Submitted:
19 February 2026
Posted:
27 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
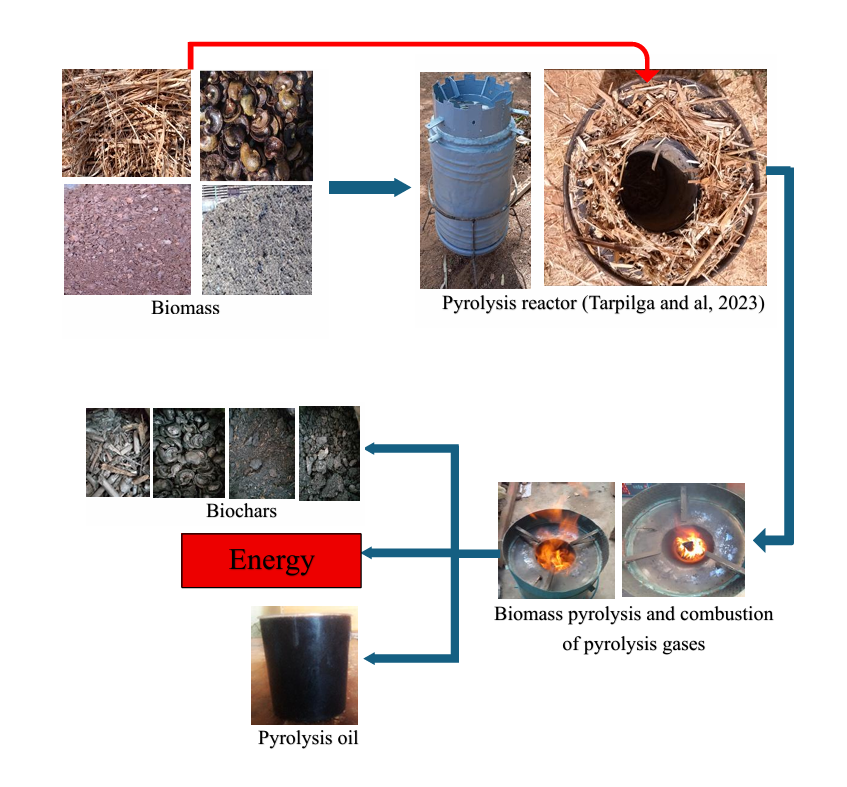
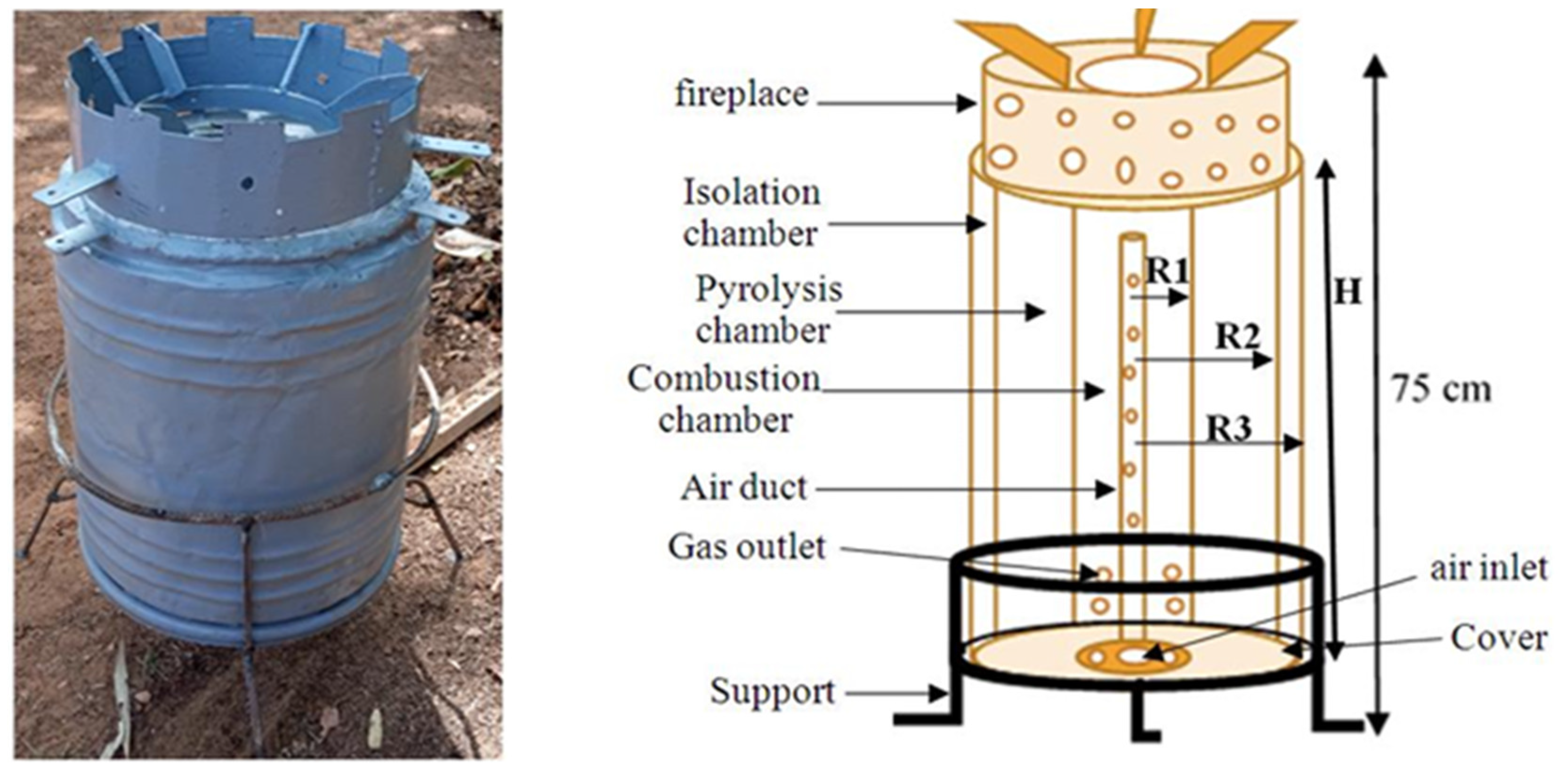
2.1. Pyrolyzer and Gas Detector
2.2. Biomasses
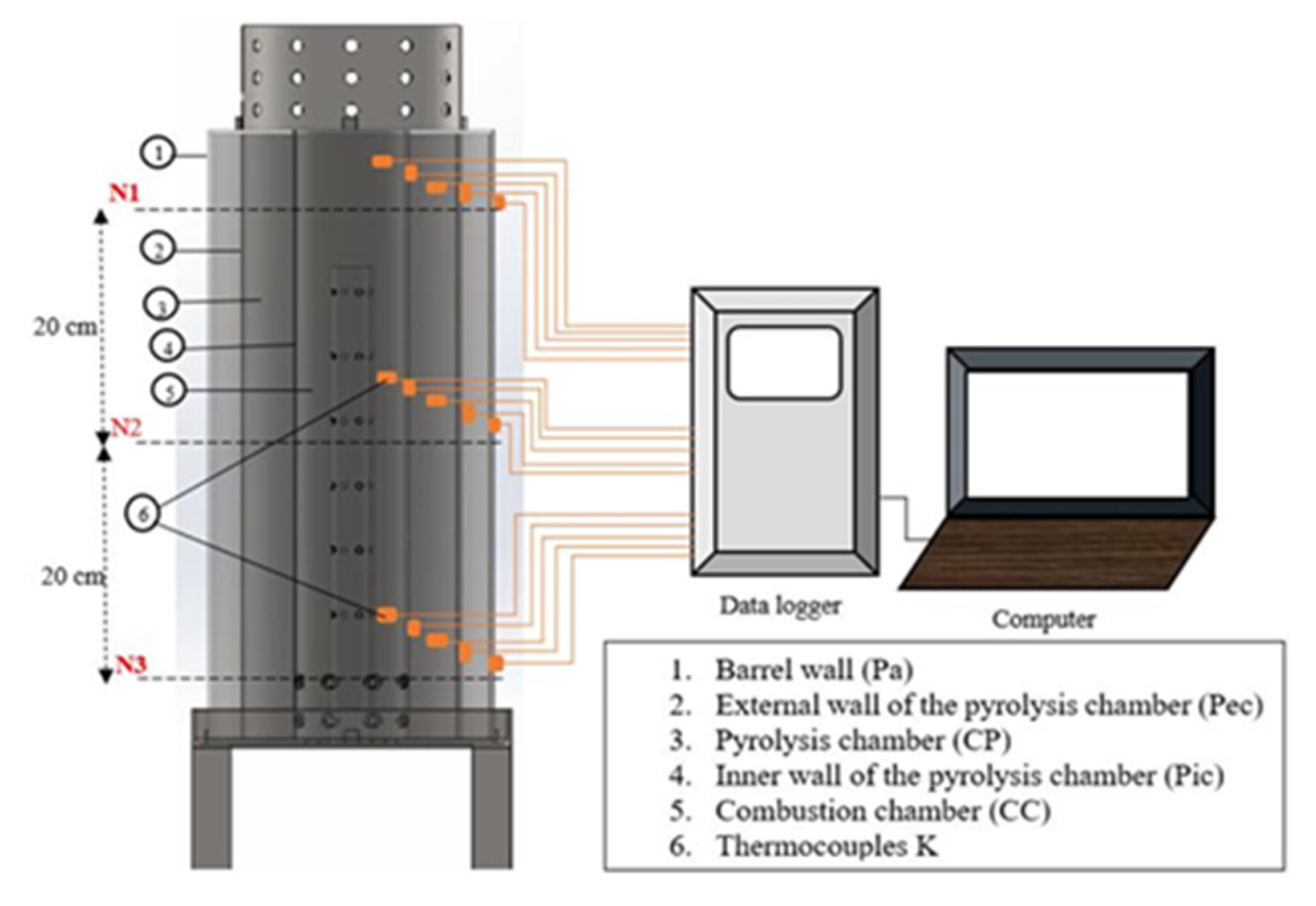
2.3. Thermocouples and Data Logger
3. Methods
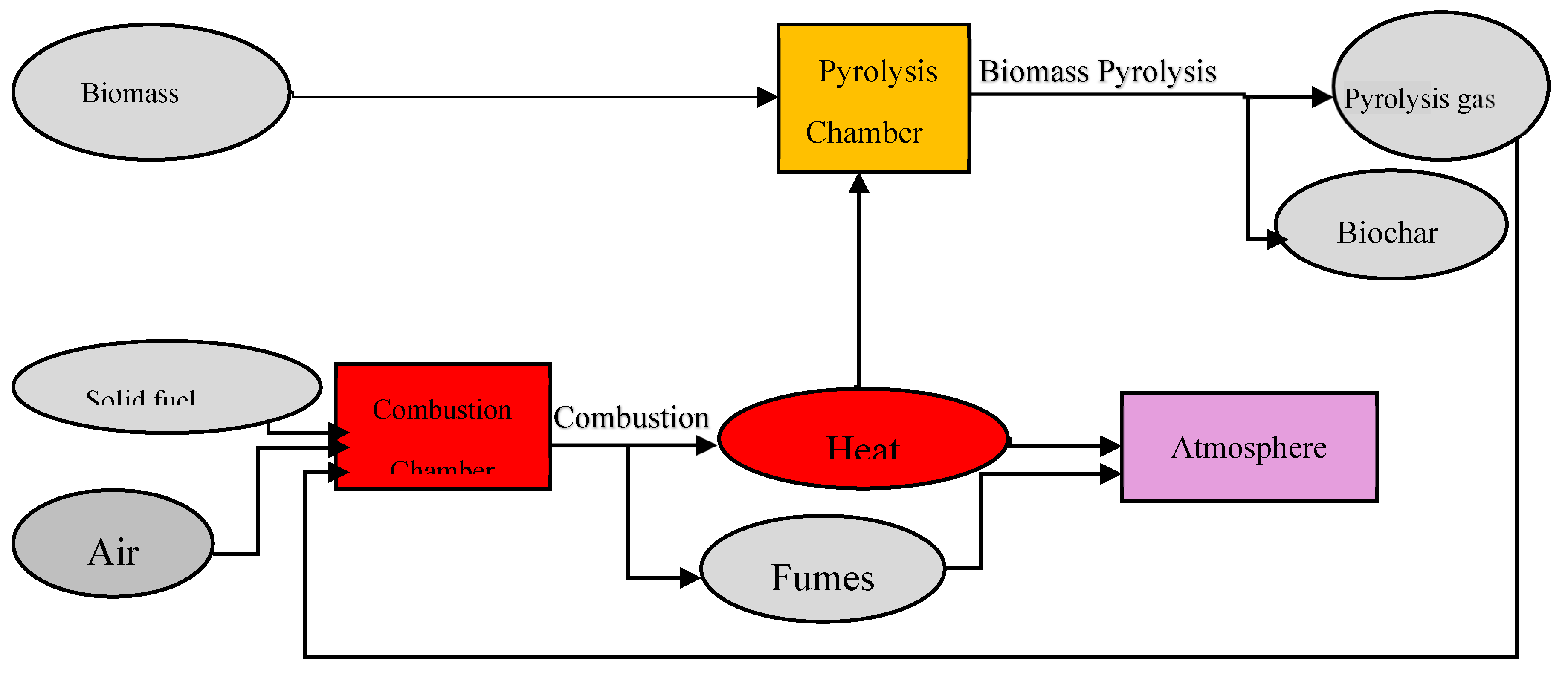
3.1. Oven Operation
3.2. Experimental Setup
3.3. Mass-Energy Balance and Performance Indicators
3.3.1. Mass and Energy Balance
3.3.2. Performance Indicators
4. Results
4.1. Pyrolysis Gas
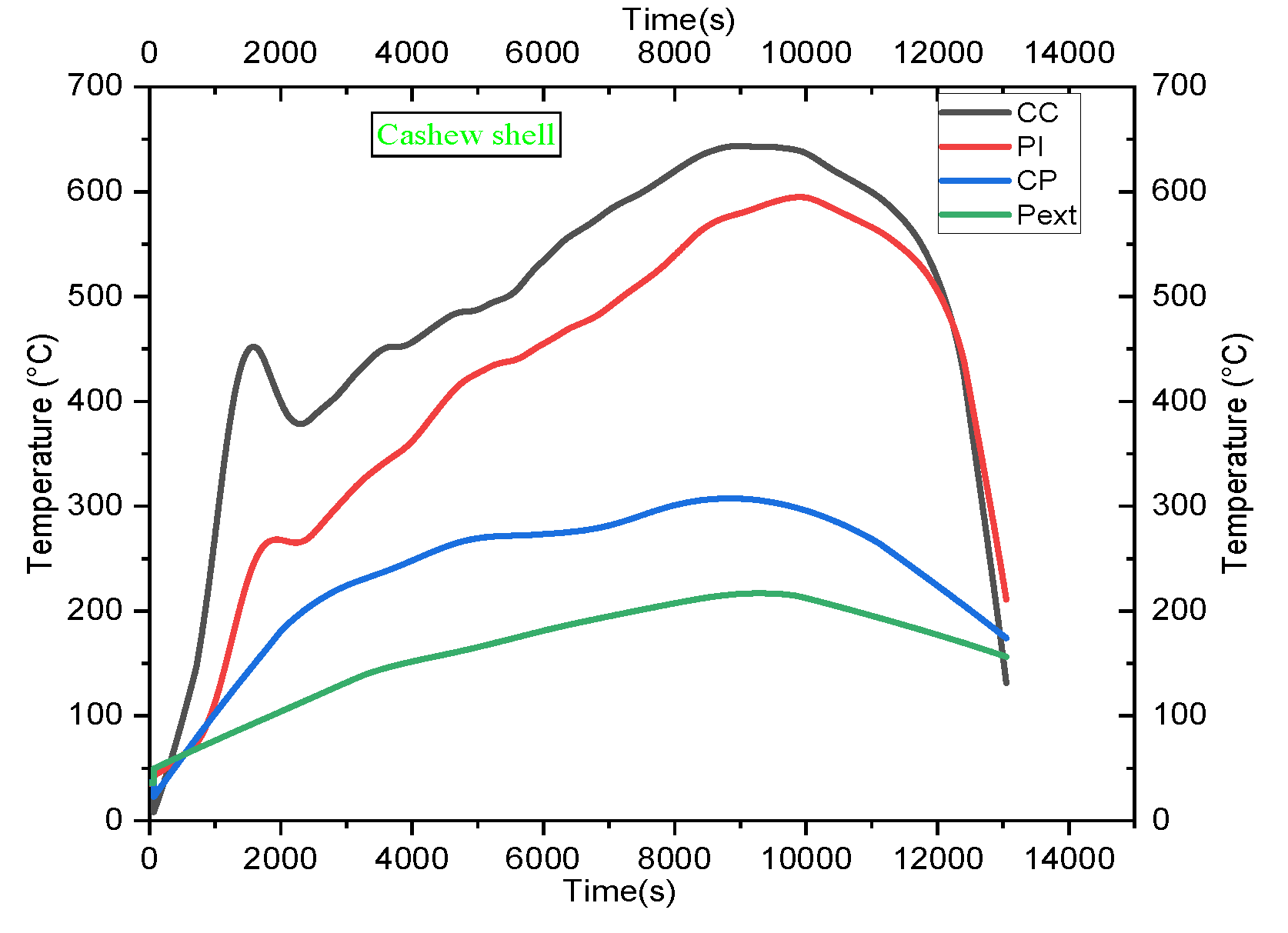
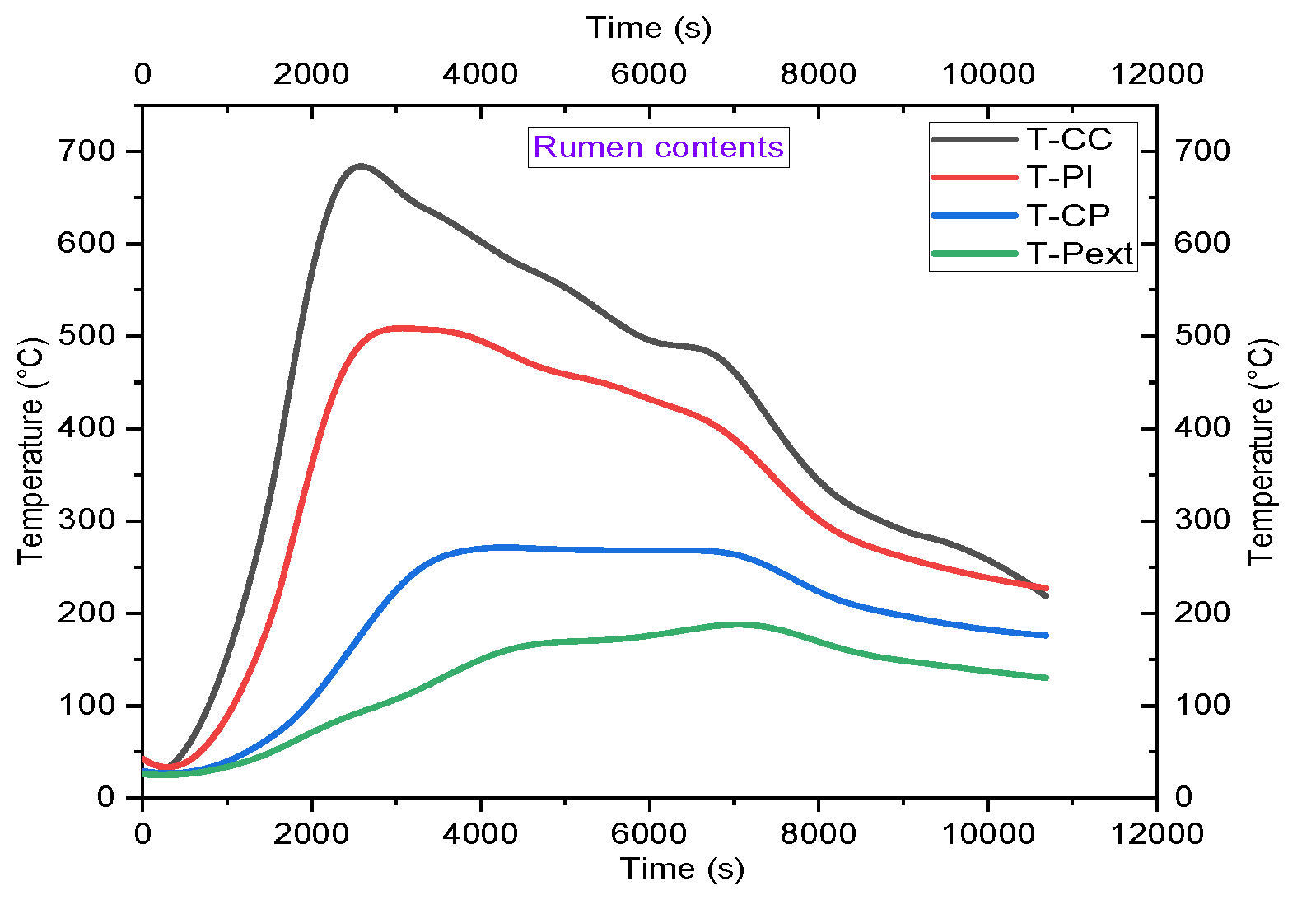
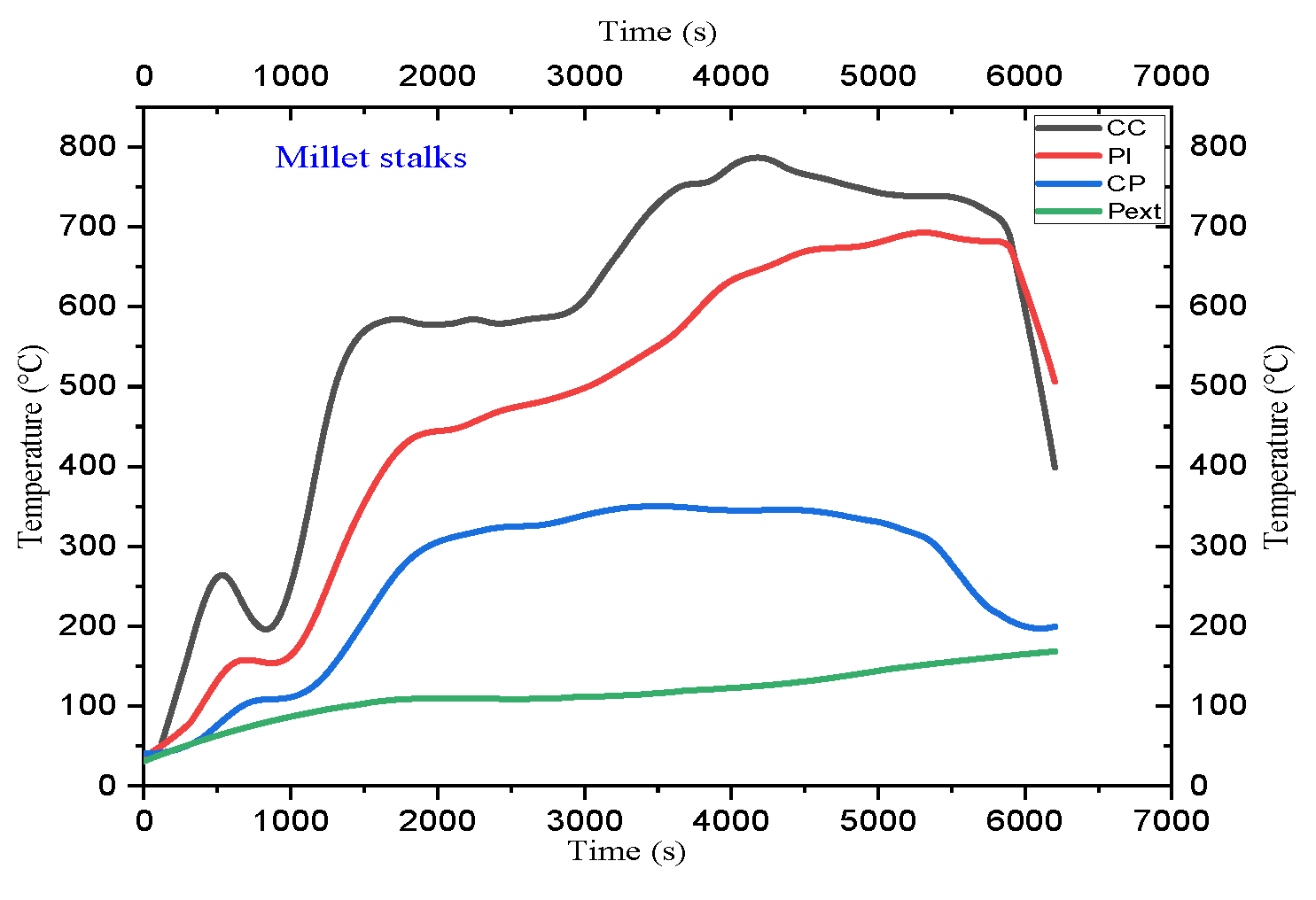
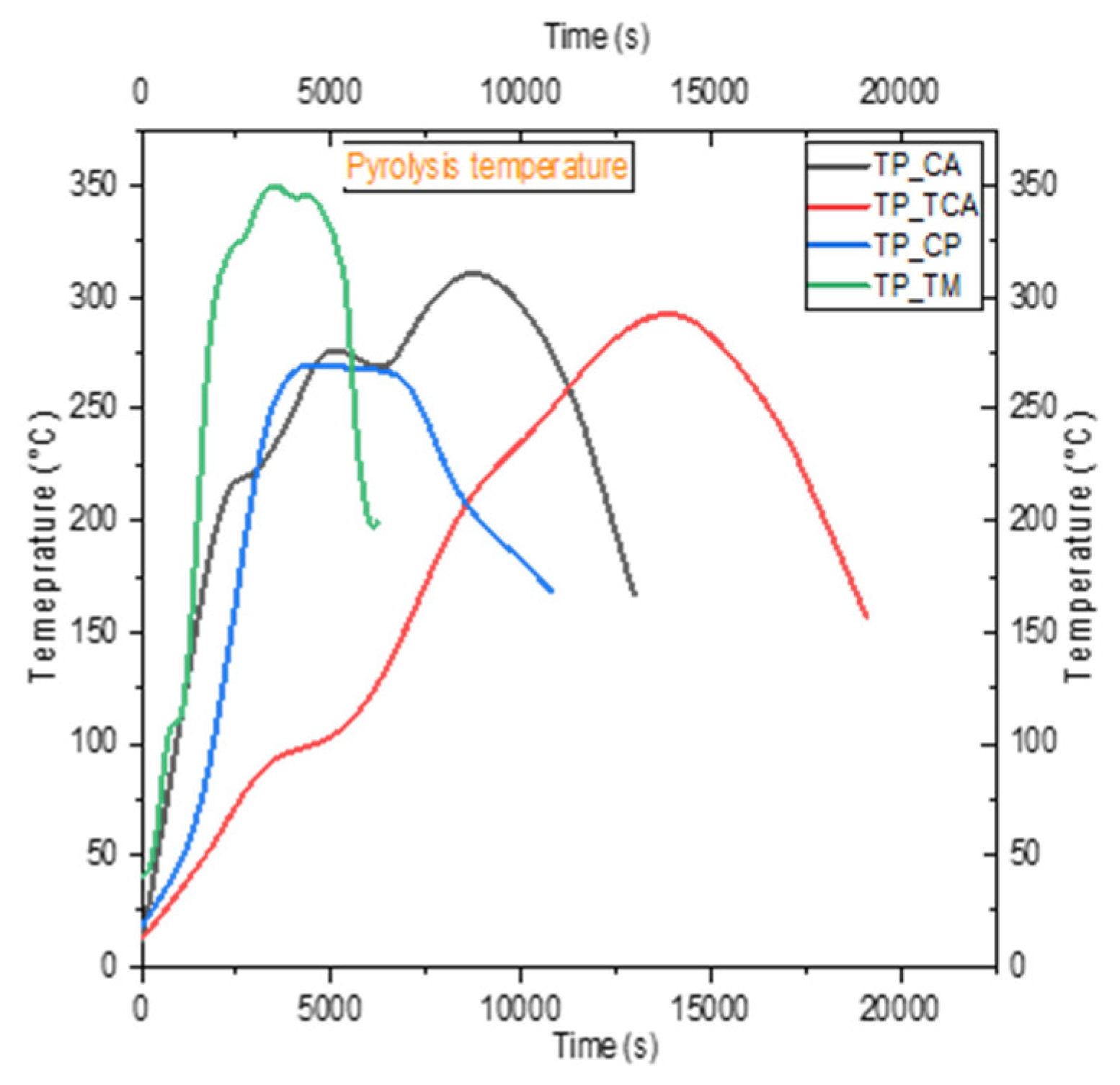
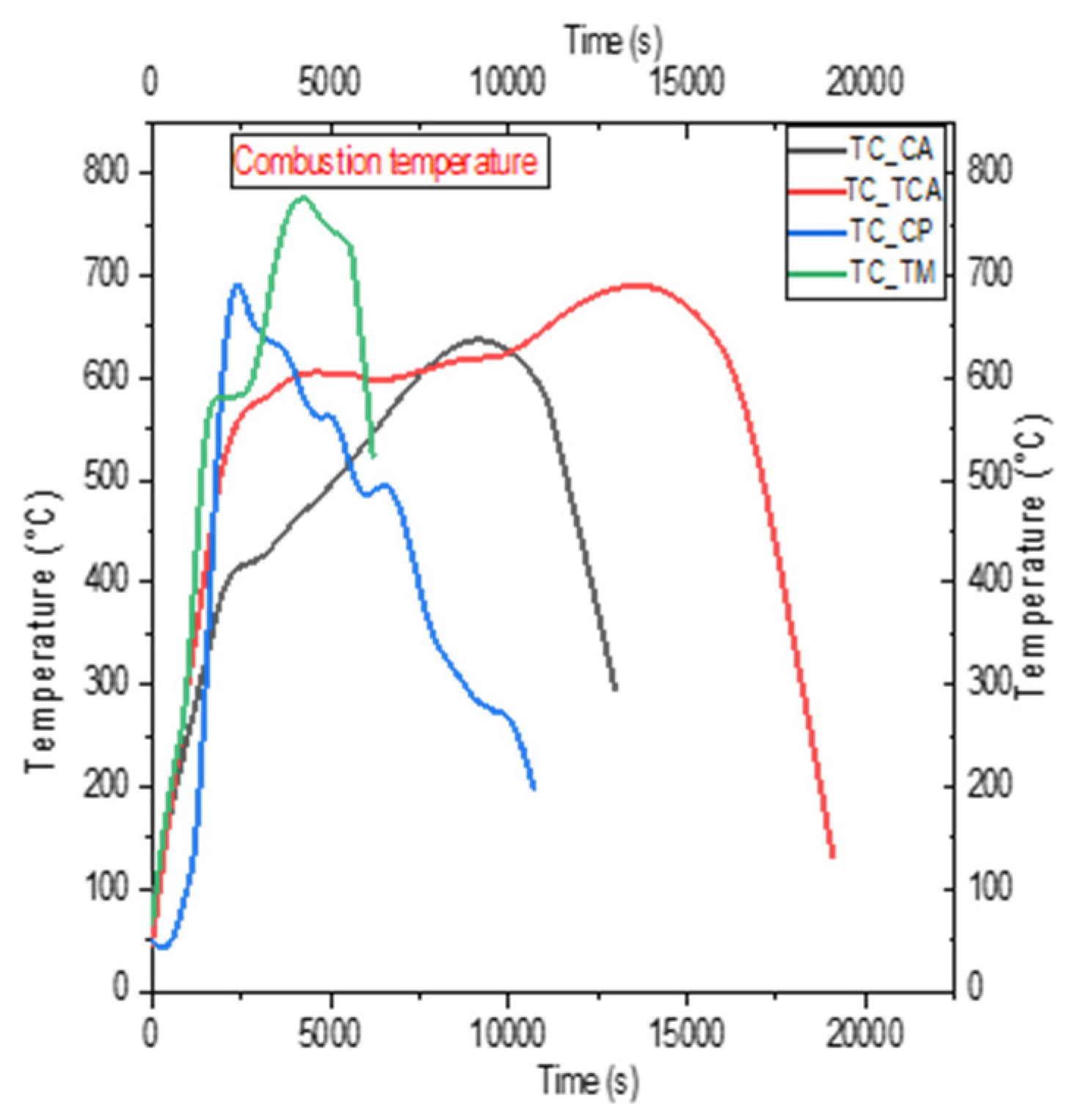
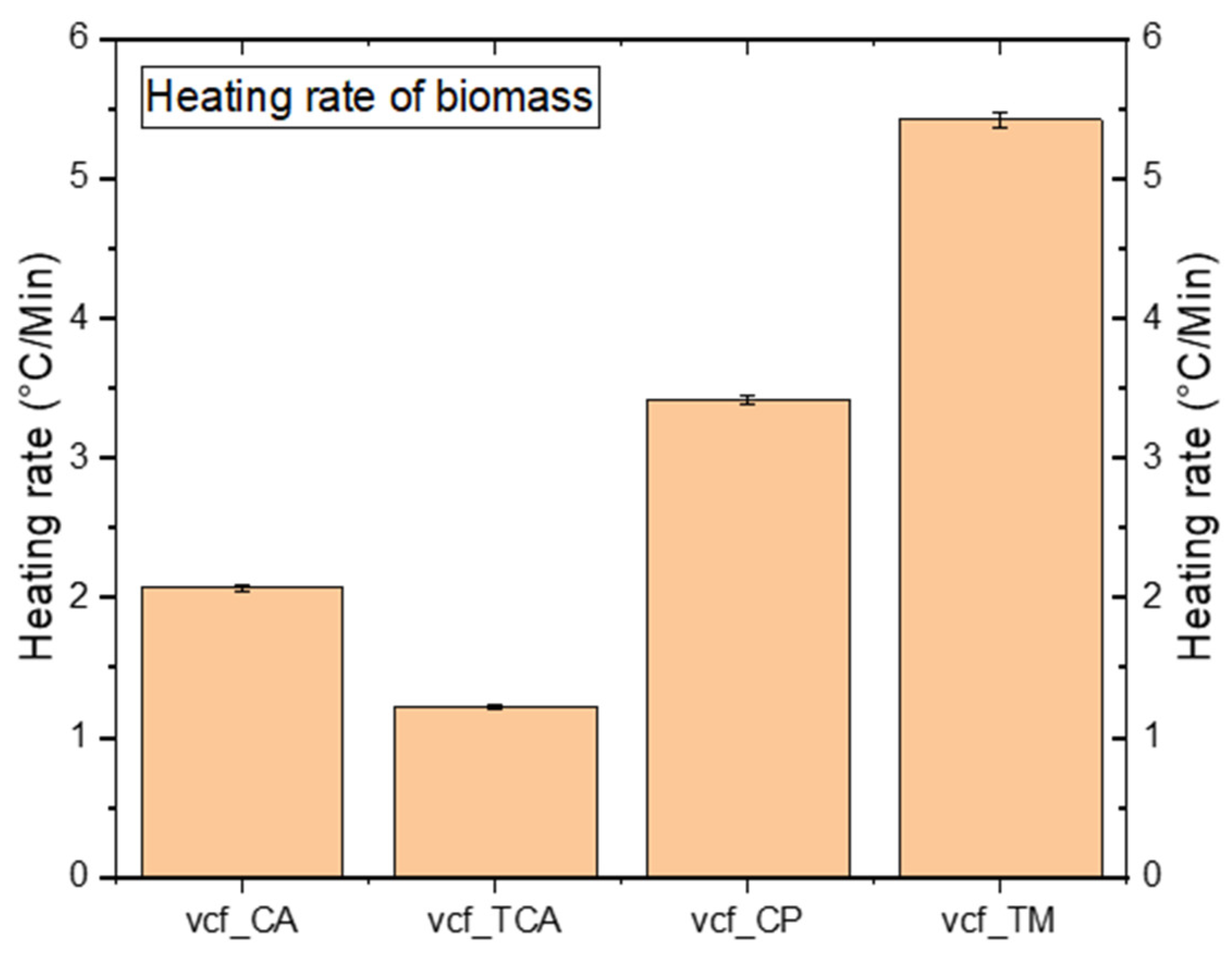
4.2. Temperature Profile
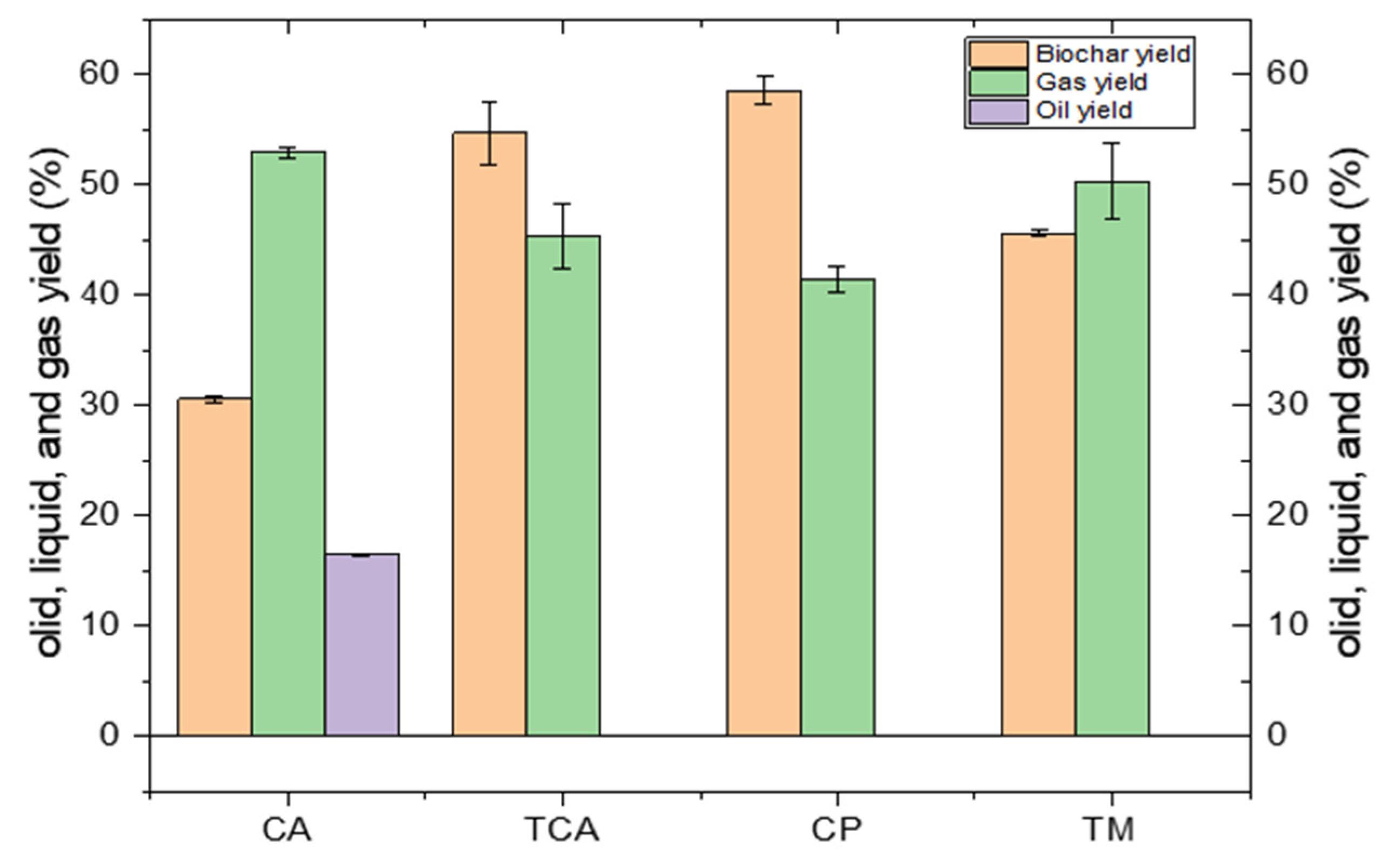
4.3. Pyrolysis efficiency
5. Discusssion
5.1. Pyrolysis Gas
5.2. Analysis of Temperature Profiles
5.3. Pyrolysis Efficiency
6. Conclusion
Conflicts of Interest
Appendix A
Appendix B

Appendix C

References
- Amaliyah, N., & Putra, A. E. E. (2021). Microwave-Assisted Pyrolysis of Cashew Nut Shell. International Journal of Design & Nature and Ecodynamics, 16(2), 227--232. [CrossRef]
- Armah, E. K., Chetty, M., Adedeji, J. A., Estrice, D. E., Mutsvene, B., Singh, N., Tshemese, Z., Armah, E. K., Chetty, M., Adedeji, J. A., Estrice, D. E., Mutsvene, B., Singh, N., & Tshemese, Z. (2022). Biochar: Production, Application and the Future. In Biochar—Productive Technologies, Properties and Applications. IntechOpen. [CrossRef]
- C.F.D.T, & agro-alimentaire, F. générale. (1981). Alternative pour aujourd’hui: La biomasse, énergie verte. FeniXX.
- Chormare, R., Moradeeya, P. G., Sahoo, T. P., Seenuvasan, M., Baskar, G., Saravaia, H. T., & Kumar, M. A. (2023). Conversion of solid wastes and natural biomass for deciphering the valorization of biochar in pollution abatement: A review on the thermo-chemical processes. Chemosphere, 339, 139760. [CrossRef]
- Chung, K. W. Y., Blin, J., Lanvin, C., Martin, E., Valette, J., & Van De Steene, L. (2024). Pyrolysis of cashew nut shells-focus on extractives. Journal of Analytical and Applied Pyrolysis, 179, 106452. [CrossRef]
- Demirbas, A. (2004). Combustion characteristics of different biomass fuels. Progress in Energy and Combustion Science, 30(2), 219--230. [CrossRef]
- Demirbas, Ayhan. (2004). Effect of initial moisture content on the yields of oily products from pyrolysis of biomass. Journal of Analytical and Applied Pyrolysis, 71(2), 803--815. [CrossRef]
- El-Sayed, S. A., Khass, T. M., & Mostafa, M. E. (2024). Thermal degradation behaviour and chemical kinetic characteristics of biomass pyrolysis using TG/DTG/DTA techniques. Biomass Conversion and Biorefinery, 14(15), 17779--17803. [CrossRef]
- Energy production from biomass (part 1): Overview of biomass. (2002). Bioresource Technology, 83(1), 37--46. [CrossRef]
- Graça, J., Kwapinska, M., Murphy, B., Duggan, T., Leahy, J. J., & Kelleher, B. (2024). Pyrolysis, a recovery solution to reduce landfilling of residual organic waste generated from mixed municipal waste. Environmental Science and Pollution Research, 31(21), 30676--30687. [CrossRef]
- Kan, T., Strezov, V., & Evans, T. J. (2016). Lignocellulosic biomass pyrolysis: A review of product properties and effects of pyrolysis parameters. Renewable and Sustainable Energy Reviews, 57, 1126--1140. [CrossRef]
- Laurent, P., Roiz, J., Wertz, J.-L., Richel, A., & Paquot, M. (2011). Le bioraffinage, une alternative prometteuse à la pétrochimie. BASE. https://popups.uliege.be/1780-4507/index.php?id=8007.
- Lewandowski, W. M., Ryms, M., & Kosakowski, W. (2020). Thermal Biomass Conversion: A Review. Processes, 8(5), 516. [CrossRef]
- Li, J., Lin, L., Ju, T., Meng, F., Han, S., Chen, K., & Jiang, J. (2024). Microwave-assisted pyrolysis of solid waste for production of high-value liquid oil, syngas, and carbon solids: A review. Renewable and Sustainable Energy Reviews, 189, 113979. [CrossRef]
- Liu, Q. D. Z. C. F. H. Y. Z. J. Y. S. G. H. (2018). Influences of Temperature and Residence Time on Secondary Reactions of Volatiles from Coal Pyrolysis. The Chinese Journal of Process Engineering, 18(1), 140. [CrossRef]
- Luo, S., Xiao, B., Hu, Z., & Liu, S. (2010). Effect of particle size on pyrolysis of single-component municipal solid waste in fixed bed reactor. International Journal of Hydrogen Energy, 35(1), 93--97. [CrossRef]
- McKendry, P. (2002). Energy production from biomass (part 1): Overview of biomass. Bioresource Technology, Reviews Issue, 83(1), 37--46. [CrossRef]
- Mehrabian, R., Scharler, R., & Obernberger, I. (2012). Effects of pyrolysis conditions on the heating rate in biomass particles and applicability of TGA kinetic parameters in particle thermal conversion modelling. Fuel, 93, 567--575. [CrossRef]
- M. Vilas-Boas, A. C., C. Tarelho, L. A., M. Oliveira, H. S., S. Silva, F. G. C., T. Pio, D., & A. Matos, M. A. (2024). Valorisation of residual biomass by pyrolysis: Influence of process conditions on products. Sustainable Energy & Fuels, 8(2), 379--396. [CrossRef]
- Nussbaumer, T. (2003). Combustion and Co-combustion of Biomass: Fundamentals, Technologies, and Primary Measures for Emission Reduction. Energy & Fuels, 17(6), 1510--1521. [CrossRef]
- Paudel, P. P., Kafle, S., Park, S., Kim, S. J., Cho, L., & Kim, D. H. (2024). Advancements in sustainable thermochemical conversion of agricultural crop residues: A systematic review of technical progress, applications, perspectives, and challenges. Renewable and Sustainable Energy Reviews, 202, 114723. [CrossRef]
- Siddiqui, M. T. H., Nizamuddin, S., Mubarak, N. M., Shirin, K., Aijaz, M., Hussain, M., & Baloch, H. A. (2019). Characterization and Process Optimization of Biochar Produced Using Novel Biomass, Waste Pomegranate Peel: A Response Surface Methodology Approach. Waste and Biomass Valorization, 10(3), 521--532. [CrossRef]
- Tarpilga et al.,2023. (s. d.). Consulté 8 mai 2025, à l’adresse https://www.elixirpublishers.com/articles/1682592793_202303002.pdf.
- Tarpilga, M., Ouedraogo, F., Gounkaou, Y., Da, F., Ouedraogo, S., & Naon, B. (2023). Temporal Temperature Evolusion during the Pyrolisis of Cotton Stalks, Corn Stalk and Rice Husk Using Multifunction Family Oven for the Biochar Production. International Journal of Plant & Soil Science, 35, 336--347. [CrossRef]
- Wang, P., Yu, J., Liu, X., & Millan, M. (2023). On the effect of pellet density on biomass pyrolysis in a pressurized fixed bed reactor. Fuel, 354, 129191. [CrossRef]


| Biomass | Constituents of the gases in the pyrolysis chamber | |||
|---|---|---|---|---|
| (%Vol) | (ppm) | (ppm) | (%LEL) | |
| Millet stalks (TM) | ||||
| Cashew shells (CA) | ||||
| Cashew nut cakes (TCA) | ||||
| Gut contents (CP) | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




