Submitted:
18 February 2026
Posted:
27 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Site

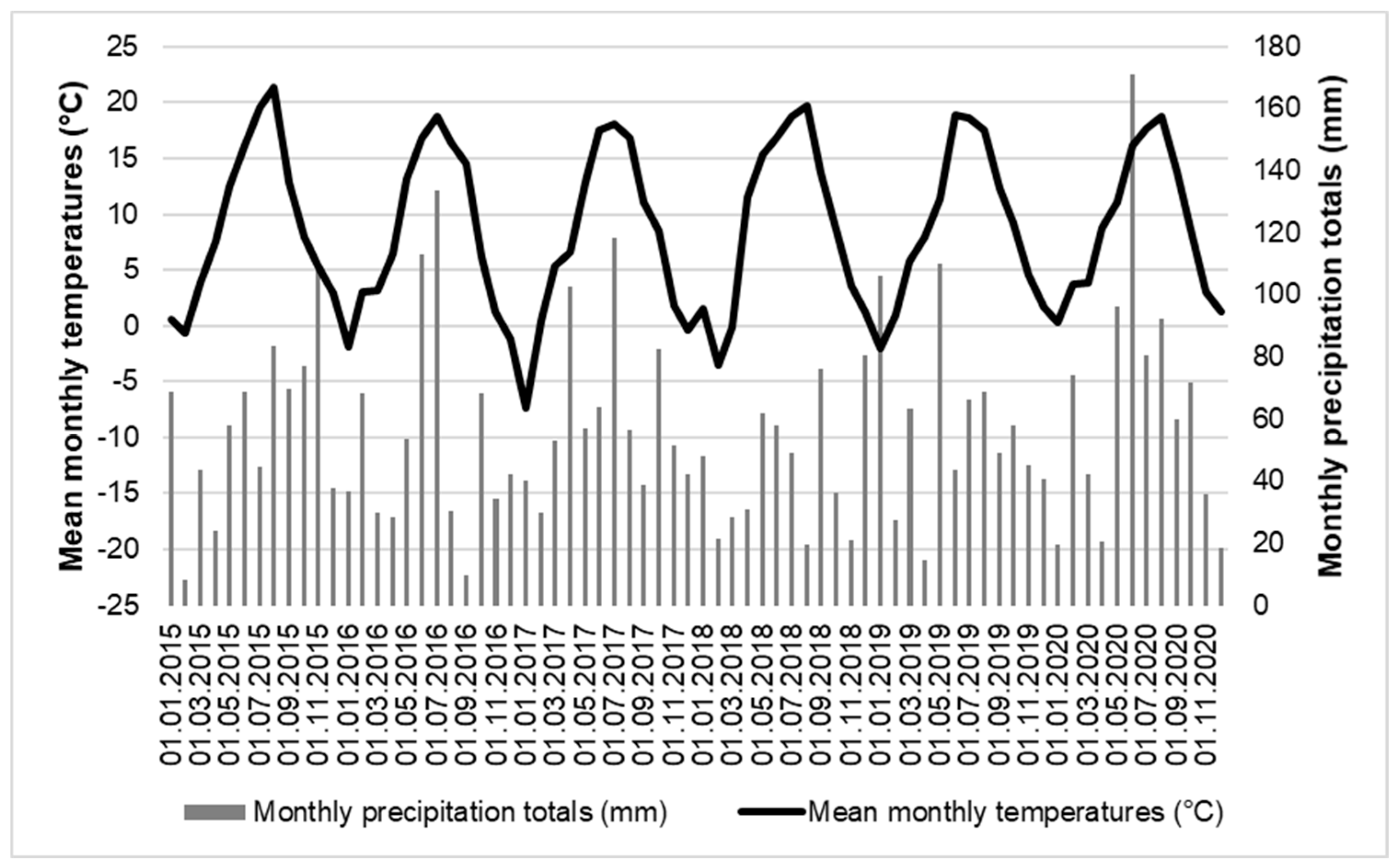
2.2. Monitoring of Microclimatic Parameters
2.3. Soil Sampling and Analysis
| Soil Parameter | Symbol | Analytical Method |
|---|---|---|
| pH in water extract | pHwater | 1:5 soil to solute; pH meter (ISO 10390) |
| pH in potassium chloride | pHKCl | |
| Total carbon | C (%) | Walkley-Black [21] |
| Total nitrogen | N | Kjeldahl [22] |
| Phosphorus content | P (mg/kg) | Göhler [18] (O horizon) Mehlich II [19] (A horizon) |
| Magnesium content | Mg (mg/kg) | |
| Calcium content | Ca (mg/kg) | |
| Potassium content | K (mg/kg) | |
| Aluminum content | Al (mg/kg) | Sokolov [20] |
2.4. Leaf Analysis and Tree Biometric Parameters
1. Statistical Analysis
1. Results
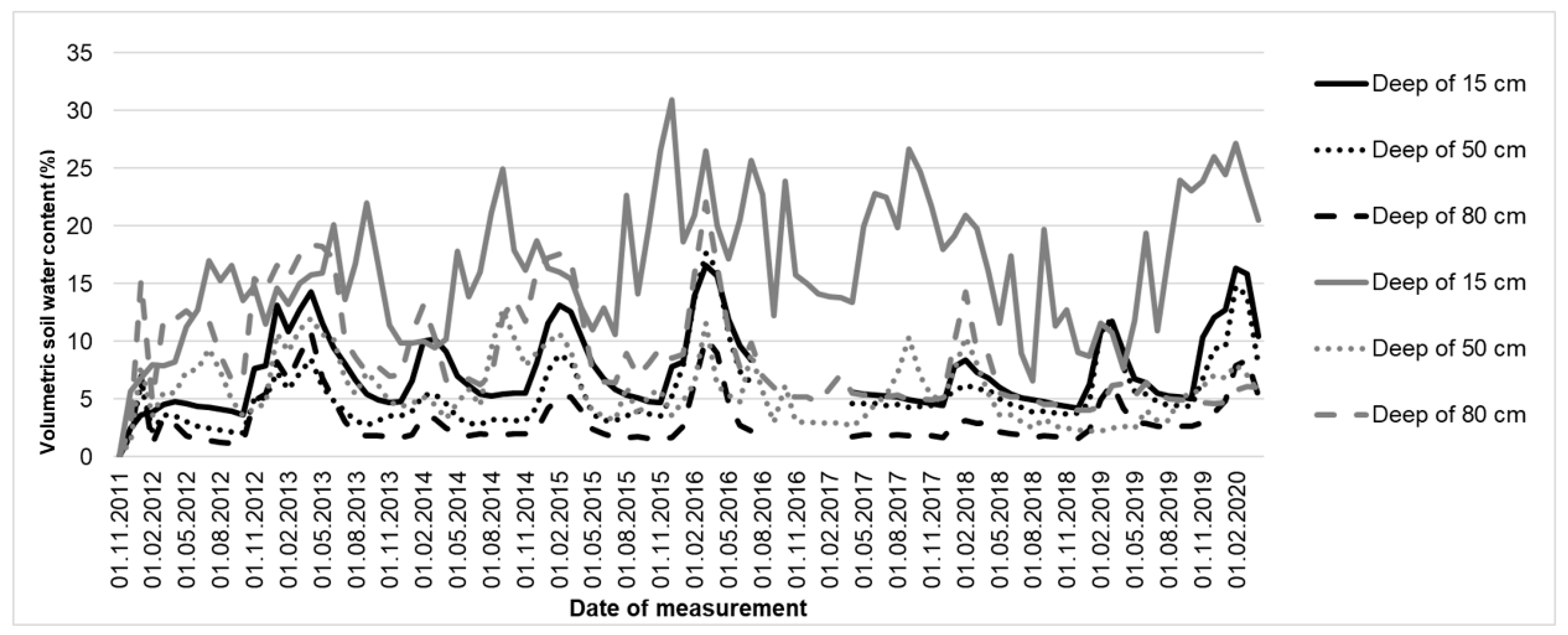
1.1. Soil Moisture
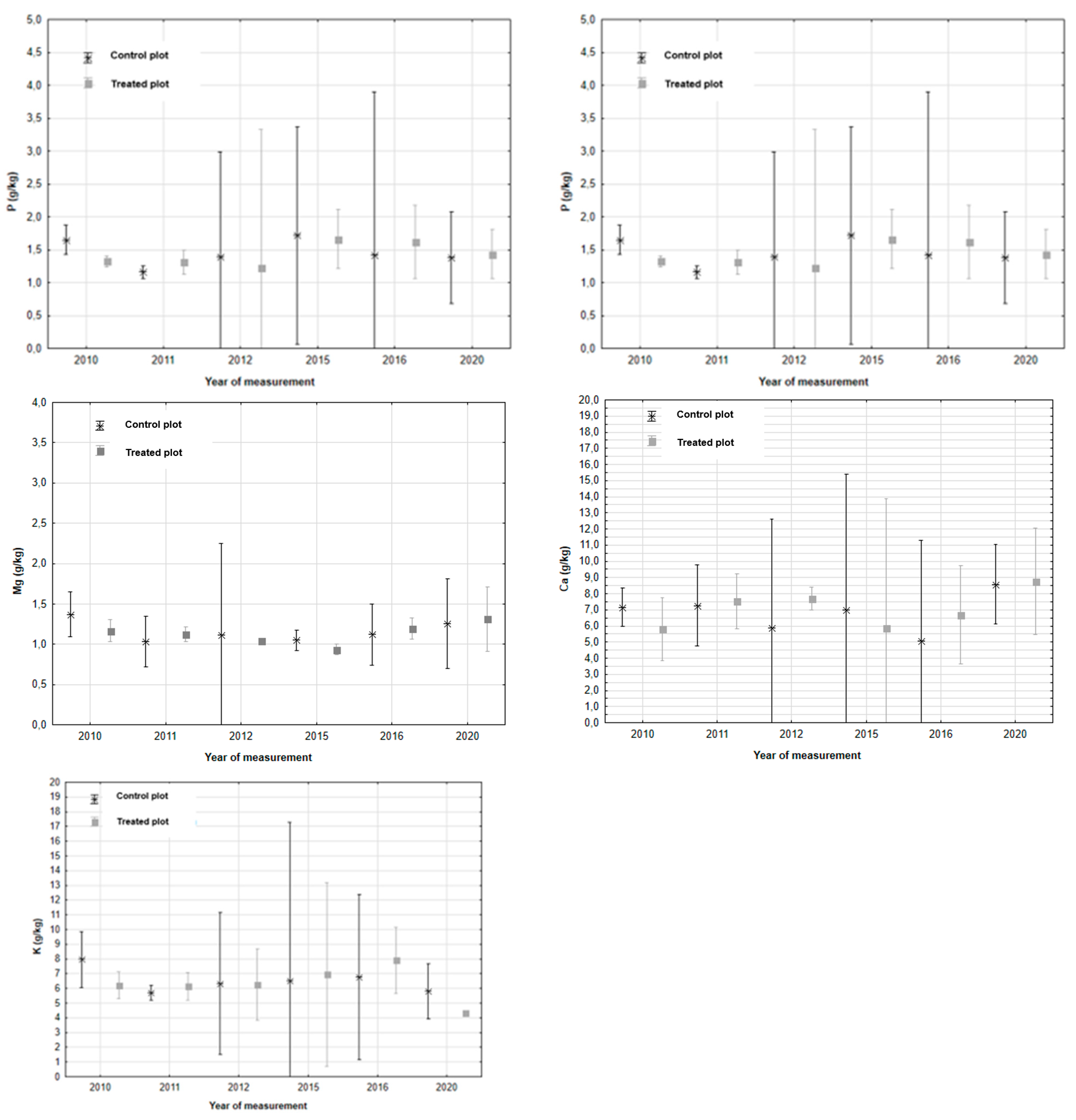
1.1. Chemical Soil Properties
| Parameter | Horizon | Treated plot | Control plot | Difference | P | |||
|---|---|---|---|---|---|---|---|---|
| Mean | SE | Mean | SE | Mean | Lower / Upper | |||
| pHwater | OH | 4.00 | ±0.04 | 4.07 | ±0.05 | -0.06 | -0.15 / 0.02 | 0.170 |
| A | 3.68 | ±0.04 | 3.66 | ±0.02 | 0.02 | -0.05 / 0.10 | 0.500 | |
| pHKCl | OH | 3.38 | ±0.04 | 3.47 | ±0.06 | -0.09 | -0.21/ 0.01 | 0.090 |
| A | 3.10 | ±0.03 | 3.08 | ±0.02 | 0.02 | -0.04 / 0.08 | 0.420 | |
| C (%) | OH | 3.43 | ±0.20 | 3.61 | ±0.10 | -0.18 | -0.53 / 0.17 | 0.290 |
| A | 1.81 | ±0.18 | 1.84 | ±0.18 | -0.03 | -0.30 / 0.24 | 0.810 | |
| C:N | OH | 20.26 | ±0.69 | 21.58 | ±0.50 | -1.31 | -0.10 / 0.12 | 0.160 |
| A | 21.25 | ±0.86 | 21.93 | ±0.98 | -0.68 | -3.19 / 1.83 | 0.570 | |
| P (mg/kg) | OH | 13.51 | ±1.47 | 16.40 | ±2.13 | -2.88 | -6.39 / 0.62 | 0.100 |
| A | 19.42 | ±0.27 | 15.20 | ±0.12 | -1.31 | -0.10 / 0.12 | 0.160 | |
| Al (mmol/kg) | OH | |||||||
| A | 84.76 | ±3.24 | 94.08 | ±1.77 | -9.32 | -17.51 / -1.13 | 0.020 | |
|
Mg (mg/kg) |
OH | 150.53 | ±6.81 | 171.46 | ±12.48 | -20.93 | -40.00 / -1.85 | 0.030 |
| A | 96.18 | ±6.44 | 77.99 | ±5.14 | 18.19 | 7.26 / 29.12 | 0.000 | |
| Ca (mg/kg) | OH | 571.94 | ±49.47 | 752.92 | ±75.04 | -180.97 | -301.80 / -60.10 | 0.000 |
| A | 408.51 | ±38.49 | 353.27 | ±28.63 | 55.24 | -32.01 / 142.49 | 0.200 | |
|
K (mg/kg) |
OH | 181.74 | ±19.22 | 185.25 | ±20.65 | -3.51 | -29.84 / 22.81 | 0.780 |
| A | 108.17 | ±10.86 | 94.59 | ±6.41 | 13.57 | -6.08 / 33.23 | 0.160 | |
3.2. Concentrations of in Norway Spruce Needles
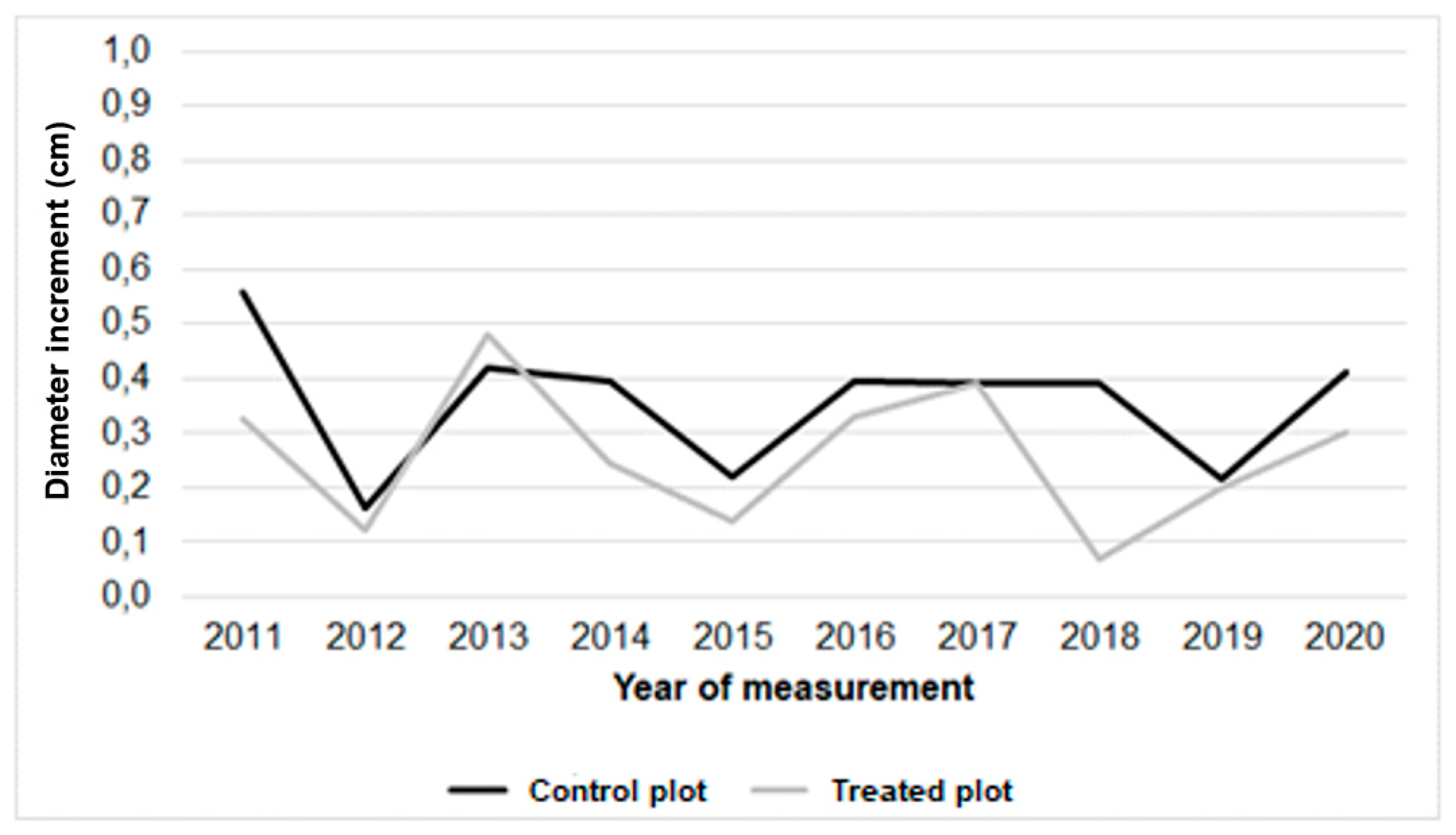
3.3. Tree Biometric Parameters
4. Discussion
5. Conclusions
- Soil chemistry responded, trees less so: Reduced rainfall produced clear shifts in soil chemistry (Mg, Ca, exchangeable Al), whereas needle chemistry and growth showed only modest short-term responses.
- Vertical nutrient reshuffling: A horizon-specific redistribution of base cations was evident (declining Ca in the O_H horizon; contrasting Mg trends in O_H vs. A), highlighting the sensitivity of near-surface layers to drying.
- Lower exchangeable Al (H2 supported): Exchangeable Al decreased in the upper soil layer, suggesting a temporary reduction in Al mobility under moisture limitation.
- Foliar stoichiometry buffered: Needle nutrient concentrations did not differ significantly between treatments; interannual growth variability tracked regional dry years rather than the manipulation itself.
- Hydrologic driver near surface: The strongest drying occurred at 0–15 cm, with deeper layers more stable; surface desiccation is exacerbated by high spruce canopy interception.
- Management implications: Prioritize routine monitoring of Ca and Mg in the O_H and A horizons; combine soil chemical indicators with tree-level physiological metrics when diagnosing drought impacts.
- Future work: Extend monitoring of exchangeable Al and vertical Ca/Mg stratification, couple soil-solution chemistry with root distribution and hydraulics, and test responses across site fertilities and species mixtures.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tate, E.L.; Gustard, A. Drought Definition: A Hydrological Perspective. In Drought and Drought Mitigation in Europe; Advances in Natural and Technological Hazards Research; Vogt, J.V., Somma, F., Eds.; Springer: Dordrecht, The Netherlands, 2000; Vol. 14, pp. 23–48. [Google Scholar] [CrossRef]
- Ionita, M.; Nagavciuc, V.; Kumar, R.; Rakovec, O. On the curious case of the recent decade, mid-spring precipitation deficit in Central Europe. npj Climate and Atmospheric Science 2020, 3, 49. [Google Scholar] [CrossRef]
- O’Geen, A.T. Soil Water Dynamics. Nature Education Knowledge 2013, 4(5), 9. [Google Scholar]
- Hsu, H.; Dirmeyer, P.A. Soil moisture–evaporation coupling shifts into new gears under increasing CO₂. Nature Communications 2023, 14, 1398. [Google Scholar] [CrossRef]
- Vinš, B.; et al. Dopady možné změny klimatu na lesy v České republice: Územní studie změny klimatu pro Českou republiku. In Element 2; Český hydrometeorologický ústav: Praha, Czech Republic, 1996. [Google Scholar]
- Nihlgård, B. Plant Biomass, Primary Production and Distribution of Chemical Elements in a Beech and a Planted Spruce Forest in South Sweden. Oikos 1972, 23, 68–91. [Google Scholar] [CrossRef]
- Jost, G.; Schume, H.; Hager, H. Factors controlling soil water-recharge in a mixed European beech (Fagus sylvatica L.)–Norway spruce (Picea abies (L.) Karst.) stand. European Journal of Forest Research 2004, 123, 93–104. [Google Scholar] [CrossRef]
- García-Palacios, P.; Prieto, I.; Ourcival, J.M.; Hättenschwiler, S. Disentangling the litter quality and soil microbial contribution to leaf and fine root litter decomposition responses to reduced rainfall. Ecosystems 2016, 19, 490–503. [Google Scholar] [CrossRef]
- De Deyn, G.B.; Kooistra, L. The role of soils in habitat creation, maintenance and restoration. Philosophical Transactions of the Royal Society B 2021, 376(1834), 20200170. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Yang, Z.; He, K.; Zhou, W.; Feng, W. Soil Fungal Community Diversity, Co-Occurrence Networks, and Assembly Processes under Diverse Forest Ecosystems. Microorganisms 2024, 12, 1915. [Google Scholar] [CrossRef] [PubMed]
- Borken, W.; Davidson, E.A.; Savage, K.; Gaudinski, J.; Trumbore, S.E. Drying and wetting effects on carbon dioxide release from organic horizons. Soil Science Society of America Journal 2003, 67, 1888–1896. [Google Scholar] [CrossRef]
- Manghabati, H.; Weis, W.; Göttlein, A. Importance of soil extractable phosphorus distribution for mature Norway spruce nutrition and productivity. European Journal of Forest Research 2018, 137, 631–642. [Google Scholar] [CrossRef]
- Niederberger, J.; Kohler, M.; Bauhus, J. Distribution of phosphorus fractions with different plant availability in German forest soils and their relationship with common soil properties and foliar P contents. SOIL 2019, 5, 189–204. [Google Scholar] [CrossRef]
- Novák, J.; Slodičák, M.; Dušek, D. Akumulace humusu a živin pod mladými porosty smrku ztepilého v ochranném pásmu vodních zdrojů v Krušných horách. Zprávy lesnického výzkumu (Reports of Forestry Research) 2009, 54(Special), 37–42. [Google Scholar]
- Hofmeister, J.; Oulehle, F.; Krám, P.; et al. Loss of nutrients due to litter raking compared to the effect of acidic deposition in two spruce stands, Czech Republic. Biogeochemistry 2008, 88, 139–151. [Google Scholar] [CrossRef]
- Jandl, R.; Alewell, C.; Prietzel, J. Calcium loss in Central European forest soils. Soil Science Society of America Journal 2004, 68(2), 588–595. [Google Scholar] [CrossRef]
- IUSS Working Group WRB. World Reference Base for Soil Resources. In ternational Soil Classification System for Naming Soils and Creating Legends for Soil Maps, 4th ed.; International Union of Soil Sciences (IUSS): Vienna, Austria, 2022; ISBN 979-8-9862451-1-9. [Google Scholar]
- Göhler, F. Arbeitsanleitung für die chemische Schnelluntersuchung und die Ergänzung von Nährlösungen der erdelosen Kultur im Produktionsbetrieb; 1960; Volume Band 8, Heft 6, p. 419. [Google Scholar] [CrossRef]
- Mehlich, A. New extractant for soil test evaluation of phosphorus, potassium, magnesium, calcium, sodium, manganese and zinc. Communications in Soil Science and Plant Analysis 1978, 9(6), 477–492. [Google Scholar] [CrossRef]
- Agrokhimicheskie metody issledovaniya pochv [Agrochemical Methods of Soil Research]; Sokolov, A.V., Askinazi, D.L., Eds.; Nauka: Moscow, Russia, 1965. [Google Scholar]
- Walkley, A.; Black, I.A. An examination of the Degtjareff method for determining soil organic matter and a proposed modification of the chromic acid titration method. Soil Science 1934, 37(1), 29–38. [Google Scholar] [CrossRef]
- Kjeldahl, J. Neue Methode zur Bestimmung des Stickstoffs in organischen Körpern. Zeitschrift für Analytische Chemie 1883, 22, 366–382. [Google Scholar] [CrossRef]
- Griesbauer, H.; DeLong, S.C.; Rogers, B.; Foord, V. Growth sensitivity to climate varies with soil moisture regime in spruce–fir forests in central British Columbia. Trees 2021, 35, 649–669. [Google Scholar] [CrossRef]
- Jamnická, G.; Fleischer, P., Jr.; Konôpková, A.; Pšidová, E.; Kučerová, J.; Kurjak, D.; Živčák, M.; Ditmarová, Ľ. Norway Spruce (Picea abies L.) Provenances Use Different Physiological Strategies to Cope with Water Deficit. Forests 2019, 10, 651. [Google Scholar] [CrossRef]
- Ogaya, R.; Peñuelas, J. Climate Change Effects in a Mediterranean Forest Following 21 Consecutive Years of Experimental Drought. Forests 2021, 12, 306. [Google Scholar] [CrossRef]
- Jevšenak, J.; Tychkov, I.; Gričar, J.; Levanič, T.; Tumajer, J.; Prislan, P.; Arnič, D.; Popkova, M.; Shishov, V.V. Growth-limiting factors and climate response variability in Norway spruce (Picea abies L.) along an elevation and precipitation gradients in Slovenia. International Journal of Biometeorology 2021, 65, 311–324. [Google Scholar] [CrossRef]
- Qin, H.; Jiao, L.; Zhou, Y.; Wu, J.; Che, X. Elevation affects the ecological stoichiometry of Qinghai spruce in the Qilian Mountains of northwest China. Frontiers in Plant Science 2022, 13, 917755. [Google Scholar] [CrossRef]
- Kiil, A.D. Distribution of Moisture in Spruce–Fir Duff and Its Relevance to Fire Danger Rating; Internal Report No. A-34; Forest Research Laboratory, Canadian Forestry Service: Edmonton, AB, Canada, 1970. [Google Scholar]
- Jančo, M.; Mezei, P.; Kvas, A.; Danko, M.; Sleziak, P.; Minďáš, J.; Škvarenina, J. Effect of mature spruce forest on canopy interception in subalpine conditions during three growing seasons. Journal of Hydrology and Hydromechanics 2021, 69, 1–14. [Google Scholar] [CrossRef]
- Sun, G.; Domec, J.-C.; Amatya, D.M. Forest evapotranspiration: Measurement and modelling at multiple scales. In Forest Evapotranspiration: Measurement and Modelling at Multiple Scales; USDA Forest Service, 2011; pp. 31–70. [Google Scholar]
- Rukh, S.; Poschenrieder, W.; Heym, M.; Pretzsch, H. Drought Resistance of Norway Spruce (Picea abies [L.] Karst) and European Beech (Fagus sylvatica [L.]) in Mixed vs. Monospecific Stands and on Dry vs. Wet Sites. From Evidence at the Tree Level to Relevance at the Stand Level. Forests 2020, 11, 639. [Google Scholar] [CrossRef]
- Kuželková, M.; Jačka, L.; Kovář, M.; Hradilek, V.; Máca, P. Tree trait–mediated differences in soil moisture regimes: A comparative study of beech, spruce, and larch in a drought-prone area of Central Europe. Research Square (preprint) 2023. [Google Scholar] [CrossRef]
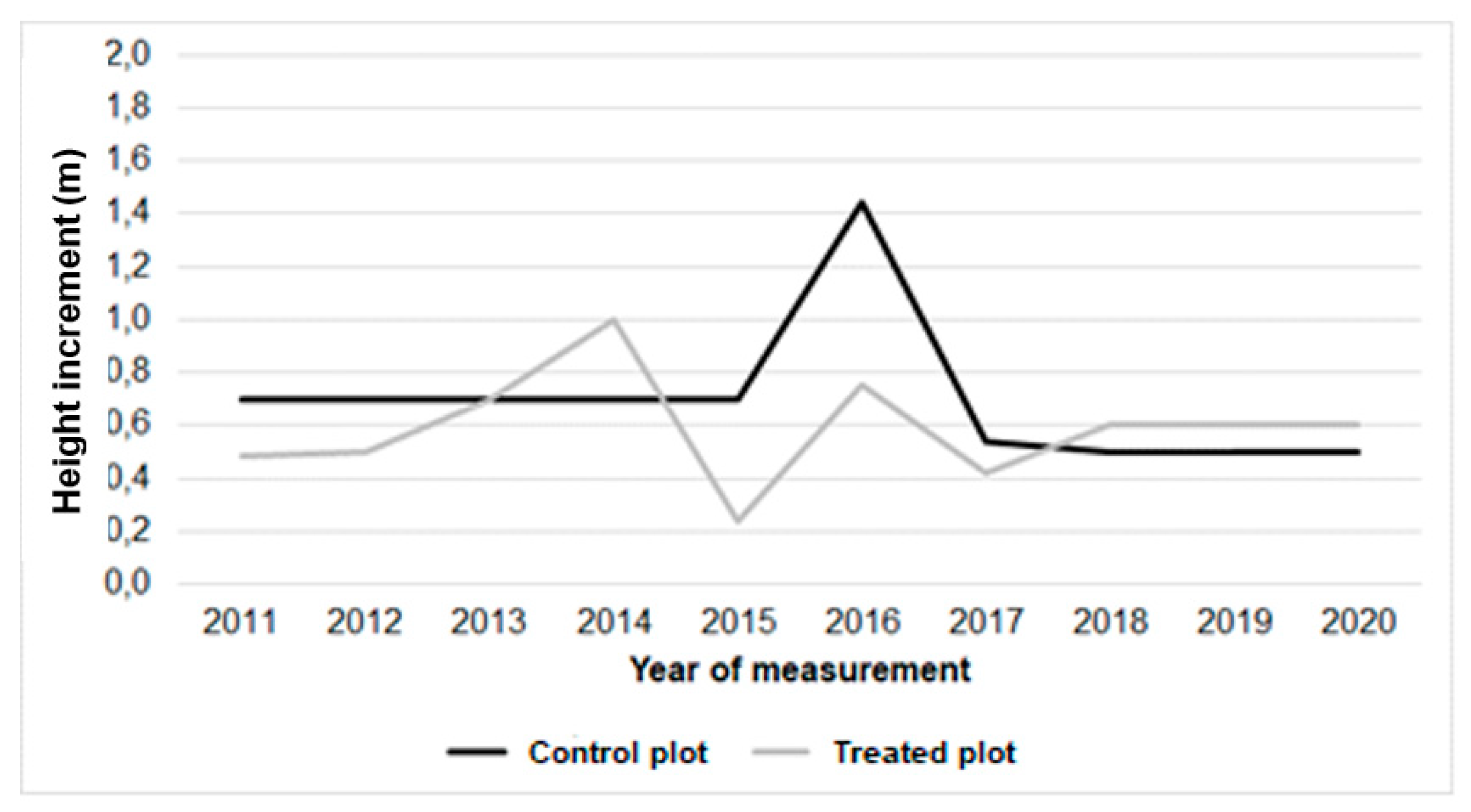
| Height of above ground part (m) in | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Variant/ year | 2010 | 2011 | 2012 | 2013 | 2014 | 2015 | 2016 | 2017 | 2018 | 2019 | 2020 |
| Control plot | 13,97±2,5 | 14,67±3,5 | 15,37±2,5 | 16,07±2,1 | 16,77±2,3 | 17,47±2,4 | 18,91±3,2 | 19,45±1,2 | 19,95±3,2 | 20,45±3,4 | 20,95±2,7 |
| Treated plot | 15,05±2,7 | 15,53±4,6 | 16,03±3,2 | 16,73±2,7 | 17,73±2,7 | 17,97±2,7 | 18,72±2,1 | 19,14±2,4 | 19,74±4,1 | 20,34±2,7 | 20,94±2,3 |
| Diameter (cm) in | |||||||||||
| Variant/ year | 2010 | 2011 | 2012 | 2013 | 2014 | 2015 | 2016 | 2017 | 2018 | 2019 | 2020 |
| Control plot | 13,63±3,3 | 13,75±3,4 | 14,47±3,8 | 14,89±3,7 | 15,32±3,8 | 15,51±3,9 | 15,95±4,1 | 16,29±3,7 | 16,68±3,2 | 16,90±3,5 | 17,70±3,2 |
| Treated plot | 14,30±3,6 | 14,33±3,6 | 14,78±4,3 | 15,26±3,9 | 15,52±4,1 | 15,62±4,1 | 16,03±4,3 | 16,37±4,1 | 16,30±3,9 | 16,50±3,7 | 16,80±4,2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





