Submitted:
24 February 2026
Posted:
25 February 2026
You are already at the latest version
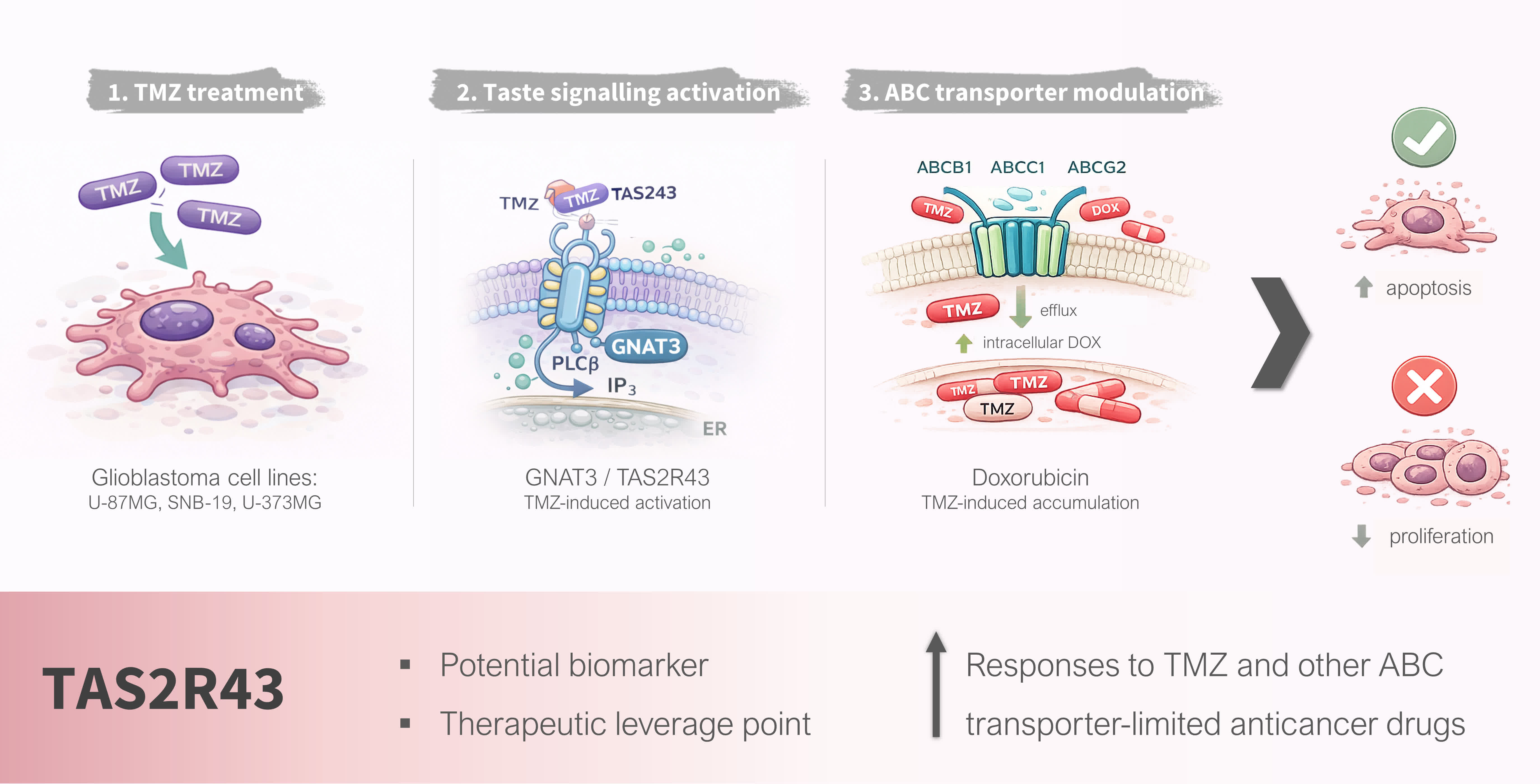
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Cell Culture
2.3. Evaluation of the Impact of TAS2R in TMZ Cytotoxicity
2.3.1. Effects of TMZ in Intracellular Ca2+ Responses of Glioblastoma Cells
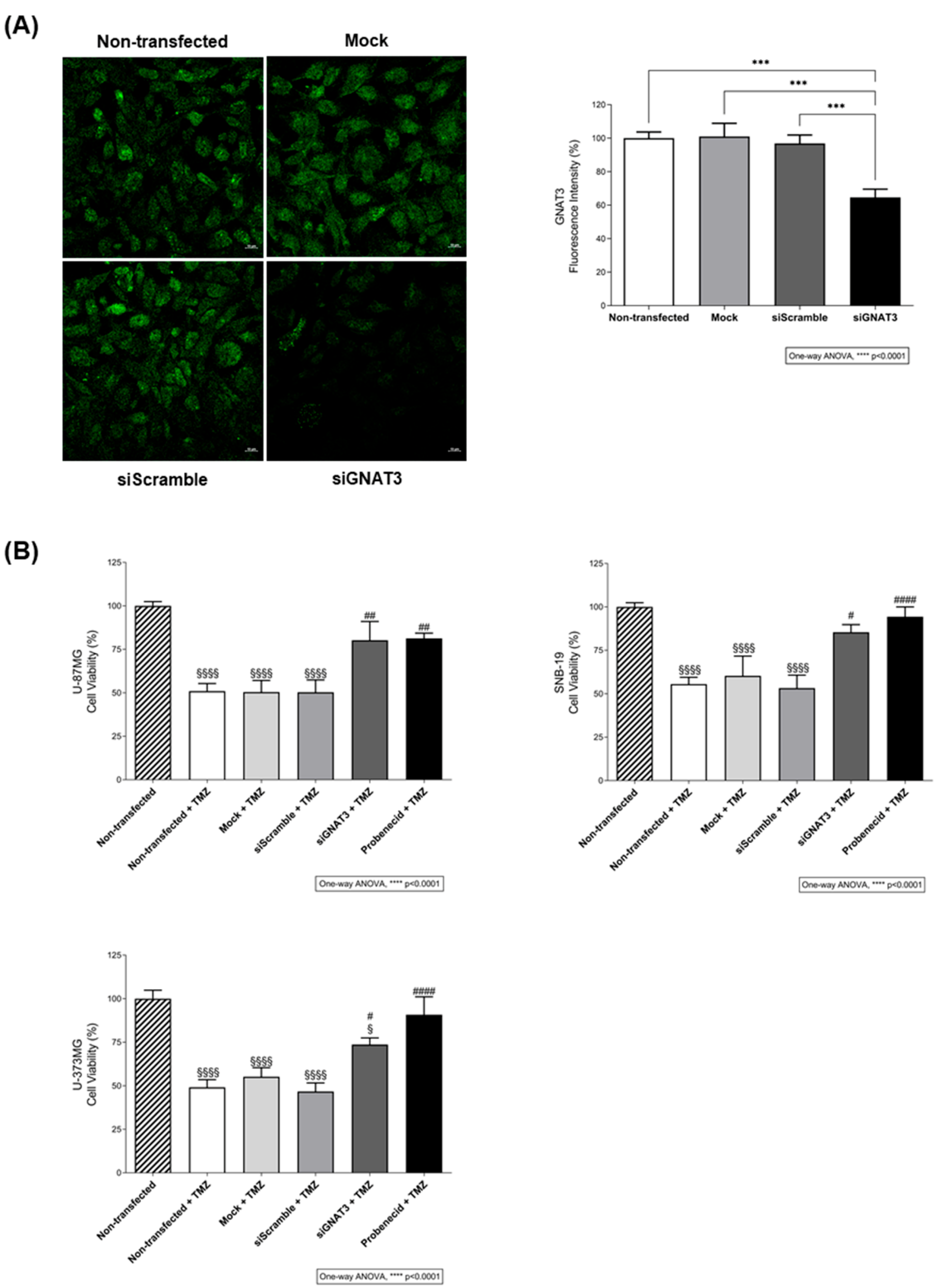
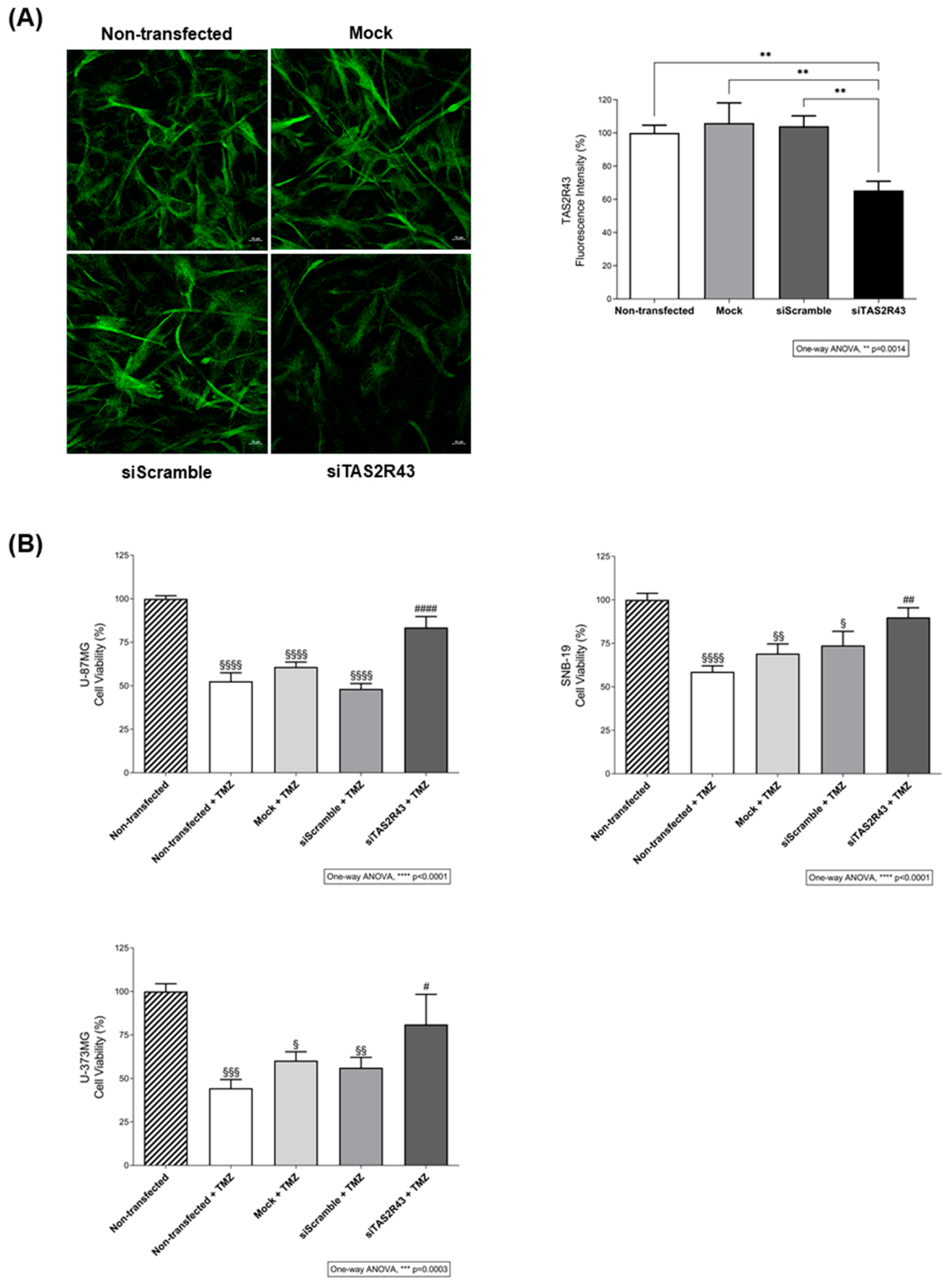
2.3.2. Evaluation of the Impact of GNAT3 Knockdown in TMZ Cytotoxicity
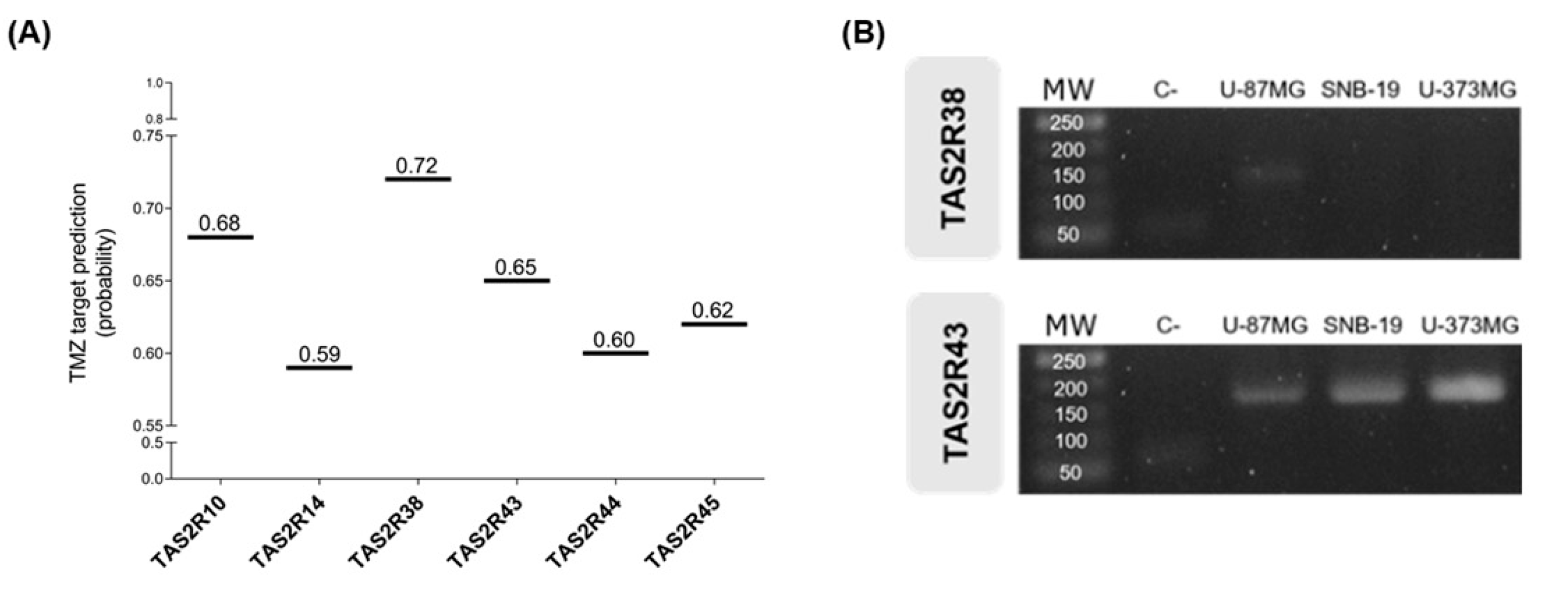
2.4. Identification of the TMZ Target TAS2R on Glioblastoma Cells
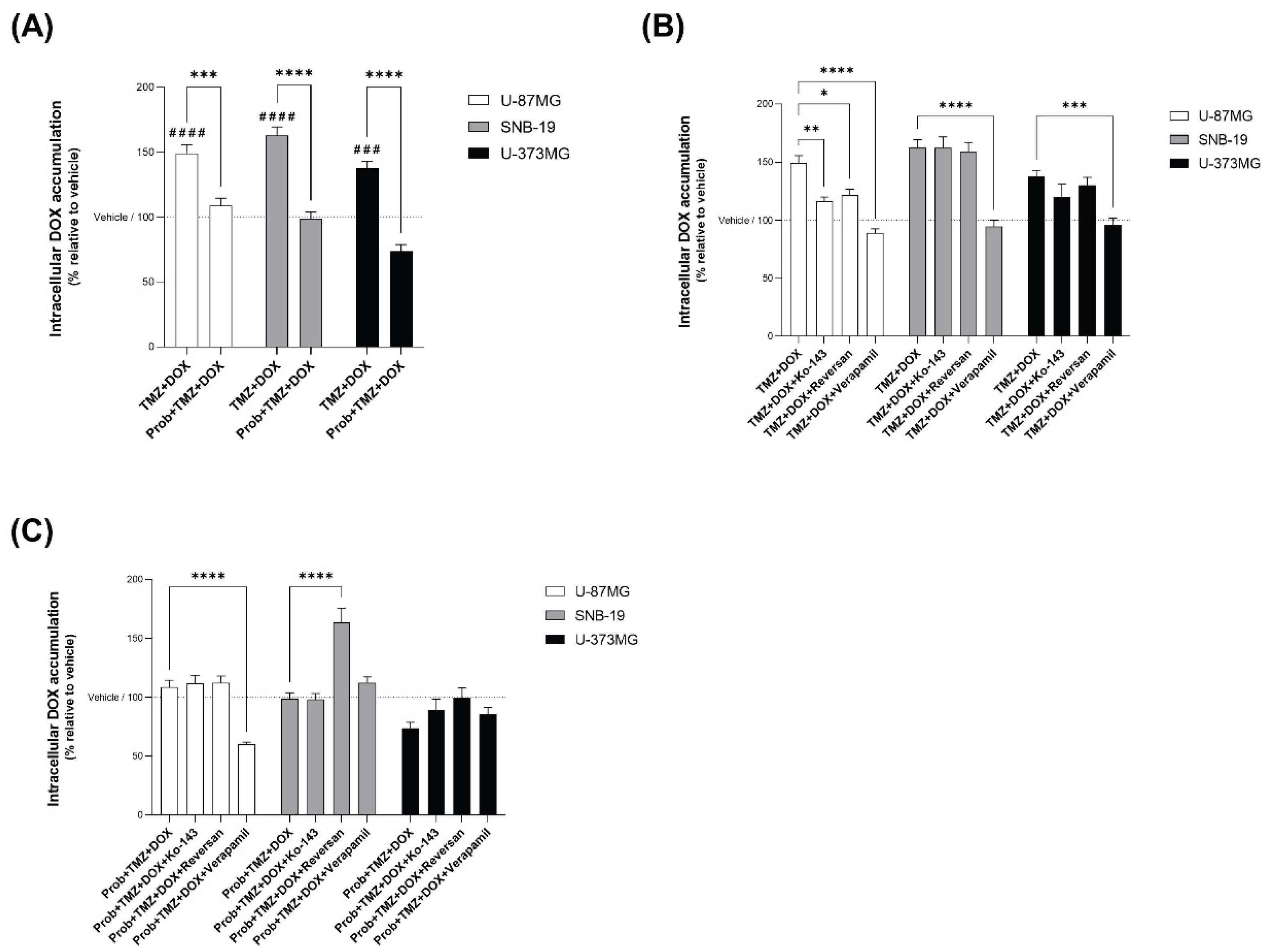
2.5. Effect of TMZ on ABC Transporters
2.6. Data Analysis
3. Results
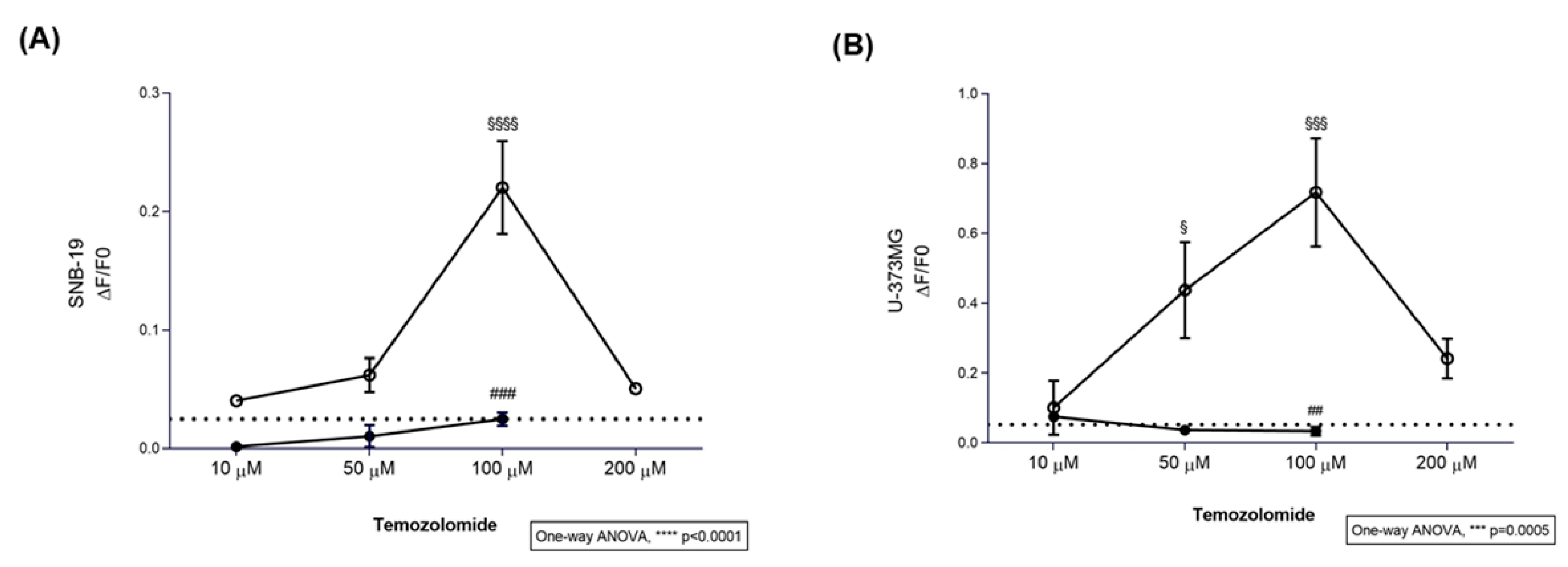
3.1. TMZ Elicits Intracellular Ca2+ Responses in Glioblastoma Cell Lines
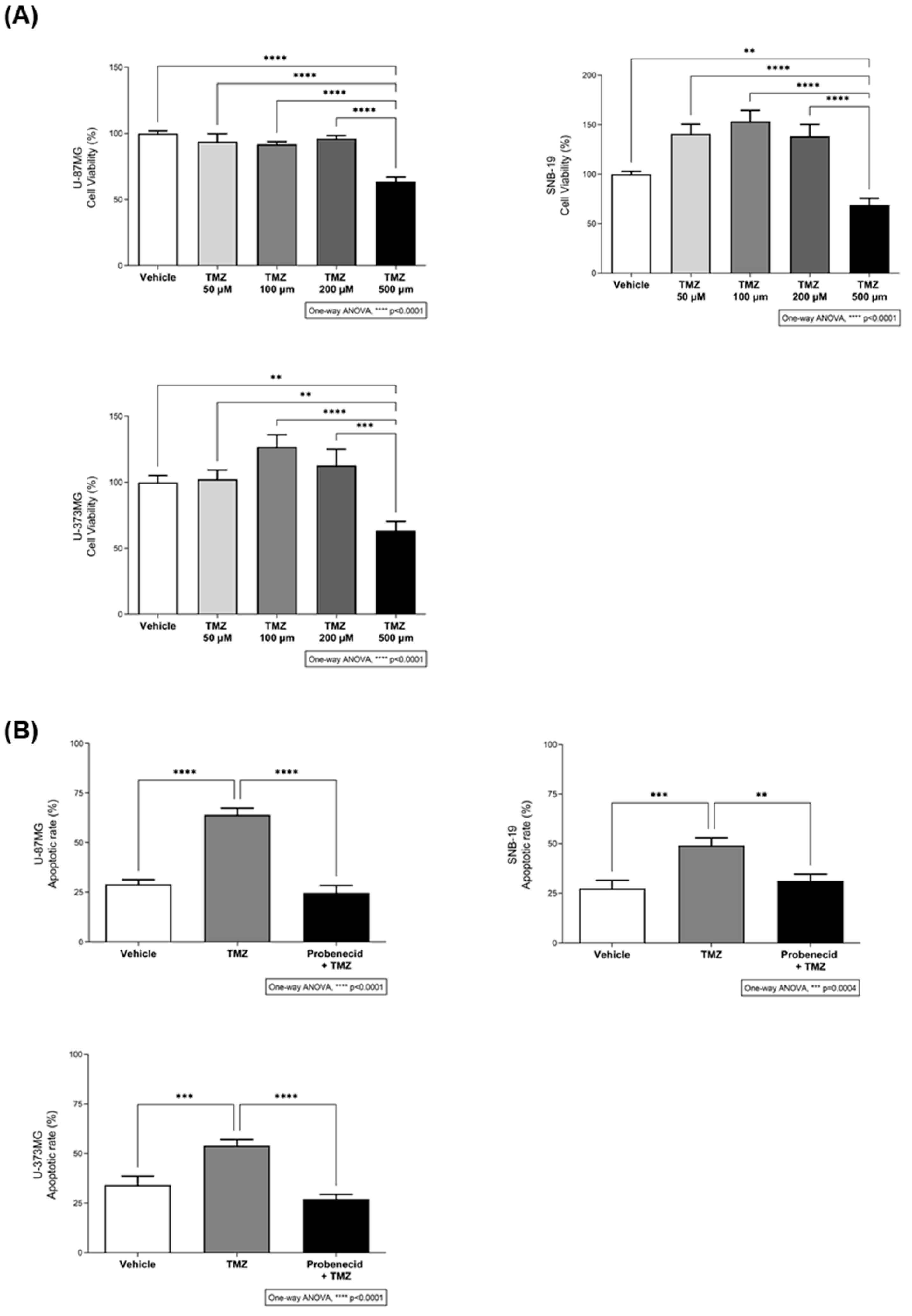
3.2. The Bitter Taste Signalling Pathway Enhances the Anticancer Effects of TMZ
3.3. TAS2R43 Mediates the Effects of TMZ in the Viability of Glioblastoma Cells
3.4. TMZ Induces Intracellular DOX Accumulation in Glioblastoma Cells
4. Discussion
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Avau, B.; Depoortere, I. The bitter truth about bitter taste receptors: beyond sensing bitter in the oral cavity. Acta Physiol. (Oxf.) 2016, 216, 407–420. [Google Scholar] [CrossRef] [PubMed]
- Duarte, A.C.; Santos, J.; Costa, A.R.; Ferreira, C.L.; Tomás, J.; Quintela, T.; et al. Bitter taste receptors profiling in the human blood-cerebrospinal fluid-barrier. Biochem Pharmacol. 2020, 177. [Google Scholar] [CrossRef] [PubMed]
- Li, F. Taste perception: from the tongue to the testis. Mol. Hum. Reprod. 2013, 19, 349–360. [Google Scholar] [CrossRef] [PubMed]
- Jaggupilli, A.; Singh, N.; Upadhyaya, J.; Sikarwar, A.S.; Arakawa, M.; Dakshinamurti, S.; et al. Analysis of the expression of human bitter taste receptors in extraoral tissues. Mol. Cell Biochem. 2017, 426, 137–147. [Google Scholar] [CrossRef]
- Dorsam, R.T.; Gutkind, J.S. G-protein-coupled receptors and cancer. Nat. Rev. Cancer 2007, 7, 79–94. [Google Scholar] [CrossRef]
- O’Hayre, M.; Degese, M.S.; Gutkind, J.S. Novel insights into G protein and G protein-coupled receptor signaling in cancer. Curr. Opin. Cell Biol. 2014, 27, 126–135. [Google Scholar] [CrossRef]
- Yang, J.; Xu, Y.; Fu, Z.; Chen, J.; Fan, W.; Wu, X. Progress in research and development of temozolomide brain-targeted preparations: a review. J. Drug Target. 2023, 31, 119–133. [Google Scholar] [CrossRef]
- Strobel, H.; Baisch, T.; Fitzel, R.; Schilberg, K.; Siegelin, M.D.; Karpel-Massler, G.; et al. Temozolomide and Other Alkylating Agents in Glioblastoma Therapy. Biomedicines 2019, 7, 69. [Google Scholar] [CrossRef]
- Kaina, B.; Christmann, M.; Naumann, S.; Roos, W.P. MGMT: Key node in the battle against genotoxicity, carcinogenicity and apoptosis induced by alkylating agents. DNA Repair (AMST) 2007, 6, 1079–1099. [Google Scholar] [CrossRef]
- Bao, S.; Wu, Q.; McLendon, R.E.; Hao, Y.; Shi, Q.; Hjelmeland, A.B.; et al. Glioma stem cells promote radioresistance by preferential activation of the DNA damage response. Nature 2006, 444, 756–760. [Google Scholar] [CrossRef]
- Chen, J.; Li, Y.; Yu, T.S.; McKay, R.M.; Burns, D.K.; Kernie, S.G.; et al. A restricted cell population propagates glioblastoma growth after chemotherapy. Nature 2012, 488, 522–526. [Google Scholar] [CrossRef] [PubMed]
- Eramo, A.; Ricci-Vitiani, L.; Zeuner, A.; Pallini, R.; Lotti, F.; Sette, G.; et al. Chemotherapy resistance of glioblastoma stem cells. Cell Death Differ. 2006, 13, 1238–1241. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Zepeda, D.; Taghi, M.; Scherrmann, J.M.; Decleves, X.; Menet, M.C. ABC Transporters at the Blood-Brain Interfaces, Their Study Models, and Drug Delivery Implications in Gliomas. Pharmaceutics 2019, 12. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.K.; Singh, P.; Ali, V.; Verma, M. Role of membrane-embedded drug efflux ABC transporters in the cancer chemotherapy. Oncol. Rev. 2020, 14, 144–151. [Google Scholar] [CrossRef]
- Hermann, D.M.; Kilic, E.; Spudich, A.; Krämer, S.D.; Wunderli-Allenspach, H.; Bassetti, C.L. Role of drug efflux carriers in the healthy and diseased brain. Ann. Neurol. 2006, 60, 489–498. [Google Scholar] [CrossRef]
- Qosa, H.; Miller, D.S.; Pasinelli, P.; Trotti, D. Regulation of ABC efflux transporters at blood-brain barrier in health and neurological disorders. Brain Res. 2015, 1628, 298–316. [Google Scholar] [CrossRef]
- Saunders, N.R.; Habgood, M.D.; Møllgård, K.; Dziegielewska, K.M. The biological significance of brain barrier mechanisms: Help or hindrance in drug delivery to the central nervous system? F1000Res 2016, 5. [Google Scholar] [CrossRef]
- Pitcher, M.R.; Quevedo, J. Tools for studying drug transport and metabolism in the brain. Expert. Opin. Drug Metab. Toxicol. 2016, 12, 161–168. [Google Scholar] [CrossRef]
- Adler, E.; Hoon, M.A.; Mueller, K.L.; Chandrashekar, J.; Ryba, N.J.P.; Zuker, C.S. A novel family of mammalian taste receptors. Cell 2000, 100, 693–702. [Google Scholar] [CrossRef]
- Chandrashekar, J.; Mueller, K.L.; Hoon, M.A.; Adler, E.; Feng, L.; Guo, W.; et al. T2Rs function as bitter taste receptors. Cell 2000, 100, 703–711. [Google Scholar] [CrossRef]
- Lang, T.; Di Pizio, A.; Risso, D.; Drayna, D.; Behrens, M. Activation Profile of TAS2R2, the 26th Human Bitter Taste Receptor. Mol Nutr Food Res. 2023, 67. [Google Scholar] [CrossRef] [PubMed]
- Diao, W.; Tong, X.; Yang, C.; Zhang, F.; Bao, C.; Chen, H.; et al. Behaviors of Glioblastoma Cells in in Vitro Microenvironments. Sci. Rep. 2019, 9, 85. [Google Scholar] [CrossRef] [PubMed]
- Memme, S.; Sukhorukov, V.L.; Höring, M.; Westerling, K.; Fiedler, V.; Katzer, A.; et al. Cell surface area and membrane folding in glioblastoma cell lines differing in PTEN and p53 status. PLoS One 2014, 9. [Google Scholar]
- Fritz, F.; Preissner, R.; Banerjee, P. VirtualTaste: a web server for the prediction of organoleptic properties of chemical compounds. Nucleic Acids Res. 2021, 49, W679–W684. [Google Scholar] [CrossRef]
- Costa, A.R.; Duarte, A.C.; Gonçalves, I.; Ferreira, C.L.; Cavaco, J.E.; Ferrer, I.; et al. Taste receptors’ profiling in glioblastoma. bioRxiv 2025, 2025.06.24.661264. [Google Scholar] [CrossRef]
- Thorn, C.F.; Oshiro, C.; Marsh, S.; Hernandez-Boussard, T.; McLeod, H.; Klein, T.E.; et al. Doxorubicin pathways: pharmacodynamics and adverse effects. Pharmacogenet Genom. 2011, 21, 440–446. [Google Scholar] [CrossRef]
- Amawi, H.; Hammad, A.M.; Hall, F.S.; Hussein, N.; Rataan, A.O.; Mrayyan, A.; et al. Revisiting strategies to target ABC transporter-mediated drug resistance in CNS cancer. Cancer Biol. Med. 2025, 22. [Google Scholar] [CrossRef]
- De Faria, G.P.; De Oliveira, J.A.; De Oliveira, J.G.P.; Romano, S.D.O.; Neto, V.M.; Maia, R.C. Differences in the expression pattern of P-glycoprotein and MRP1 in low-grade and high-grade gliomas. Cancer Invest. 2008, 26, 883–889. [Google Scholar] [CrossRef]
- Roy, L.O.; Lemelin, M.; Blanchette, M.; Poirier, M.B.; Aldakhil, S.; Fortin, D. Expression of ABCB1, ABCC1 and 3 and ABCG2 in glioblastoma and their relevance in relation to clinical survival surrogates. J. Neurooncol. 2022, 160, 601–609. [Google Scholar] [CrossRef]
- Schulz, J.A.; Hartz, A.M.S.; Bauer, B. ABCB1 and ABCG2 Regulation at the Blood-Brain Barrier: Potential New Targets to Improve Brain Drug Delivery. Pharmacol. Rev. 2023, 75, 815–853. [Google Scholar] [CrossRef]
- Engle, K.; Kumar, G. Cancer multidrug-resistance reversal by ABCB1 inhibition: A recent update. Eur. J. Med. Chem. 2022, 239. [Google Scholar] [CrossRef]
- Poku, V.O.; Iram, S.H. A critical review on modulators of Multidrug Resistance Protein 1 in cancer cells. PeerJ 2022, 10. [Google Scholar] [CrossRef] [PubMed]
- Toyoda, Y.; Takada, T.; Suzuki, H. Inhibitors of Human ABCG2: From Technical Background to Recent Updates With Clinical Implications. Front Pharmacol. 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Greene, T.A.; Alarcon, S.; Thomas, A.; Berdougo, E.; Doranz, B.J.; Breslin, P.A.S.; et al. Probenecid inhibits the human bitter taste receptor TAS2R16 and suppresses bitter perception of salicin. PLoS One 2011, 6. [Google Scholar] [CrossRef] [PubMed]
- Wölfle, U.; Haarhaus, B.; Kersten, A.; Fiebich, B.; Hug, M.J.; Schempp, C.M. Salicin from Willow Bark can Modulate Neurite Outgrowth in Human Neuroblastoma SH-SY5Y Cells. Phytother. Res. 2015, 29, 1494–1500. [Google Scholar] [CrossRef]
- Anwar, S.; Malik, J.A.; Ahmed, S.; Kameshwar, V.A.; Alanazi, J.; Alamri, A.; et al. Can Natural Products Targeting EMT Serve as the Future Anticancer Therapeutics? Molecules 2022, 27. [Google Scholar] [CrossRef]
- Costa, A.R.; Duarte, A.C.; Costa-Brito, A.R.; Gonçalves, I.; Santos, C.R.A. Bitter taste signaling in cancer. Life Sci. 2023, 315. [Google Scholar] [CrossRef]
- Luís, Â.; Marcelino, H.; Rosa, C.; Domingues, F.; Pereira, L.; Cascalheira, J.F. The effects of cannabinoids on glioblastoma growth: A systematic review with meta-analysis of animal model studies. Eur. J. Pharmacol. 2020, 876. [Google Scholar] [CrossRef]
- Banerjee, P.; Preissner, R. BitterSweetForest: A Random Forest Based Binary Classifier to Predict Bitterness and Sweetness of Chemical Compounds. Front Chem. 2018, 6. [Google Scholar] [CrossRef]
- Chen, T.C.; Wang, W.; Golden, E.B.; Thomas, S.; Sivakumar, W.; Hofman, F.M.; et al. Green tea epigallocatechin gallate enhances therapeutic efficacy of temozolomide in orthotopic mouse glioblastoma models. Cancer Lett. 2011, 302, 100–108. [Google Scholar] [CrossRef]
- Grube, S.; Ewald, C.; Kögler, C.; Lawson McLean, A.; Kalff, R.; Walter, J. Achievable Central Nervous System Concentrations of the Green Tea Catechin EGCG Induce Stress in Glioblastoma Cells in Vitro. Nutr. Cancer 2018, 70, 1145–1158. [Google Scholar] [CrossRef] [PubMed]
- Xie, C.R.; You, C.G.; Zhang, N.; Sheng, H.S.; Zheng, X.S. Epigallocatechin Gallate Preferentially Inhibits O6-Methylguanine DNA-Methyltransferase Expression in Glioblastoma Cells Rather than in Nontumor Glial Cells. Nutr. Cancer 2018, 70, 1339–1347. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, S.X.; Ma, J.W.; Li, H.Y.; Ye, J.C.; Xie, S.M.; et al. EGCG inhibits properties of glioma stem-like cells and synergizes with temozolomide through downregulation of P-glycoprotein inhibition. J. Neurooncol. 2015, 121, 41–52. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Oh, J.H.; Zhang, W.; Rathi, S.; Le, J.; Talele, S.; et al. How Much is Enough? Impact of Efflux Transporters on Drug delivery Leading to Efficacy in the Treatment of Brain Tumors. Pharm. Res. 2023, 40, 2731–2746. [Google Scholar] [CrossRef]
- Duarte, A.C.; Rosado, T.; Costa, A.R.; Santos, J.; Gallardo, E.; Quintela, T.; et al. The bitter taste receptor TAS2R14 regulates resveratrol transport across the human blood-cerebrospinal fluid barrier. Biochem Pharmacol. 2020, 177. [Google Scholar] [CrossRef]
- Lin, F.; De Gooijer, M.C.; Roig, E.M.; Buil, L.C.M.; Christner, S.M.; Beumer, J.H.; et al. ABCB1, ABCG2, and PTEN determine the response of glioblastoma to temozolomide and ABT-888 therapy. Clin. Cancer Res. 2014, 20, 2703–2713. [Google Scholar] [CrossRef]
- Munoz, J.L.; Walker, N.D.; Scotto, K.W.; Rameshwar, P. Temozolomide competes for P-glycoprotein and contributes to chemoresistance in glioblastoma cells. Cancer Lett. 2015, 367, 69–75. [Google Scholar] [CrossRef]
- Wu, H.; Gao, W.; Chen, P.; Wei, Y.; Zhao, H.; Wang, F. Research progress of drug resistance mechanism of temozolomide in the treatment of glioblastoma. Heliyon 2024, 10. [Google Scholar] [CrossRef]
- de Gooijer, M.C.; de Vries, N.A.; Buckle, T.; Buil, L.C.M.; Beijnen, J.H.; Boogerd, W.; etal. Improved Brain Penetration and Antitumor Efficacy of Temozolomide by Inhibition of ABCB1 and ABCG2. Neoplasia 2018, 20, 710–720. [Google Scholar] [CrossRef]
- Li, Y.; Liu, Y.; Ren, J.; Deng, S.; Yi, G.; Guo, M.; et al. miR-1268a regulates ABCC1 expression to mediate temozolomide resistance in glioblastoma. J. Neurooncol. 2018, 138, 499–508. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



