Submitted:
23 February 2026
Posted:
24 February 2026
You are already at the latest version
Abstract
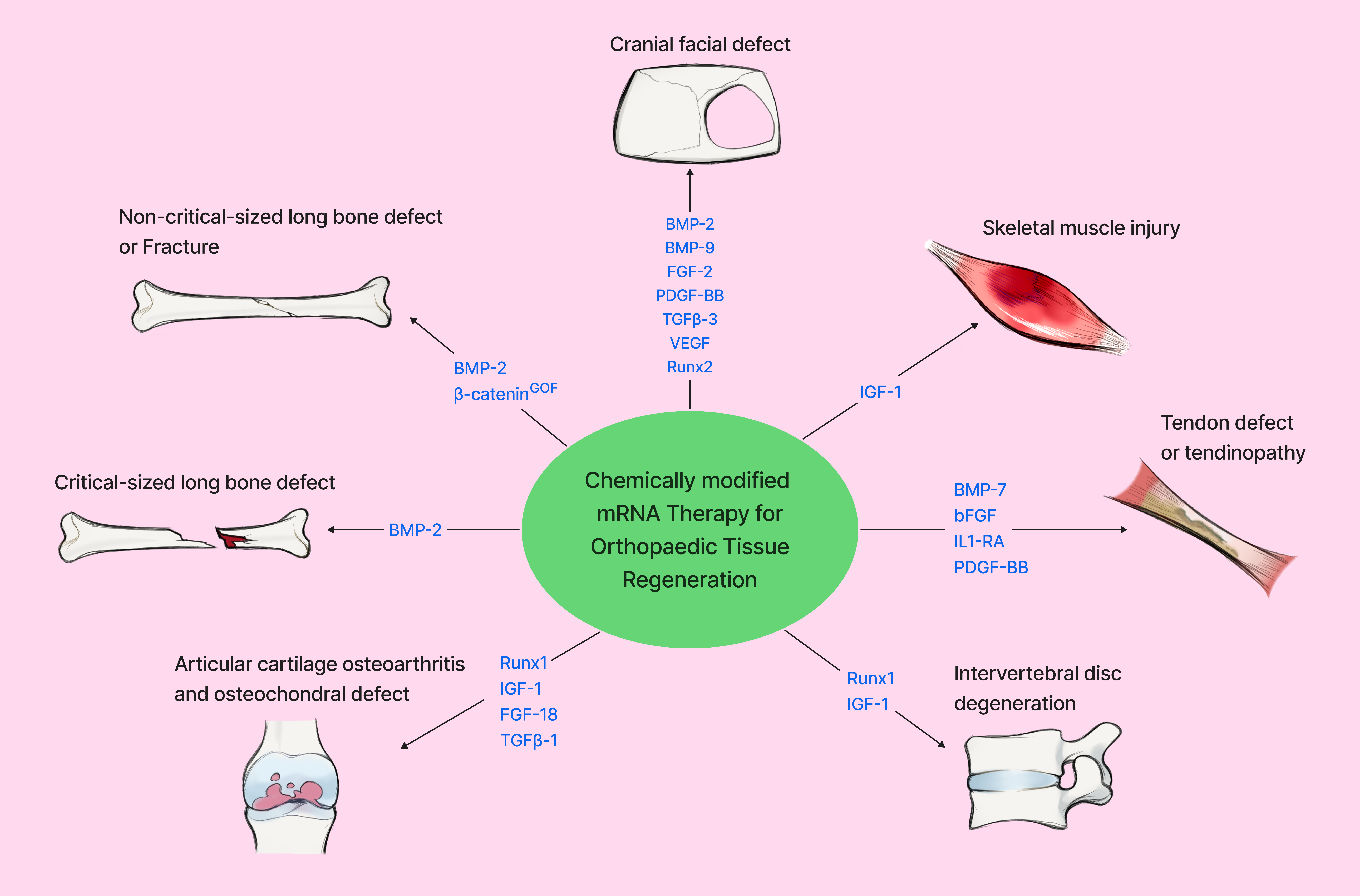
Keywords:
1. Introduction
2. Mrna for Bone Regeneration
2.1. Mrna-Based Therapeutic Strategies for Craniomaxillofacial Bone Regeneration
2.1.1. Bmp-Encoding Mrna Delivery for Calvarial Bone Regeneration
2.1.2. Cmrna Encoding Other Angiogenic and Osteogenic Genes for Craniofacial Bone Repair
Co-Delivery of BMP mRNA with Additional Growth Factor mRNAs for Craniofacial Bone Defect Repair
cmRNA Encoding RUNX2 and VEGF for Craniofacial Bone Defect Repair
2.2. Mrna Therapy for the Repair of Long Bone Defects
2.2.1. Cmrna for Repair of Non-Critical Sized Long Bone Defects or Fractures
2.2.2. Mrna Therapy for Repair of Critical Sized Segmental Long Bone Defects
3. Mrna Therapy for Tendon Regeneration and Healing
4. Mrna Therapy for the Regeneration of Articular Cartilage
4.1. Mrna for Traumatic Osteoarthritis Repair
4.2. Cmrna for Osteochondral Defect Repair
5. Mrna for Intervertebral Disc Regeneration
6. Mrna Therapy for Skeletal Muscle Injury and Repair
7. Advantages and Disadvantages of Protein Encoding Mrna Therapy for Musculoskeletal Tissue Repair
8. Perspective
9. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data
Acknowledgments
Conflicts: of Interest
Declaration of using AI for generating manuscript
References
- Palazzo, C.; Ravaud, J. F.; Papelard, A.; Ravaud, P.; Poiraudeau, S. The burden of musculoskeletal conditions. PLoS One 2014, 9((3)), e90633. [Google Scholar] [CrossRef]
- Ehlen, Q. T.; Costello, J. P., 2nd; Mirsky, N. A.; Slavin, B. V.; Parra, M.; Ptashnik, A.; Nayak, V. V.; Coelho, P. G.; Witek, L. Treatment of Bone Defects and Nonunion via Novel Delivery Mechanisms, Growth Factors, and Stem Cells: A Review. ACS Biomater Sci Eng 2024, 10((12)), 7314–7336. [Google Scholar] [CrossRef]
- Gao, X.; Ruzbarsky, J. J.; Layne, J. E.; Xiao, X.; Huard, J. Stem Cells and Bone Tissue Engineering. Life (Basel) 2024, 14((3)). [Google Scholar] [CrossRef]
- Deng, Z.; Li, Y.; Gao, X.; Lei, G.; Huard, J. Bone morphogenetic proteins for articular cartilage regeneration. Osteoarthr. Cartil. 2018, 26, 1153–1161. [Google Scholar] [CrossRef]
- Gillman, C.E.; Jayasuriya, A.C. FDA-approved bone grafts and bone graft substitute devices in bone regeneration. Mater. Sci. Eng. C 2021, 130, 112466–112466. [Google Scholar] [CrossRef]
- Hsu, W.K.; Wang, J.C. The use of bone morphogenetic protein in spine fusion. Spine J. 2008, 8, 419–425. [Google Scholar] [CrossRef] [PubMed]
- Watson-Levings, R.S.; Palmer, G.D.; Levings, P.P.; Dacanay, E.A.; Evans, C.H.; Ghivizzani, S.C. Gene Therapy in Orthopaedics: Progress and Challenges in Pre-Clinical Development and Translation. Front. Bioeng. Biotechnol. 2022, 10, 901317. [Google Scholar] [CrossRef] [PubMed]
- Tierney, E. G.; Duffy, G. P.; Cryan, S. A.; Curtin, C. M.; O'Brien, F. J. Non-viral gene-activated matrices: next generation constructs for bone repair. Organogenesis 2013, 9((1)), 22–8. [Google Scholar] [CrossRef]
- De la Vega, R.E.; Atasoy-Zeybek, A.; Panos, J.A.; Van Griensven, M.; Evans, C.H.; Balmayor, E.R. Gene therapy for bone healing: lessons learned and new approaches. Transl. Res. 2021, 236, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Chu, X.; Xiong, Y.; Lu, L.; Wang, Y.; Wang, J.; Zeng, R.; Hu, L.; Yan, C.; Zhao, Z.; Lin, S.; et al. Research progress of gene therapy combined with tissue engineering to promote bone regeneration. APL Bioeng. 2024, 8, 031502. [Google Scholar] [CrossRef]
- Ge, H.; Shi, Z.; Liu, C.; Lu, J.; Yao, Y.; Cheng, B. Viral and non-viral vectors for gene therapy in the treatment of bone-related disorders: molecular insights and clinical perspectives. Mol. Asp. Med. 2025, 105, 101400. [Google Scholar] [CrossRef]
- Mancino, C.; Franke, M.; Greco, A.; Sontam, T.; Mcculloch, P.; Corbo, C.; Taraballi, F. RNA therapies for musculoskeletal conditions. J. Control. Release 2024, 377, 756–766. [Google Scholar] [CrossRef] [PubMed]
- Surisaeng, T.; Wisitrasameewong, W.; Champaiboon, C.; Sa-Ard-Iam, N.; Chanamuangkon, T.; Thongnuek, P.; Tam, Y.K.; Muramatsu, H.; Weissman, D.; Pardi, N.; et al. BMP-2 mRNA-transfected BMSCs promote superior calvarial bone regeneration. Sci. Rep. 2025, 15, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Dimitriou, R.; Jones, E.; McGonagle, D.; Giannoudis, P.V. Bone regeneration: current concepts and future directions. BMC Med. 2011, 9, 66–66. [Google Scholar] [CrossRef] [PubMed]
- Jagadale, S.; Damle, M.; Joshi, M.G. Bone Tissue Engineering: From Biomaterials to Clinical Trials. Adv Exp Med Biol 2025, 1479, 73–115. [Google Scholar]
- Santos, W.; Benito-Ramal, E.; Toledano-Serrabona, J.; Sánchez-Garcés, M. Efficacy of bone morphogenetic protein in comparison with autogenous bone in regeneration of ameloblastoma bone defects. A systematic review. Med. Oral Patol. Oral Y Cirugia Bucal 2024, 29, e782–e790. [Google Scholar] [CrossRef]
- Gao, X.; Hwang, M.P.; Wright, N.; Lu, A.; Ruzbarsky, J.J.; Huard, M.; Cheng, H.; Mullen, M.; Ravuri, S.; Wang, B.; et al. The use of heparin/polycation coacervate sustain release system to compare the bone regenerative potentials of 5 BMPs using a critical sized calvarial bone defect model. Biomaterials 2022, 288, 121708–121708. [Google Scholar] [CrossRef]
- Schoonraad, S.A.; Jaimes, A.A.; Singh, A.J.; Croland, K.J.; Bryant, S.J. Osteogenic effects of covalently tethered rhBMP-2 and rhBMP-9 in an MMP-sensitive PEG hydrogel nanocomposite. Acta Biomater. 2023, 170, 53–67. [Google Scholar] [CrossRef] [PubMed]
- Elangovan, S.; Khorsand, B.; Do, A.-V.; Hong, L.; Dewerth, A.; Kormann, M.; Ross, R.D.; Sumner, D.R.; Allamargot, C.; Salem, A.K. Chemically modified RNA activated matrices enhance bone regeneration. J. Control. Release 2015, 218, 22–28. [Google Scholar] [CrossRef]
- Khorsand, B.; Elangovan, S.; Hong, L.; Kormann, M.S.D.; Salem, A.K. A bioactive collagen membrane that enhances bone regeneration. J. Biomed. Mater. Res. Part B: Appl. Biomater. 2018, 107, 1824–1832. [Google Scholar] [CrossRef]
- Khorsand, B.; Elangovan, S.; Hong, L.; Dewerth, A.; Kormann, M.S.D.; Salem, A.K. A Comparative Study of the Bone Regenerative Effect of Chemically Modified RNA Encoding BMP-2 or BMP-9. AAPS J. 2017, 19, 438–446. [Google Scholar] [CrossRef]
- Geng, Y.; Duan, H.; Xu, L.; Witman, N.; Yan, B.; Yu, Z.; Wang, H.; Tan, Y.; Lin, L.; Li, D.; et al. BMP-2 and VEGF-A modRNAs in collagen scaffold synergistically drive bone repair through osteogenic and angiogenic pathways. Commun. Biol. 2021, 4, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Tsou, H.-K.; Wu, C.-H.; Chan, L.Y.; Kataoka, K.; Itokazu, N.; Tsuzuki, M.; Hu, H.; Zhuo, G.-Y.; Itaka, K.; Lin, C.-Y. Administration of mRNA-Nanomedicine-Augmented Calvarial Defect Healing via Endochondral Ossification. Pharmaceutics 2023, 15, 1965. [Google Scholar] [CrossRef] [PubMed]
- Friedlaender, G.E.; Lin, S.; Solchaga, L.A.; Snel, L.B.; Lynch, S.E. The Role of Recombinant Human Platelet-derived Growth Factor-BB (rhPDGF-BB) in Orthopaedic Bone Repair and Regeneration. Curr. Pharm. Des. 2013, 19, 3384–3390. [Google Scholar] [CrossRef] [PubMed]
- Jia, T.; Jacquet, T.; Dalonneau, F.; Coudert, P.; Vaganay, E.; Exbrayat-Heritier, C.; Vollaire, J.; Josserand, V.; Ruggiero, F.; Coll, J. L.; Eymin, B. FGF-2 promotes angiogenesis through a SRSF1/SRSF3/SRPK1-dependent axis that controls VEGFR1 splicing in endothelial cells. BMC Biol 2021, 19((1)), 173. [Google Scholar] [CrossRef]
- Zhang, M.; Fukushima, Y.; Nozaki, K.; Nakanishi, H.; Deng, J.; Wakabayashi, N.; Itaka, K. Enhancement of bone regeneration by coadministration of angiogenic and osteogenic factors using messenger RNA. Inflamm. Regen. 2023, 43, 1–16. [Google Scholar] [CrossRef]
- Marsell, R.; Einhorn, T. A. The biology of fracture healing. Injury 2011, 42((6)), 551–5. [Google Scholar] [CrossRef]
- Nicholson, J.; Makaram, N.; Simpson, A.; Keating, J. Fracture nonunion in long bones: A literature review of risk factors and surgical management. Injury 2021, 52, S3–S11. [Google Scholar] [CrossRef]
- Balmayor, E.R.; Geiger, J.P.; Aneja, M.K.; Berezhanskyy, T.; Utzinger, M.; Mykhaylyk, O.; Rudolph, C.; Plank, C. Chemically modified RNA induces osteogenesis of stem cells and human tissue explants as well as accelerates bone healing in rats. Biomaterials 2016, 87, 131–146. [Google Scholar] [CrossRef]
- Badieyan, Z.S.; Berezhanskyy, T.; Utzinger, M.; Aneja, M.K.; Emrich, D.; Erben, R.; Schüler, C.; Altpeter, P.; Ferizi, M.; Hasenpusch, G.; et al. Transcript-activated collagen matrix as sustained mRNA delivery system for bone regeneration. J. Control. Release 2016, 239, 137–148. [Google Scholar] [CrossRef]
- Nelson, A.L.; Mancino, C.; Gao, X.; Choe, J.A.; Chubb, L.; Williams, K.; Czachor, M.; Marcucio, R.; Taraballi, F.; Cooke, J.P.; et al. β-catenin mRNA encapsulated in SM-102 lipid nanoparticles enhances bone formation in a murine tibia fracture repair model. Bioact. Mater. 2024, 39, 273–286. [Google Scholar] [CrossRef]
- Dumic-Cule, I.; Pecina, M.; Jelic, M.; Jankolija, M.; Popek, I.; Grgurevic, L.; Vukicevic, S. Biological aspects of segmental bone defects management. Int. Orthop. 2015, 39, 1005–1011. [Google Scholar] [CrossRef]
- De La Vega, R.E.; van Griensven, M.; Zhang, W.; Coenen, M.J.; Nagelli, C.V.; Panos, J.A.; Silva, C.J.P.; Geiger, J.; Plank, C.; Evans, C.H.; et al. Efficient healing of large osseous segmental defects using optimized chemically modified messenger RNA encoding BMP-2. Sci. Adv. 2022, 8, eabl6242. [Google Scholar] [CrossRef] [PubMed]
- Andarawis-Puri, N.; Flatow, E.L.; Soslowsky, L.J. Tendon basic science: Development, repair, regeneration, and healing. J. Orthop. Res. 2015, 33, 780–784. [Google Scholar] [CrossRef] [PubMed]
- Groth, K.; Berezhanskyy, T.; Aneja, M.; Geiger, J.; Schweizer, M.; Maucksch, L.; Pasewald, T.; Brill, T.; Tigani, B.; Weber, E.; et al. Tendon healing induced by chemically modified mRNAs. 2017, 33, 294–307. [Google Scholar] [CrossRef]
- Herbst, E.; Imhoff, F.B.; Foehr, P.; Milz, S.; Plank, C.; Rudolph, C.; Hasenpusch, G.; Geiger, J.P.; Aneja, M.K.; Groth, K.; et al. Chemically Modified Messenger RNA: Modified RNA Application for Treatment of Achilles Tendon Defects. Tissue Eng. Part A 2019, 25, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Sturm, L.; Schwemberger, B.; Menzel, U.; Hackel, S.; Albers, C. E.; Plank, C.; Rip, J.; Alini, M.; Traweger, A.; Grad, S.; Basoli, V. In Vitro Evaluation of a Nanoparticle-Based mRNA Delivery System for Cells in the Joint. Biomedicines 2021, 9((7)). [Google Scholar] [CrossRef]
- Faustini, B.; Lettner, T.; Wagner, A.; Tempfer, H.; Cesur, N.P.; Lehner, C.; Brouwer, C.; Roelofs, K.; Mykhailyk, O.; Plank, C.; et al. Improved tendon repair with optimized chemically modified mRNAs: Combined delivery of Pdgf-BB and IL-1Ra using injectable nanoparticles. Acta Biomater. 2025, 195, 451–466. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, X.; Li, H.; Zhang, R.; Zhang, T.; Juma, T.; Zhou, Y.; Guo, Q.; Zhao, H.; Cao, Y. Anti-inflammatory therapy for tendinopathy using Il1rn mRNA encapsulated in SM102 lipid nanoparticles. Front. Bioeng. Biotechnol. 2025, 13, 1641236. [Google Scholar] [CrossRef]
- Hunter, D. J.; Bierma-Zeinstra, S. Osteoarthritis. Lancet 2019, 393((10182)), 1745–1759. [Google Scholar] [CrossRef]
- Kong, K.; Li, B.; Chang, Y.; Zhao, C.; Qiao, H.; Jin, M.; Wu, X.; Fan, W.; Wang, L.; Qi, Y.; et al. Delivery of FGF18 using mRNA-LNP protects the cartilage against degeneration via alleviating chondrocyte senescence. J. Nanobiotechnology 2025, 23, 1–21. [Google Scholar] [CrossRef]
- Zhang, W.; Ouyang, H.; Dass, C.R.; Xu, J. Current research on pharmacologic and regenerative therapies for osteoarthritis. Bone Res. 2016, 4, 15040. [Google Scholar] [CrossRef]
- Yano, F.; Ohba, S.; Murahashi, Y.; Tanaka, S.; Saito, T.; Chung, U.-I. Runx1 contributes to articular cartilage maintenance by enhancement of cartilage matrix production and suppression of hypertrophic differentiation. Sci. Rep. 2019, 9, 7666. [Google Scholar] [CrossRef]
- LeBlanc, K.T.; Walcott, M.E.; Gaur, T.; O'COnnell, S.L.; Basil, K.; Tadiri, C.P.; Mason-Savas, A.; Silva, J.A.; van Wijnen, A.J.; Stein, J.L.; et al. Runx1 Activities in Superficial Zone Chondrocytes, Osteoarthritic Chondrocyte Clones and Response to Mechanical Loading. J. Cell. Physiol. 2014, 230, 440–448. [Google Scholar] [CrossRef]
- Zhang, Y.; Zuo, T.; McVicar, A.; Yang, H.-L.; Li, Y.-P.; Chen, W. Runx1 is a key regulator of articular cartilage homeostasis by orchestrating YAP, TGFβ, and Wnt signaling in articular cartilage formation and osteoarthritis. Bone Res. 2022, 10, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Aini, H.; Itaka, K.; Fujisawa, A.; Uchida, H.; Uchida, S.; Fukushima, S.; Kataoka, K.; Saito, T.; Chung, U.-I.; Ohba, S. Messenger RNA delivery of a cartilage-anabolic transcription factor as a disease-modifying strategy for osteoarthritis treatment. Sci. Rep. 2016, 6, srep18743. [Google Scholar] [CrossRef]
- Pezzotti, G.; Zhu, W.; Terai, Y.; Marin, E.; Boschetto, F.; Kawamoto, K.; Itaka, K. Raman spectroscopic insight into osteoarthritic cartilage regeneration by mRNA therapeutics encoding cartilage-anabolic transcription factor Runx1. Mater. Today Bio 2022, 13, 100210. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Peng, Z.; Xu, Y.; Sheng, Z.; Liu, Y.; Liao, Y.; Wang, Y.; Wen, Y.; Yi, J.; Xie, C.; et al. Engineered adipose-derived stem cells with IGF-1-modified mRNA ameliorates osteoarthritis development. Stem Cell Res. Ther. 2022, 13, 1–15. [Google Scholar] [CrossRef]
- Song, Z.; Li, Y.; Shang, C.; Shang, G.; Kou, H.; Li, J.; Chen, S.; Liu, H. Sprifermin: Effects on Cartilage Homeostasis and Therapeutic Prospects in Cartilage-Related Diseases. Front. Cell Dev. Biol. 2021, 9, 786546. [Google Scholar] [CrossRef]
- Huang, K.; Liu, X.; Qin, H.; Li, Y.; Zhu, J.; Yin, B.; Zheng, Q.; Zuo, C.; Cao, H.; Tong, Z.; et al. FGF18 encoding circular mRNA-LNP based on glycerolipid engineering of mesenchymal stem cells for efficient amelioration of osteoarthritis. Biomater. Sci. 2024, 12, 4427–4439. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.; Ma, B.; Pan, Z.; Zhao, Y.; Tian, L.; Fan, Y.; Kong, W.; Wang, J.; Xu, B.; Ao, Y.; et al. Targeted Therapy of Osteoarthritis via Intra-Articular Delivery of Lipid-Nanoparticle-Encapsulated Recombinant Human FGF18 mRNA. Adv. Heal. Mater. 2024, 13, e2400804. [Google Scholar] [CrossRef]
- Plaas, A.; Velasco, J.; Gorski, D.; Li, J.; Cole, A.; Christopherson, K.; Sandy, J. The relationship between fibrogenic TGFβ1 signaling in the joint and cartilage degradation in post-injury osteoarthritis. Osteoarthr. Cartil. 2011, 19, 1081–1090. [Google Scholar] [CrossRef]
- Fontana, G.; Nemke, B.; Lu, Y.; Chamberlain, C.; Lee, J.-S.; Choe, J.A.; Jiao, H.; Nelson, M.; Amitrano, M.; Li, W.-J.; et al. Local delivery of TGF-β1-mRNA decreases fibrosis in osteochondral defects. Bioact. Mater. 2025, 45, 509–519. [Google Scholar] [CrossRef]
- Lin, C.-Y.; Crowley, S.T.; Uchida, S.; Komaki, Y.; Kataoka, K.; Itaka, K. Treatment of Intervertebral Disk Disease by the Administration of mRNA Encoding a Cartilage-Anabolic Transcription Factor. Mol. Ther. - Nucleic Acids 2019, 16, 162–171. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.-C.; Tsou, H.-K.; Chang, H.-H.; Chan, L.Y.; Zhuo, G.-Y.; Maeda, T.; Lin, C.-Y. Runx1 Messenger RNA Delivered by Polyplex Nanomicelles Alleviate Spinal Disc Hydration Loss in a Rat Disc Degeneration Model. Int. J. Mol. Sci. 2022, 23, 565. [Google Scholar] [CrossRef]
- Antony, J.S.; Birrer, P.; Bohnert, C.; Zimmerli, S.; Hillmann, P.; Schaffhauser, H.; Hoeflich, C.; Hoeflich, A.; Khairallah, R.; Satoh, A.T.; et al. Local application of engineered insulin-like growth factor I mRNA demonstrates regenerative therapeutic potential in vivo. Mol. Ther. - Nucleic Acids 2023, 34, 102055. [Google Scholar] [CrossRef]
- Lee, S.S.; Huang, B.J.; Kaltz, S.R.; Sur, S.; Newcomb, C.J.; Stock, S.R.; Shah, R.N.; Stupp, S.I. Bone regeneration with low dose BMP-2 amplified by biomimetic supramolecular nanofibers within collagen scaffolds. Biomaterials 2013, 34, 452–459. [Google Scholar] [CrossRef]
- Matsumoto, T.; Cooper, G.M.; Gharaibeh, B.; Meszaros, L.B.; Li, G.; Usas, A.; Fu, F.H.; Huard, J. Cartilage repair in a rat model of osteoarthritis through intraarticular transplantation of muscle-derived stem cells expressing bone morphogenetic protein 4 and soluble flt-1. Arthritis Rheum. 2009, 60, 1390–1405. [Google Scholar] [CrossRef]
- Gao, X.; Cheng, H.; Awada, H.; Tang, Y.; Amra, S.; Lu, A.; Sun, X.; Lv, G.; Huard, C.; Wang, B.; et al. A comparison of BMP2 delivery by coacervate and gene therapy for promoting human muscle-derived stem cell-mediated articular cartilage repair. Stem Cell Res. Ther. 2019, 10, 1–13. [Google Scholar] [CrossRef]
- Gao, X.; Wright, N.; Huard, M.; Tan, J.; Ruzbarsky, J.; Lu, A.; Chubb, L.; Tuan, R.; Philippon, M.J.; Wang, Y.; et al. Comparison of 5 BMPs for their chondrogenic potentials and microfracture-mediated cartilage repair using heparin/PEAD coacervate sustained release polymer. Bioact. Mater. 2025, 52, 588–603. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



