphoma involving tonsils: a clinicopathologic study of 83 cases. Hum Pathol. 2021, 118, 60–68. [Google Scholar] [CrossRef]
- Georgaki, M; Theofilou, VI; Pettas, E; Piperi, E; Stoufi, E; Panayiotidis, P; Nikitakis, NG. Blastoid Mantle Cell Lymphoma of the Palate: Report of a Rare Aggressive Entity and Review of the Literature. Head Neck Pathol. 2022, 16, 631–642. [Google Scholar] [CrossRef]
- de Andrade, BAB; Fontes, MD; Roza, ALOC; Vargas, PA; Agostini, M; Canedo, NHS; Ramos, DD; Morais, JC; Milito, CB; Romañach, MJ. Anaplastic Large Cell Lymphoma with Oral Manifestation: A Series of Four Cases and Literature Review. Head Neck Pathol. 2020, 14, 991–1000. [Google Scholar] [CrossRef]
- de Leval, L; Feldman, AL; Pileri, S; Nakamura, S; Gaulard, P. Extranodal T- and NK-cell lymphomas. Virchows Arch. 2023, 482, 245–264. [Google Scholar] [CrossRef] [PubMed]
- Danielson, DT; Aguilera, NS; Auerbach, A. Head and Neck Classic Hodgkin, T and NK Lymphomas with Eosinophilia. Head Neck Pathol. 2025, 19, 10. [Google Scholar] [CrossRef] [PubMed]
- Iyengar, P; Mazloom, A; Shihadeh, F; et al. Hodgkin lymphoma involving extranodal and nodal head and neck sites: characteristics and outcomes. Cancer 2010, 116, 3825–3829. [Google Scholar] [CrossRef] [PubMed]
- Pereira, MAS; Santos, GR; Legarrea, JMA. A rare development of classical Hodgkin lymphoma in the head and neck region: Case report and review of the literature. J Am Dent Assoc. 2024, 155, 781–786. [Google Scholar] [CrossRef]
| ANTİJEN | CLONE | SOURCE | DİLUTİON |
|---|---|---|---|
| ALK | ALK1 | DAKO | RTU |
| BCL-2 | 124 | DAKO | RTU |
| BCL-6 | PG-B6p | DAKO | RTU |
| CD3 | M7254 | DAKO | RTU |
| CD5 | 4C7 | DAKO | RTU |
| CD8 | C8/144B | DAKO | RTU |
| CD10 | 56C6 | DAKO | RTU |
| CD20 | L26 | DAKO | 1/200 |
| CD30 | Ber-H2 | DAKO | RTU |
| CD43 | DF-T1 | DAKO | RTU |
| CD45 | 2B11+PD7/26 | DAKO | RTU |
| CD56 | 123C3 | DAKO | RTU |
| CD79a | JCB117 | DAKO | RTU |
| CYCLİN D1 | EP12 | DAKO | RTU |
| EMA | E29 | DAKO | 1/200 |
| TİA1 | TİA1 | DAKO | RTU |
| Granzym B | GrB-7 | DAKO | RTU |
| CD30 | M0751 | DAKO | 1/100 |
| CD15 | M3631 | DAKO | 1/100 |
| MUM1 | MUM1p | DAKO | RTU |
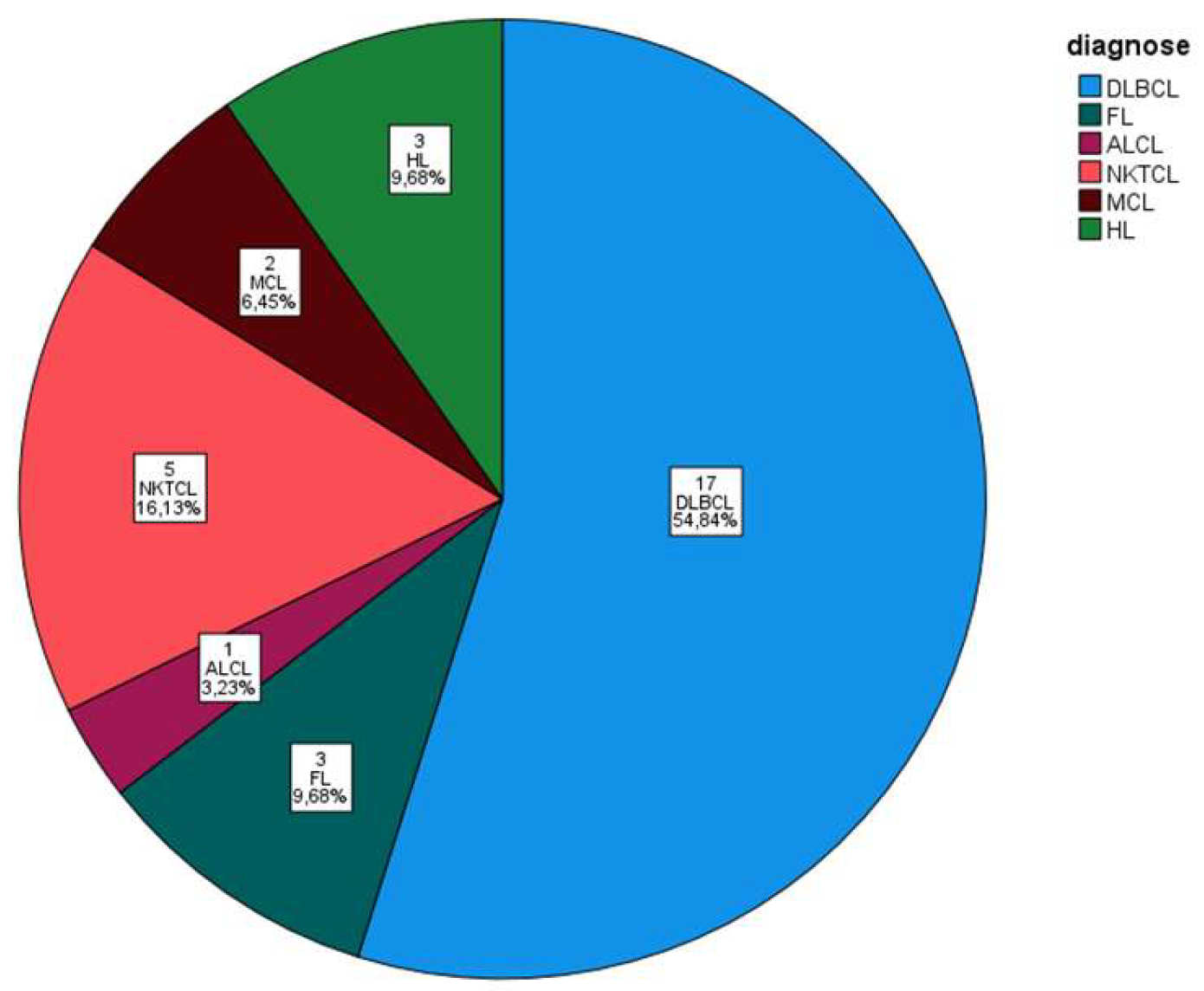
| Tonsil | Naso-pharynx | Oral cavite | Nasal cavite | Salivary gland | Thyroid | Total | |
|---|---|---|---|---|---|---|---|
| DLBCL | 6 | 3 | 4 | 1 | 2 | 1 | 17 |
| FL | 2 | 1 | 0 | 0 | 0 | 0 | 3 |
| ALCL | 1 | 0 | 0 | 0 | 0 | 0 | 1 |
| NKTCL | 1 | 1 | 1 | 2 | 0 | 0 | 5 |
| MCL | 2 | 0 | 0 | 0 | 0 | 0 | 2 |
| HL | 0 | 3 | 0 | 0 | 0 | 0 | 3 |
| Total | 12 | 8 | 5 | 3 | 2 | 1 | 31 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




