Submitted:
18 February 2026
Posted:
25 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
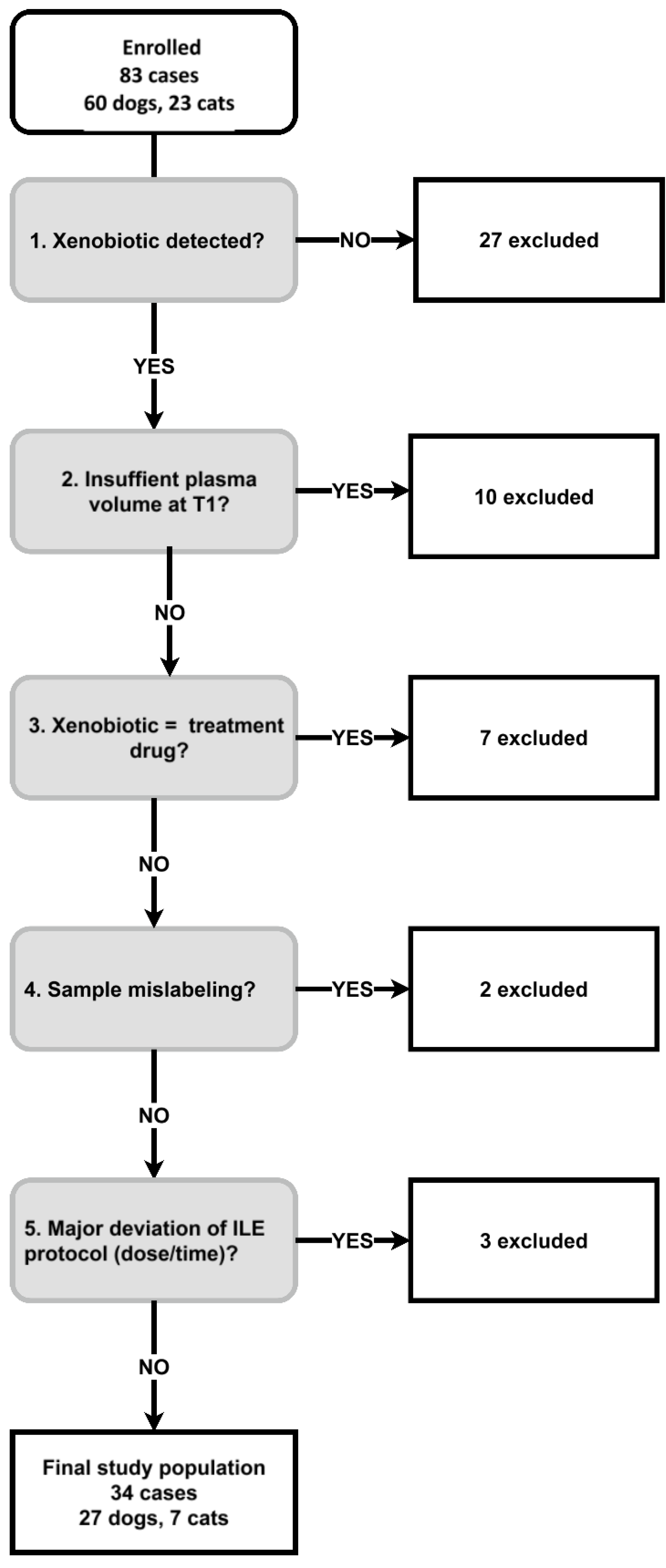
2.1. Study Design
2.2. Study Population
2.3. Treatment Protocol
2.4. Blood Sampling and Processing
2.5. Xenobiotic Identification and Quantification
2.6. Physicochemical Data
2.7. Clinical Outcome
2.8. Data Handling and Statistical Analysis
3. Results
3.1. Study Population
3.2. Treatments
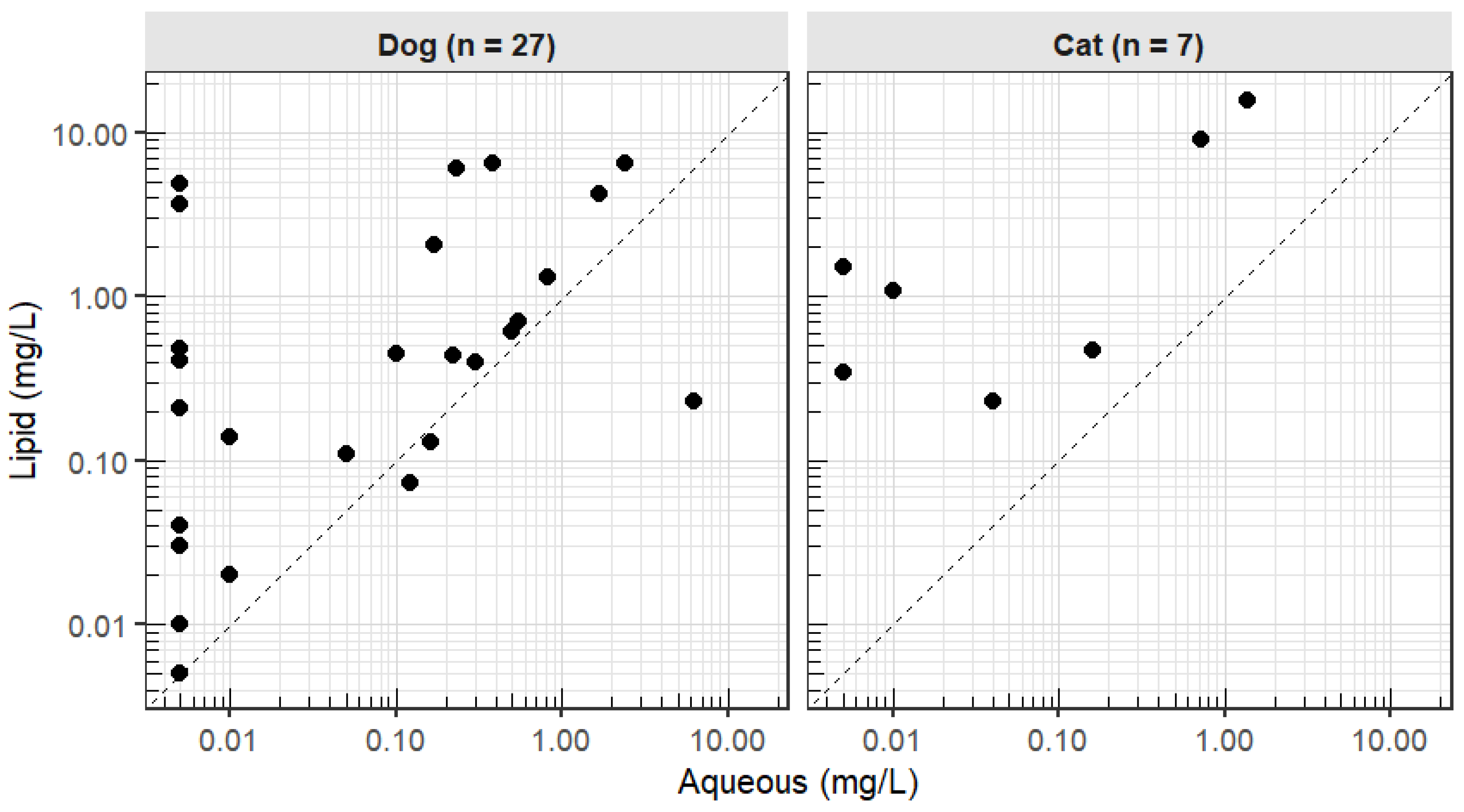
3.3. Xenobiotic Identification, Quantification and Physicochemistry
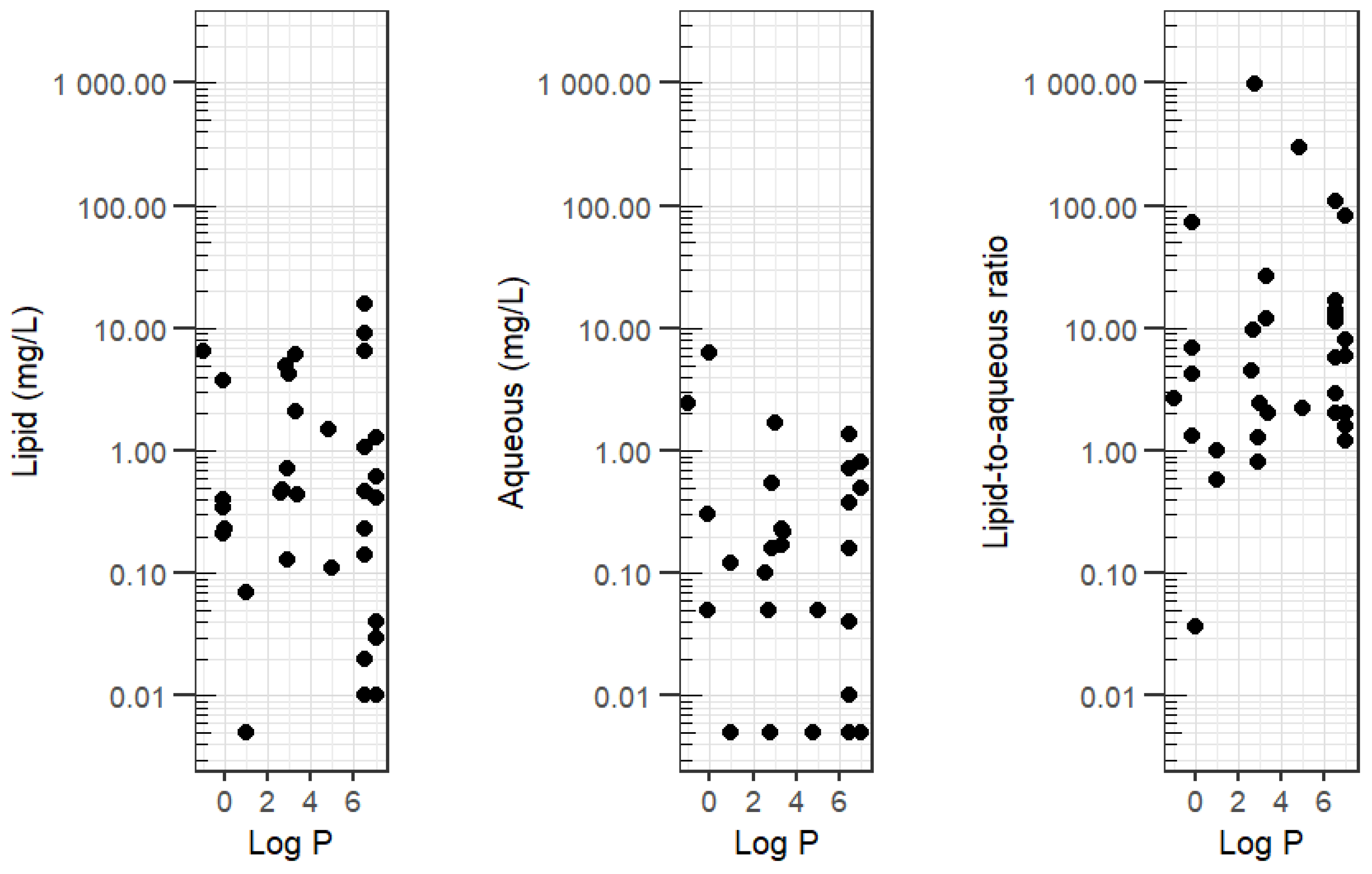
3.4. Log P and Xenobiotic Concentrations
- Log P = base-10 logarithm of the n-octanol/water partition coefficient (derived from PubChem)
- Lipid-to-aqueous ratio = lipid fraction concentration divided by the aqueous fraction concentration
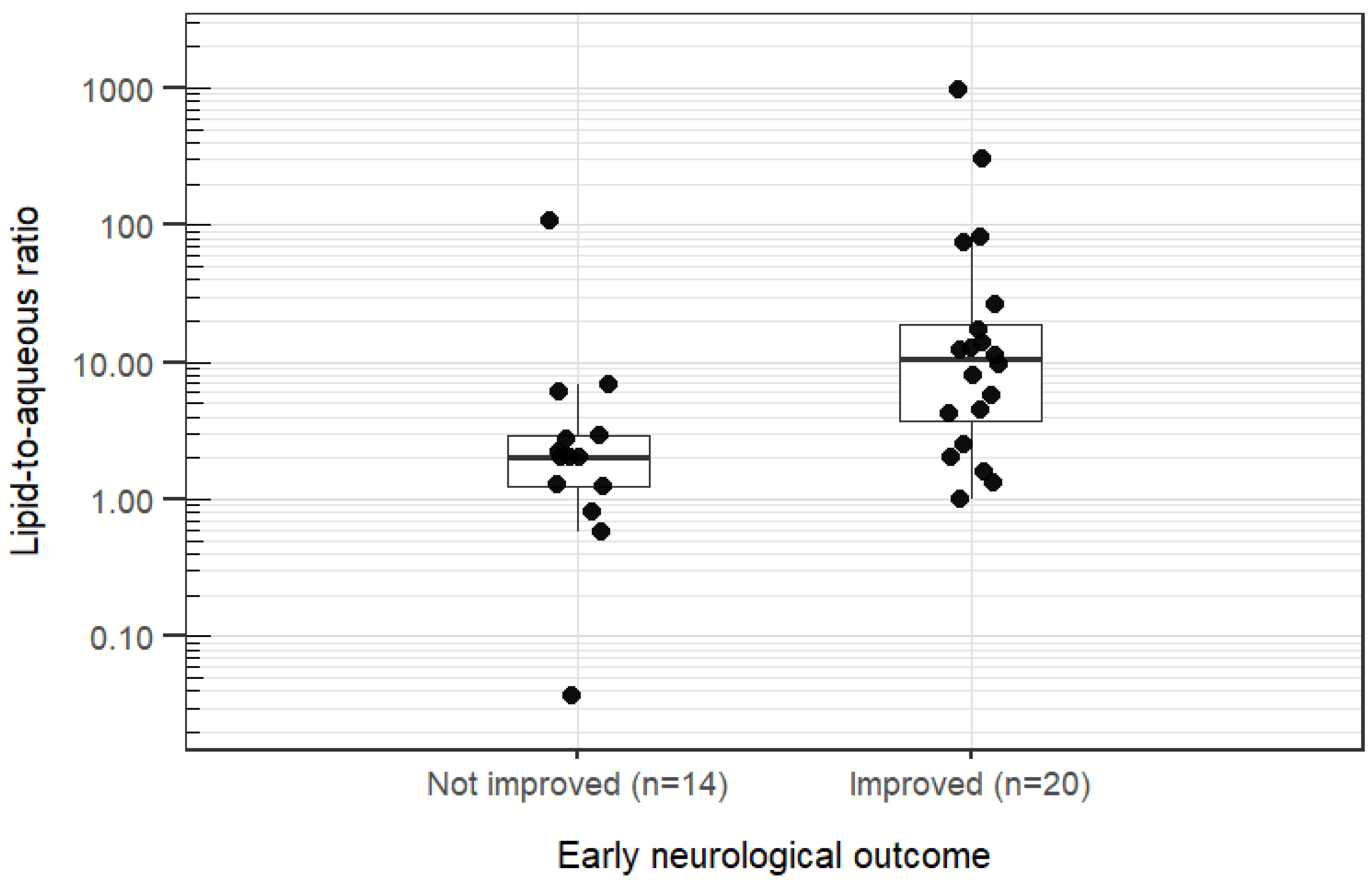
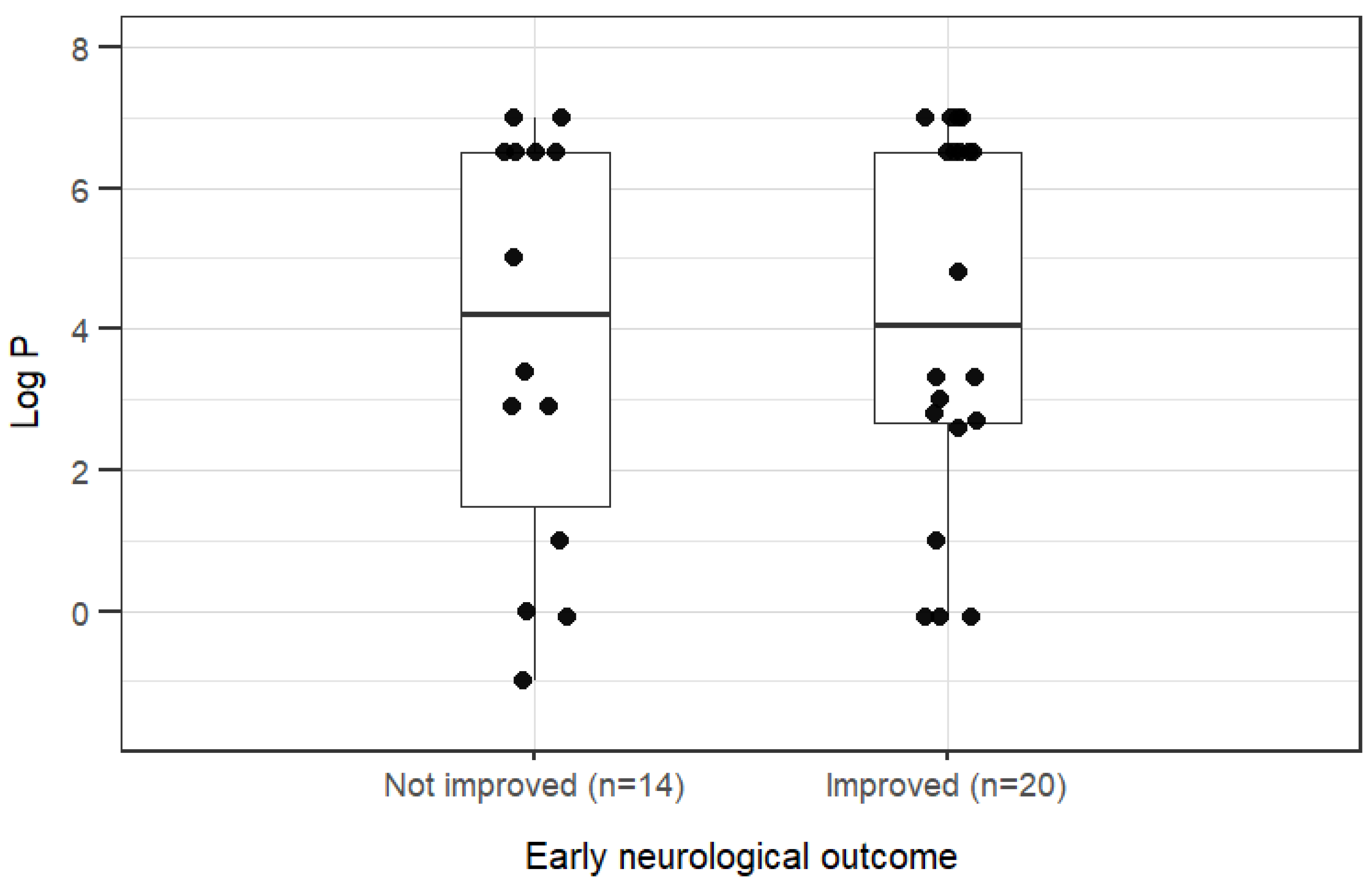
3.5. Short-Term Neurological Outcome, Xenobiotic Concentrations, and log P
- Lipid-to-aqueous ratio = lipid fraction concentration divided by the aqueous fraction concentration.
- Boxes show median and range from the first to the third quartile; whiskers extend to values within 1.5 times the interquartile range.
- Early neurological outcome was defined as the neurological improvement from the start of intravenous lipid emulsion administration to 4–6 hours afterwards. Outcome was assessed independently by two observers; disagreements were resolved by consensus.
- Log P = base-10 logarithm of the n-octanol/water partition coefficient (derived from PubChem)
- Boxes show median and range from the first to the third quartile; whiskers extend to values within 1.5 times the interquartile range.
- Early neurological outcome was defined as the neurological improvement from the start of intravenous lipid emulsion administration to 4–6 hours afterwards. Outcome was assessed independently by two observers; disagreements were resolved by consensus.
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Curti, R.; Kupper, J.; Kupferschmidt, H.; et al. Von Hanf bis Schokolade: Tiervergiftungen im Wandel der Zeit. Eine retrospektive Aufarbeitung der Kasuistik des Schweizerischen Toxikologischen Informationszentrums (1997–2006). [A retrospective study of animal poisoning reports to the Swiss Toxicological Information Centre (1997–2006)]. Schweiz Arch Tierheilkd. 2009, 151, 265–273. German. [CrossRef]
- Berny, P.; Caloni, F.; Croubels, S.; et al. Animal poisoning in Europe. Part 2: Companion animals. Vet J. 2010, 183, 255–259.
- Means, C.; Wismer, T. An overview of trends in animal poisoning cases in the United States: 2011 to 2017. Vet Clin North Am Small Anim Pract. 2018, 48, 899–907.
- Markert, C.; Heilmann, R.M.; Kiwitz, D.; et al. Intravenous lipid emulsion for the treatment of poisonings in 313 dogs and 100 cats (2016–2020). Front Vet Sci. 2023, 10, 1272705. [CrossRef]
- Taftachi, F.; Sanaei-Zadeh, H.; Sepehrian, B.; et al. Lipid emulsion improves Glasgow Coma Scale and decreases blood glucose level in the setting of acute non-local anesthetic drug poisoning—a randomized controlled trial. Eur Rev Med Pharmacol Sci. 2012, 16 (Suppl 1), 38–42.
- Robben, J.H.; Dijkman, M.A. Lipid therapy for intoxications. Vet Clin North Am Small Anim Pract. 2017, 47, 435–450.
- Gwaltney-Brant, S.; Meadows, I. Intravenous lipid emulsions in veterinary clinical toxicology. Vet Clin North Am Small Anim Pract. 2018, 48, 933–942.
- Bilvanisi, S.; Gülen, M.; Sabak, M.; et al. Intravenous lipid emulsion therapy in drug overdose and poisoning: An updated review. Eurasian J Med. 2024, 56, 205–212. [CrossRef]
- Hallberg, D.; Holm, I.; Obel, A.L.; et al. Fat emulsions for complete intravenous nutrition. Postgrad Med J. 1967, 43, 307–316.
- Weinberg, G.L.; VadeBoncouer, T.; Ramaraju, G.A.; et al. Pretreatment or resuscitation with a lipid infusion shifts the dose-response to bupivacaine-induced asystole in rats. Anesthesiology. 1998, 88, 1071–1075.
- Weinberg, G.; Ripper, R.; Feinstein, D.L.; et al. Lipid emulsion infusion rescues dogs from bupivacaine-induced cardiac toxicity. Reg Anesth Pain Med. 2003, 28, 198–202.
- Rosenblatt, M.A.; Abel, M.; Fischer, G.W.; et al. Successful use of a 20% lipid emulsion to resuscitate a patient after a presumed bupivacaine-related cardiac arrest. Anesthesiology. 2006, 105, 217–218.
- Neal, J.M.; Barrington, M.J.; Fettiplace, M.R.; et al. The third American Society of Regional Anesthesia and Pain Medicine practice advisory on local anesthetic systemic toxicity: Executive summary 2017. Reg Anesth Pain Med. 2018, 43, 113–123.
- Sirianni, A.J.; Osterhoudt, K.C.; Calello, D.P.; et al. Use of lipid emulsion in the resuscitation of a patient with prolonged cardiovascular collapse after overdose of bupropion and lamotrigine. Ann Emerg Med. 2008, 51, 412–415, 415.e1. [CrossRef]
- Cave, G.; Harvey, M. Intravenous lipid emulsion as antidote beyond local anesthetic toxicity: A systematic review. Acad Emerg Med. 2009, 16, 815–824.
- Crandell, D.E.; Weinberg, G.L. Moxidectin toxicosis in a puppy successfully treated with intravenous lipids. J Vet Emerg Crit Care (San Antonio). 2009, 19, 181–186.
- Cao, D.J.; Heard, K.; Foran, M.; et al. Intravenous lipid emulsion in the emergency department: A systematic review of recent literature. J Emerg Med. 2015, 48, 387–397. [CrossRef]
- Tampakis, K.; Vogiatzakis, N.; Kontogiannis, C.; et al. Intravenous lipid emulsion as an antidote in clinical toxicology: A systematic review. Eur Rev Med Pharmacol Sci. 2020, 24, 7138–7148.
- Jaffal, K.; Chevillard, L.; Mégarbane, B. Lipid emulsion to treat acute poisonings: Mechanisms of action, indications, and controversies. Pharmaceutics. 2023, 15, 1396. [CrossRef]
- Fettiplace, M.R.; Weinberg, G. Lipid emulsion for xenobiotic overdose: Pro. Br J Clin Pharmacol. 2023, 89, 1708–1718.
- Peacock, R.E.; Hosgood, G.; Swindells, K.L.; et al. A randomized, controlled clinical trial of intravenous lipid emulsion as an adjunctive treatment for permethrin toxicosis in cats. J Vet Emerg Crit Care (San Antonio). 2015, 25, 597–605.
- Lundgren, S.; Dreimanis, K.; Engdahl, K.; et al. Alpha-chloralose poisoning in 25 cats: Clinical picture and evaluation of treatment with intravenous lipid emulsion. J Feline Med Surg. 2024, 26, 1098612X241235776. [CrossRef]
- Levine, M.; Hoffman, R.S.; Lavergne, V.; et al. Systematic review of the effect of intravenous lipid emulsion therapy for non-local anesthetics toxicity. Clin Toxicol (Phila). 2016, 54, 194–221.
- Behnoush, A.H.; Alizadeh, N.; Emami, M.; et al. Effects of intravenous lipid emulsion administration in acute tramadol poisoning: A randomized controlled trial. J Emerg Med. 2024, 66, 154–162.
- Gosselin, S.; Hoegberg, L.C.G.; Hoffman, R.S.; et al. Evidence-based recommendations on the use of intravenous lipid emulsion therapy in poisoning. Clin Toxicol (Phila). 2016, 54, 899–923.
- Shi, K.; Xia, Y.; Wang, Q.; et al. The effect of lipid emulsion on pharmacokinetics and tissue distribution of bupivacaine in rats. Anesth Analg. 2013, 116, 804–809.
- Litonius, E.; Tarkkila, P.; Neuvonen, P.J.; Rosenberg, P.H. Effect of intravenous lipid emulsion on bupivacaine plasma concentration in humans. Anaesthesia. 2012, 67, 600–605. [CrossRef]
- Heinonen, J.A.; Litonius, E.; Backman, J.T.; Neuvonen, P.J.; Rosenberg, P.H. Intravenous lipid emulsion entraps amitriptyline into plasma and can lower its brain concentration—an experimental intoxication study in pigs. Basic Clin Pharmacol Toxicol. 2013, 113, 193–200.
- Barker, K.; Stewart, M.; Rutter, A.; Tranter, S.; Fellows, L. An in vitro study to determine the impact of lipid emulsion on partitioning of a broad spectrum of drugs associated with overdose. BJA Open. 2024, 10, 100292. [CrossRef]
- Jones, E.; Walton, S.A.; Davis, J.; Council-Troche, M. An in vitro evaluation of intravenous lipid emulsion on three common canine toxicants. Front Vet Sci. 2024, 11, 1482871. [CrossRef]
- Hwang, Y.; Sohn, J.T. Effect of lipid emulsion on neuropsychiatric drug-induced toxicity: A narrative review. Medicine (Baltimore). 2024, 103, e37612. [CrossRef]
- French, D.; Armenian, P.; Ruan, W.; Wong, A.; Drasner, K.; Olson, K.R.; et al. Serum verapamil concentrations before and after Intralipid therapy during treatment of an overdose. Clin Toxicol (Phila). 2011, 49, 340–344. [CrossRef]
- Tikhomirov, M.; Jajor, P.; Śniegocki, T.; et al. Predicting the efficacy of opioid sequestration by intravenous lipid emulsion using biologically relevant in vitro models of drug distribution. Sci Rep. 2022, 12, 18683. [CrossRef]
- Ruan, W.; French, D.; Wong, A.; Drasner, K.; Wu, A.H.B. A mixed (long- and medium-chain) triglyceride lipid emulsion extracts local anesthetic from human serum in vitro more effectively than a long-chain emulsion. Anesthesiology. 2012, 116, 334–339.
- Samuels, T.L.; Willers, J.W.; Uncles, D.R.; et al. In vitro suppression of drug-induced methaemoglobin formation by Intralipid in whole human blood: Observations relevant to the lipid sink theory. Anaesthesia. 2012, 67, 60–67.
- Niiya, T.; Litonius, E.; Petäjä, L.; Neuvonen, P.J.; Rosenberg, P.H. Intravenous lipid emulsion sequesters amiodarone in plasma and eliminates its hypotensive action in pigs. Ann Emerg Med. 2010, 56, 402–408.e2. [CrossRef]
- Lemarchand, C.; Rosoux, R.; Pénide, M.E.; Berny, P. Tissue concentrations of pesticides, PCBs and metals among ospreys (Pandion haliaetus) collected in France. Bull Environ Contam Toxicol. 2012, 88, 89–93. [CrossRef]
- R Core, T.e.a.m. R: A language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing;2023.
- Liu, Y.; Zhang, J.; Yu, P.; et al. Mechanisms and efficacy of intravenous lipid emulsion treatment for systemic toxicity from local anesthetics. Front Med (Lausanne). 2021, 8, 756866.
- Lee, S.H.; Sohn, J.T. Mechanisms underlying lipid emulsion resuscitation for drug toxicity: A narrative review. Korean J Anesthesiol. 2023, 76, 171–182.
- Mazoit, J.X.; Le Guen, R.; Beloeil, H.; et al. Binding of long-lasting local anesthetics to lipid emulsions. Anesthesiology. 2009, 110, 380–386.
- Laine, J.; Lokajová, J.; Parshintsev, J.; Kaukonen, A.M.; Kostiainen, R.; Wiedmer, S.K. Interaction of a commercial lipid dispersion and local anesthetics in human plasma: Implications for drug trapping by lipid-sinks. Anal Bioanal Chem. 2010, 396, 2599–2607. [CrossRef]
- Lee, S.H.; Kim, S.; Sohn, J.T. Lipid emulsion treatment for drug toxicity caused by non-local anesthetic drugs in pediatric patients: A narrative review. Pediatr Emerg Care. 2023, 39, 53–59.
- Bhal, S.K.; Kassam, K.; Peirson, I.G.; Payne, A.J. The rule of five revisited: Applying log D in place of log P in drug-likeness filters. Mol Pharm. 2007, 4, 556–560.
- Fernandez, A.L.; Lee, J.A.; Rahilly, L.; et al. The use of intravenous lipid emulsion as an antidote in veterinary toxicology. J Vet Emerg Crit Care (San Antonio). 2011, 21, 309–320.
- Markert, C.; Heilmann, R.M.; Kiwitz, D.; et al. A retrospective evaluation of confirmed and suspected poisonings in 166 cats between 2016 and 2020. Vet World. 2023, 16, 1940–1951.
- Doak, B.C.; Over, B.; Giordanetto, F.; Kihlberg, J. Oral druggable space beyond the rule of 5: Insights from drugs and clinical candidates. Chem Biol. 2014, 21, 1115–1142.
- Manallack, D.T. The pK(a) distribution of drugs: Application to drug discovery. Perspect Med Chem. 2007, 1, 25–38.
- Li, Z.; Li, M.; Sun, H.; et al. Prediction of drug capturing by lipid emulsions in vivo for the treatment of a drug overdose. J Control Release. 2022, 346, 148–157.
- Kiwitz, D.; Markert, C.; Dörfelt, R. Clinical effects and adverse effects of intravenous lipid emulsion treatment in dogs and cats with suspected poisoning. PLoS One. 2024, 19, e0298828.
| Xenobiotic | Dogs and Cats (n) | Dogs (n) | Cats (n) | Log P | CAS RN | PubChem CID |
| Permethrin | 9 | 4 | 5 | 6.5 | 52645-53-1 | 40326 |
| Δ9-Tetrahydrocannabinol | 6 | 6 | 0 | 7.0 | 1972-08-3 | 16078 |
| Diethyl phosphate | 2 | 1 | 1 | -0.2b | 598-02-7 | 654 |
| Alpha-chloralose | 2 | 2 | 0 | 1.0 | 15879-93-3 | 7057995 |
| Pyrilamine | 2 | 2 | 0 | 3.3 | 91-84-9 | 4992 |
| Caffeine | 2 | 2 | 0 | -0.1 | 58-08-2 | 2519 |
| Menthone | 1 | 1 | 0 | 3.0 | 89-80-5 | 26447 |
| Baclofen | 1 | 1 | 0 | -1.0 | 1134-47-0 | 2284 |
| Amitriptyline | 1 | 1 | 0 | 4.9 | 50-48-6 | 2160 |
| Metronidazole | 1 | 1 | 0 | 0b | 443-48-1 | 4173 |
| Geraniol | 1 | 1 | 0 | 2.9b | 106-24-1 | 637566 |
| Menthol | 1 | 1 | 0 | 3.2 | 89-78-1 | 16666 |
| 2,5-Dimethoxy-p-cymene | 1 | 1 | 0 | 3.4b | 14753-08-3 | 6427071 |
| Tramadol | 1 | 1 | 0 | 2.6b | 27203-92-5 | 33741 |
| Olanzapine | 1 | 1 | 0 | 3.0 | 132539-06-1 | 4585 |
| 3,6-Dimethoxy-9H-carbazole | 1 | 1 | 0 | 2.8b | 57103-01-2 | 644464 |
| Pyriproxyfen | 1 | 0 | 1 | 4.8b | 95737-68-1 | 91753 |
| Total | 34 | 27 | 7 | 4.1(-1.0 to 7.0)c |
| ρc | pValue | |
| Dogs (n = 27) | ||
| lipid | -0.24 | 0.22 |
| aqueous | -0.31 | 0.11 |
| lipid-to-aqueous ratio | 0.14 | 0.48 |
| Dogs + Cats (N = 34) | ||
| lipid | -0.12 | 0.50 |
| aqueous | -0.26 | 0.14 |
| lipid-to-aqueous ratio | 0.16 | 0.36 |
| GMR (95% CI)b | pValue | |
| Dogs (n = 27) | ||
| lipid | 0.80 (0.60–1.07) | 0.14 |
| aqueous | 0.71 (0.54–0.94) | 0.02 |
| lipid-to-aqueous ratioc | 1.13 (0.86–1.48) | 0.39 |
| Dogs + cats (N = 34) | ||
| lipid | 0.91 (0.70–1.19) | 0.50 |
| aqueous | 0.78 (0.61–1.01) | 0.07 |
| lipid-to-aqueous ratio | 1.17 (0.92–1.48) | 0.22 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




