Submitted:
24 February 2026
Posted:
26 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Contrast-to-Noise Ratio (CNR) - Definition and Analytical Model
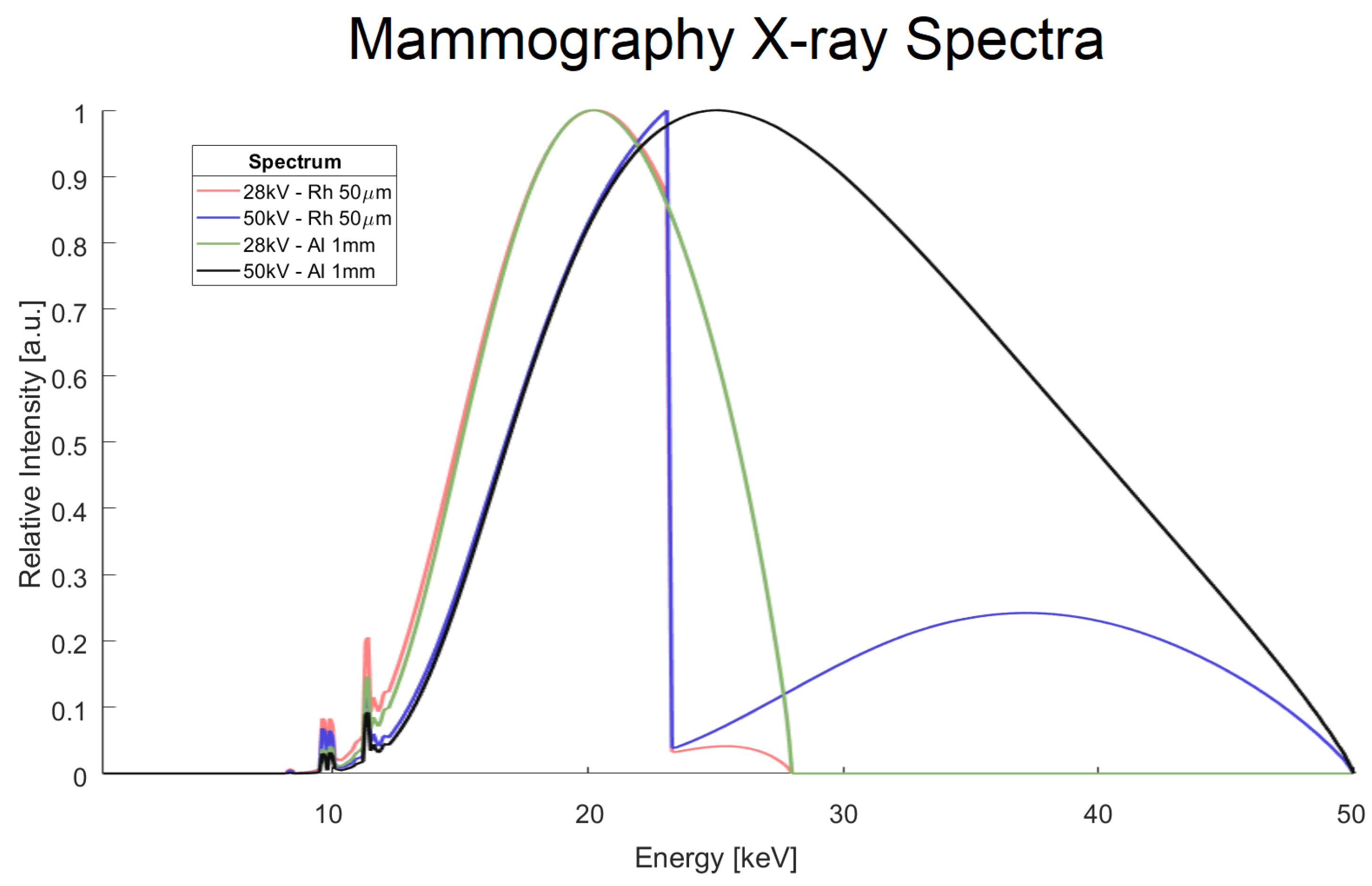
2.2. Mammography X-Ray Spectra
- Standard mammography spectrum (): 28 kVp tube voltage, 50 µm Rh filter;
- Filter-modified spectrum (): with 1 mm Al filter instead;
- Voltage-modified spectrum (): 50 kVp tube voltage instead;
- Filter-Voltage-modified spectrum (): 50 kVp tube voltage, 1 mm Al filter.
2.3. Mammography Simulations and Analysis
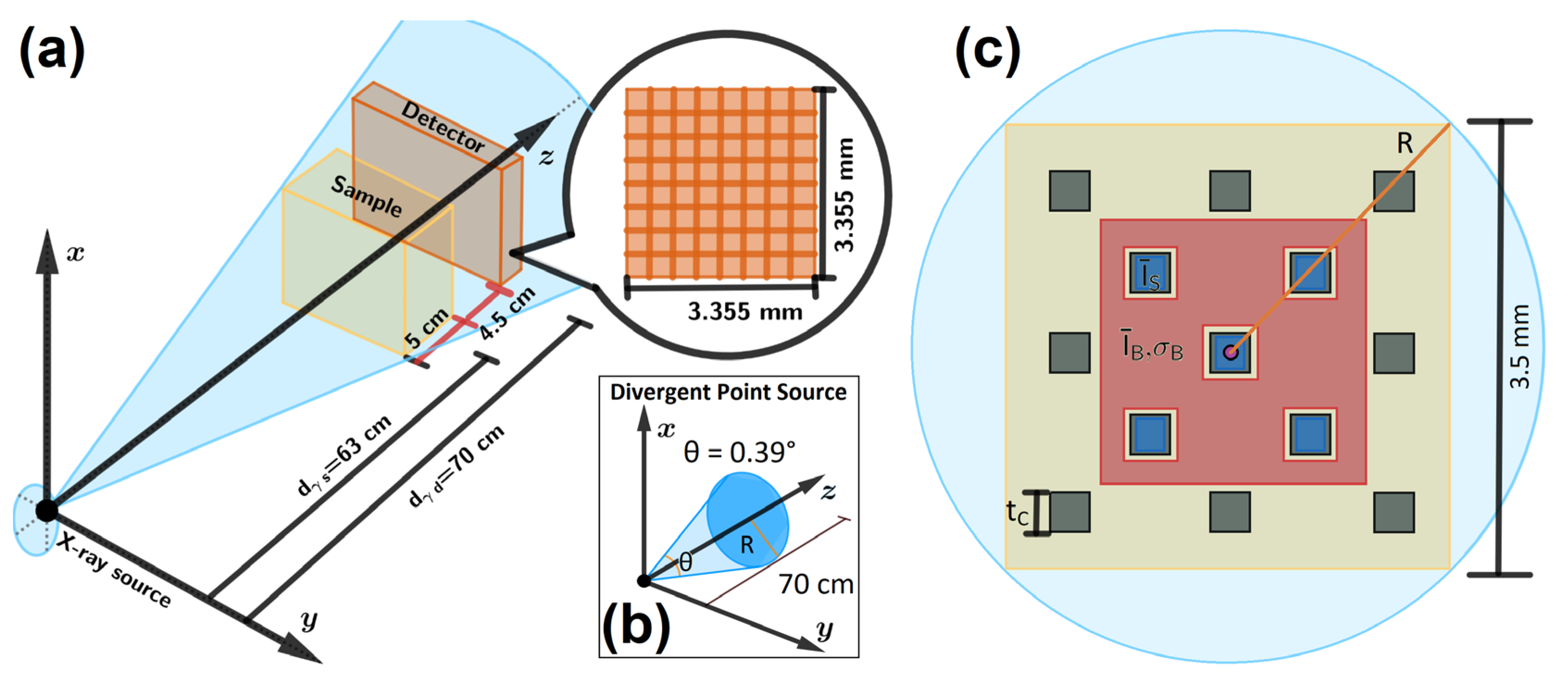
2.3.1. Imaging Geometry
2.3.2. X-Ray Source
2.3.3. Phantom
2.3.4. Simulation Statistics
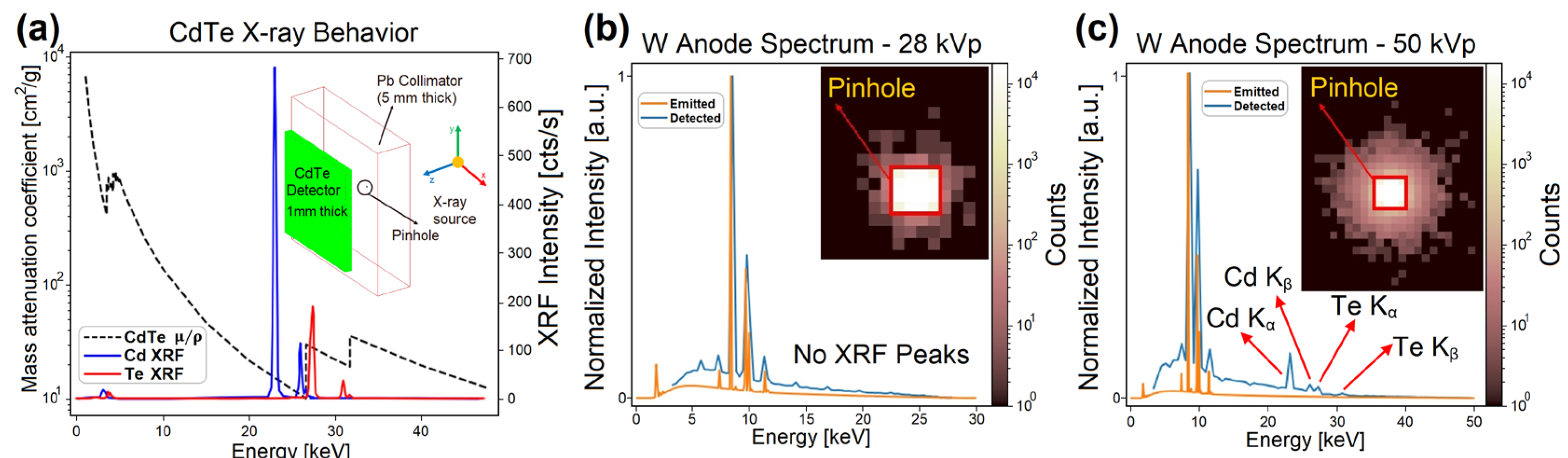
2.3.5. Spectroscopic Analysis
2.4. Monte Carlo Validation Tests
2.5. Experimental Validation - Breast Implant X-Ray Imaging
3. Results
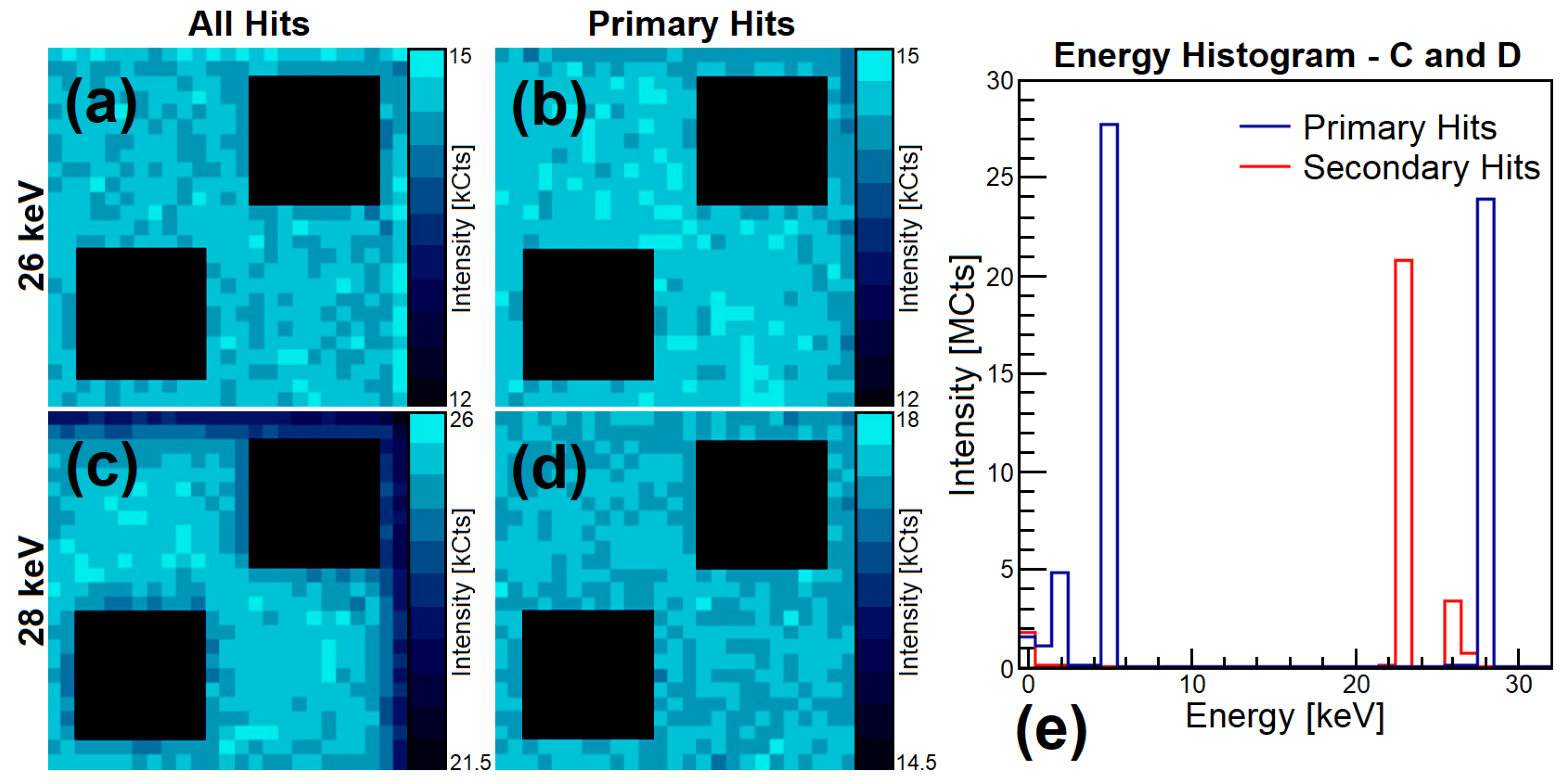
3.1. Monte Carlo Validation Tests
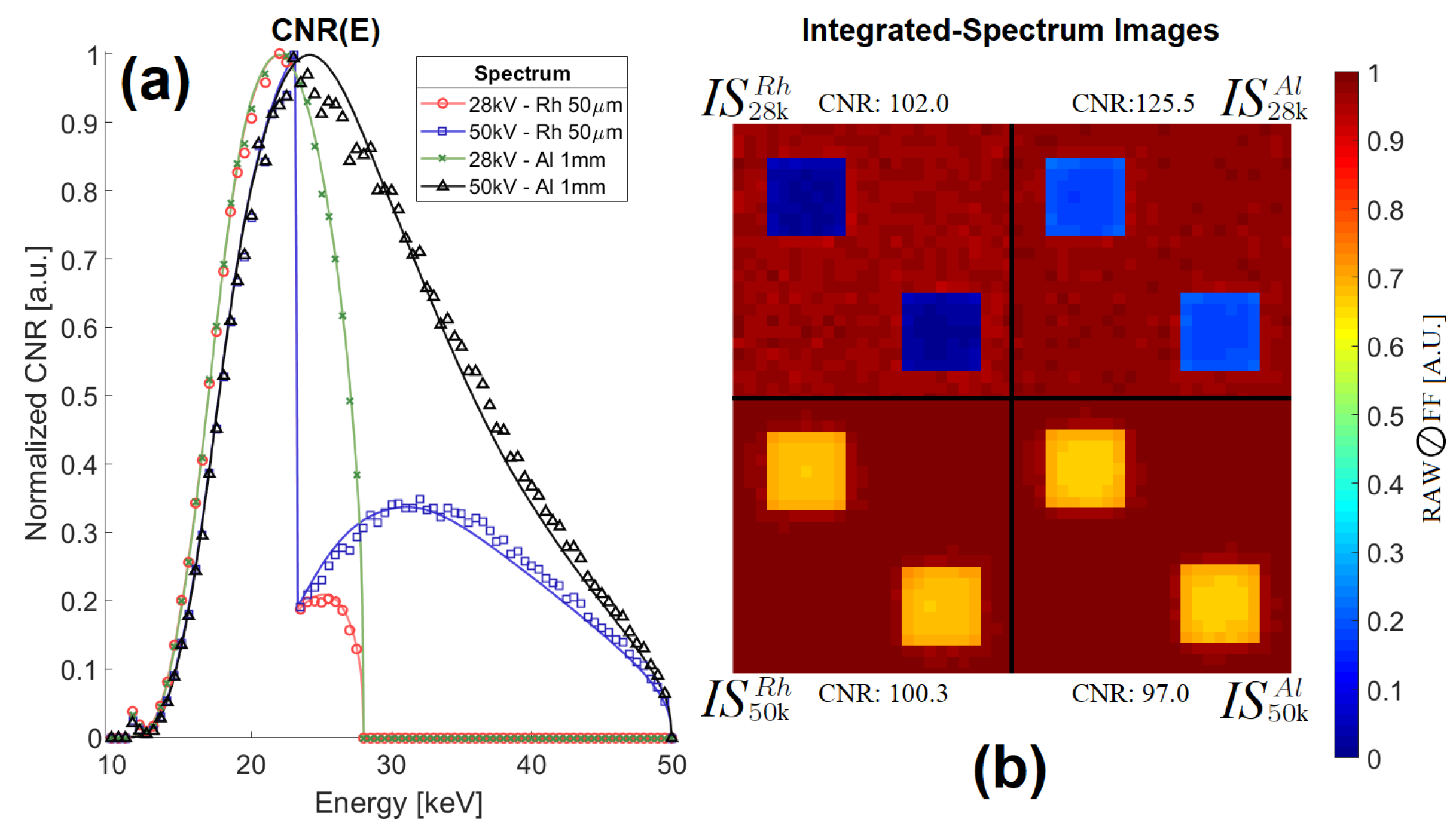
3.2. Mammography Simulations
3.3. Experimental Validation – Breast Implant Imaging
4. Discussion
4.1. Modified Mammography X-Ray Spectra
4.2. Mammography Simulations
4.3. Experimental Validation – Breast Implant X-Ray Imaging
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| CNR | Contrast-to-Noise Ratio |
| Rh | Rhodium |
| CdTe | Cadmium Telluride |
| Al | Aluminum |
| ESD | Entrance Surface Dose |
| HA | Calcium Hydroxyapatite |
| MRI | Magnetic Resonance Imaging |
| PCD | Photon-Counting Detector |
| SPCD | Spectroscopic Photon-Counting Detector |
| NIST | National Institute of Standards and Technology (USA) |
| W | Tungsten |
| GATE | Geant4 Application for Tomographic Emission |
| PET | Positron Emission Tomography |
| CT | Computer Tomography |
| BI-RADS | Breast Imaging Reporting and Data System |
Appendix A. Simulation Validation Table
| Monte Carlo Software Used | GATE. Version 9.2. Based on GEANT4, v11.0.3 |
| Simulation Times |
|
| Hardware |
|
| Physics and Transport | emstandard_opt4 physics list |
| Travel Cut-offs | Gamma, Electrons:
|
| Digitizer Energy Threshold | 3 keV |
| Number of primaries, all simulations | 1010 primary photons per energy bin. 1.62 x 1012 total photons generated |
Appendix B. Monte Carlo Mammography Spectra
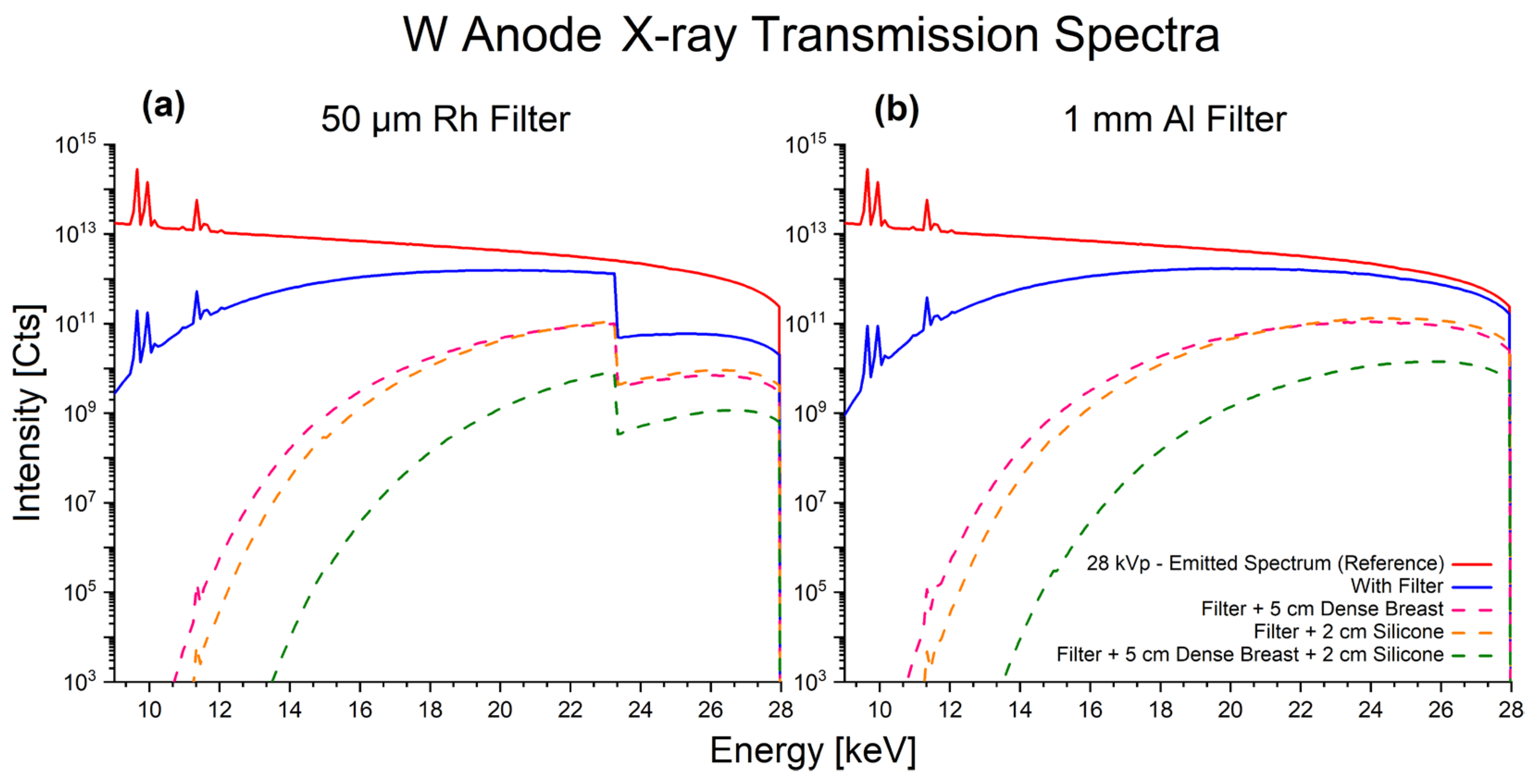
References
- Li, X.; Weaver, O.; Desouki, M.M.; Dabbs, D.; Shyum, S.; et al. Microcalcification Is an Important Factor in the Management of Breast Intraductal Papillomas Diagnosed on Core Biopsy. Am J Clin Pathol 2012, 138, 789–95. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, L.; Thomas, V.; Sharma, N. Microcalcification on Mammography: Approaches to Interpretation and Biopsy. Brit J Radiol 2016, 90. [Google Scholar] [CrossRef]
- Dance, D.R.; Christofides, S.; Maidment, A.D.A.; McLean, I.D.; Ng, K.H. Diagnostic Radiology Physics – A Handbook for Teachers and students; International Atomic Energy Agency: Vienna, 2014; ISBN 978-92-0-131010-1. [Google Scholar]
- Bantick, G.; Taggart, I. Mammography and Breast Implants. Brit J Plast Surg 1995, 48, 49–52. [Google Scholar] [CrossRef]
- Siu, A. Screening for Breast Cancer: U.S. Preventive Services Task Force Recommendation Statement. Ann Intern Med 2016, 164, 279–96. [Google Scholar] [CrossRef]
- McCormack, V.A.; dos Santos Silva, I. Breast Density and Parenchymal Patterns as Markers of Breast Cancer Risk: A Meta-analysis. Cancer Epidem Biomar 2006, 15, 1159–1169. [Google Scholar] [CrossRef] [PubMed]
- Boyd, N.; Martin, L.; Bronskill, M.; Yaffe, M.; Duric, N.; Minkin, S. Breast Tissue Composition and Susceptibility to Breast Cancer. JNCI-J Natl Cancer I 2010, 102, 1224–1237. [Google Scholar] [CrossRef]
- Vachon, C.M.; Van Gils, C.H.; Sellers, T.A.; Ghosh, K.; Pruthi, S.; et al. Mammographic Density, Breast Cancer Risk and Risk Prediction. Breast Cancer Res 2007, 9, 1–9. [Google Scholar] [CrossRef]
- Sprague, B.L.; Gangnon, R.E.; Burt, V.; Trentham-Dietz, A.; Hampton, J.M.; et al. Prevalence of Mammographically Dense Breasts in the United States. JNCI-J Natl Cancer I 2014, 106. [Google Scholar] [CrossRef]
- Jackson, V.P.; Hendrick, R.E.; Feig, S.A.; Kopans, D.B. Imaging of the Radiographically Dense Breast. Radiology 1993, 188, 297–301. [Google Scholar] [CrossRef] [PubMed]
- Scheel, J.R.; Lee, J.M.; Sprague, B.L.; Lee, C.I.; Lehman, C.D. Screening Ultrasound as an Adjunct to Mammography in Women with Mammographically Dense Breasts. Am J Obstet Gynecol 2015, 212, 9–17. [Google Scholar] [CrossRef]
- Rebolj, M.; Assi, V.; Brentnall, A.; Parmar, D.; Duffy, S.W. Addition of Ultrasound to Mammography in the Case of Dense Breast Tissue: Systematic Review and Meta-analysis. Brit J Cancer 2018, p118, 1559–1570. [Google Scholar] [CrossRef]
- Sieber, D.A.; Adams, J.; William, P. What’s Your Micromort? A Patient-Oriented Analysis of Breast Implant-Associated Anaplastic Large Cell Lymphoma (BIA-ALCL). Aesthet Surg J 2017, 37, 887–891. [Google Scholar] [CrossRef]
- Kalaaji, A. ISAPS International Survey on Aesthetic/Cosmetic Procedures Performed in 2024. International Society of Aesthetic Plastic Surgery. 2025. Available online: https://www.isaps.org/media/oogpzodr/isaps-global-survey_2024.pdf (accessed on 18 February 2026).
- Eklund, G.; Busby, R.; Miller, S.; Job, J. Improved Imaging of the Augmented Breast. Am J Roentengol 1988, 151, 469–473. [Google Scholar] [CrossRef]
- Smalley, S.M. Breast Implants and Breast Cancer Screening. J Midwifery Wom Heal 2003, 48, 329–337. [Google Scholar] [CrossRef]
- Miglioretti, D.L.; Rutter, C.M.; Geller, B.M.; Cutter, G.; Barlow, W.E.; et al. Effect of Breast Augmentation on the Accuracy of Mammography and Cancer Characteristics. JAMA-J Am Med Assoc 2004, 291, 442–450. [Google Scholar] [CrossRef] [PubMed]
- Berger, N.; Marcon, M.; Saltybaeva, N.; Kalender, W.A.; Alkadhi, H.; Frauenfelder, T.; Boss. Dedicated Breast Computed Tomography with a Photon-Counting Detector: Initial Results of Clinical In Vivo Imaging. Invest Radiol 2019, 7. [Google Scholar] [CrossRef]
- Poikela, T.; Plosila, J.; Westerlund, T.; Campbell, M.; De Gaspari, M.; et al. Timepix3: a 65K Channel Hybrid Pixel Readout Chip with Simultaneous ToA/ToT and Sparse Readout. J Inst 2014, 9. [Google Scholar] [CrossRef]
- Roque, G.; Avila, C.; Pérez-Lara, M.L.; Mendoza, L.; Procz, S. Study of Contrast-to-Noise Ratio Performance of a Medipix3RX CdTe Detector for Low Dose Mammography Imaging. Nucl Instrum Meth A 2021, 992. [Google Scholar] [CrossRef]
- Ballabriga, R.; Alozy, J.; Blaj, G.; Campbell, M.; Fiederle, M.; et al. The Medipix3RX: a High Resolution, Zero Dead-time Pixel Detector Readout Chip Allowing Spectroscopic Imaging. J Inst 2014, 8. [Google Scholar] [CrossRef]
- Ballabriga, R.; Alozy, J.; Campbell, M.; Frojdh, E.; Heijne, E.H.M.; et al. Review of Hybrid Pixel Detector Readout ASICs for Spectroscopic X-ray Imaging. J Inst 2016, 11. [Google Scholar] [CrossRef]
- Procz, S.; Roque, G.; Avila, C.; Racedo, J.; Rueda, R.; et al. Investigation of CdTe, GaAs, Se and Si as Sensor Materials for Mammography. IEEE T Med Imaging 2020, 39, 3766–3777. [Google Scholar] [CrossRef]
- Hemdal, B.; Hernsdorf, L.; Andersson, I.; Bengtsson, G.; Heddson, B.; Olsson, M. Average Glandular Dose in Routine Mammography Screening using a Sectra Microdose Mammography Unit. Radiat Prot Dosim 2005, 114, 436–443. [Google Scholar] [CrossRef]
- Ghammraoui, B.; Glick, J. Investigating the Feasibility of Classifying Breast Pixel Using Photon-counting Spectral Mammography: A Simulation Study. Med Phys 2017, 44, 1–9. [Google Scholar] [CrossRef]
- Siemens Healthineers AG. Photon-counting CT - A quantum leap in computed tomography 2025. Available online: https://www.siemens-healthineers.com/computed-tomography/ct-technologies-and-innovations/photon-counting-ct (accessed on 18 February 2026).
- Canon Medical Systems Corporation. Photon Counting CT - A new era in computed tomography 2025. Available online: https://global.medical.canon/products/computed-tomography/pcct (accessed on 18 February 2026).
- Zhan, X.; Zhang, R.; Niu, X.; Hein, I.; Budden, B.; et al. Comprehensive Evaluations of a Prototype Full Field-of-view Photon Counting CT System through Phantom Studies. Phys Med Biol 2023, 68. [Google Scholar] [CrossRef] [PubMed]
- Koninklijke Philips, N.V. See beyond the conventional to the next generation of multi-energy CT. Available online: https://www.philips.com.my/healthcare/sites/spectral-ct-learning-center (accessed on 18 February 2026).
- CIRS, Inc.Mammographic Accreditation Phantom - Model 015. Computerized Imaging Reference Systems – CIRS, 2013.
- Swinehart, D.F. The Beer-Lambert Law. J Chem Educ 1962, 39. [Google Scholar] [CrossRef]
- Hubbell, J.; Seltzer, S. Tables of X-Ray Mass Attenuation Coefficients and Mass Energy-Absorption Coefficients 1 keV to 20 MeV for Elements Z = 1 to 92 and 48 Additional Substances of Dosimetric Interest; NIST: USA, 2008. [Google Scholar] [CrossRef]
- Poludniowski, G.; Omar, A.; Bujila, R.; Andreo, P. Technical Note: SpekPy v2.0—A Software Toolkit for Modeling X-ray Tube Spectra. Med Phys 2021, 48, 3630–3637. [Google Scholar] [CrossRef] [PubMed]
- Vorbau, R.; Poludniowski, G. Technical Note: SpekPy Web—Online X-ray Spectrum Calculations using an Interface to the SpekPy Toolkit. J Appl Clin Med Phys 2024, 25, 1–5. [Google Scholar] [CrossRef]
- Jakubek, J. Data Processing and Image Reconstruction Methods for Pixel Detectors. Nucl Instrum Meth A 2007, 576, 223–234. [Google Scholar] [CrossRef]
- Sarrut, D.; Arbor, N.; Baudier, T.; Borys, D.; Etxebeste, A.; et al. The OpenGATE Ecosystem for Monte Carlo Simulation in Medical Physics. Phys Med Biol 2022, 67. [Google Scholar] [CrossRef]
- Allison, J.; Amako, K.; Apostolakis, J.; Arce, P.; Asai, M.; et al. Recent Developments in Geant4. Nucl Instrum Meth A 2016, 835. [Google Scholar] [CrossRef]
- Hologic, Inc. Selenia Dimensions with Acquisition Workstation 5000; Hologic, Inc., 2011. [Google Scholar]
- Harms, A.A.; Zeilinger, A. A New Formulation of Total Unsharpness in Radiography. Phys Med Biol 1977, 22, 70–80. [Google Scholar] [CrossRef]
- Spak, D.A.; Plaxco, J.S.; Santiago, L.; Dryen, M.J.; Dogan, B.E. BI-RADS® fifth edition: A summary of changes. Diagn Interv Imag 2017, 98, 179–190. [Google Scholar] [CrossRef]
- Destounis, S.V.; Friedewald, S.M.; Grimm, L.J.; Poplack, S.P.; Sung, J.S. Mammography. In ACR BI-RADS® v2025 Manual; Newell, M.S., Destounis, S.V, Eds.; American College of Radiology: Reston, VA (USA), 2025. [Google Scholar]
- Oliveira, A.; Meguerian, B.A.; Mesquita, C.T. Influence of Breast Characteristics in Myocardial Scintigraphy through the Monte Carlo Method. Arq Bras Cardiol 2011, 96. [Google Scholar] [CrossRef]
- CIRS, INC.Tissue-Equivalent Phantom for Mammography - Model 011A. Computerized Imaging Reference Systems - CIRS, 2013 .
- Rose, A. Vision - Human and Electronic, 1st ed.; Springer: New York, USA, 1973; ISBN 978-1-4684-2039-5. [Google Scholar] [CrossRef]
- Burgess, A.E. The Rose Model, Revisited. J Opt Soc Am A 1999, 16. [Google Scholar] [CrossRef]
- Bearden, J.A. X-Ray Wavelengths. Rev Mod Phys 1967, 39, 78–124. [Google Scholar] [CrossRef]
- Krause, M.O. Atomic Radiative and Radiationless Yields for K and L Shells. J Phys Chem Ref Data 1979, 8, 307–327. [Google Scholar] [CrossRef]
- Anton, G.; Durst, J.; Firsching, M.; Giersch, J.; Korn, A.; et al. Imaging Theory for X-ray Pixel Detectors. Nucl Instrum Meth A 2006, 563, 116–123. [Google Scholar] [CrossRef]
- Giersch, J.; Niederlöhner, D.; Anton, G. The Influence of Energy Weighting on X-ray Imaging Quality. Nucl Instrum Meth A 2004, 531, 68–74. [Google Scholar] [CrossRef]
- Roque, G.; Schütz, M.K.; Fey, J.; Useche Parra, J.S.; Fiederle, M.; Procz, S. Sub-pixel Energy-weighting Techniques for Metallic Contaminant Highlighting in a Pharmaceutical Hard Capsule using a Timepix3 CdZnTe Hybrid Pixel Detector. J Inst 2022, 17. [Google Scholar] [CrossRef]
- Schopoulos, I.; Rogers, D.W.O.; Bazalova-Carter, M.; Bolch, W.; Heath, E.; et al. RECORDS: improved Reporting of montE CarlO RaDiation transport Studies: Report of the AAPM Research Committee Task Group 268. Med Phys 2017, 45, 1–5. [Google Scholar] [CrossRef]

| Material | Adipose tissue | Glandular tissue |
|---|---|---|
| Density [] | 0.92 | 1.02 |
| Element | Weight Fraction | |
| Hydrogen | 0.120 | 0.106 |
| Carbon | 0.640 | 0.332 |
| Nitrogen | 0.008 | 0.030 |
| Oxygen | 0.229 | 0.527 |
| Sodium | 0 | 0.001 |
| Sulfur | 0 | 0.002 |
| Chlorine | 0 | 0.001 |
| Phosphor | 0.002 | 0.001 |
| Calcium | 0.001 | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




