Submitted:
22 February 2026
Posted:
27 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Patients and Treatment
2.2. Evaluation and Laboratory Testing
2.3. Statistical Analysis
3. Results
3.1. Subsection
3.1.1. Comparative Analysis of the General Characteristics of the Three Groups
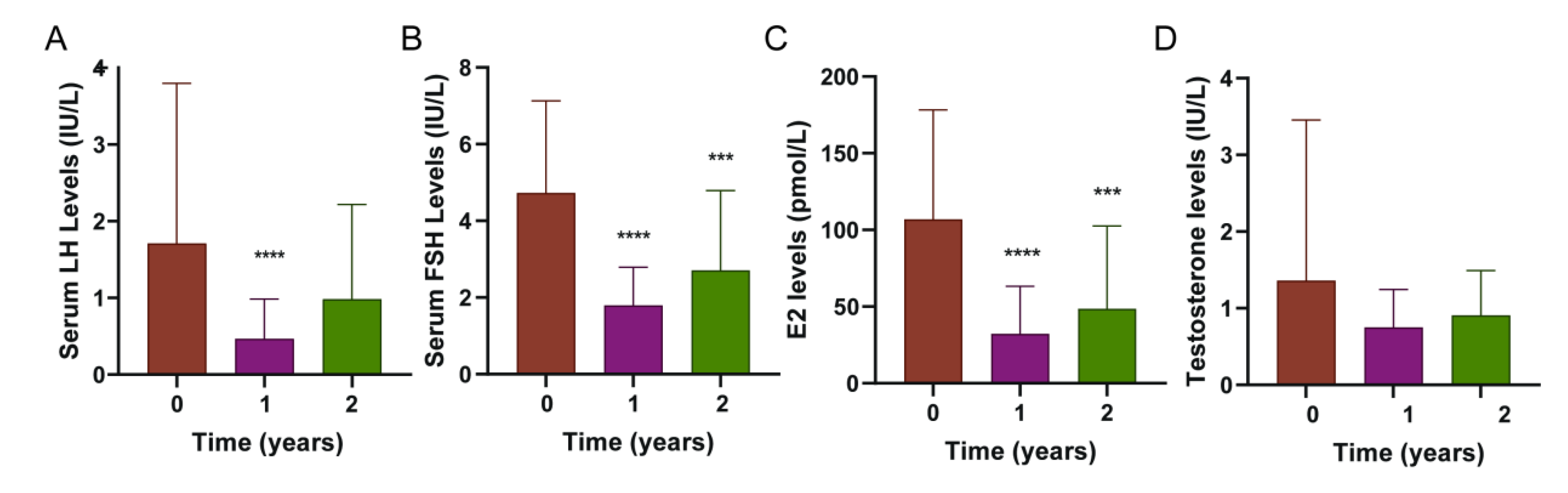
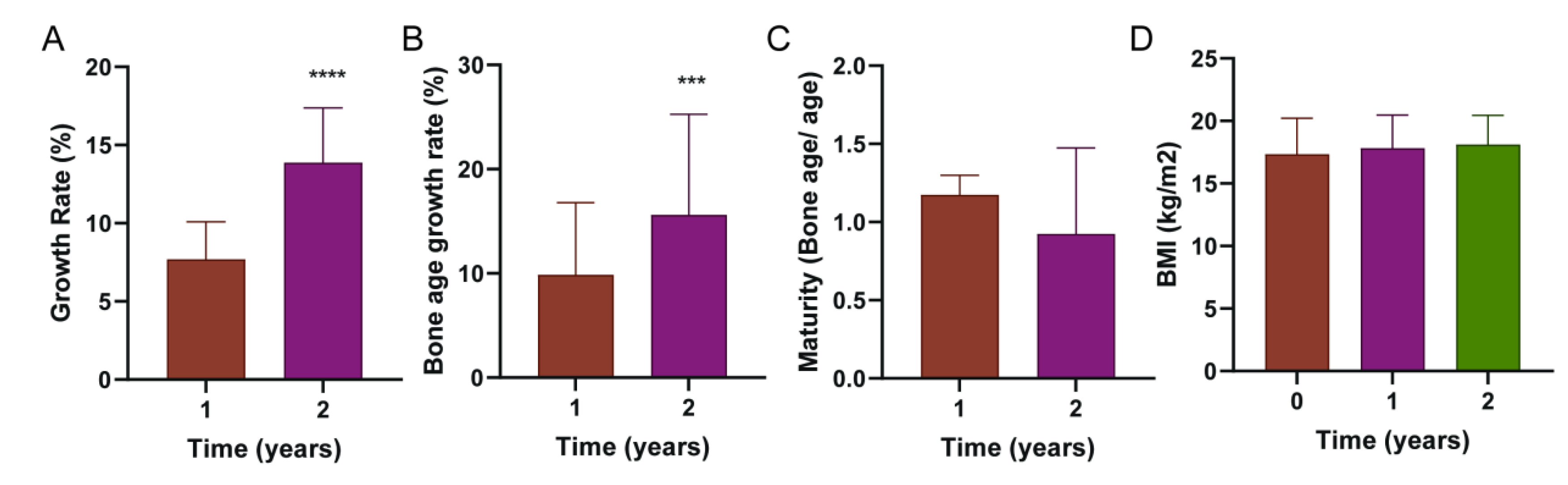
3.1.2. Comparative Analysis of the Serum Sex Hormones and Growth Indexes in the Three Participant Groups After 1 Year
3.1.3. Comparative Analysis of the Serum Sex Hormones and Growth Indexes in the Three Participant Groups After 2 Years
3.1.4. Comparative Analysis of the Serum Sex Hormones
4. Discussion
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zevin, EL; Eugster, EA. Central precocious puberty: a review of diagnosis, treatment, and outcomes. Lancet Child Adolesc Health 2023, 7(12), 886–896. [Google Scholar] [CrossRef]
- Soriano-Guillén, L; Argente, J. Pubertad precoz central: aspectos epidemiológicos, etiológicos y diagnóstico-terapéuticos Central precocious puberty: epidemiology, etiology, diagnosis and treatment. Ann Paediatr (Barc);Spanish 2011, 74(5), 336.e1–336.e13. [Google Scholar] [CrossRef] [PubMed]
- Kim, EY; Lee, MI. Psychosocial aspects in girls with idiopathic precocious puberty. Psychiatry Investig. 2012, 9(1), 25–28. [Google Scholar] [CrossRef]
- Pasquino, AM; Pucarelli, I; Accardo, F; et al. Long-term observation of 87 girls with idiopathic central precocious puberty treated with gonadotropin-releasing hormone analogs: impact on adult height, body mass index, bone mineral content, and reproductive function. J Clin Endocrinol Metab. 2008, 93(1), 190–195. [Google Scholar] [CrossRef] [PubMed]
- Park, J; Kim, JH. Change in body mass index and insulin resistance after 1-year treatment with gonadotropin-releasing hormone agonists in girls with central precocious puberty. Ann Pediatr Endocrinol Metab 2017, 22(1), 27–35. [Google Scholar] [CrossRef]
- Poomthavorn, P; Suphasit, R; Mahachoklertwattana, P. Adult height, body mass index and time of menarche of girls with idiopathic central precocious puberty after gonadotropin releasing hormone analogue treatment. Gynecol Endocrinol 2011, 27(8), 524–528. [Google Scholar] [CrossRef] [PubMed]
- Klein, KO; Mauras, N; Nayak, S; et al. Efficacy and safety of leuprolide acetate 6-month depot for the treatment of central precocious puberty: a phase 3 study. J Endocr Soc. 2023, 7(7), bvad071. [Google Scholar] [CrossRef]
- Vatopoulou, A; Roos, E; Daniilidis, A; Dinas, K. Long-term effects of treatment of central precocious puberty with gonadotropin-releasing hormone analogs every three months. Gynecol Endocrinol 2020, 36(12), 1124–1126. [Google Scholar] [CrossRef]
- Popovic, J; Geffner, ME; Rogol, AD; et al. Gonadotropin-releasing hormone analog therapies for children with central precocious puberty in the United States. Front Pediatr 2022, 10, 968485. [Google Scholar] [CrossRef]
- Lin, WD; Wang, CH; Tsai, FJ. Genetic screening of the makorin ring finger 3 gene in girls with idiopathic central precocious puberty. Clin Chem Lab Med. 2016, 54(3), e93–e96. [Google Scholar] [CrossRef]
- Yoo, E; Kim, S; Jung, HL; et al. Impact of 6-month triptorelin formulation on predicted adult height and basal gonadotropin levels in patients with central precocious puberty. Front Endocrinol (Lausanne) 2023, 14, 1134977. [Google Scholar] [CrossRef]
- Micangeli, G; Paparella, R; Tarani, F; et al. Clinical management and therapy of precocious puberty in the Sapienza university pediatrics hospital of Rome, Italy. Children (Basel) 2023, 10(10), 1672. [Google Scholar] [CrossRef]
- Dong, Y; Dai, L; Dong, Y; et al. Analysis of risk factors of precocious puberty in children. BMC Pediatr 2023, 23(1), 456. [Google Scholar]
- Choe, Y; Cha, JH; Kim, YJ; et al. Rapid weight gain in early life is associated with central precocious puberty in girls, not in boys - a nationwide population-based study in Korea. Front Endocrinol (Lausanne) 2023, 14, 1210995. [Google Scholar] [CrossRef]
- Benedetto, M; Riveros, V; Eymann, A; Terrasa, S; Alonso, G. Analysis of the incidence of central precocious puberty treated with gonadotropin-releasing hormone analogs. Impact of the COVID-19 pandemic. Arch Argent Pediatr 2023, 121(3), e202202849. [Google Scholar] [PubMed]
- Jang, S; Kim, SJ; Lee, M; et al. Comparison of the effect of gonadotropin-releasing hormone agonist dosage in girls with central precocious puberty. Ann Pediatr Endocrinol Metab 2023, 28(4), 283–288. [Google Scholar] [CrossRef] [PubMed]
- Cipolla, C; Sodero, G; Pane, LC; et al. Auxological and metabolic parameters of children undergoing the gonadotropin-releasing hormone stimulation test: correlations with the final diagnosis of central precocious puberty in a single-center study. Biomedicines 2023, 11(6), 1678. [Google Scholar] [CrossRef] [PubMed]
- Bangalore Krishna, KK; Fuqua, JS; Rogol, AD; et al. Use of gonadotropin-releasing hormone analogs in children: update by an international consortium. Horm Res Paediatr. 2019, 91(6), 357–372. [Google Scholar] [CrossRef]
- Yang, J; Song, Q; Gao, S; et al. Efficacy of leuprorelin 3-month depot (11.25 mg) compared to 1-month depot (3.75 mg) for central precocious puberty in Chinese girls: A prospective cohort study. Int J Endocrinol 2022, 2022, 1043293. [Google Scholar] [CrossRef]
- Dotremont, H; France, A; Heinrichs, C; et al. Efficacy and safety of a 4-year combination therapy of growth hormone and gonadotropin-releasing hormone analogue in pubertal girls with short predicted adult height. Front Endocrinol (Lausanne) 2023, 14, 1113750. [Google Scholar] [CrossRef]
- Soliman, AT; Alaaraj, N; De Sanctis, V; et al. Long-term health consequences of central precocious/early puberty (CPP) and treatment with Gn-RH analogue: a short update. Acta Biomed. 2023, 94(6), e2023222. [Google Scholar] [PubMed]
- Valenzise, M; Nasso, C; Scarfone, A; et al. Leuprolide and triptorelin treatment in children with idiopathic central precocious puberty: an efficacy/tolerability comparison study. Front Pediatr 2023, 11, 1170025. [Google Scholar] [CrossRef] [PubMed]
- Yang, EH; Jo, HY; Park, SJ; et al. Effect of gonadotropin-releasing hormone agonist treatment on near final height in girls with central precocious puberty and early puberty. Ann Pediatr Endocrinol Metab 2023, 28(1), 49–53. [Google Scholar] [CrossRef] [PubMed]
- Luo, X; Zhang, C; Yang, Y; et al. Efficacy and safety of triptorelin 3-month formulation in Chinese children with central precocious puberty: A Phase 3, open-label, single-arm study. Adv Ther. 2023, 40(10), 4574–4588. [Google Scholar] [CrossRef]
| Characteristic | N | Overall, N = 601 |
Triptorelingroup , N = 161 |
Leuprolide group, N = 181 | Mixed group, N = 261 |
p-value2 |
|---|---|---|---|---|---|---|
| Age | 60 | 9.00(8.00,10.00) | 8.50(8.00,10.00) | 10.00(8.00,10.75) | 9.00(8.00,9.75) | 0.39 |
| Sex | 60 | 0.34 | ||||
| Female | 52 (87%) | 14 (88%) | 14 (78%) | 24 (92%) | ||
| Male | 8 (13%) | 2 (13%) | 4 (22%) | 2 (7.7%) | ||
| Birth height | 60 | 50.00(49.00,50.00) | 50.00(49.75,50.00) | 50.00(49.25,50.00) | 50.00(49.25,50.00) | 0.89 |
| Birth weight | 60 | 3.25(2.95,3.46) | 3.20(2.94,3.33) | 3.15(2.85,3.43) | 3.35(3.00,3.50) | 0.37 |
| Height | 60 | 137(7) | 136(6) | 140(8) | 136(6) | 0.09 |
| Weight | 60 | 33(7) | 32(6) | 34(7) | 33(8) | 0.69 |
| BMI | 60 | 17.27(15.09,18.69) | 16.63(15.01,19.19) | 17.57(15.35,17.98) | 17.11(15.13,18.73) | 0.92 |
| BA | 60 | 10.57(1.32) | 10.30(1.61) | 11.04(1.35) | 10.42(1.04) | 0.20 |
| 25-OHD | 60 | 23.9(6.2) | 23.1(5.3) | 23.0(5.7) | 25.0(6.9) | 0.46 |
| Ca | 60 | 2.53(0.09) | 2.51(0.07) | 2.54(0.09) | 2.53(0.09) | 0.36 |
| TSH | 59 | 1.97(1.16,2.45) | 2.01(1.58,2.43) | 1.68(0.85,2.95) | 1.91(1.26,2.32) | 0.88 |
| T4 | 60 | 108(98,121) | 119(99,125) | 108(98,118) | 108(99,118) | 0.60 |
| ACTH | 60 | 11(8,18) | 15(7,19) | 11(10,21) | 10(8,15) | 0.68 |
| LH | 60 | 0.98(0.47,1.91) | 1.17(0.51,2.00) | 1.30(0.79,1.99) | 0.62(0.31,1.55) | 0.07 |
| Fsh | 60 | 4.42(2.83,6.31) | 5.17(4.12,5.66) | 4.25(2.86,6.53) | 4.17(2.71,6.13) | 0.58 |
| PRL | 60 | 168(134,235) | 166(132,215) | 195(136,271) | 164(135,236) | 0.59 |
| Testosterone | 57 | 0.80(0.41,1.50) | 0.58(0.30,1.12) | 1.32(0.73,2.28) | 0.80(0.40,1.15) | 0.04 |
| E2 | 57 | 86(52,135) | 104(74,132) | 87(53,183) | 74(39,124) | 0.26 |
| Characteristic | N | Overall, N = 601 |
Triptorelin group, N = 161 |
Leuprolide group, N = 181 |
Mixed group, N = 261 |
p-value2 |
|---|---|---|---|---|---|---|
| LH- one year | 50 | 0.29(0.21,0.60) | 0.29(0.24,0.36) | 0.38(0.21,0.79) | 0.29(0.19,0.66) | 0.88 |
| LH- two year | 33 | 0.98(1.23) | 0.43(0.28) | 1.44(1.66) | 0.88(1.04) | 0.23 |
| Fsh- one year | 50 | 1.57(1.10,2.24) | 1.28(0.82,1.79) | 1.53(1.13,2.07) | 1.88(1.28,2.82) | 0.18 |
| Fsh- two year | 33 | 2.71(2.08) | 1.87(1.31) | 2.47(1.75) | 3.18(2.46) | 0.40 |
| Testosterone-one year | 48 | 0.76(0.42,1.15) | 0.65(0.37,1.18) | 0.85(0.55,1.13) | 0.73(0.25,1.10) | 0.61 |
| Testosterone- two year | 29 | 0.91(0.58) | 1.20(0.64) | 0.82(0.48) | 0.87(0.63) | 0.57 |
| E2-one year | 44 | 28(11,43) | 24(9,54) | 23(13,32) | 34(14,48) | 0.59 |
| E2- two year | 29 | 49(54) | 57(75) | 69(72) | 31(16) | 0.81 |
| Height -one year | 60 | 144.9(6.5) | 144.3(6.0) | 147.5(7.6) | 143.5(5.7) | 0.083 |
| Height- two year | 44 | 150(6) | 149(8) | 153(7) | 149(5) | 0.28 |
| Weight-one year | 60 | 38(7) | 37(7) | 39(7) | 37(8) | 0.73 |
| Weight- two year | 44 | 41(7) | 43(7) | 41(6) | 40(7) | 0.48 |
| BMI- one year | 60 | 17.82(2.65) | 17.91(2.87) | 17.61(2.09) | 17.92(2.94) | 0.89 |
| BMI- two year | 44 | 18.11(2.35) | 19.31(1.90) | 17.48(1.64) | 18.04(2.82) | 0.12 |
| BA-one year | 60 | 11.55(1.01) | 11.36(1.25) | 11.92(1.04) | 11.40(0.77) | 0.17 |
| BA- two year | 45 | 12.10(0.89) | 11.81(0.96) | 12.39(0.92) | 11.99(0.81) | 0.17 |
| Growth rate-one year | 60 | 7.20(6.28,9.13) | 7.15(6.48,9.25) | 7.40(6.55,8.30) | 6.65(6.00,9.18) | 0.63 |
| Growth rate- two year | 44 | 13.9(3.5) | 13.8(2.8) | 13.5(4.3) | 14.2(3.2) | 0.62 |
| Maturity3-one year | 60 | 1.18(1.08,1.26) | 1.13(1.08,1.29) | 1.17(1.10,1.21) | 1.20(1.06,1.26) | 0.96 |
| Maturity3- two year | 60 | 0.92(0.55) | 0.70(0.65) | 1.09(0.42) | 0.95(0.54) | 0.59 |
| GBA4-one year | 60 | 9(4,14) | 9(5,15) | 7(4,12) | 9(4,15) | 0.65 |
| GBA4- two year | 45 | 16(10) | 14(7) | 14(11) | 17(10) | 0.46 |
| Characteristic | N | Overall, N = 601 |
Triptorelin group, N = 161 |
Leuprolide group, N = 181 |
Mixed,group, N = 261 |
p-value2 |
|---|---|---|---|---|---|---|
| LH- one year | 33 | -0.52(-1.25,-0.11) | -1.39(-2.59,-0.32) | -0.70(-1.18,-0.34) | -0.19(-0.74,-0.04) | 0.27 |
| LH- two year | 33 | -0.38(-0.90,0.60) | -1.42(-2.42,-0.42) | -0.20(-0.75,0.24) | -0.24(-0.65,0.71) | 0.28 |
| Fsh- one year | 33 | -2.17(-4.35,-0.83) | -2.30(-3.03,-2.00) | -3.04(-4.48,-0.48) | -1.53(-4.34,-0.82) | 0.91 |
| Fsh- two year | 33 | -1.67(-3.87,0.24) | -2.34(-3.13,-1.76) | -1.59(-3.84,0.32) | -1.57(-3.95,0.78) | 0.81 |
| Testosterone-one year | 28 | -0.07(-0.42,0.28) | 0.01(-0.02,0.27) | -0.28(-0.47,-0.18) | 0.09(-0.27,0.44) | 0.20 |
| Testosterone- two year | 28 | 0.23(-0.31,0.41) | 0.38(0.26,0.38) | -0.24(-0.80,0.01) | 0.30(-0.21,0.48) | 0.20 |
| E2- one year | 25 | -32(-85,-12) | -97(-118,-86) | -24(-77,-12) | -29(-71,-17) | 0.23 |
| E2- two year | 25 | -53(-91,26) | -115(-131,-84) | -14(-75,37) | -46(-59,-3) | 0.085 |
| Growth- one year | 60 | 7.20(6.28,9.13) | 7.15(6.48,9.25) | 7.40(6.55,8.30) | 6.65(6.00,9.18) | 0.63 |
| Growth- two year | 44 | 13.6(11.9,15.3) | 14.2(13.3,15.6) | 13.3(10.4,14.8) | 13.5(12.6,15.0) | 0.62 |
| Mature- one year | 60 | 1.18(1.08,1.26) | 1.13(1.08,1.29) | 1.17(1.10,1.21) | 1.20(1.06,1.26) | 0.96 |
| Mature- two year | 60 | 1.18(0.74,1.24) | 1.02(0.00,1.30) | 1.16(1.07,1.27) | 1.21(1.10,1.23) | 0.59 |
| BMI- one year | 44 | 0.43(-0.47,1.44) | 1.32(0.22,1.71) | 0.71(-0.16,1.26) | 0.02(-0.90,0.99) | 0.28 |
| BMI- two year | 44 | 0.82(-0.21,1.65) | 1.49(-0.14,1.71) | 0.68(-0.25,1.27) | 0.72(-0.01,1.32) | 0.81 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




