Submitted:
18 February 2026
Posted:
27 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
2.1. Palinological Results
2.2. Statistical Results
3. Discussion
| 1a. Pollen grains 5-porate ................................................................……………………….Alnus glutinosa 1b. Pollen grains 3-porate or 4–5-porate ..................................................................................................... 2 2a. Pollen grains 4–5-porate; P ≈ 31 µm; E ≈ 36 µm ......................................................... Carpinus betulus 2b. Pollen grains strictly 3-porate ………………………………………………………………................. 3 3a. Polar axis (P) > 27 µm …………………………………………………………………............................ 4 3b. Polar axis (P) < 27 µm ................................................................................................................................ 5 4a. P ≈ 28 µm; exine thickness ≈ 1.54 µm ………………………………………………….. Corylus colurna 4b. P ≈ 27 µm; pore length ≈ 2.59 µm ……………………………………………........... Carpinus orientalis 5a. Exine thickness < 1.0 µm ……………………………………………………..................... Betula pendula 5b. Exine thickness ≥ 1.0 µm ……………………………………………..................................................... 6 6a. Exine thickness ≈ 1.32 µm; E ≈ 27 µm ……………………………………………….... Corylus avellana 6b. Exine thickness ≈ 1.21 µm; E ≈ 25 µm ........................................................................ Ostrya carpinifolia |
4. Materials and Methods
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| µm | Micrometre |
| SEM | Scanning Electron Microscopy |
| ANOVA | ANalysis Of VAriance |
| Tukey’s HSD | Tukey’s Honestly Significant Difference |
| PCA | Principal Component Analysis |
| P | Polar axis length |
| E | Equatorial diameter |
| plg | Pore length |
| plt | Pore width |
| Ex | Exine thickness |
| LM | Light Microscopy |
References
- Erdtman, G. Pollen Morphology and Plant Taxonomy; Almqvist & Wiksell: Angiosperms, 1952. [Google Scholar]
- Punt, W.; Hoen, P.P.; Blackmore, S.; Nilsson, S.; Le Thomas, A. Glossary of pollen and spore terminology. Rev. Palaeobot. Palynol. 2007, 143, 1–81. [Google Scholar] [CrossRef]
- Karlıoğlu Kılıç, N.; Yılmaz Dağdeviren, R.; Caner, H.; Akkemik, Ü. Türkiye’de kullanılmakta olan palinoloji ve polen terimleri üzerine bir değerlendirme ve öneriler. Avrasya Terim Dergisi 2020, 8, 98–108. [Google Scholar] [CrossRef]
- Moore, P. D.; Webb, J. A. An Illustrated Guide to Pollen Analysis; Hodder and Stoughton: London, UK, 1978. [Google Scholar]
- Türkiye’nin Bütün Ağaçları ve Çalıları; Akkemik, Ü., Ed.; Türkiye İş Bankası Kültür Yayınları: Türkiye, 2023. [Google Scholar]
- Yaltırık, F.; Efe, A. Dendroloji Ders Kitabı; Çantay Kitabevi: İstanbul, Türkiye, 2000. [Google Scholar]
- Kayacık, H. Orman ve Park Ağaçlarının Özel Sistematiği: Angiospermae (Kapalı Tohumlular); İstanbul Üniversitesi Orman Fakültesi Yayınları: İstanbul, Türkiye, 1975. [Google Scholar]
- Wodehouse, R. P. Pollen Grains; McGraw-Hill Book Company: New York, 1935. [Google Scholar]
- Kuprianova, L. A. On a hitherto undescribed family belonging to the Amentiferae. Taxon 1963, 12, 12–13. [Google Scholar] [CrossRef]
- Praglowski, J. R. Notes on the pollen morphology of Swedish trees and shrubs. Grana Palynologica 1962, 3, 45–76. [Google Scholar] [CrossRef]
- Aytuğ, B. İstanbul Çevresi Bitkilerinin Polen Atlası; İstanbul Üniversitesi Orman Fakültesi Yayınları: İstanbul, Türkiye, 1971. [Google Scholar]
- Blackmore, S.; Steinmann, J.A.J.; Hoen, P. P.; Punt, W. Betulaceae and Corylaceae. Rev. Palaeobot. Palynol. 2003, 123, 71–98. [Google Scholar] [CrossRef]
- Alan, Ş. Türkiye Corylaceae ve Betulaceae Familyaları Polenlerinin Biyokimyasal ve Morfolojik Özellikleri. In Doktora Tezi; Ankara Üniversitesi: Ankara, 2010. [Google Scholar]
- Erdtman, G. An Introduction to Pollen Analysis; The Cnronica Botanica Company: Leyden, The Netherlands, 1943. [Google Scholar]
- Leopold, E.B.; Birkebak, J.; Reinink-Smith, L.; Jayachandar, A.P.; Narváez, P.; Zaborac-Reed, S. Pollen morphology of the three subgenera of Alnus. Palynology 2012, 36, 131–151. [Google Scholar] [CrossRef]
- Shayanmehr, F.; Jalali, S.H.; Hosseinzadeh Colagar, A.; Yousefzadeh, H.; Zare, H. Pollen morphology of the genus Alnus Mill. In Hyrcanian Forests, North of Iran. Appl. Environ. Res. 2014, 13, 819–831. [Google Scholar] [CrossRef]
- Kizilpinar Temizer, İ.; Turkmen, Z. Pollen morphology of A. glutinosa (L.) Gaertner subsp. glutinosa (Betulaceae). Kastamonu Üniversitesi Orman Fakültesi Dergisi 2016, 16, 423–426. [Google Scholar] [CrossRef]
- Sahli, A.; Kassout, J.; Boselli, V.A.; Ennouni, H.; Chakkour, S.; Kadaoui, K.; Houssni, M.; Ater, M. Pollen variability of Alnus glutinosa (L.) Gaerth. (Betulaceae) from southern range edge populations in Northern Morocco. Int. J. Plant Biol. 2023, 14, 797–810. [Google Scholar] [CrossRef]
- Kedves, M.; Pardutz, A. Scanning electron microscopy of some selected recent Amentiflorae pollen II. Acta Biol. Szeged. 1983, 29, 67–76. [Google Scholar]
- Mäkelä, E. M. Size distinctions between Betula pollen types-a review. Grana 1996, 35, 248–256. [Google Scholar] [CrossRef]
- Krizo, M.; Slobodnik, B. Changes in the size of pollen grains and structure of the tapetal layer in Betula pendula (Roth) during the maturation of pollen. Bull. Polish Acad. Sci. Biol. Sci 1997, 45, 2–4. [Google Scholar]
- Tetiana, S.; Kateryna, G.; Jan, B.; Radovan, O. Morphological characteristics and antioxidant activity of pollen silver birch (Betula pendula Ehrh.). Pharmacogn. Commun. 2014, 4, 25–34. [Google Scholar] [CrossRef]
- Bıçakçı, A.; Saatçioğlu, G.; Tosunoğlu, A. Allerjenik Betula (huş ağacı) polenlerinin Türkiye’deki dağılımları. Astım Allerji İmmünoloji 2017, 15, 142–153. [Google Scholar] [CrossRef]
- Payamnoor, V.; Sattarian, A.; Kordalivand, A.; Mohammadi, J. Pollen morphology and trichome types of Betula spp. in the Hyrcanian forests of northern Iran. Journal of Forestry Research 2019, 30, 1191–1198. [Google Scholar] [CrossRef]
- Acar Şahin, A.; Pınar, N.M. Pollen morphology of selected allergenıc specıes at beşevler 10. year campus, Ankara Unıversıty, Turkey. Communications Faculty of Sciences University of Ankara Series C Biology 2020, 29, 318–337. [Google Scholar]
- Kedves, M. Scanning electron microscopy of some selected recent Amentiflorae pollens I. Acta Bot. Acad. Sci. Hung. 1979, 25, 75–82. [Google Scholar]
- Akhondnezhad, S.; Nejadsattari, T.; Sattarian, A.; Asri, Y.; Bagheriieh, N.M. Pollen morphology of the genus Carpinus L. (Corylaceae) in Iran. Iran. J. Bot. 2011, 17, 233–237. [Google Scholar] [CrossRef]
- Nikolaieva, N.; Brindza, J.; Garkava, K.; Ostrovsky, R. Pollen features of hazelnut (Corylus avellana L.) from different habitats. Modern Phytomorphology 2014, 6, 53–58. [Google Scholar] [CrossRef]
- Hesse, M.; Halbritter, H.; Zetter, R.; Weber, M.; Buchner, R.; Frosch-Radivo, A.; Ulrich, S. Pollen Terminology an Illustrated Handbook, 1st ed.; Springer: Vienna, Austria, 2009. [Google Scholar]
- Beug, H.J. Leitfaden der Pollenbestimmung für Mitteleuropa und angrenzende Gebiete; Verlag Dr. Friedrich Pfeil: München, 2015. [Google Scholar]
- Python Software Foundation. Python (Version 3.10). 2023. Available online: https://www.python.org.
- McKinney, W. Data structures for statistical computing in Python. SciPy 2010, 445, 51–56. [Google Scholar] [CrossRef]
- Virtanen, P.; Gommers, R.; Oliphant, T. E.; Haberland, M.; Reddy, T.; Cournapeau, D.; Van Mulbregt, P. SciPy 1.0: fundamental algorithms for scientific computing in Python. Nature methods 2020, 17, 261–272. [Google Scholar] [CrossRef] [PubMed]
- Seabold, S.; Perktold, J. Statsmodels: econometric and statistical modeling with python. SciPy 2010, 7, 92–96. [Google Scholar] [CrossRef]
- Pedregosa, F.; Varoquaux, G.; Gramfort, A.; Michel, V.; Thirion, B.; Grisel, O.; Duchesnay, É. Scikit-learn: Machine learning in Python. Journal of Machine Learning Research 2011, 12, 2825–2830. Available online: https://inria.hal.science/hal-00650905v2.
- Hunter, J. D. Matplotlib: A 2D graphics environment. Computing in Science & Engineering 2007, 9, 90–95. [Google Scholar] [CrossRef]
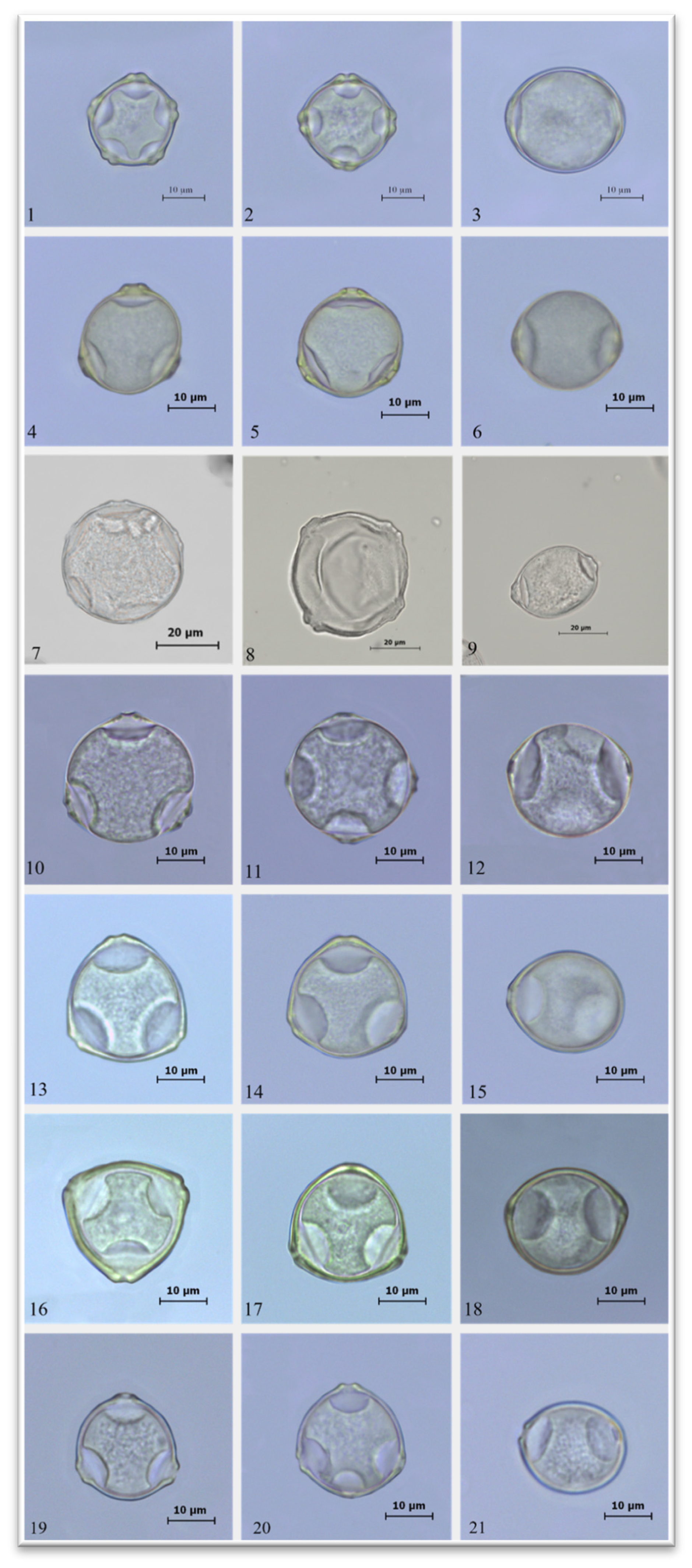
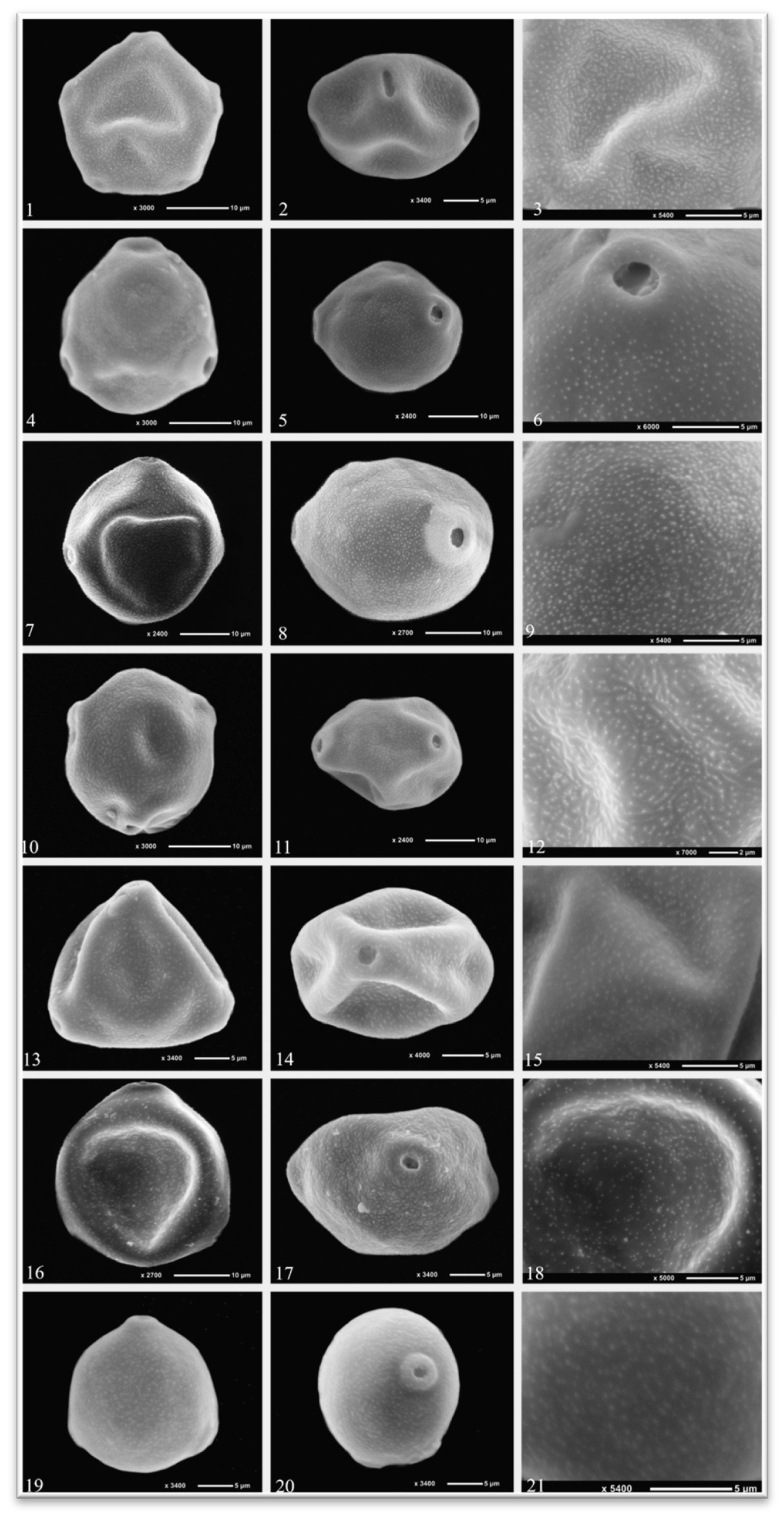
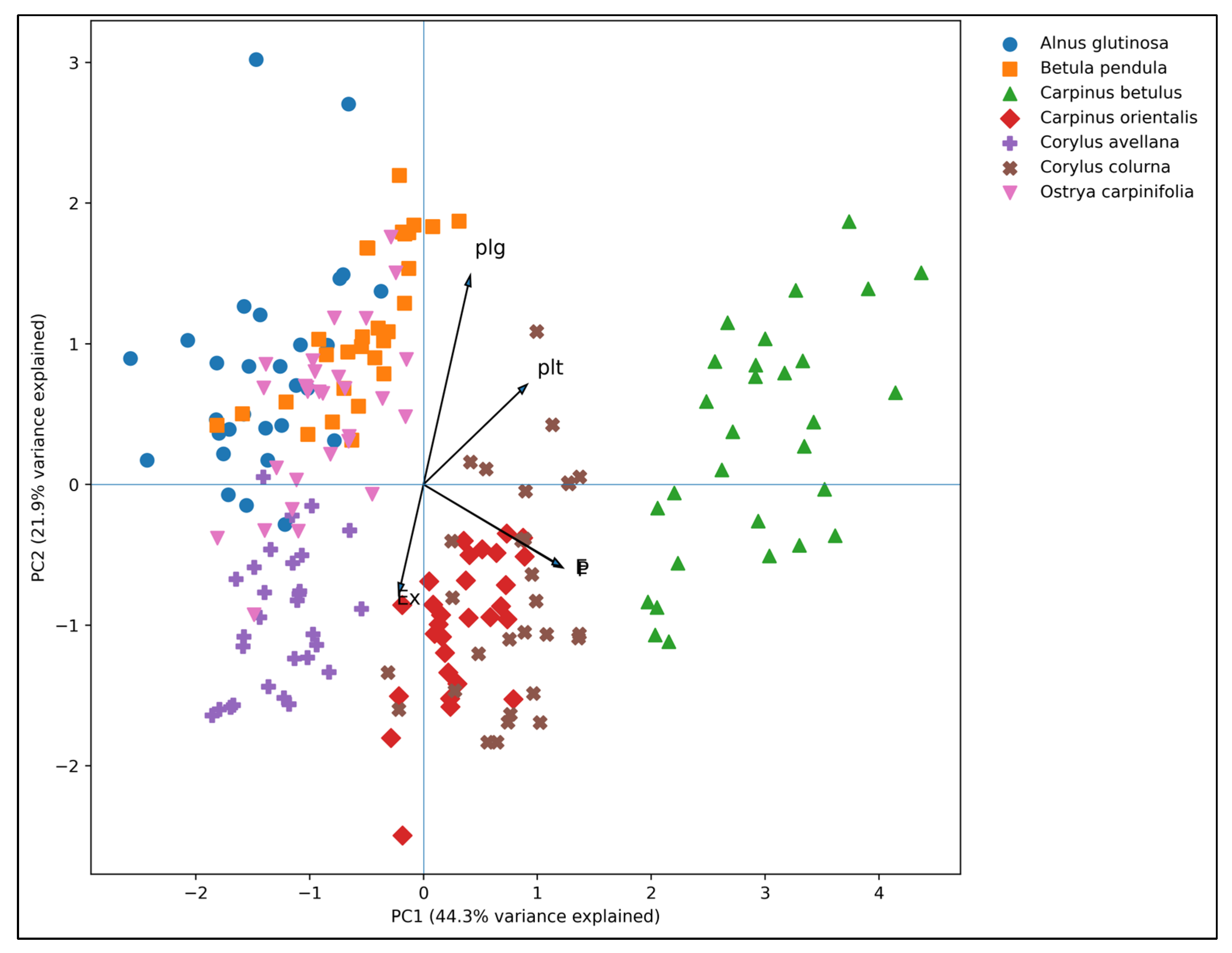
| Species | P (µm) |
E (µm) |
P/E | Plg (µm) | Plt (µm) | Ex (µm) | Number of pores | Ornamentation | |
|---|---|---|---|---|---|---|---|---|---|
| LM | SEM | ||||||||
|
Alnus glutinosa |
17.53-24.71 | 20.96-29.50 | 0.88 | 3.05-4.22 | 1.27-2.85 | 1.03-1.65 | 5 (Stephanoporate) |
Psilate | Microechinate |
|
Betula pendula |
20.64-24.82 | 23.27-26.96 | 0.89 | 2.58-3.51 | 2.03-3.24 | 0.63-1.16 | 3 (Triporate) |
Psilate | Microechinate |
| Carpinus betulus | 28.52-33.72 | 34.42-39.67 | 0.86 | 2.86-4.33 | 2.32-3.72 | 0.84-1.38 | 4-5 (Tetraporate, Stephanoporate) |
Psilate | Microechinate |
| Carpinus orientalis | 25.53-29.40 | 30-33.91 | 0.88 | 2.07-2.98 | 1.57-2.36 | 0.72-1.18 | 3 (Triporate) |
Psilate | Microechinate |
| Corylus avellana | 20.71-23.70 | 25.26-30.90 | 0.83 | 2.08-2.95 | 1.82-2.71 | 1.06-1.53 | 3 (Triporate) |
Psilate | Microechinate |
|
Corylus colurna |
26.01-29.33 | 27.8-32.46 | 0.93 | 2.36-3.76 | 2.11-3.25 | 1.09-1.92 | 3 (Triporate) |
Psilate | Microechinate |
| Ostrya carpinifolia | 21.09-23.92 | 22.71-26.91 | 0.91 | 2.26-3.49 | 2.01-2.96 | 0.89-1.52 | 3 (Triporate) |
Psilate | Microechinate |
| Species | P mean |
sd |
E mean |
sd |
plg mean |
sd |
plt mean |
sd |
Ex mean |
sd |
|---|---|---|---|---|---|---|---|---|---|---|
| Alnus glutinosa | 21.77 | 1.92 | 24.71 | 2.13 | 3.50 | 0.36 | 2.06 | 0.41 | 1.27 | 0.15 |
| Betula pendula | 22.64 | 1.07 | 25.34 | 0.83 | 3.06 | 0.26 | 2.76 | 0.30 | 0.86 | 0.15 |
| Carpinus betulus | 31.35 | 1.63 | 36.54 | 1.55 | 3.47 | 0.47 | 3.17 | 0.43 | 1.04 | 0.13 |
| Carpinus orientalis | 27.62 | 0.99 | 31.55 | 1.00 | 2.59 | 0.24 | 2.10 | 0.22 | 0.94 | 0.12 |
| Corylus avellana | 22.63 | 0.80 | 27.03 | 1.46 | 2.48 | 0.24 | 2.21 | 0.26 | 1.32 | 0.15 |
| Corylus colurna | 28.16 | 1.00 | 30.35 | 1.29 | 2.99 | 0.36 | 2.76 | 0.34 | 1.54 | 0.21 |
| Ostrya carpinifolia | 22.62 | 0.78 | 25.00 | 1.37 | 3.05 | 0.30 | 2.56 | 0.32 | 1.21 | 0.17 |
| Variable | ||
|---|---|---|
| P | 274.74 | p < 0.001 |
| E | 283.16 | p < 0.001 |
| plg | 42.02 | p < 0.001 |
| plt | 45.59 | p < 0.001 |
| Ex | 67.91 | p < 0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





