Submitted:
21 February 2026
Posted:
26 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
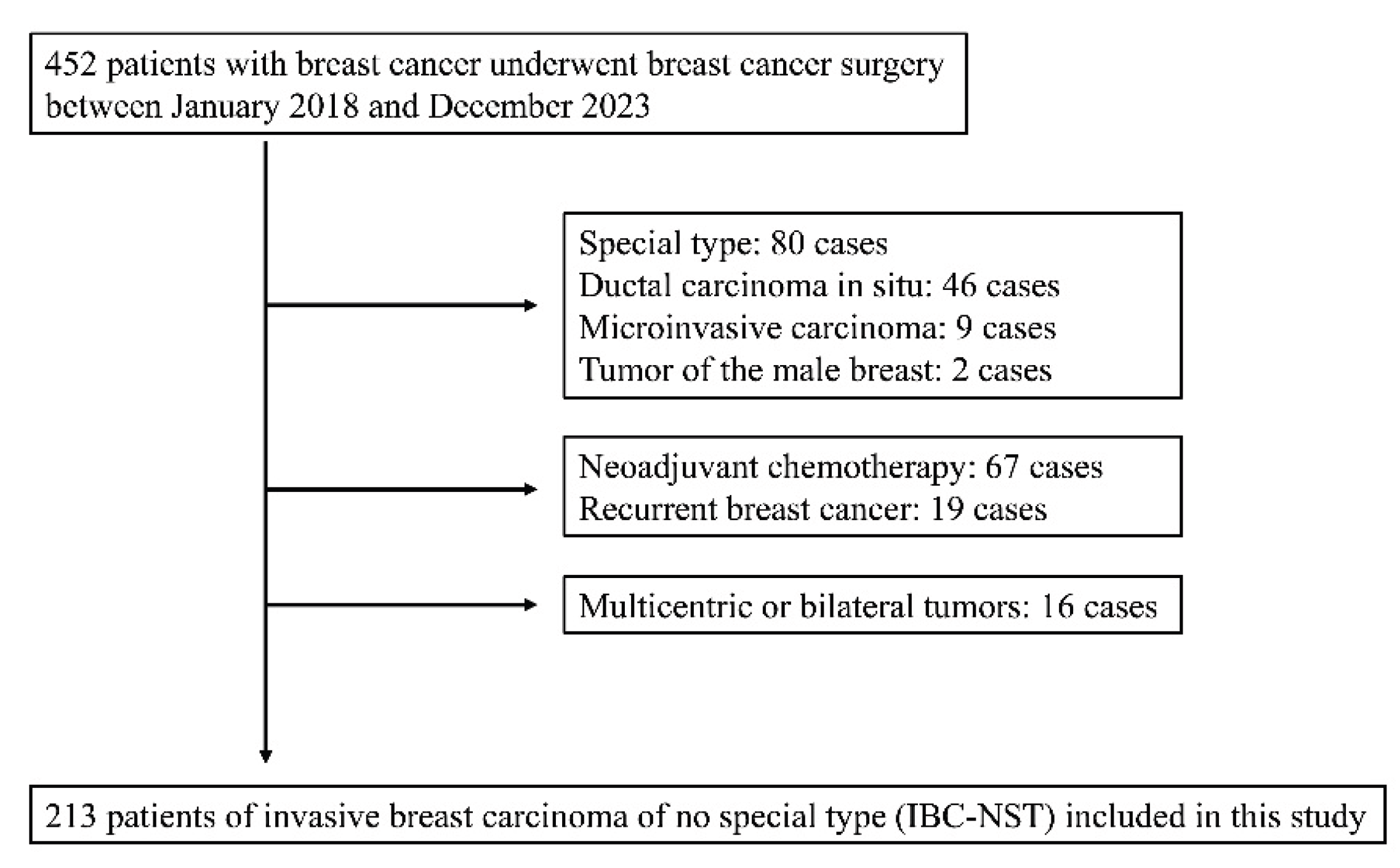
2.1. Case Selection
2.2. Clinical and Pathological Data Collection
2.3. Preparation of Specimens
2.4. Definition of Pathological Parameters
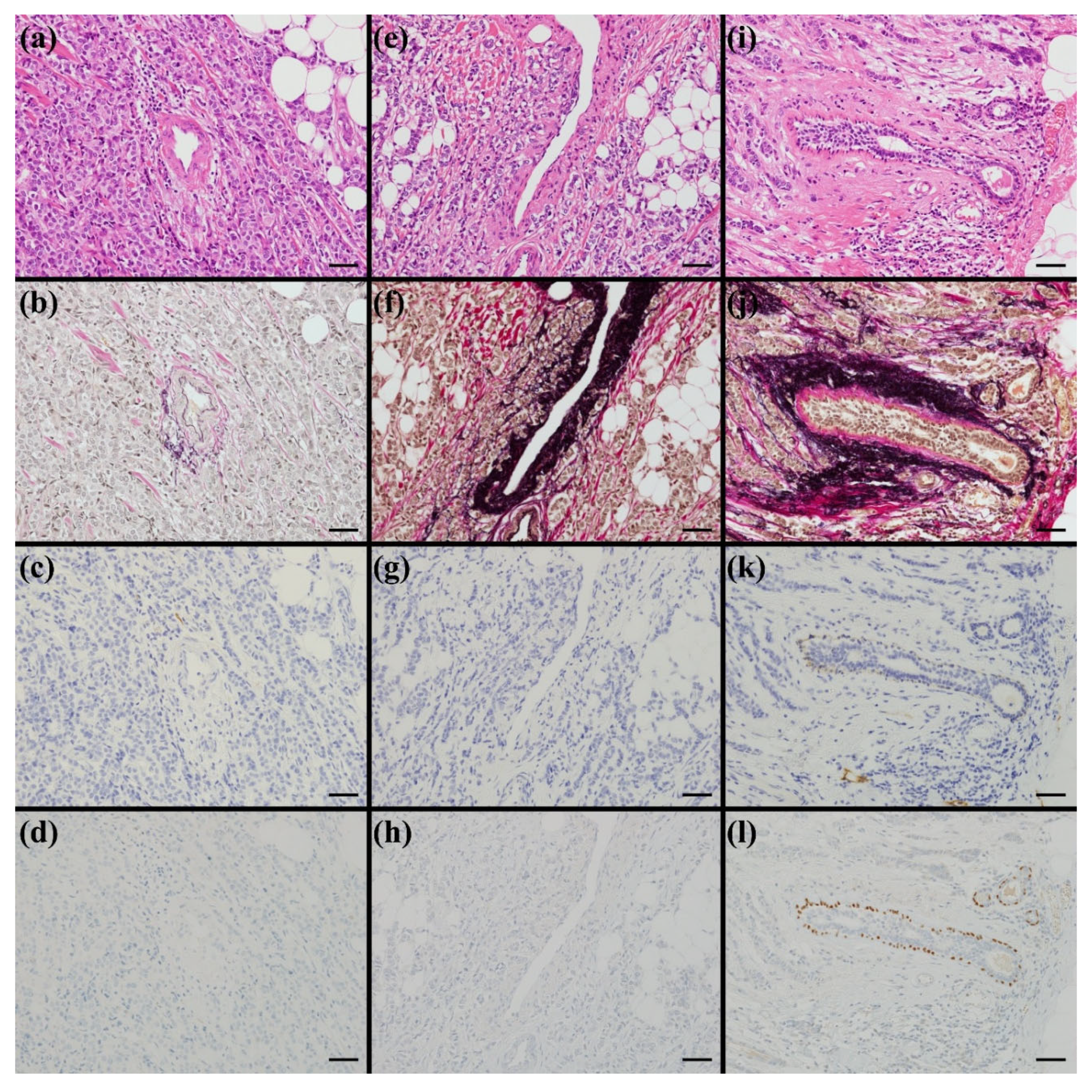
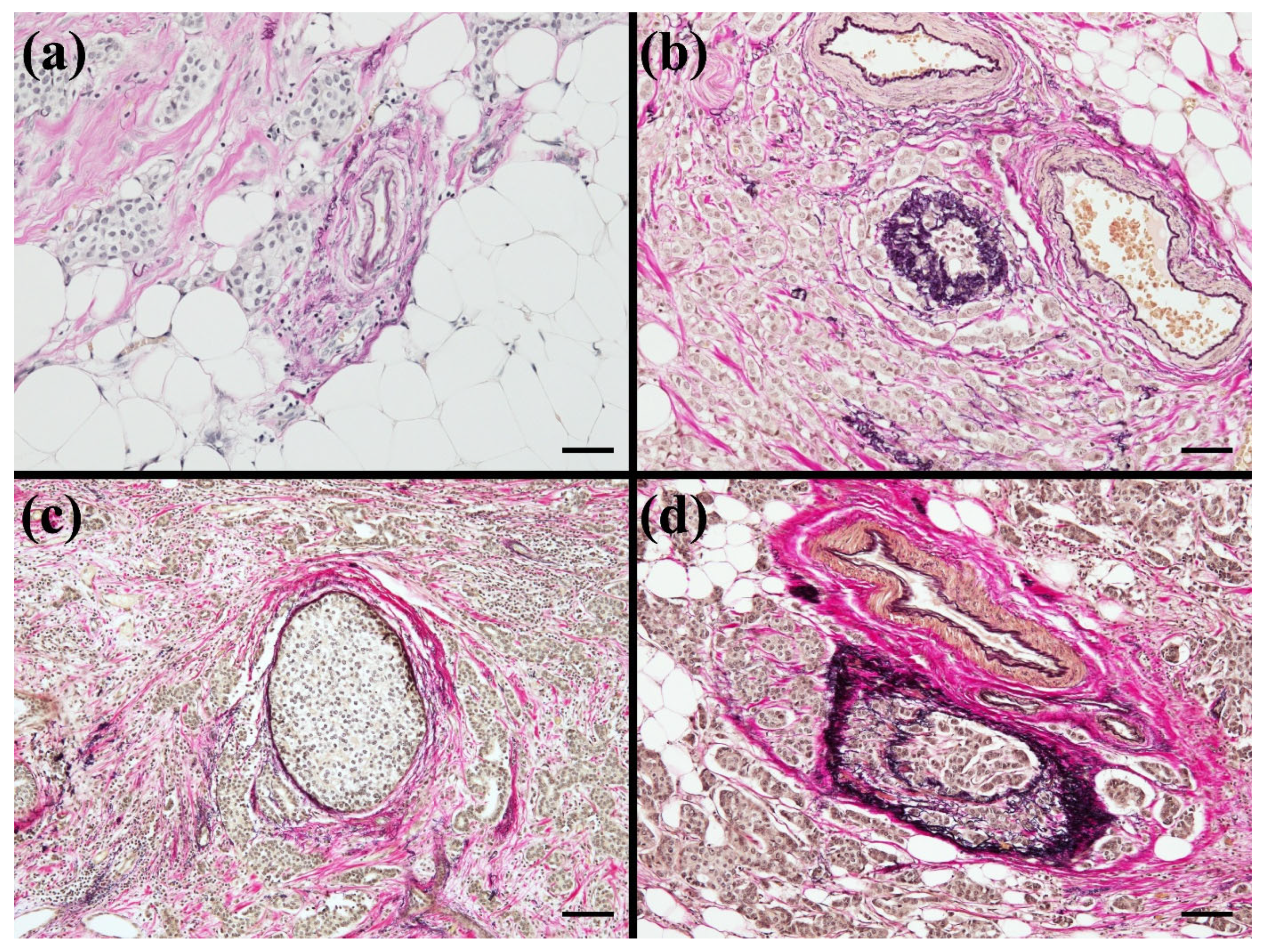
2.5. Lymphatic invasion
2.6. Venous Invasion
2.7. Perivascular and Periductal Invasion
2.8. Retraction Artifact and Perineural Invasion
2.9. Pathological Evaluation and Consensus
2.10. Statistical Analyses
3. Results
3.1. Patient Characteristics
3.2. Correlation Between Lymph Node Metastasis and Clinicopathological Characteristics
3.3. Comparative Analysis of Histological Surrogates for Predicting LNM
3.4. Stratified Analysis by Tumor size (Table 4)
| pT1 | pT2‒4 | ||||||
| N0 (n=99) Number (%) |
N0(i+) and N1-3 (n=31) Number (%) |
P-value | N0 (n=27) Number (%) |
N0(i+) and N1-3 (n=56) Number (%) |
P-value | ||
| Periarterial or perivenous invasion (periA/V) | Periarterial or perivenous invasion (periA/V) | ||||||
| (+) | 72 (72.7) | 31 (100.0) | < 0.001 | (+) | 25 (92.6) | 54 (96.4) | 0.593 |
| (-) | 27 (27.3) | 0 (0.0) | (-) | 2 (7.4) | 2 (3.6) | ||
| Lymphatic invasion in HE staining | Lymphatic invasion in HE staining | ||||||
| (+) | 11 (11.1) | 11 (35.5) | 0.004 | (+) | 9 (33.3) | 18 (32.1) | 1.000 |
| (-) | 88 (88.9) | 20 (64.5) | (-) | 18 (66.7) | 38 (67.9) | ||
| Lymphatic invasion in D2-40 staining | Lymphatic invasion in D2-40 staining | ||||||
| (+) | 14 (14.1) | 10 (32.3) | 0.033 | (+) | 9 (33.3) | 21 (37.5) | 0.810 |
| (-) | 85 (85.9) | 21 (67.7) | (-) | 18 (66.7) | 35 (62.5) | ||
| Retraction artifact | Retraction artifact | ||||||
| (+) | 31 (31.3) | 12 (38.7) | 0.513 | (+) | 13 (48.1) | 39 (69.6) | 0.089 |
| (-) | 68 (68.7) | 19 (61.3) | (-) | 14 (51.9) | 17 (30.4) | ||
| Perineural Invasion | Perineural Invasion | ||||||
| (+) | 19 (19.2) | 11 (35.5) | 0.086 | (+) | 10 (37.0) | 20 (35.7) | 1.000 |
| (-) | 80 (80.8) | 20 (64.5) | (-) | 17 (63.0) | 36 (64.3) | ||
| EVG, Elastica‒van Gieson; HE, hematoxylin and eosin | |||||||
3.5. Stratified Logistic Regression Analysis by Tumor Size (Table 5)
3.5.1. Associated factors in the pT1 group (≤20 mm)
3.5.2. Associated factors in the pT2–4 group (>20 mm)
| pT1 | |||||||
| Univariate | Multivariate | VIF | |||||
| Odds ratio | 95% CI | P-value | Odds ratio | 95% CI | P-value | ||
| periA/V with EVG stain | 23.897 | 3.161 – 3063.945 | < 0.001 | 16.080 | 2.019 – 2081.931 | 0.004 | 1.000 |
| Lymphatic invasion with HE stain | 4.317 | 1.669 – 11.297 | 0.003 | 2.790 | 0.789 – 10.229 | 0.110 | 1.747 |
| Lymphatic invasion with D2-40 | 2.880 | 1.126 – 7.262 | 0.028 | 1.452 | 0.381 – 5.127 | 0.571 | 1.737 |
| Retraction artifact | 1.394 | 0.600 – 3.166 | 0.434 | 1.030 | 0.398 – 2.554 | 0.950 | 1.092 |
| Perineural Invasion | 2.316 | 0.950 – 5.541 | 0.064 | 1.822 | 0.718 – 4.617 | 0.204 | 1.043 |
| pT2-4 | |||||||
| Univariate | Multivariate | VIF | |||||
| Odds ratio | 95% CI | P-value | Odds ratio | 95% CI | P-value | ||
| periA/V with EVG stain | 2.160 | 0.288 – 16.2 | 0.454 | 2.630 | 0.306 – 22.60 | 0.378 | 1.086 |
| Lymphatic invasion with HE stain | 0.947 | 0.357 – 2.52 | 0.914 | 0.626 | 0.179 – 2.19 | 0.464 | 1.559 |
| Lymphatic invasion with D2-40 | 1.200 | 0.457 – 3.15 | 0.711 | 1.270 | 0.375 – 4.31 | 0.699 | 1.508 |
| Retraction artifact | 2.470 | 0.960 – 6.36 | 0.061 | 2.800 | 1.010 – 7.75 | 0.047 | 1.137 |
| Perineural Invasion | 0.944 | 0.364 – 2.45 | 0.906 | 1.160 | 0.412 – 3.25 | 0.781 | 1.111 |
| Elastica van Gieson (EVG), Hematoxylin and eosin (HE), confidence interval (CI), variance inflation factor (VIF) | |||||||
3.6. Subgroup Analysis Within pT1 Categories (Table 6)
3.6.1. pT1a Group (≤5 mm)
3.6.2. pT1b Group (6–10 mm)
3.6.3. pT1c Group (11–20 mm)
| pT1a | pT1b | pT1c | |||||||||
| N0 (n=11) Number (%) |
N0(i+) and N1-3 (n=0) Number (%) |
P-value | N0 (n=39) Number (%) |
N0(i+) and N1-3 (n=5) Number (%) |
P-value | N0 (n=49) Number (%) |
N0(i+) and N1-3 (n=26) Number (%) |
P-value | |||
| Periarterial or perivenous invasion (periA/V) | Periarterial or perivenous invasion (periA/V) | Periarterial or perivenous invasion (periA/V) | |||||||||
| (+) | 3 (27.3) | 0 | - | (+) | 32 (82.1) | 5 (100) | 0.574 | (+) | 37 (75.5) | 26 (100) | 0.006 |
| (-) | 8 (72.7) | 0 | (-) | 7 (17.9) | 0 (0) | (-) | 12 (24.5) | 0 (0) | |||
| Lymphatic invasion in HE staining | Lymphatic invasion in HE staining | Lymphatic invasion in HE staining | |||||||||
| (+) | 1 (9.1) | 0 | - | (+) | 3 (7.7) | 2 (40) | 0.091 | (+) | 7 (14.3) | 9 (34.6) | 0.073 |
| (-) | 10 (90.9) | 0 | (-) | 36 (92.3) | 3 (60) | (-) | 42 (85.7) | 17 (65.4) | |||
| Lymphatic invasion in D2-40 staining | Lymphatic invasion in D2-40 staining | Lymphatic invasion in D2-40 staining | |||||||||
| (+) | 1 (9.1) | 0 | - | (+) | 4 (10.3) | 1 (20) | 0.470 | (+) | 9 (18.4) | 9 (34.6) | 0.157 |
| (-) | 10 (90.9) | 0 | (-) | 35 (89.7) | 4 (80) | (-) | 40 (81.6) | 17 (65.4) | |||
| Retraction artifact | Retraction artifact | Retraction artifact | |||||||||
| (+) | 3 (27.3) | 0 | - | (+) | 6 (15.4) | 2 (40) | 0.219 | (+) | 22 (44.9) | 10 (38.5) | 0.632 |
| (-) | 8 (72.7) | 0 | (-) | 33 (84.6) | 3 (60) | (-) | 27 (55.1) | 16 (61.5) | |||
| Perineural Invasion | Perineural Invasion | Perineural Invasion | |||||||||
| (+) | 0 (0) | 0 | - | (+) | 6 (15.4) | 2 (40) | 0.219 | (+) | 13 (26.5) | 9 (34.6) | 0.595 |
| (-) | 11 (100) | 0 | (-) | 33 (84.6) | 3 (60) | (-) | 36 (73.5) | 17 (65.4) | |||
| EVG, Elastica‒van Gieson; HE, hematoxylin and eosin | |||||||||||
| pT1b | |||||||
| Univariate | Multivariate | VIF | |||||
| Odds ratio | 95% CI | P-value | Odds ratio | 95% CI | P-value | ||
| periA/V with EVG stain | 2.538 | 0.240 – 346.838 | 0.498 | 2.408 | 0.131 – 489.364 | 0.604 | 1.000 |
| Lymphatic invasion with HE stain | 7.449 | 0.970 – 56.312 | 0.053 | 8.695 | 0.801 – 130.15 | 0.074 | 1.665 |
| Lymphatic invasion with D2-40 | 2.630 | 0.227 – 19.430 | 0.390 | 1.889 | 0.101 – 29.734 | 0.647 | 1.438 |
| Retraction artifact | 3.681 | 0.525 – 23.396 | 0.177 | 7.055 | 0.798 – 88.376 | 0.078 | 1.470 |
| Perineural Invasion | 3.681 | 0.525 – 23.396 | 0.177 | 2.757 | 0.328 – 22.638 | 0.335 | 1.127 |
| pT1c | |||||||
| Univariate | Multivariate | VIF | |||||
| Odds ratio | 95% CI | P-value | Odds ratio | 95% CI | P-value | ||
| periA/V with EVG stain | 17.667 | 2.153 – 2300.087 | 0.003 | 14.700 | 1.687 – 1935.315 | 0.010 | 1.000 |
| Lymphatic invasion with HE stain | 3.076 | 1.023 – 9.61 | 0.045 | 2.922 | 0.568 – 16.79 | 0.198 | 2.179 |
| Lymphatic invasion with D2-40 | 2.314 | 0.798 – 6.784 | 0.121 | 1.558 | 0.305 – 7.509 | 0.578 | 1.974 |
| Retraction artifact | 0.778 | 0.294 – 2.004 | 0.604 | 0.430 | 0.116 – 1.359 | 0.155 | 1.454 |
| Perineural Invasion | 1.468 | 0.528 – 4.018 | 0.457 | 1.063 | 0.357 – 3.142 | 0.911 | 1.048 |
| Elastica van Gieson (EVG), Hematoxylin and eosin (HE), confidence interval (CI), variance inflation factor (VIF) | |||||||
3.7. Logistic Regression Analysis by pT1 Subcategories (Table 7)
3.7.1. pT1b Group (6–10 mm)
3.7.2. pT1c Group (11–20 mm)
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| IBC-NST | Invasive breast carcinoma of no special type |
| LNM | Lymph node metastasis |
| periA/V | Periarterial or perivenous invasion |
| periA | Periarterial invasion |
| periV | Perivenous invasion |
| periD | Periductal invasion |
| LI | Lymphatic invasion |
| VI | Venous invasion |
| PNI | Perineural invasion |
| EVG | Elastica–van Gieson |
| H&E (HE) | Hematoxylin and eosin |
| D2-40 | Podoplanin (lymphatic endothelial marker) |
| DCIS | Ductal carcinoma in situ |
| ER | Estrogen receptor |
| PgR | Progesterone receptor |
| HER2 | Human epidermal growth factor receptor 2 |
| FISH | Fluorescence in situ hybridization |
| SSM | Skin-sparing mastectomy |
| NSM | Nipple-sparing mastectomy |
| OR | Odds ratio |
| CI | Confidence interval |
| NPV | Negative predictive value |
| PPV | Positive predictive value |
| LR+ | Positive likelihood ratio |
| LR− | Negative likelihood ratio |
| VIF | Variance inflation factor |
References
- Shioya, A.; Takata, M.; Kumagai, M.; Hoshi, D.; Han, J.; Oyama, T.; Haba, Y.; Morioka, E.; Inokuchi, M.; Noguchi, M.; et al. Periarterial or perivenous invasion is an independent indicator of lymph node metastasis in invasive breast carcinoma of no special type. Pathol. Res. Pract. 2024, 260, 155407. [Google Scholar] [CrossRef]
- Houvenaeghel, G.; Cohen, M.; Classe, J.M.; Reyal, F.; Mazouni, C.; Chopin, N.; Martinez, A.; Daraï, E.; Coutant, C.; Colombo, P.E.; et al. Lymphovascular invasion has a significant prognostic impact in patients with early breast cancer, results from a large, national, multicenter, retrospective cohort study. ESMO Open 2021, 6, 100316. [Google Scholar] [CrossRef] [PubMed]
- Ejlertsen, B.; Jensen, M.B.; Rank, F.; Rasmussen, B.B.; Christiansen, P.; Kroman, N.; Kvistgaard, M.E.; Overgaard, M.; Toftdahl, D.B.; Mouridsen, H.T. Population-based study of peritumoral lymphovascular invasion and outcome among patients with operable breast cancer. J. Natl Cancer Inst. submitted; accepted. 2009, 101, 729–735. [Google Scholar] [CrossRef]
- Song, Y.J.; Shin, S.H.; Cho, J.S.; Park, M.H.; Yoon, J.H.; Jegal, Y.J. The role of lymphovascular invasion as a prognostic factor in patients with lymph node-positive operable invasive breast cancer. J. Breast Cancer 2011, 14, 198–203. [Google Scholar] [CrossRef]
- Rakha, E.A.; Martin, S.; Lee, A.H.S.; Morgan, D.; Pharoah, P.D.P.; Hodi, Z.; Macmillan, D.; Ellis, I.O. The prognostic significance of lymphovascular invasion in invasive breast carcinoma. Cancer 2012, 118, 3670–3680. [Google Scholar] [CrossRef] [PubMed]
- He, K.W.; Sun, J.J.; Liu, Z.B.; Zhuo, P.Y.; Ma, Q.H.; Liu, Z.Y.; Yu, Z.Y. Prognostic significance of lymphatic vessel invasion diagnosed by D2-40 in Chinese invasive breast cancers. Med. (Baltim.) 2017, 96, e8490. [Google Scholar] [CrossRef]
- Lee, S.J.; Go, J.; Ahn, B.S.; Ahn, J.H.; Kim, J.Y.; Park, H.S.; Kim, S.I.; Park, B.W.; Park, S. Lymphovascular invasion is an independent prognostic factor in breast cancer irrespective of axillary node metastasis and molecular subtypes. Front. Oncol. 2023, 13, 1269971. [Google Scholar] [CrossRef]
- Woo, C.S.; Silberman, H.; Nakamura, S.K.; Ye, W.; Sposto, R.; Colburn, W.; Waisman, J.R.; Silverstein, M.J. Lymph node status combined with lymphovascular invasion creates a more powerful tool for predicting outcome in patients with invasive breast cancer. Am. J. Surg. 2002, 184, 337–340. [Google Scholar] [CrossRef]
- Takada, K.; Kashiwagi, S.; Asano, Y.; Goto, W.; Kouhashi, R.; Yabumoto, A.; Morisaki, T.; Shibutani, M.; Takashima, T.; Fujita, H.; et al. Prediction of lymph node metastasis by tumor-infiltrating lymphocytes in T1 breast cancer. BMC Cancer 2020, 20, 598. [Google Scholar] [CrossRef]
- Gajdos, C.; Tartter, P.I.; Bleiweiss, I.J. Lymphatic invasion, tumor size, and age are independent predictors of axillary lymph node metastases in women with T1 breast cancers. Ann. Surg. 1999, 230, 692–696. [Google Scholar] [CrossRef]
- Karaman, S.; Detmar, M. Mechanisms of lymphatic metastasis. J. Clin. Invest. 2014, 124, 922–928. [Google Scholar] [CrossRef]
- Dieterich, L.C.; Tacconi, C.; Ducoli, L.; Detmar, M. Lymphatic vessels in cancer. Physiol. Rev. 2022, 102, 1837–1879. [Google Scholar] [CrossRef] [PubMed]
- Nathanson, S.D. Insights into the mechanisms of lymph node metastasis. Cancer 2003, 98, 413–423. [Google Scholar] [CrossRef]
- Mohammed, R.A.A.; Martin, S.G.; Gill, M.S.; Green, A.R.; Paish, E.C.; Ellis, I.O. Improved methods of detection of lymphovascular invasion demonstrate that it is the predominant method of vascular invasion in breast cancer and has important clinical consequences. Am. J. Surg. Pathol. 2007, 31, 1825–1833. [Google Scholar] [CrossRef]
- Kahn, H.J.; Marks, A. A new monoclonal antibody, D2-40, for detection of lymphatic invasion in primary tumors. Lab. Invest. 2002, 82, 1255–1257. [Google Scholar] [CrossRef]
- Kanner, W.A.; Galgano, M.T.; Atkins, K.A. Podoplanin expression in basal and myoepithelial cells: utility and potential pitfalls. Appl. Immunohistochem. Mol. Morphol. 2010, 18, 226–230. [Google Scholar] [CrossRef] [PubMed]
- Ordóñez, N.G. Value of podoplanin as an immunohistochemical marker in tumor diagnosis: a review and update. Appl. Immunohistochem. Mol. Morphol. 2014, 22, 331–347. [Google Scholar] [CrossRef]
- Ren, S.; Abuel-Haija, M.; Khurana, J.S.; Zhang, X. D2-40: an additional marker for myoepithelial cells of breast and the precaution in interpreting tumor lymphovascular invasion. Int. J. Clin. Exp. Pathol. 2011, 4, 175–182. [Google Scholar] [PubMed]
- Jokinen, C.H.; Dadras, S.S.; Goldblum, J.R.; van de Rijn, M.; West, R.B.; Rubin, B.P. Diagnostic implications of podoplanin expression in peripheral nerve sheath neoplasms. Am. J. Clin. Pathol. 2008, 129, 886–893. [Google Scholar] [CrossRef]
- Hosoya, K.; Wakahara, M.; Ikeda, K.; Umekita, Y. Perineural invasion predicts unfavorable prognosis in patients with invasive breast cancer. Cancer Diagn. Progn. 2023, 3, 208–214. [Google Scholar] [CrossRef]
- Narayan, P.; Flynn, J.; Zhang, Z.; Gillespie, E.F.; Mueller, B.; Xu, A.J.; Cuaron, J.; McCormick, B.; Khan, A.J.; Cahlon, O.; et al. Perineural invasion as a risk factor for locoregional recurrence of invasive breast cancer. Sci. Rep. 2021, 11, 12781. [Google Scholar] [CrossRef] [PubMed]
- Acs, G.; Dumoff, K.L.; Solin, L.J.; Pasha, T.; Xu, X.; Zhang, P.J. Extensive retraction artifact correlates with lymphatic invasion and nodal metastasis and predicts poor outcome in early stage breast carcinoma. Am. J. Surg. Pathol. 2007, 31, 129–140. [Google Scholar] [CrossRef] [PubMed]
- Zaorsky, N.G.; Patil, N.; Freedman, G.M.; Tuluc, M. Differentiating lymphovascular invasion from retraction artifact on histological specimen of breast carcinoma and their implications on prognosis. J. Breast Cancer 2012, 15, 478–480. [Google Scholar] [CrossRef] [PubMed]
- Kanda, Y. Investigation of the freely available easy-to-use software “EZR” for medical statistics. Bone Marrow Transplant. 2013, 48, 452–458. [Google Scholar] [CrossRef]
- Kurosumi, M. Immunohistochemical assessment of hormone receptor status using a new scoring system (J-score) in breast cancer. Breast Cancer 2007, 14, 189–193. [Google Scholar] [CrossRef]
- Wolff, A.C.; Hammond, M.E.H.; Allison, K.H.; Harvey, B.E.; Mangu, P.B.; Bartlett, J.M.S.; Bilous, M.; Ellis, I.O.; Fitzgibbons, P.; Hanna, W.; et al. Human epidermal growth factor receptor 2 testing in breast cancer: American Society of Clinical Oncology/College of American Pathologists clinical practice guideline focused update. Arch. Pathol. Lab. Med. 2018, 142, 1364–1382. [Google Scholar] [CrossRef]
- Breslin, J.W.; Yang, Y.; Scallan, J.P.; Sweat, R.S.; Adderley, S.P.; Murfee, W.L. Lymphatic vessel network structure and physiology. Compr. Physiol. 2018, 9, 207–299. [Google Scholar] [CrossRef]
- Jesinger, R.A. Breast anatomy for the interventionalist. Tech. Vasc. Interv. Radiol. 2014, 17, 3–9. [Google Scholar] [CrossRef]
- Kuhn, E.; Gambini, D.; Despini, L.; Asnaghi, D.; Runza, L.; Ferrero, S. Updates on lymphovascular invasion in breast cancer. Biomedicines 2023, 11, 968. [Google Scholar] [CrossRef]
- Kazlouskaya, V.; Malhotra, S.; Lambe, J.; Idriss, M.H.; Elston, D.; Andres, C. The utility of elastic Verhoeff-Van Gieson staining in dermatopathology. J. Cutan. Pathol. 2012, 40, 211–225. [Google Scholar] [CrossRef]
- Huang, J.C.; Bhaskar, S.M.M. Clot morphology in acute ischemic stroke decision making. Int. J. Mol. Sci. 2022, 23, 12373. [Google Scholar] [CrossRef]
| Characteristic | Patients (n=213) | Characteristic | Patients (n=213) | Characteristic | Patients (n=213) |
| Age-yrs | Invasive tumor size (greatest dimension, mm) | ER (J-score) | |||
| Average | 60.7 | Average | 19.8 | 3b | 193 |
| Median | 61 | Median | 17 | 3a | 6 |
| Range | 23-92 | Range | 2-90 | 2 | 1 |
| premenopausal | 72 | Total tumor size (mm) | 1 | 2 | |
| postmenopausal | 141 | Average | 37.9 | 0 | 11 |
| Sex | Median | 28 | PgR (J-score) | ||
| Male | 0 | Range | 5-135 | 3b | 130 |
| Female | 213 | Pathological T categories | 3a | 51 | |
| Operative method | 1 (up to 20 mm) | 130 | 2 | 10 | |
| mastectomy (including SSM and NSM) | 107 | 1a (1‒5 mm) | 11 | 1 | 1 |
| partial mastectomy | 106 | 1b (6‒10 mm) | 44 | 0 | 21 |
| Lymph node dissection | 1c (11‒20 mm) | 75 | HER2 score | ||
| performed | 52 | 2 (21‒50 mm) | 73 | 3+ | 10 |
| not performed | 161 | 3 (>50 mm) | 6 | 2+ and FISH(+) | 8 |
| primary tumor location | 4 | 4 | 2+ and FISH(-) | 27 | |
| Left breast | 106 | Regional lymph node metastasis | 1+ | 130 | |
| Right breast | 107 | N0 | 126 | 0 | 38 |
| Anatomical Subsites | N+ (including N0(i+), N1mi, N1-3) | 87 | Ki-67 labeling index (hot spot) | ||
| Upper-inner quadrant | 51 | N0(i+) | 7 | Average (%) | 25.9 |
| Lower-inner quadrant | 28 | N1mi | 14 | Median (%) | 21.1 |
| Upper-outer quadrant | 90 | N1 | 46 | Range (%) | 0.5-88.6 |
| Lower-outer quadrant | 27 | N2 | 14 | >20% | 113 |
| Central portion | 12 | N3 | 6 | ≤ 20% | 100 |
| Axillary tail | 5 | Distant metastasis | |||
| Nuclear grade | M0 | 211 | |||
| 1 | 87 | M1 | 2 | ||
| 2 | 37 | pTNM stage | |||
| 3 | 89 | IA | 104 | ||
| Histological grade | IB | 5 | |||
| I | 73 | IIA | 45 | ||
| II | 84 | IIB | 32 | ||
| III | 56 | IIIA | 17 | ||
| Unifocal or multifocal | IIIB | 3 | |||
| Unifocal | 140 | IIIC | 5 | ||
| multifocal | 73 | IV | 2 | ||
| SSM, skin sparing mastectomy; NSM, nipple sparing mastectomy; ER, estrogen receptor; PgR, progesterone receptor; HER2, Human Epidermal Growth Factor Receptor 2; FISH, Fluorescence in situ hybridization | |||||
|
N0 (n=126) Number (%) |
N0(i+) and N1-3 (n=87) Number (%) |
P-value |
N0 (n=126) Number (%) |
N0(i+) and N1-3 (n=87) Number (%) |
P-value | |||||
| Age | Periarterial invasion (periA) | |||||||||
| Premenopausal | 39 (31.0) | 33 (37.9) | 0.306 | (+) | 68 (54.0) | 60 (69.0) | 0.033 | |||
| Postmenopausal | 87 (69.0) | 54 (62.1) | (-) | 58 (46.0) | 27 (31.0) | |||||
| Primary tumor location | Perivenous invasion (periV) | |||||||||
| Left breast | 66 (52.4) | 40 (46.0) | 0.404 | (+) | 90 (71.4) | 83 (95.4) | <0.001 | |||
| Right breast | 60 (47.6) | 47 (54.0) | (-) | 36 (28.6) | 4 (4.6) | |||||
| Anatomical Subsites | Periarterial or perivenous invasion (periA/V) | |||||||||
| Upper-inner quadrant | 33 (26.2) | 18 (20.7) | 0.417 | (+) | 97 (77.0) | 85 (97.7) | <0.001 | |||
| Lower-inner quadrant | 13 (10.3) | 15 (17.2) | (-) | 29 (23.0) | 2 (2.3) | |||||
| Upper-outer quadrant | 56 (44.4) | 34 (39.1) | Periductal invasion (periD) | |||||||
| Lower-outer quadrant | 13 (10.3) | 14 (16.1) | (+) | 93 (73.8) | 74 (85.1) | 0.062 | ||||
| Central portion | 7 (5.6) | 5 (5.7) | (-) | 33 (26.2) | 13 (14.9) | |||||
| Axillary tail | 4 (3.2) | 1 (1.1) | Lymphatic invasion in HE staining | |||||||
| Nuclear grade | (+) | 20 (15.9) | 29 (33.3) | 0.005 | ||||||
| 1 | 67 (53.2) | 20 (23.0) | <0.001 | (-) | 106 (84.1) | 58 (66.7) | ||||
| 2 | 20 (15.9) | 17 (19.5) | Lymphatic invasion in D2-40 staining | |||||||
| 3 | 39 (31.0) | 50 (57.5) | (+) | 23 (18.3) | 31 (35.6) | 0.006 | ||||
| Histological grade | (-) | 103 (81.7) | 56 (64.4) | |||||||
| I | 56 (44.4) | 17 (19.5) | <0.001 | Venous invasion in HE staining | ||||||
| II | 45 (35.7) | 39 (44.8) | (+) | 9 (7.1) | 18 (20.7) | 0.006 | ||||
| III | 25 (19.8) | 31 (35.6) | (-) | 117 (92.9) | 69 (79.3) | |||||
| Unifocal or Multifocal | Venous invasion in EVG staining | |||||||||
| Unifocal | 93 (73.8) | 47 (54.0) | 0.003 | (+) | 49 (38.9) | 66 (75.9) | <0.001 | |||
| Multifocal | 33 (26.2) | 40 (46.0) | (-) | 77 (61.1) | 21 (24.1) | |||||
| Invasive tumor size (mm) | Retraction artifact | |||||||||
| >20 mm | 28 (22.2) | 56 (64.4) | <0.001 | (+) | 44 (34.9) | 51 (58.6) | <0.001 | |||
| ≤20 mm | 98 (77.8) | 31 (35.6) | (-) | 82 (65.1) | 36 (41.4) | |||||
| Perineural Invasion | ||||||||||
| (+) | 29 (23.0) | 31 (35.6) | 0.062 | |||||||
| (-) | 97 (77.0) | 56 (64.4) | ||||||||
| EVG, Elastica‒van Gieson; HE, hematoxylin and eosin | ||||||||||
| Sensitivity | Specificity | PPV | NPV | LR+ | LR- | |
| Periarterial invasion (periA) | 69.00% | 46.00% | 46.90% | 68.20% | 1.28 | 0.67 |
| Perivenous invasion (periV) | 95.40% | 28.60% | 48.00% | 90.00% | 1.34 | 0.16 |
| Periarterial or perivenous invasion (periA/V) | 97.70% | 23.00% | 46.70% | 93.50% | 1.27 | 0.1 |
| Periductal invasion (periD) | 85.10% | 26.20% | 44.30% | 71.70% | 1.15 | 0.57 |
| Lymphatic invasion in HE staining | 33.30% | 84.10% | 59.20% | 64.60% | 2.1 | 0.79 |
| Lymphatic invasion in D2-40 staining | 35.60% | 81.70% | 57.40% | 64.80% | 1.95 | 0.79 |
| Venous invasion in HE staining | 20.70% | 92.90% | 66.70% | 62.90% | 2.9 | 0.85 |
| Venous invasion in EVG staining | 75.90% | 61.10% | 57.40% | 78.60% | 1.95 | 0.39 |
| Retraction artifact | 58.60% | 65.10% | 53.70% | 69.50% | 1.68 | 0.64 |
| Perineural Invasion | 35.60% | 77.00% | 51.70% | 63.40% | 1.55 | 0.84 |
| PPV, positive predictive value; NPV, negative predictive value; LR+, positive likelihood ratio; LR-, Negative likelihood ratio | ||||||
| Univariate | Multivariate | ||||||
| Odds ratio | 95% CI | P-value | Odds ratio | 95% CI | P-value | VIF | |
| Periarterial invasion (periA) with EVG stain | 1.900 | 1.070 – 3.360 | 0.029 | ||||
| Perivenous invasion (periV) with EVG stain | 8.300 | 2.8300 – 24.300 | 0.000 | ||||
| Periarterial or perivenous invasion (periA/V) with EVG stain | 12.700 | 2.9400 – 54.800 | 0.001 | 9.690 | 2.160 – 43.500 | 0.003 | 1.031 |
| Periductal invasion (periD) with EVG stain | 2.020 | 0.992 – 4.110 | 0.053 | ||||
| Lymphatic invasion with HE stain | 2.650 | 1.380 – 5.090 | 0.003 | 1.330 | 0.572 – 3.110 | 0.505 | 1.540 |
| Lymphatic invasion with D2-40 | 2.480 | 1.320 – 4.650 | 0.005 | 1.500 | 0.659 – 3.400 | 0.335 | 1.528 |
| Retraction artifact | 2.640 | 1.500 – 4.63 | 0.001 | 2.410 | 1.300 – 4.480 | 0.005 | 1.077 |
| Perineural Invasion | 1.850 | 1.010 – 3.390 | 0.046 | 1.570 | 0.820 – 3.020 | 0.173 | 1.048 |
| EVG, Elastica‒van Gieson; HE, hematoxylin and eosin; CI, confidence interval; VIF, variance inflation factor | |||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




