Submitted:
23 February 2026
Posted:
25 February 2026
You are already at the latest version
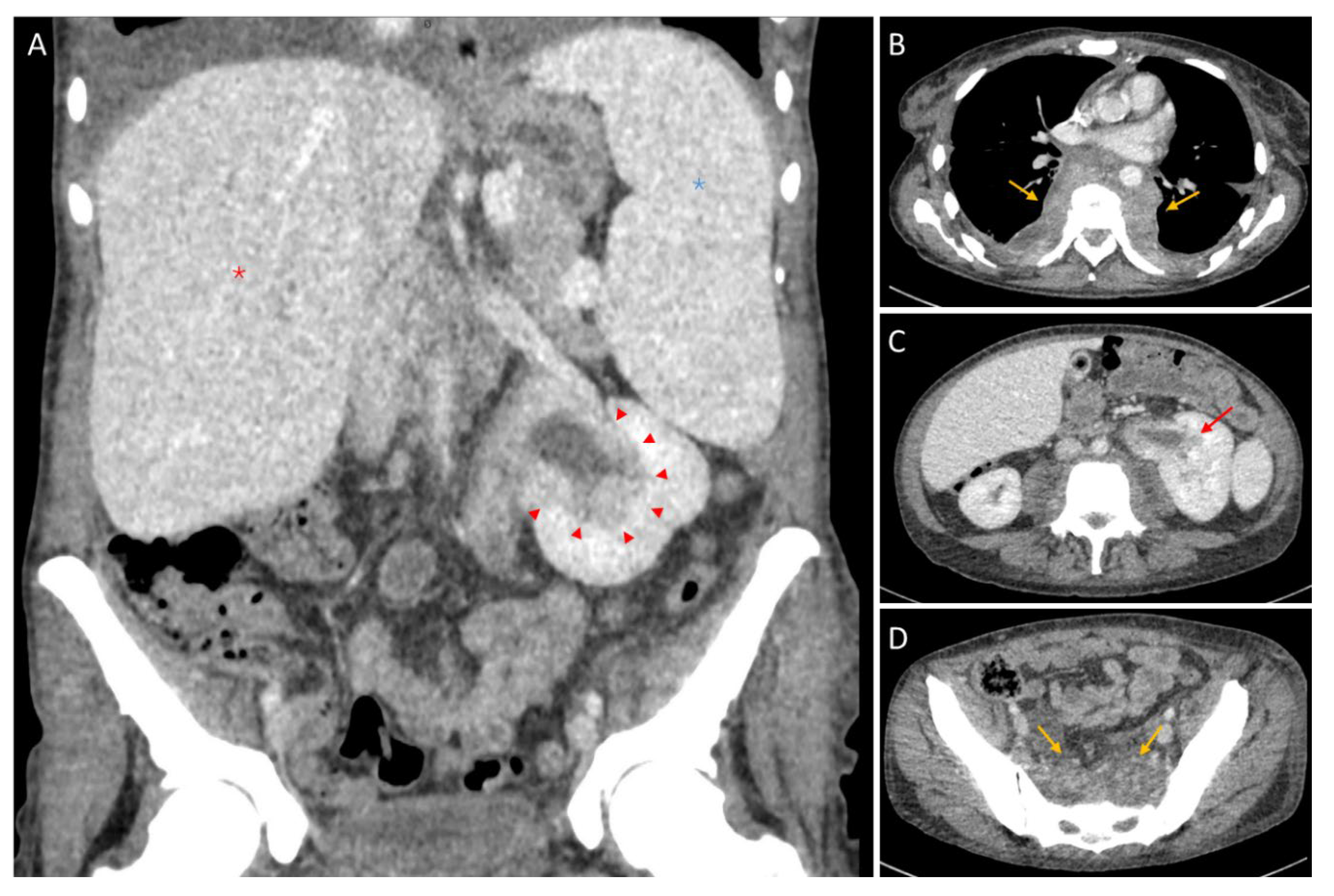
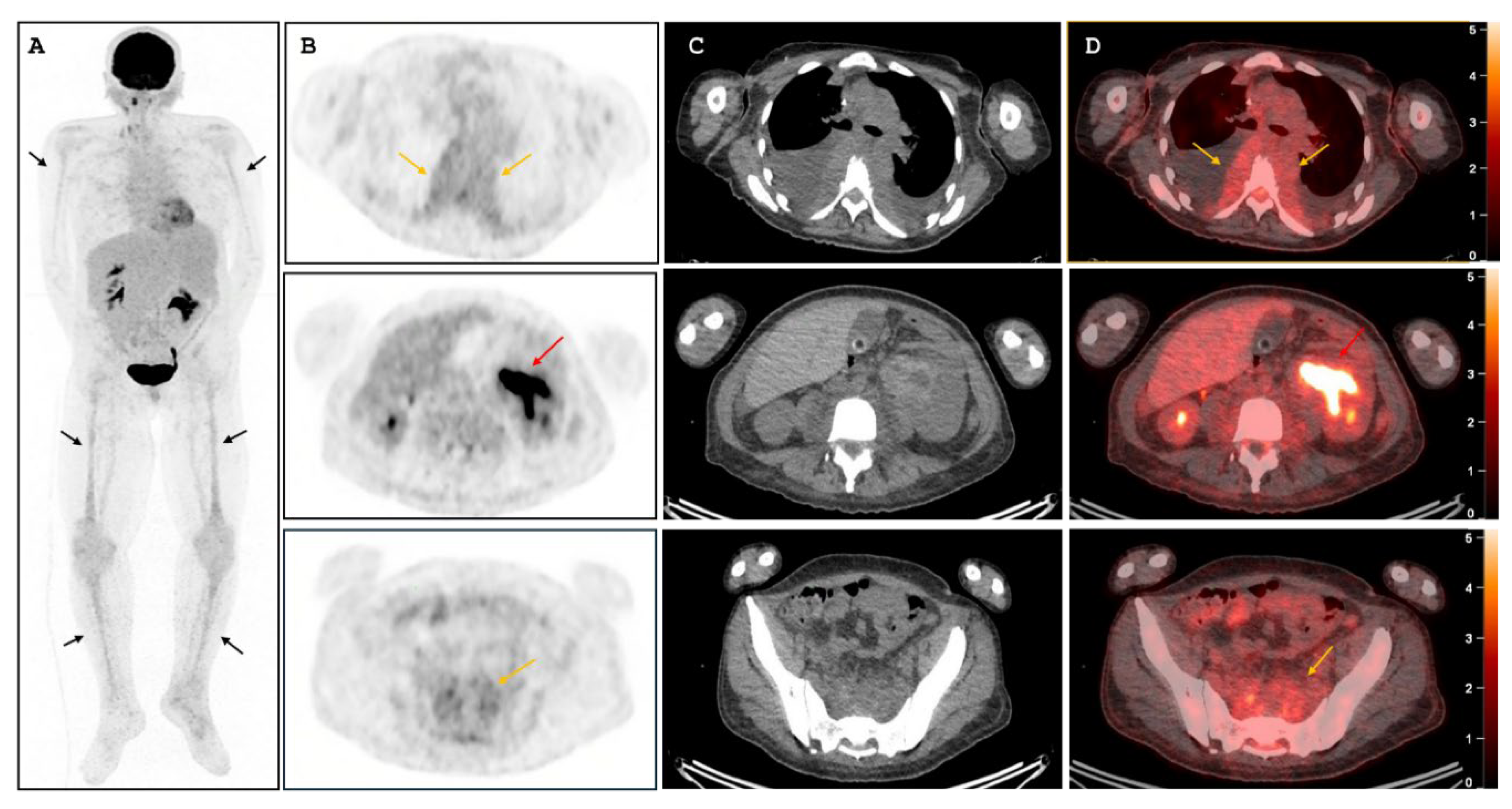
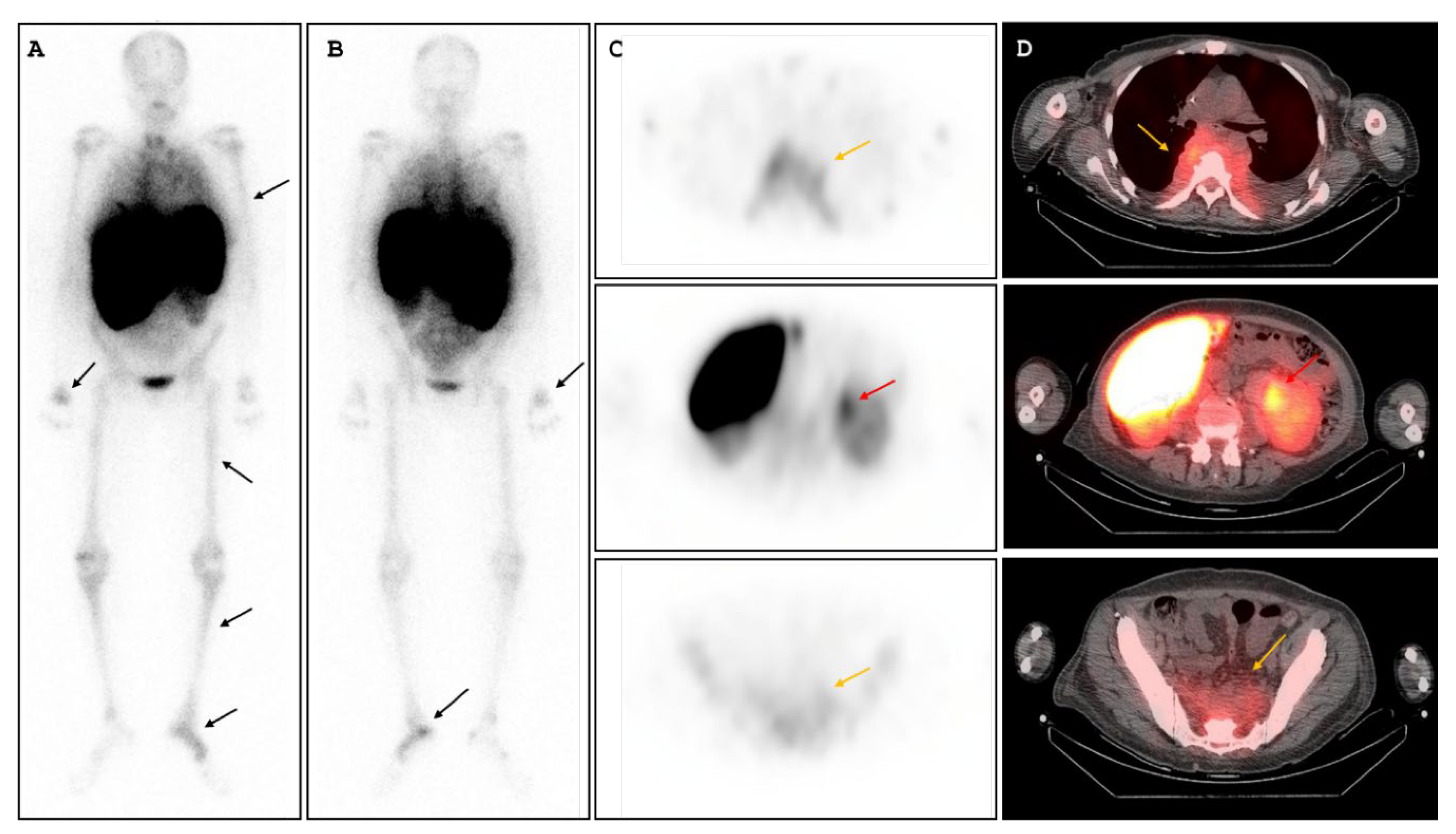
Abstract
Keywords:
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Malla S, Razik A, Das CJ, Naranje P, Kandasamy D, Kumar R. Marrow outside marrow: imaging of extramedullary haematopoiesis. Clin Radiol. 2020;75(8):565-578. [CrossRef]
- Roberts AS, Shetty AS, Mellnick VM, Pickhardt PJ, Bhalla S, Menias CO. Extramedullary haematopoiesis: radiological imaging features. Clin Radiol. 2016;71(9):807-814. [CrossRef]
- Gryspeerdt S, Oyen R, Van Hoe L, Baert AL, Boogaerts M. Extramedullary hematopoiesis encasing the pelvicalyceal system: CT findings. Ann Hematol. 1995;71(1):53-56. [CrossRef]
- La Fianza A, Torretta L, Spinazzola A. Extramedullary hematopoiesis in chronic myelofibrosis encasing the pelvicaliceal system and perirenal spaces: CT findings. Urol Int. 2005;75(3):281-284. [CrossRef]
- Galperin-Aizenberg M, Volchek Y, Even-Sapir E, Vasserman M, Maizlin ZV, Cohenpour M, et al. Renal extramedullary haematopoiesis mimicking renal lymphoma on computed tomography. Clin Radiol. 2006;61(10):896-898. [CrossRef]
- Boellaard R, Delgado-Bolton R, Oyen WJG, Giammarile F, Tatsch K, Eschner W, et al. FDG PET/CT: EANM procedure guidelines for tumour imaging: version 2.0. Eur J Nucl Med Mol Imaging. 2015;42(2):328-354. [CrossRef]
- Van Son PM, Evers D, Aarntzen EHJG. The value of FDG-PET CT scans to evaluate bone marrow in haemato-oncological conditions. Neth J Med. 2019;77(7):234-242.
- Dahlsgaard-Wallenius SE, Juul-Jensen K, Nielsen AL, Hildebrandt MG. Extramedullary hematopoiesis visualized on FDG-PET/CT in a patient with beta-thalassemia. Nucl Med Mol Imaging. 2022;56(6):328-330. [CrossRef]
- Yang M, Roarke M, Kelly J, et al. Diffuse pulmonary extramedullary hematopoiesis in myelofibrosis diagnosed with technetium-99m sulfur colloid bone marrow scintigraphy and single photon emission computerized tomography/CT. Am J Hematol. 2017;92(3):323-324. [CrossRef]
- Ojeda-Uribe M, Morel O, Ungureanu C, et al. Assessment of sites of marrow and extramedullary hematopoiesis by hybrid imaging in patients with primary myelofibrosis. Cancer Med. 2016;5(9):2378-2384. [CrossRef]
- Zul Khairul Azwadi I, Norhayati MN, Abdullah MS. Percutaneous nephrostomy versus retrograde ureteral stenting for acute upper obstructive uropathy: a systematic review and meta-analysis. Sci Rep. 2021 Mar 23;11(1):6613. [CrossRef]
- Townsley DM, Scheinberg P, Winkler T, et al. Eltrombopag added to standard immunosuppression for aplastic anemia. N Engl J Med. 2017 Apr 20;376(16):1540-1550. [CrossRef]
- Lee JW, Lee SE, Jung CW, et al. Romiplostim in patients with refractory aplastic anaemia previously treated with immunosuppressive therapy: a dose-finding and long-term treatment phase 2 trial. Lancet Haematol. 2019 Nov;6(11):e562-e572. [CrossRef]
- Kröger N, Bacigalupo A, Barbui T, et al. Indication and management of allogeneic haematopoietic stem-cell transplantation in myelofibrosis: updated recommendations by the EBMT/ELN International Working Group. Lancet Haematol. 2024 Jan;11(1):e62-e74. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




