Submitted:
14 April 2026
Posted:
15 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Plant Collection
2.3. Methods
2.3.1. Preparation of Plant Extract
2.3.2. Determination of Total Phenolic Content (TPC)
2.3.3. Determination of Total Flavonoids Content (TFC)
2.3.4. DPPH Radical Scavenging Activity
2.3.5. ABTS Scavenging Activity
2.3.6. Reducing Power
2.3.7. Superoxide Radical Scavenging Activity
2.3.8. Superoxide Dismutase (SOD) Activity Assay
2.3.9. Experimental Design to Optimize Extraction of Secondary Metabolites
2.3.10. Intrinsic Synchronous Fluorescence Spectroscopy (SFS) of Plant Extracts
2.3.11. Intrinsic Synchronous Phosphorescence Spectroscopy (SPS) of Plant Extracts
2.3.12. Intrinsic 3D Fluorescence Spectra Measurements of Plant Extracts
2.3.13. FTIR Analysis of Plant Extracts
2.3.14. Statistical Analysis
3. Results and Discussion
3.1. Phytochemical Assays of Plant Extracts
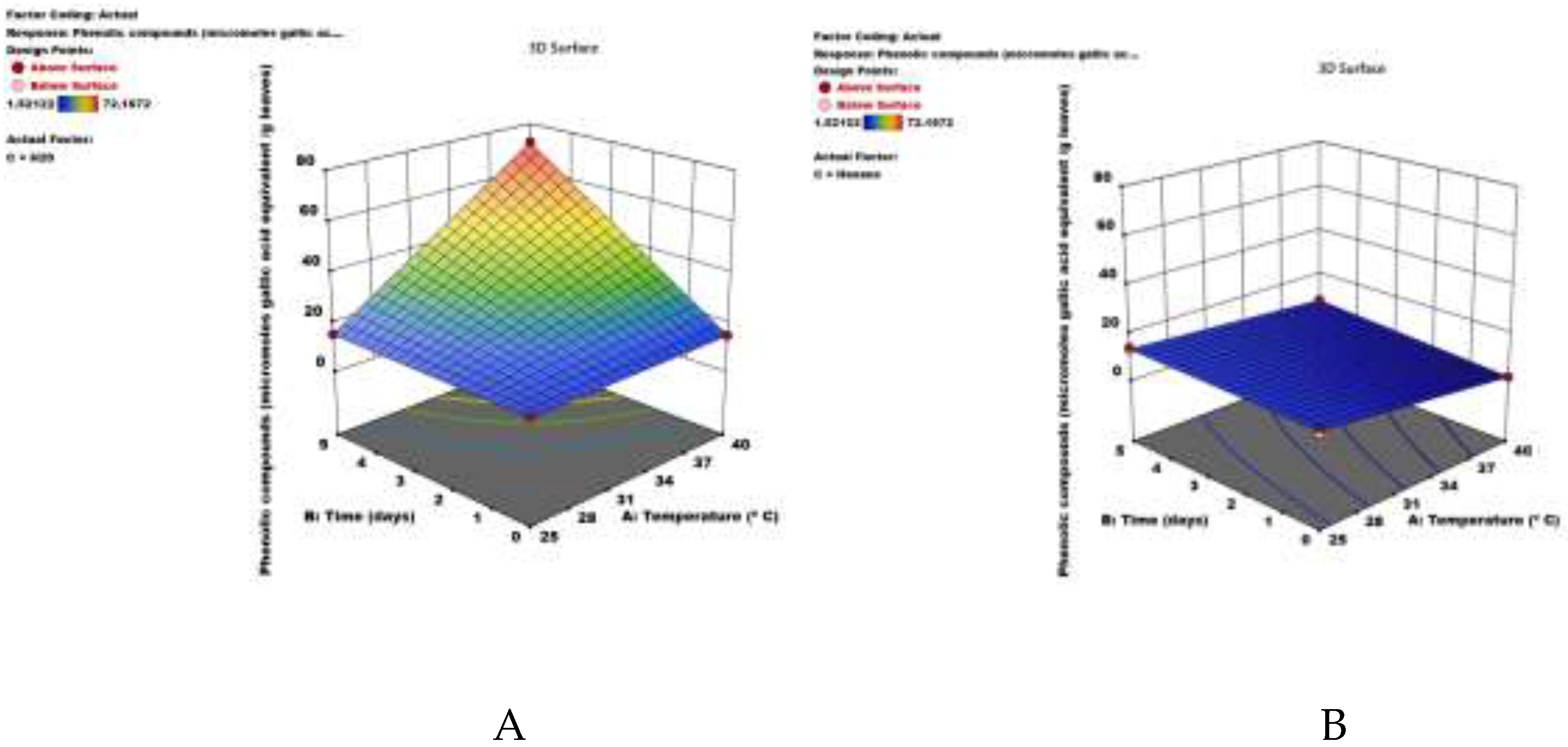
3.2. Optimization of Extraction of Secondary Metabolites from Plant Extracts
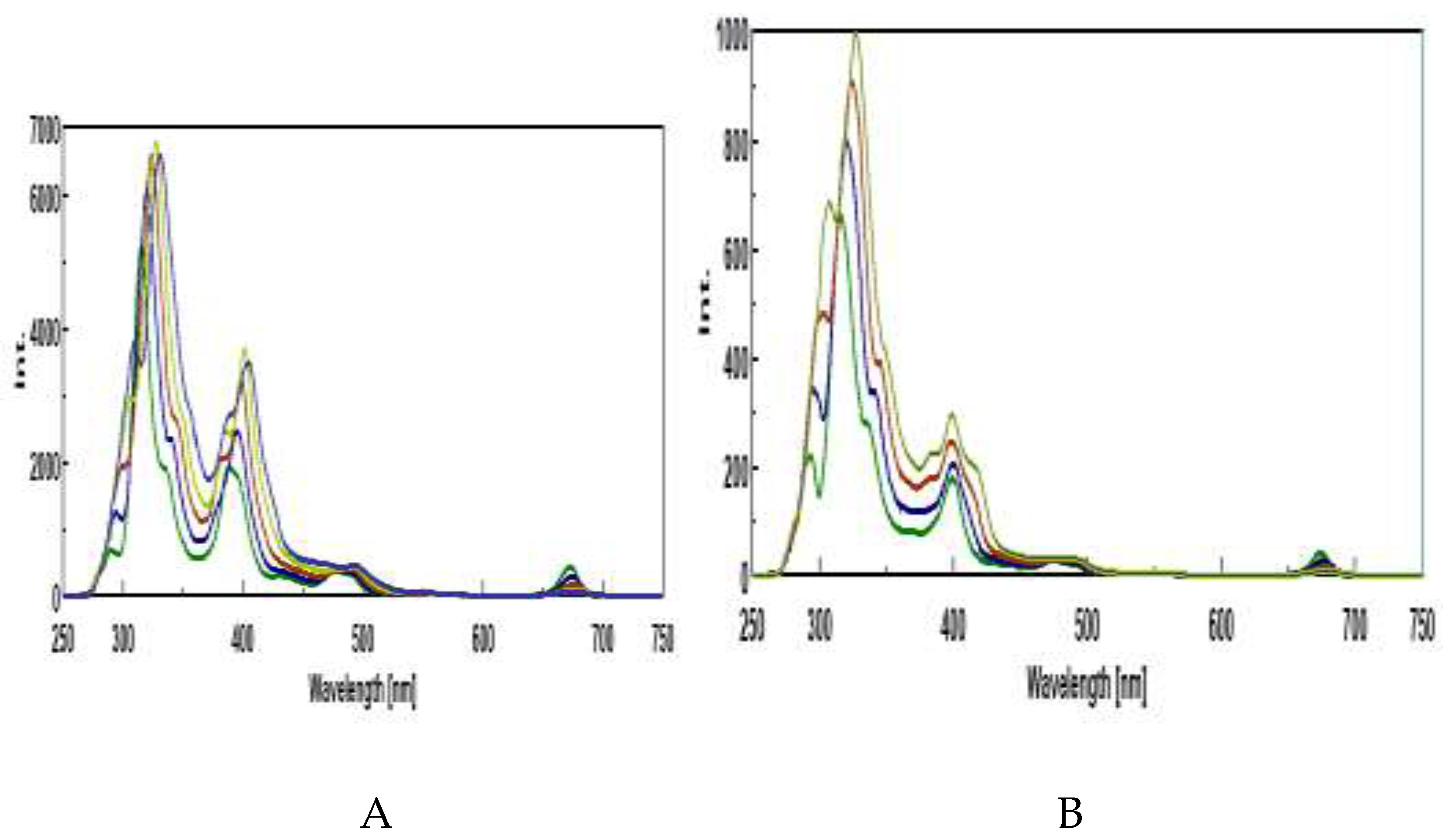
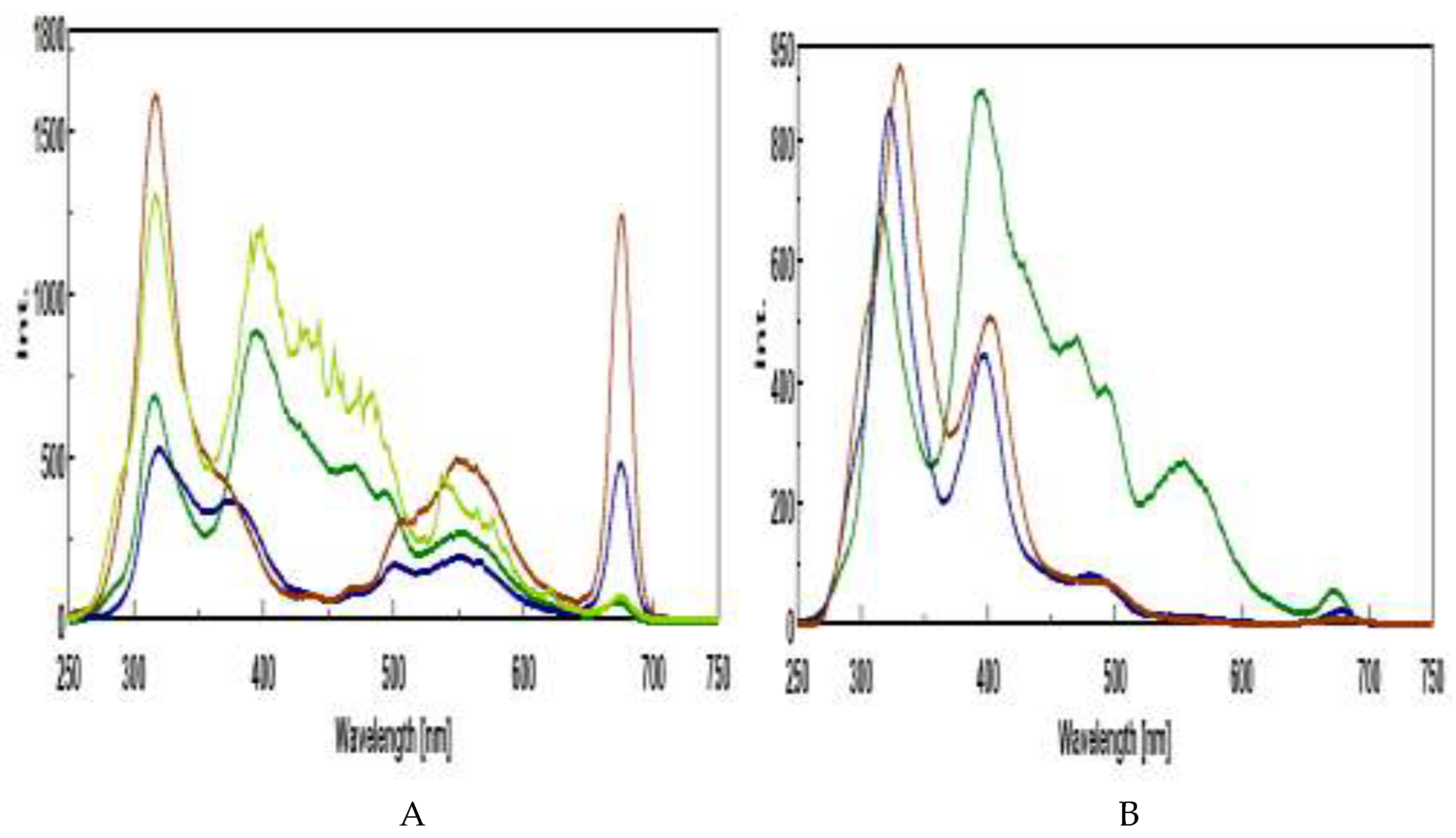
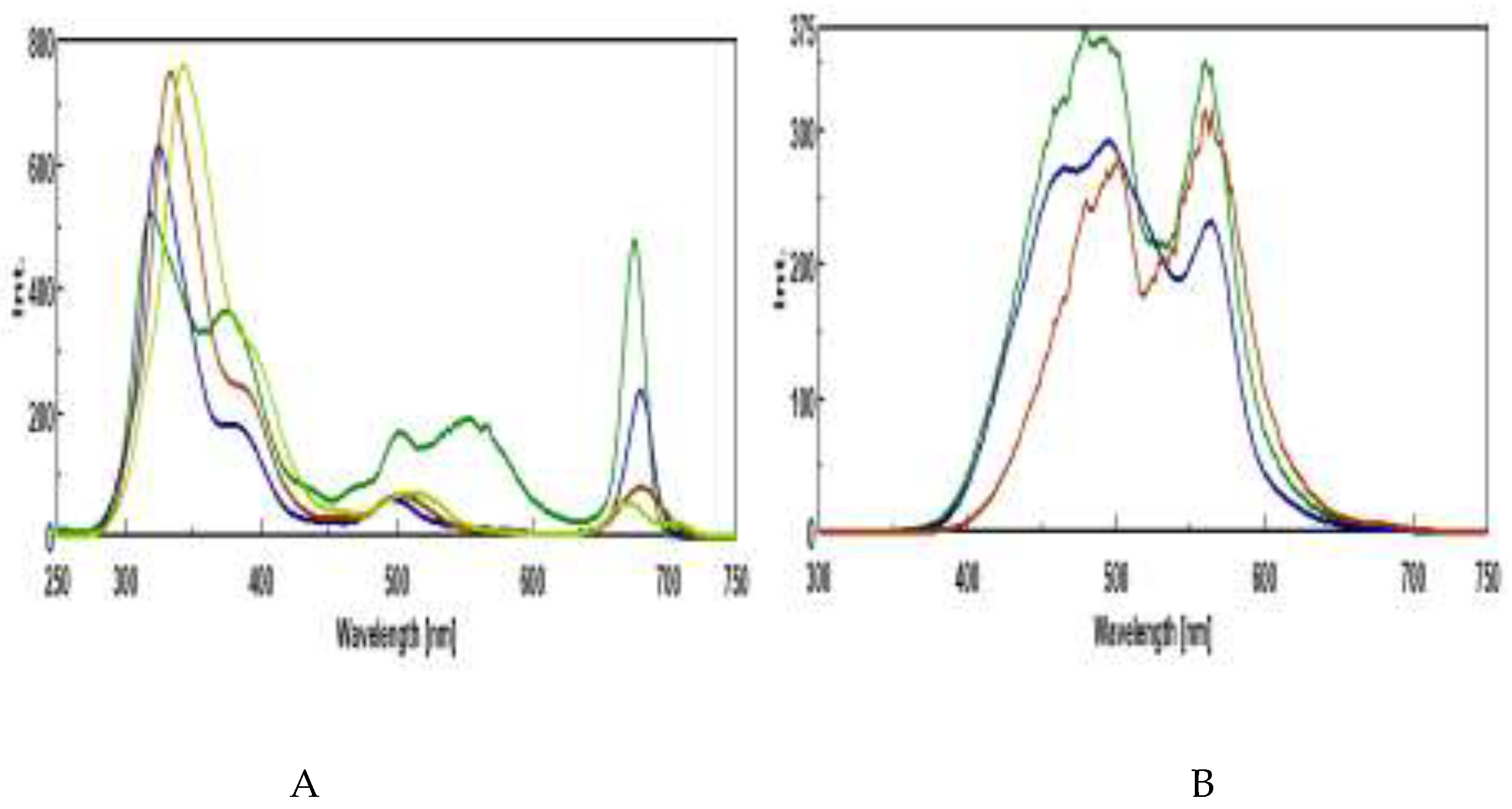
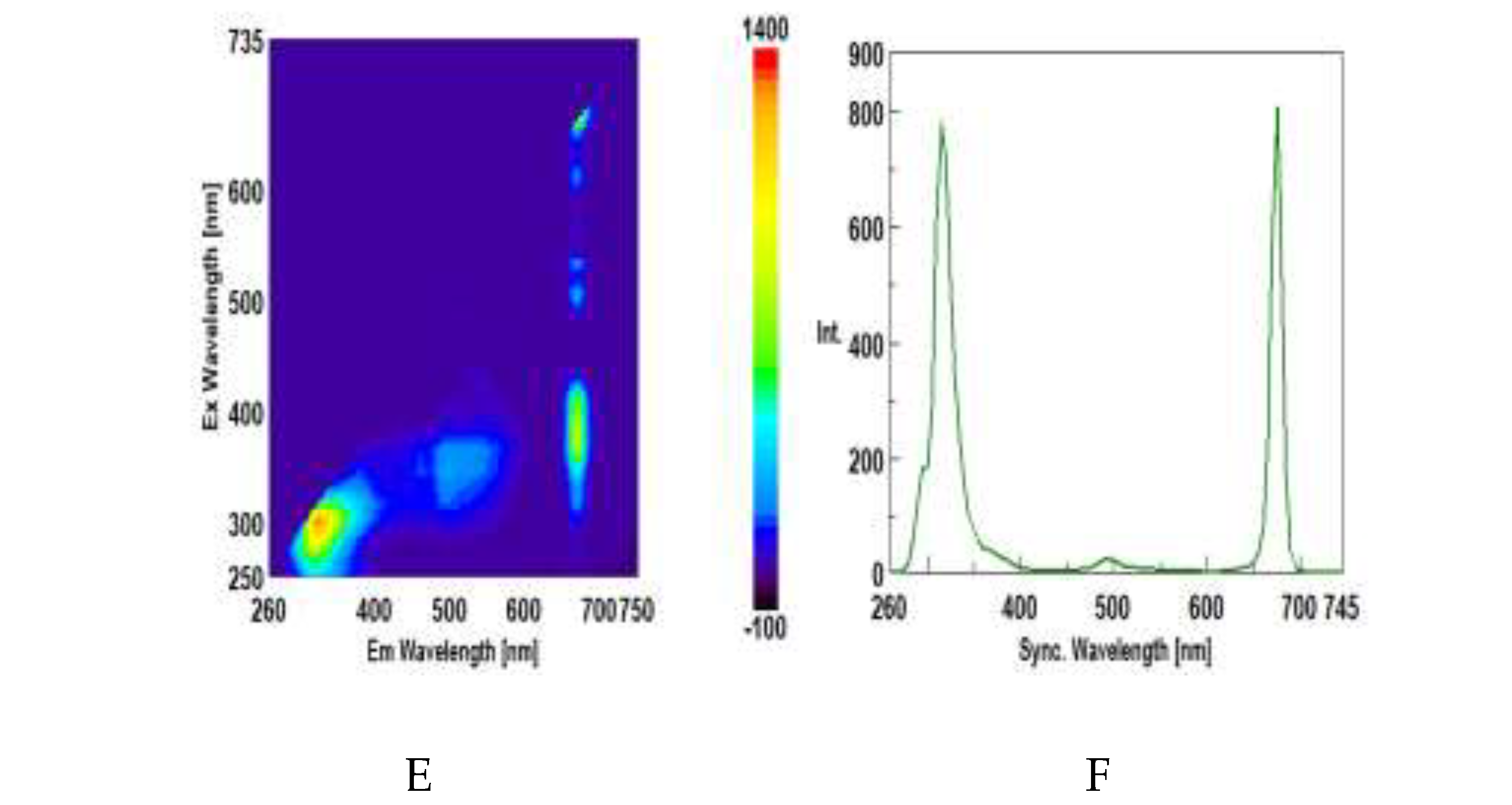
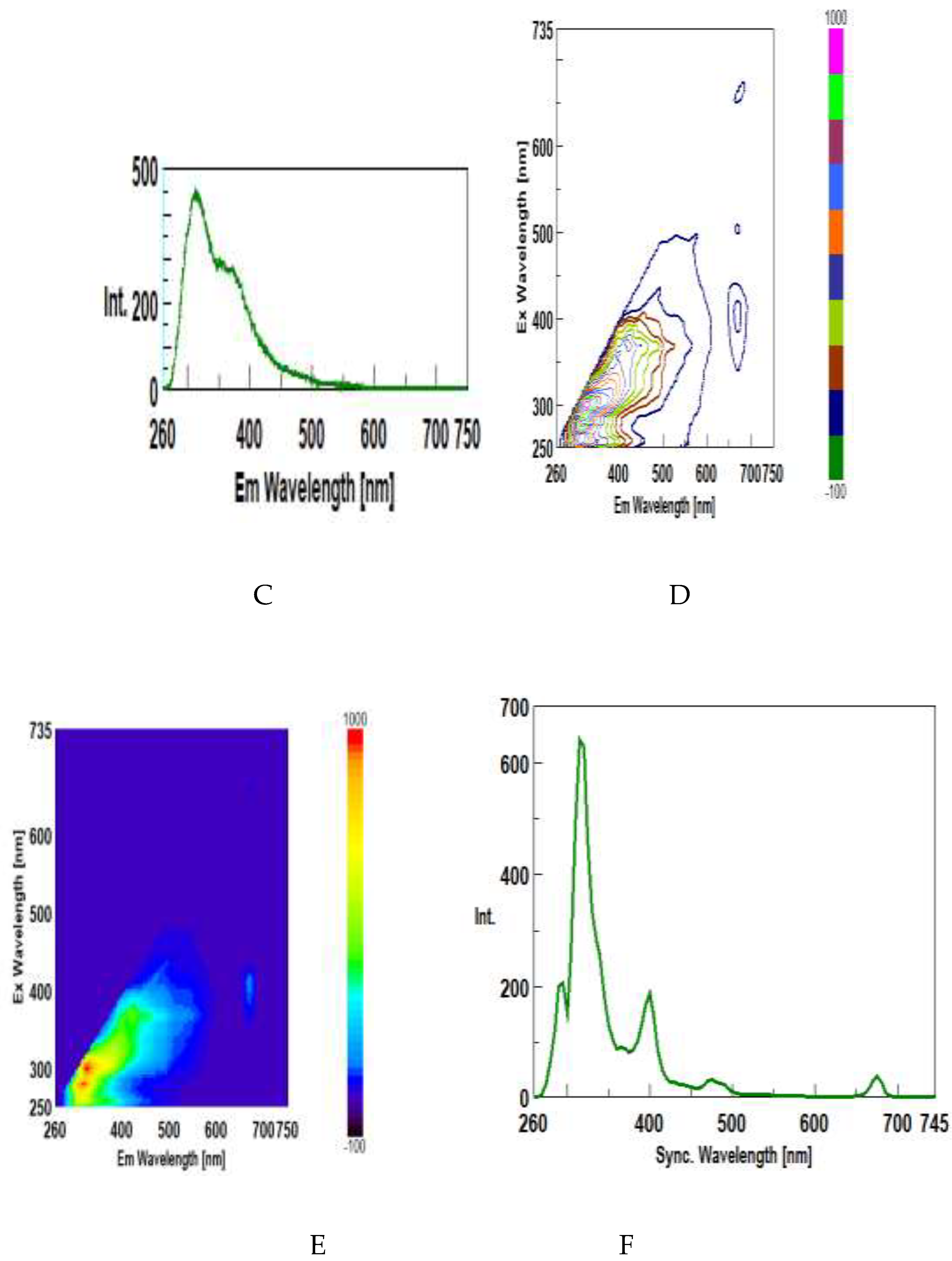
3.3. Synchronous Fluorescence Spectroscopy (SFS)
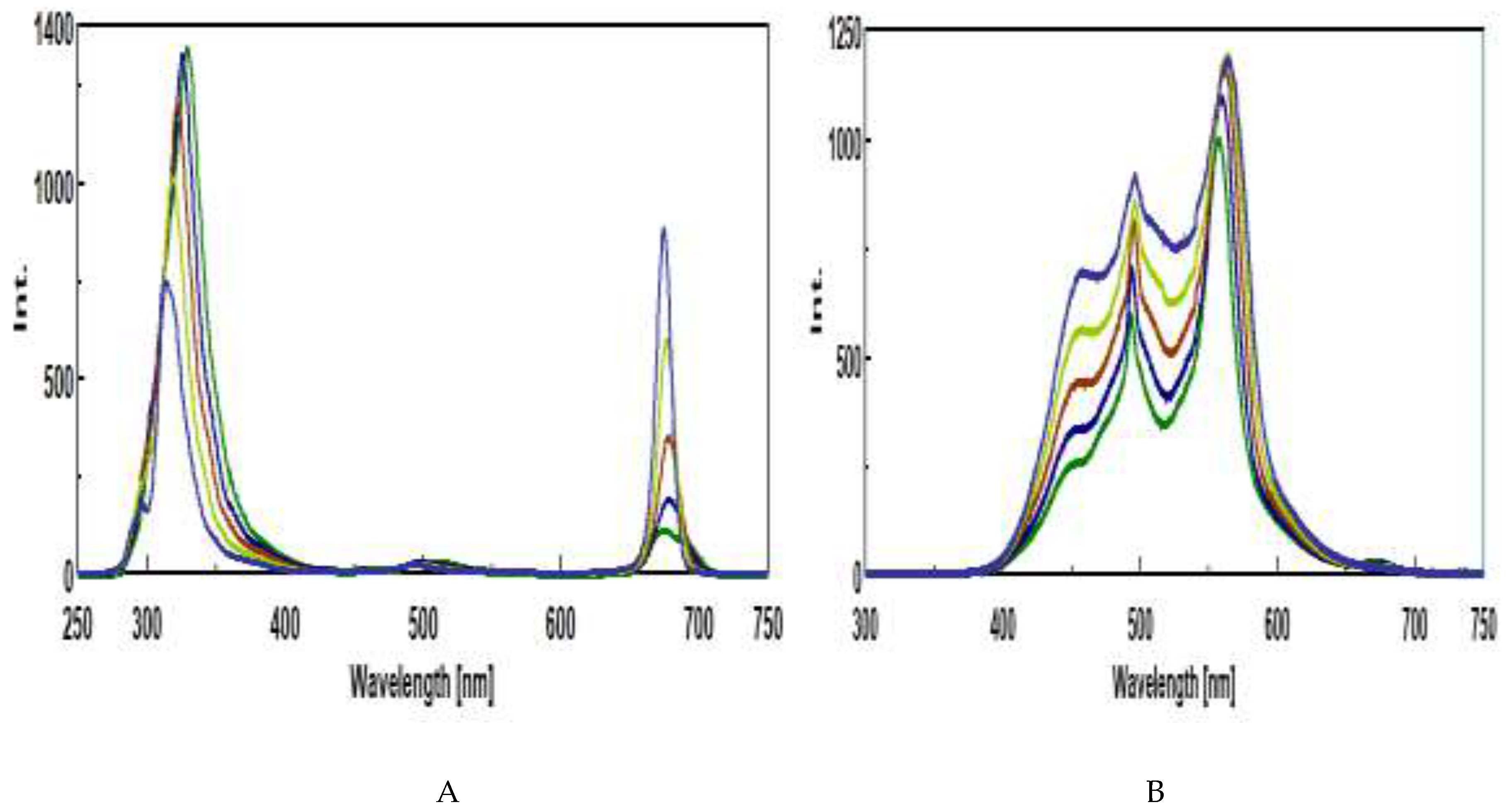
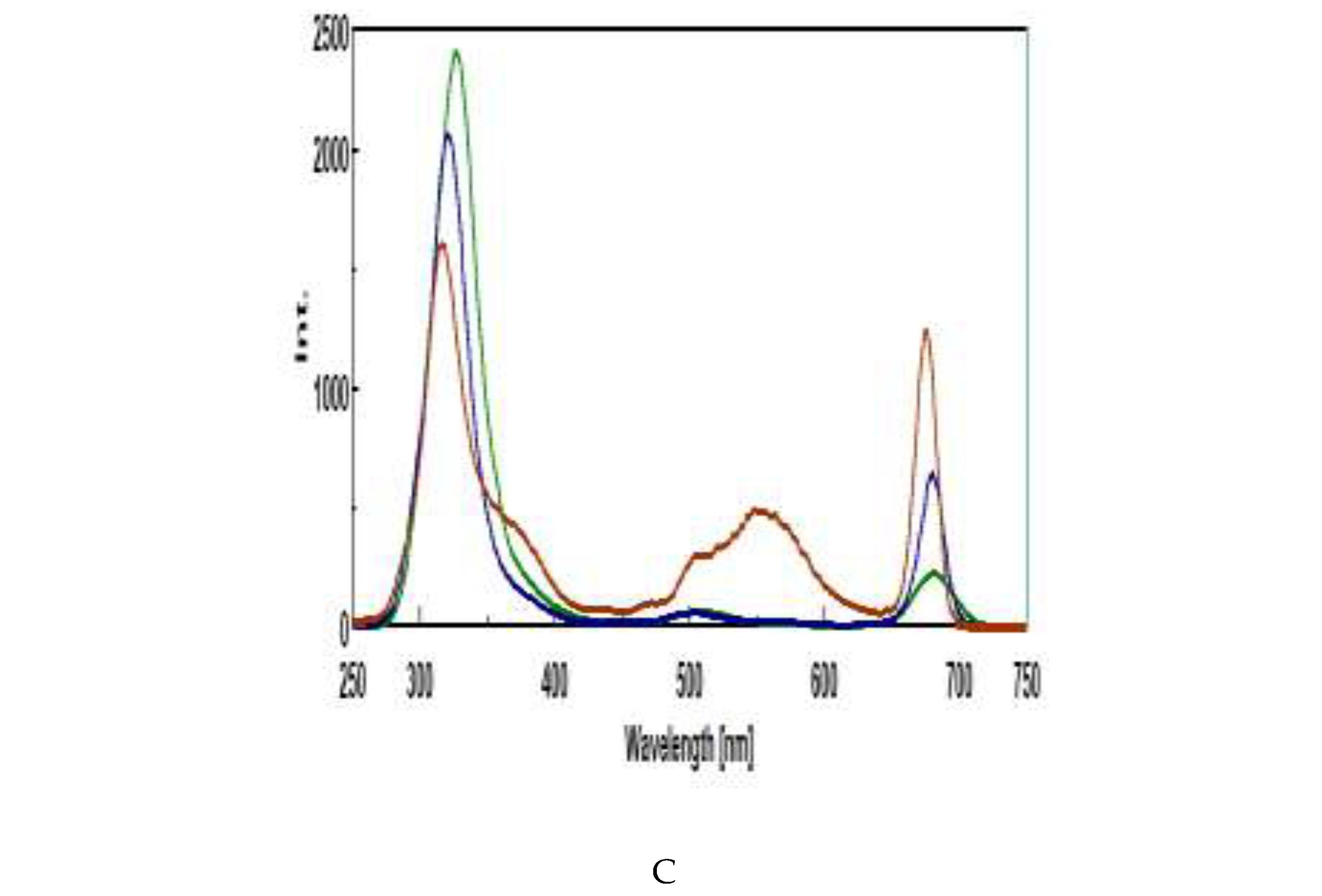
3.4. Intrinsic Synchronous Phosphorescence Spectroscopy (SPS)
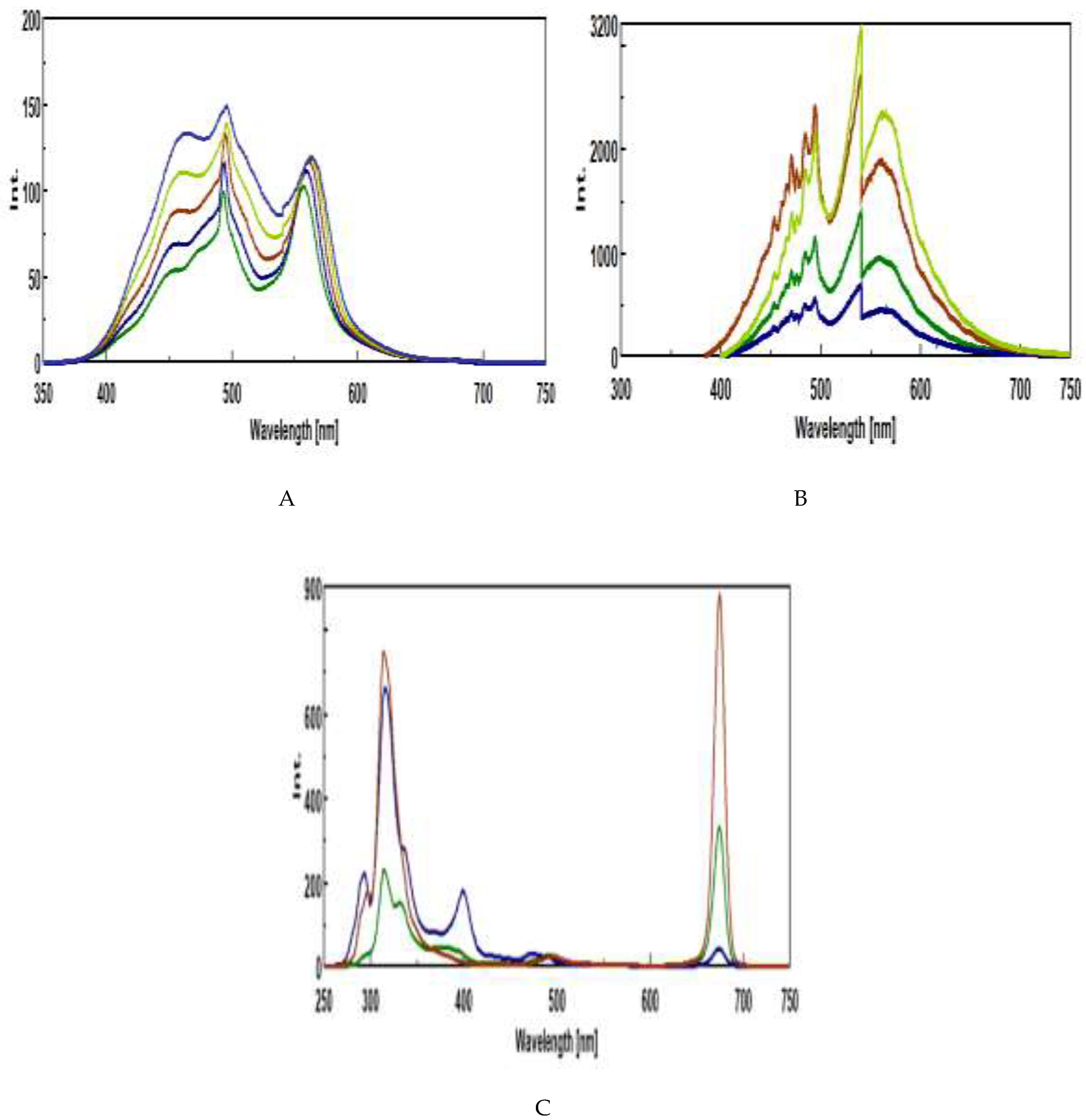
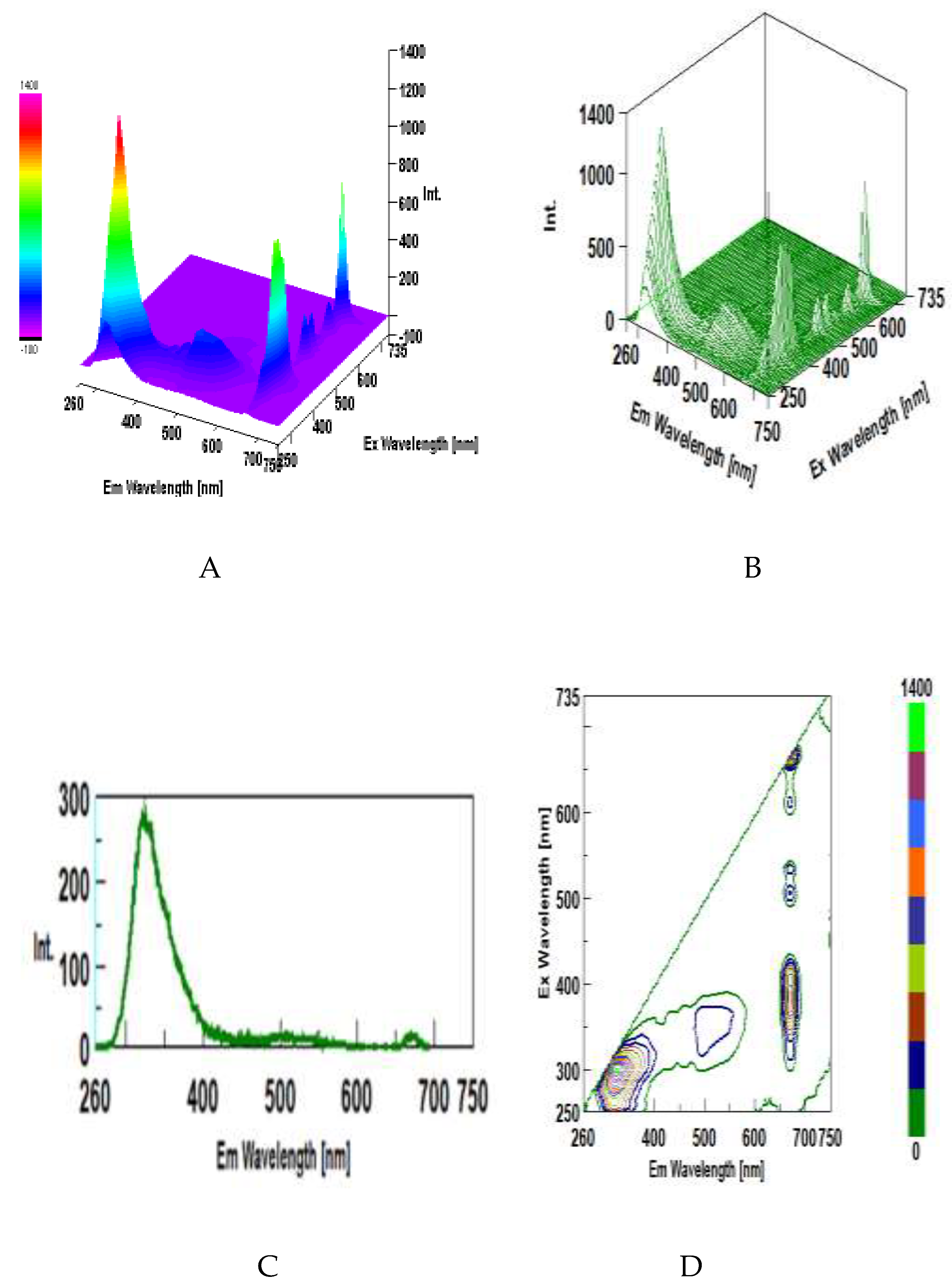
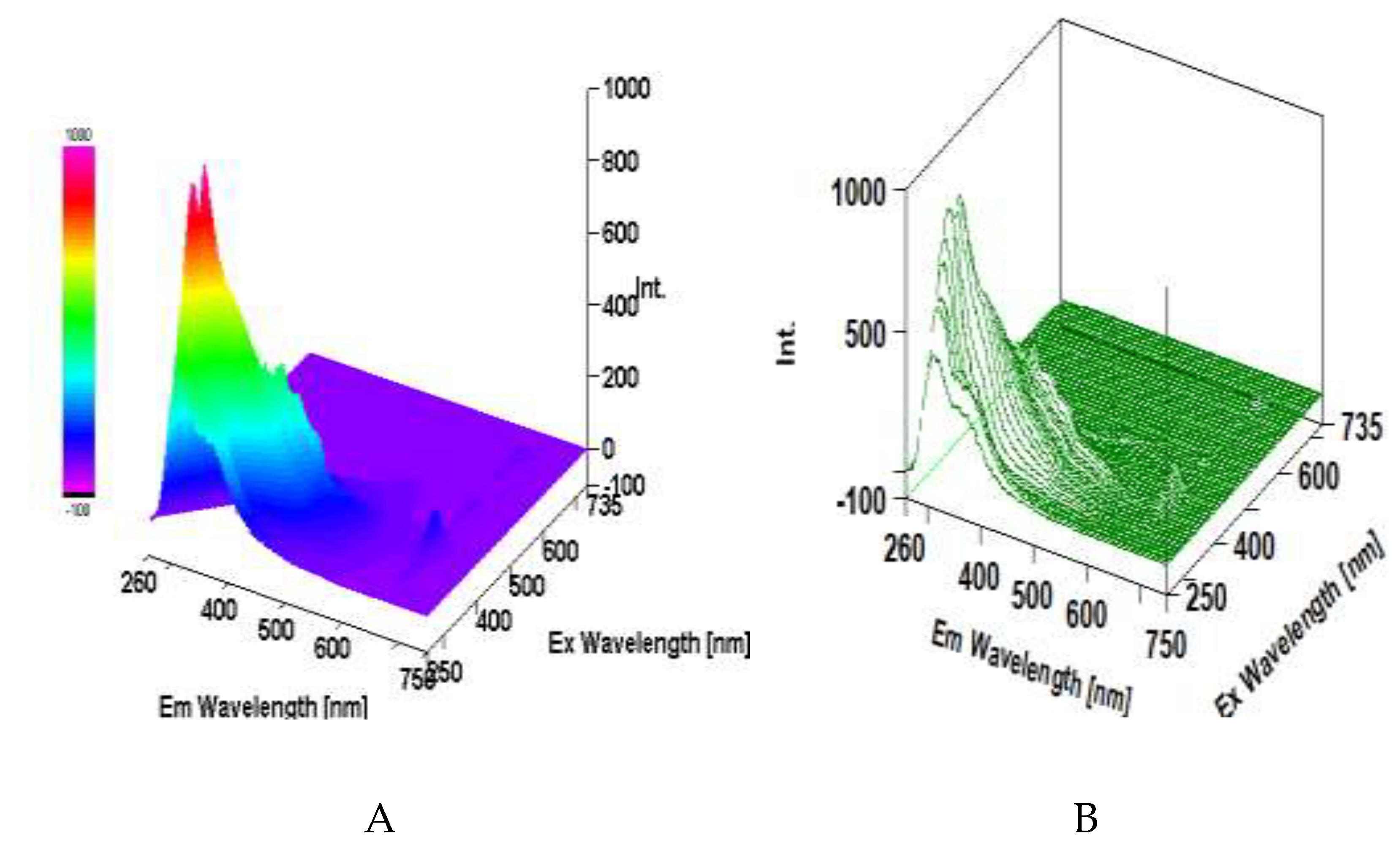
3.5. Intrinsic 3D Fluorescence Spectroscopy
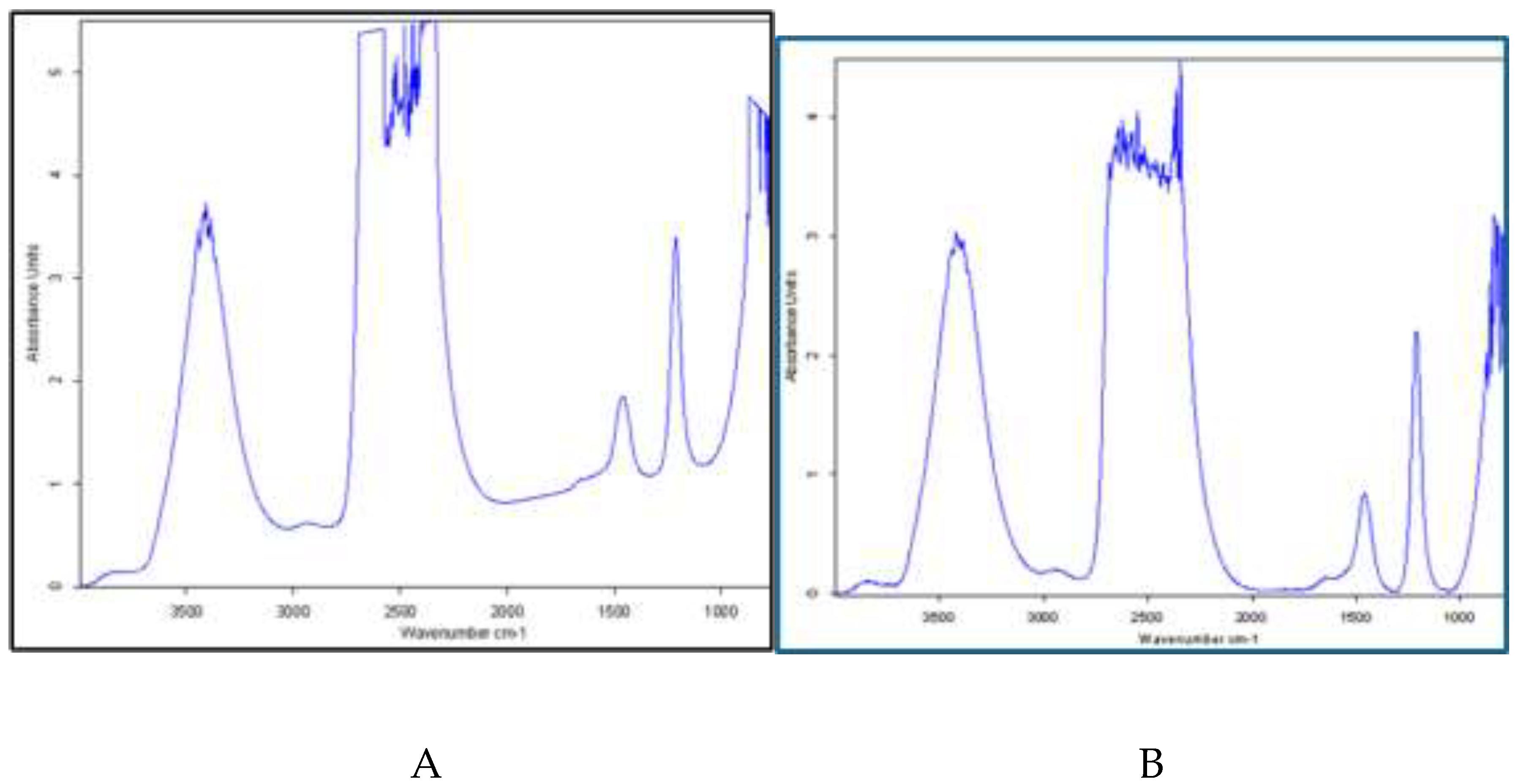
3.6. FTIR Analysis
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| ABTS – 2,2′-azinobis-3-ethylbenzothiazoline-6-sulphonic acid DPPH- 2,2-Diphenyl-1-picrylhydrazyl 3D-SFS- 3D synchronous fluorescence spectroscopy NBT-Nitro blue tetrazolium PMS- Phenazine methosulfate SFS- Synchronous Fluorescence Spectroscopy SOD – Superoxide dismutase SPS- Synchronous Phosphorescence Spectroscopy TCA- Trichloroacetic acid TPC- Total phenolic content TFC- Total flavonoids content |
References
- Naseer, M.; Adil, M. Phytochemical profiling, HPLC analysis, and antimicrobial potential of Curio radicans (L. f.) P.V. Heath. Sci Rep 2025, 15, 34753. [Google Scholar] [CrossRef]
- Jain, D.; Meena, M.; Janmeda, P.; Seth, C.S.; Arora, J. Analysis of Quantitative phytochemical content and antioxidant activity of leaf, stem and bark of Gymnosporia senegalensis (Lam.) Loes. Plants 2024, 13, 1425. [Google Scholar] [CrossRef]
- Maheshwaran, L.; Nadarajah, L.; Senadeera, S.P.N.N.; Ranaweera, C.B.; Chandana, A.K.; Pathirana, R.N. 1. Phytochemical Testing Methodologies and Principles for Preliminary Screening/ Qualitative Testing. Asian Plant Research Journal 2024, 12, 11–38. [Google Scholar] [CrossRef]
- Ceríaco, L.M.P.; Lima, R.F.; Melo, M.; Bell, R.C. 2022 Biodiversity of the Gulf of Guinea Oceanic Islands. In Science and Conservation; Publisher Springer Cham. [CrossRef]
- Omokhua, A. G.; Abdalla, M. A.; Van Staden, J.; McGaw, L. J. A comprehensive study of the potential phytomedicinal use and toxicity of invasive Tithonia species in South Africa 06 Biological Sciences 0605 Microbiology. BMC Complementary and Alternative Medicine 2018, 18(1), 1–15. [Google Scholar] [CrossRef]
- Chagas-Paula, D. A.; Oliveira, R. B.; Rocha, B. A.; Da Costa, F. B. Ethnobotany, chemistry, and biological activities of the genus Tithonia (Asteraceae). Chemistry and Biodiversity 2012, 9(2), 210–235. [Google Scholar] [CrossRef]
- Currais, A.; Chiruta, C.; Goujon-Svrzic, M.; Costa, G.; Santos, T.; Batista, M. T.; Paiva, J.; Madureira, M.C.; Maher, P. Screening and identification of neuroprotective compounds relevant to Alzheimer’s disease from medicinal plants of S. Tomé e Príncipe. Journal of Ethnopharmacology 2014, 155(1), 830–840. [Google Scholar] [CrossRef] [PubMed]
- Madureira, M.C.; Martins, A.P.; Gomes, M.; Paiva, J.; Cunha, A.P.; Rosario. Antimalarial activity of medicinal plants used in traditional medicine in S. Tomé and Príncipe. Journal of Ethnopharmacology 2002, 81, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Ajao, A.A.; Moteetee, A. N. Review Tithonia diversifolia (Hemsl) A. Gray. (Asteraceae: Heliantheae), an invasive plant of significant ethnopharmacological importance. South African Journal of Botany 2017, 113, 396–403. [Google Scholar] [CrossRef]
- Di Giacomo, C.; Vanella, L.; Sorrenti, V.; Santangelo, R.; Barbagallo, I.; Calabrese, G.; Genovesem, C.; Mastrojeni, S.; Ragusa, S.; Acquaviva, R. Effects of Tithonia diversifolia (Hemsl.) A. Gray Extract on Adipocyte Differentiation of Human Mesenchymal Stem Cells. PLoS ONE 2015, 10(4), e0122320. [Google Scholar] [CrossRef]
- Goffin, E.; Ziemons, E.; De Mol, P.; De Madureira, M. D. C.; Martins, A. P.; Proença da Cunha, A.; Frederich, M. In vitro antiplasmodial activity of Tithonia diversifolia and identification of its main active constituent: Tagitinin C. Planta Medica 2002, 68(6), 543–545. [Google Scholar] [CrossRef]
- Gama; R, M.; Ruggiero, A.A. Andrade.G.P.; Armando-Junior, J.; Pinhal, M.A.S. Optimizing bioactive compounds extraction and cell viability effect of Tithonia diversifolia (Hemsl.) A. Gray dry flower extracts. 2025 Journal of Pharmacy & Pharmacognosy Research 13(5), 1313–1326, 2025. [CrossRef]
- John-Dewole, O.O.; Oni, S.O. Phytochemical and Antimicrobial Studies of Extracts from the Leaves of Tithonia Diversifolia for Pharmaceutical Importance. IOSR Journal of Pharmacy and Biological Sciences 2013, 6(4), 21–25. [Google Scholar] [CrossRef]
- Farias, A.L.F.; Rodrigues, A. B. L.; Martins, R.L.; Rabelo, E.M.; Farias, C.W.F.; Almeida, S.s. M. S. Chemical Characterization, Antioxidant, Cytotoxic and Microbiological Activities of the Essential Oil of Leaf of Tithonia Diversifolia (Hemsl) A. Gray (Asteraceae). Pharmaceuticals 2019, 12(1), 34. [Google Scholar] [CrossRef]
- Barboza, B. R.; Da Silva Barros, B. R.; Ramos, B. D. A.; De Moura, M. C.; Napoleão, T. H.; Dos Santos Correia, M. T.; Coelho, L.-C.-B.-B.; Filho, I.-J.-C.; Maior, A.-M.-S.; Silva, T.-D.; Nerys, L.-C.-R.; Santana, E.-R.-B.; Lima, C.-S.-A.; Lorena, V.-M.-B.; De Melo, C. M. L. Phytochemical bioprospecting, antioxidant, antimicrobial and cytotoxicity activities of saline extract from Tithonia diversifolia (Hemsl) A. Gray leaves. Asian Pacific Journal of Tropical Biomedicine 2018, 8(5), 245–253. [Google Scholar] [CrossRef]
- Marques, L.; Karmali, A. Experimental Planning for Production of β-D-Glucan: Purification and Fluorescence Properties from Basidiomycete Strains. Separations 2025, 12, 336. [Google Scholar] [CrossRef]
- Sun, S.; Yu, Y.; Jo, Y.; Han, J.H.; Xue, Y.; Cho, M.; BaeS, -J.; Ryu, D.; Park, W.; Ha, K.-T.; Zhuang, S. Impact of extraction techniques on phytochemical composition and bioactivity of natural product mixtures. Front. Pharmacol. 2025, 16, 1615338. [Google Scholar] [CrossRef]
- Popescu, S.A.; Peled, A. Optimized RED spectral band Fluorescence of edible plants leaves extracts. Applied Surface Science Advances 2023, 13, 100385. [Google Scholar] [CrossRef]
- Tian, S.; Zhang, Y.; Wang, J.; Zhang, R.; Wu, W.; He, Y.; Wu, X.; Sun, W.; Li, D.; Xiao, Y.; et al. New 3-D Fluorescence Spectral Indices for Multiple Pigment Inversions of Plant Leaves via 3-D Fluorescence Spectra. Remote Sens. 2024, 16, 1885. [Google Scholar] [CrossRef]
- Herald, T.J.; Gadgil, P.; Perumal, R.; Bean, S.R.; Wilson, J.D. High-throughput micro-plate HCl-vanillin assay for screening tannin content in sorghum grain. J. Sci. Food Agric. 2014, 94, 2133–2136. [Google Scholar] [CrossRef] [PubMed]
- Attard, E. A rapid microtitre plate Folin-Ciocalteu method for the assessment of polyphenols. Open Life Sci. 2013, 8, 48–53. [Google Scholar] [CrossRef]
- Bobo-García, G.; Davidov-Pardo, G.; Arroqui, C.; Vírseda, P.; Marín-Arroyo, M.R.; Navarro, M. Intra-laboratory validation of microplate methods for total phenolic content and antioxidant activity on polyphenolic extracts, and comparison with conventional spectrophotometric methods. J. Sci. Food Agric. 2015, 95, 204–209. [Google Scholar] [CrossRef]
- Reis, F.S.; Pereira, E.; Barros, L.; Sousa, M.J.; Martins, A.; Ferreira, I.C.F.R. Biomolecule profiles in inedible wild mushrooms with antioxidant value. Molecules 2011, 16, 4328–4338. [Google Scholar] [CrossRef] [PubMed]
- Khair-ul-Bariyah, S.; Ahmed, D.; Ikram, M. Ocimum basilicum: A review on phytochemical and pharmacological studies. Pak. J. Chem. 2012, 2, 78–85. [Google Scholar] [CrossRef]
- Nishanthini, A.; Mohan, V.R. Antioxidant activites of Xanthosoma sagittifolium Schott using various in vitro assay models. Asian Pacific Journal of Tropical Biomedicine 2, Supplement 2012, Pages S1701–S1706. [Google Scholar] [CrossRef]
- Boonmee, A.; Srisomsap, C.; Karnchanatatc A. Sangvanicha, P. An antioxidant protein in Curcuma comosa Roxb. Rhizomes, Food Chemistry 2011, 124, 476–480. [Google Scholar] [CrossRef]
- Karmali, A. Fluorescence and Phosphorescence Assay of β-D Glucans from. Mushrooms Processes 14, 442. [CrossRef]
- Popovic, L.; Rijkers, G.T. Phytochemicals: Principles and Practice. Biology 2026, 15(1), 18. [Google Scholar] [CrossRef]
- Kumar, A.; P, N.; Kumar, M.; Jose, A.; Tomer, V.; Oz, E.; Proestos, C.; Zeng, M.; Elobeid, T.; Sneha, K.; Oz, F. Major Phytochemicals: Recent Advances in Health Benefits and Extraction Method. Molecules 2023, 28, 887. [Google Scholar] [CrossRef] [PubMed]
- Hiransai, P.; Tangpong, J.; Kumbuar, C.; Hoonheang, N.; Rodpech, O.; Sangsuk, P.; Kajklangdon, U.; Inkaow, W. Anti-nitric oxide production, anti-proliferation and antioxidant effects of the aqueous extract from Tithonia diversifolia. Asian Pac J Trop Biomed 2016, 6, 950–956. [Google Scholar] [CrossRef]
- Pulido, K.D.P.; Dulcey, A.J.C.; Martinez, J.H.I. New caffeic acid derivative from Tithonia diversifolia (Hemsl.) A. Gray butanolic extract and its antioxidant activity. Food Chem. Toxicol. 2017. [Google Scholar] [CrossRef]
- Jeevitha, M.; Ravi, P.V.; Subramaniyam, V.; Pichumani, M.; Sripathi, S.K. Exploring the phyto- and physicochemical evaluation, fluorescence characteristics and antioxidant activities of Acacia ferruginea Dc: an endangered medicinal plant. Future Journal of Pharmaceutical Sciences 2021, 7, 228. [Google Scholar] [CrossRef]
- Lang, M.; Stober, F.; Lichtenthaler, H.K. Fluorescence emission spectra of plant leaves and plant constituents. Radiat Environ Biophys 1991, 30, 333–347. [Google Scholar] [CrossRef]
- Krasnovsky, A.A.; Kovalev, Y.V. Review. Spectral and Kinetic Parameters of Phosphorescence of Triplet Chlorophyll a in the Photosynthetic Apparatus of Plants. Biochemistry (Moscow) 2014, 79, 349–361. [Google Scholar] [CrossRef]
- Chen, W.; Zhu, Z. Ultralong luminescence lifetime imaging of edible plant tissue for humidity sensing in food packaging by a smartphone. Food Chemistry 2024, 454, 139778. [Google Scholar] [CrossRef] [PubMed]
- Tian, S.; Zhang, Y.; Wang, J.; Zhang, R.; Wu, W.; He, Y.; Wu, X.; Sun, W.; Li, D.; Xiao, Y.; et al. New 3-D Fluorescence Spectral Indices for Multiple Pigment Inversions of Plant Leaves via 3-D Fluorescence Spectra. Remote Sens. 2024, 16, 1885. [Google Scholar] [CrossRef]
- Kua, Y.G.; Baea, J.H.; Martinez-Ayalac, A.L.; Vearasilp, S.; Namiesnike, J.; Paskof, P.; Katrichh, E.; Gorinstein, S. Efficient three-dimensional fluorescence measurements for characterization of binding properties in some. Sensors and Actuators B 2017, 248, 777–784. [Google Scholar] [CrossRef]
- Zhou, T.; Fu, Y.; Zhang, Y.; Meng, Z.-Y.; Xu, H.-D.; Tian, R.T.; Wang, C.; Wang, T.-Y.; Deng, X.-Y.; Zhang, Y.; Wang, L. Application of three-dimensional fluorescence spectral characterization and chemometrics in the analysis of traceability of Paeoniae Radix Rubra. PLoS One 2025, 20(8), e0328834. [Google Scholar] [CrossRef]
- Ku, Y.G.; Kim, H.C.; Bae, J.H.; Kang, B.S.; Nemirovski, A.; Barasch, D.; Gorinstein, S. Antioxidant capacities and polyphenols in autumn-growing cultivar of Chinese cabbage (Brassica rapa L. ssp. pekinensis cv. Bulam Plus). European Food Research and Technology 2019, 245, 1871–1879. [Google Scholar] [CrossRef]
- Stanley, O.; Uche, C.A.; Chidi, N.; Constance, N. Gas chromatography mass spectrometry/Fourier transform infrared (GC-MS/FTIR) spectral analyses of Tithonia diversifolia (Hemsl.) A. Gray leaves. Journal of Medicinal Plants Research 2017, 11, 345–350. [Google Scholar] [CrossRef]
| Plant extracts | TPC | TFC | Reducing power | ABTS inhibition | DPPH inhibition | Superoxide radical | SOD activity |
| (mmoles gallic acid | (mmoles catechin | (mmoles TE equivalent/g | IC50 | IC50 | scavenging activity | (Units/g leaves) | |
| equivalent/g leaves) | equivalent/g leaves) | leaves) | (mg extract/mL | (mg extract/mL) | IC50 (mg extract/mL) | ||
| H20,25ºC, 0 days | 14.24±0.52a | 3.41±0.24a | 21.63±2.17b | 0.90±0.01a | 2.20±0.02a | 1.35±0.01a | 1101.95±34.14a |
| H20,25ºC, 5 days | 15.32±0.01a | 5.61±0.93b | 26.63±0.36a | 0.09±0.01c | 1.35±0.02b | 0.90±0.02c | 1107.72±91.15c |
| Hexane,25ºC,0 days 12.04±1.31a | 6.56±0.82a | 4.82±0.15a | 6.63±0.05b | 6.48±0.03b | 1.06±0.01b | 576.47±40.71c | |
| Hexane,25ºC,5 days 13.46±0.88a | 3.23±0.15a | 0.39±0.03b | 3.19±0.01c | 2.65±0.09d | 0.65±0.01d | 388.26±25.42c | |
| H20,40ºC, 0 days | 14.87±0.12b | 4.63±0.38c | 0.87±0.20a | 0.52±0.01a | 2.23±0.07c | 1.06±0.01b | 145.62±23.07d |
| H20,40ºC, 5 days | 71.95±0.30c | 6.20±0.31d | 12.13±0.20d | 0.32±0.004d | 1.51±0.07d | 0.92±0.03b | 1249.53±160.04c |
| Hexane, 40ºC,0 days 1.53±0.02b | 4.36±0.34b | 2.08±0.15e |
10.87±0.07e | 8.78±0.14c | 9.09±0.02d | 549.05±260.88f | |
| Hexane, 40ºC,5 days 7.62±0.33c | 5.22±0.38d | 2.63±0.25e |
4.07±0.04f | 7.61±0.36b | 8.23±0.02d | 237.00±18.37e | |
| Sum of Squares | Degree of Freedom | Mean Square |
F Value |
p- value | ||
| Model | 6740.36 | 7 | 962.91 | 2558.67 | < 0.0001 | |
| A-Temperature | 418.77 | 1 | 418.77 | 1112.76 | < 0.0001 | |
| B-Time | 1077.58 | 1 | 1077.58 | 2863.39 | < 0.0001 | |
| C-Solvent | 1668.91 | 1 | 1668.91 | 4434.70 | < 0.0001 | |
| AB | 920.44 | 1 | 920.44 | 2445.83 | < 0.0001 | |
| AC | 1354.75 | 1 | 1354.75 | 3599.90 | < 0.0001 | |
| BC | 640.57 | 1 | 640.57 | 1702.16 | < 0.0001 | |
| ABC | 659.32 | 1 | 659.32 | 1751.98 | < 0.0001 | |
| Pure Error | 3.01 | 8 | 0.3763 | |||
| Cor Total | 6743.37 | 15 |
|
Factor Coefficient Estimate df Standard Error 95% CI Low 95% CI High VIF Intercept 18.88 1 0.1534 18.53 19.24 A-Temperature 5.12 1 0.1534 4.76 5.47 1.0000 B-Time 8.21 1 0.1534 7.85 8.56 1.0000 C-Solvent -10.21 1 0.1534 -10.57 -9.86 1.0000 AB 7.58 1 0.1534 7.23 7.94 1.0000 AC -9.20 1 0.1534 -9.56 -8.85 1.0000 BC -6.33 1 0.1534 -6.68 -5.97 1.0000 ABC -6.42 1 0.1534 -6.77 -6.07 1.0000 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




