Submitted:
18 February 2026
Posted:
18 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
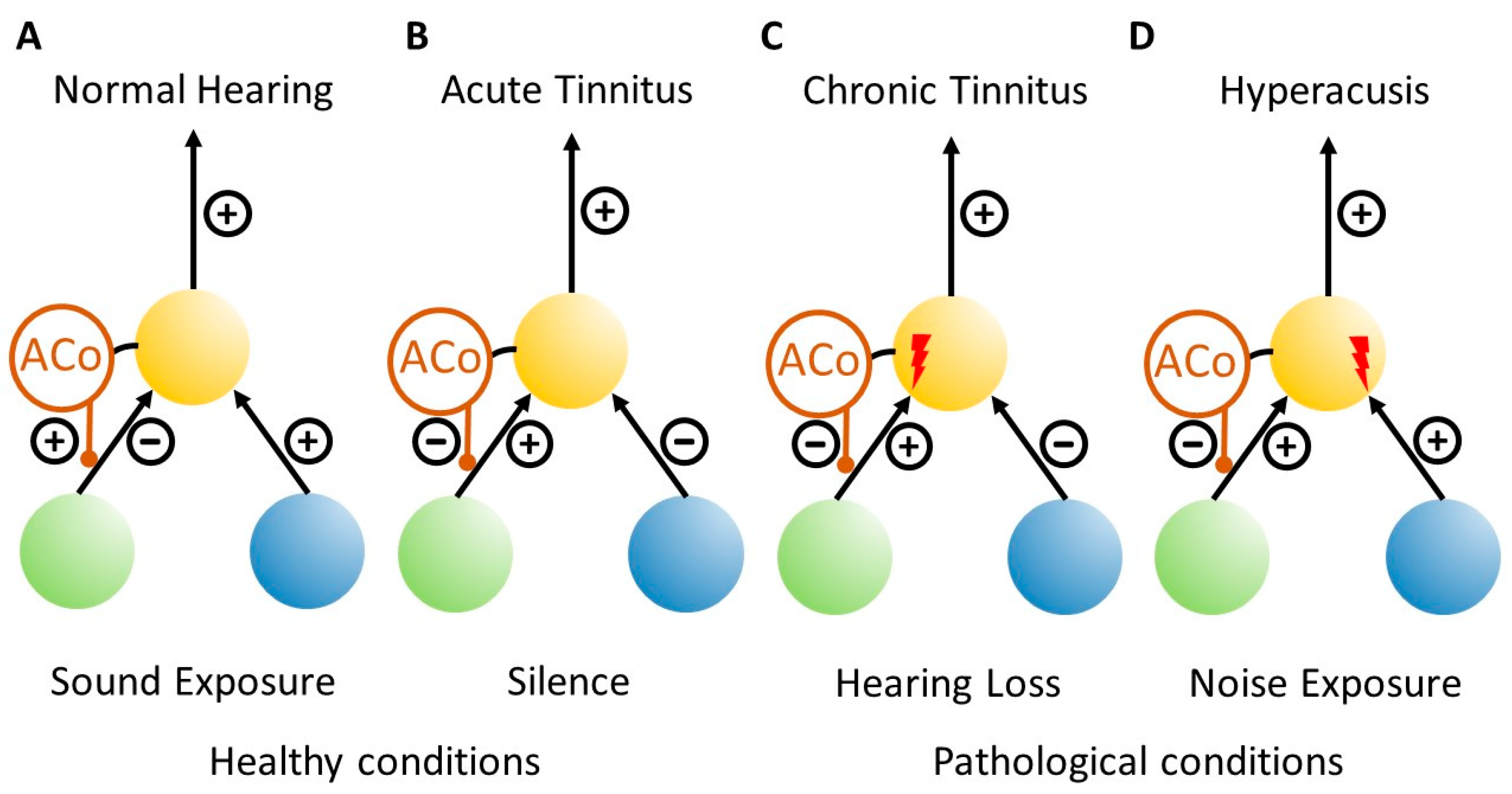
1.1. The Erlangen Model of Tinnitus Development
1.2. Classical Conditioning
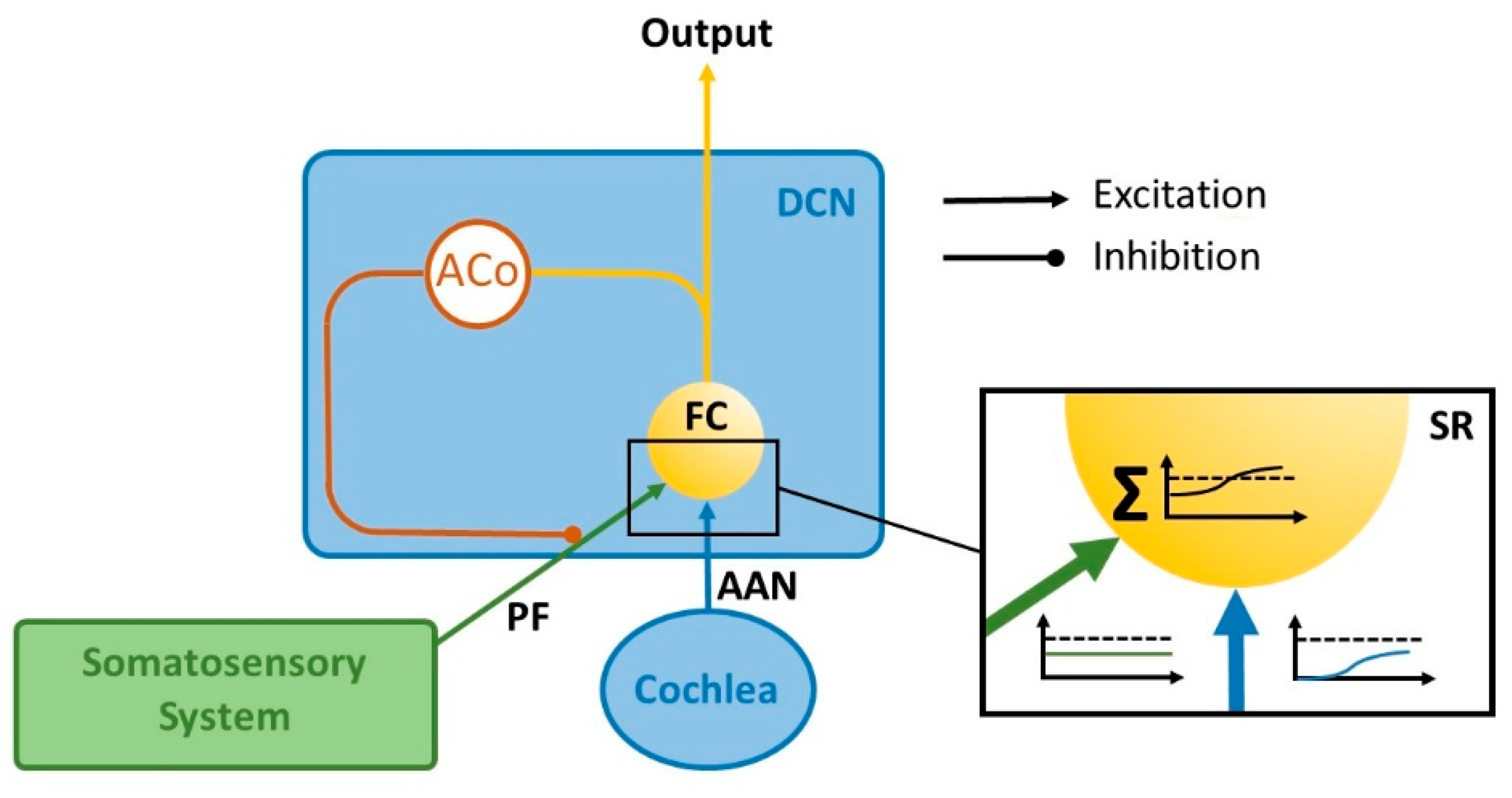
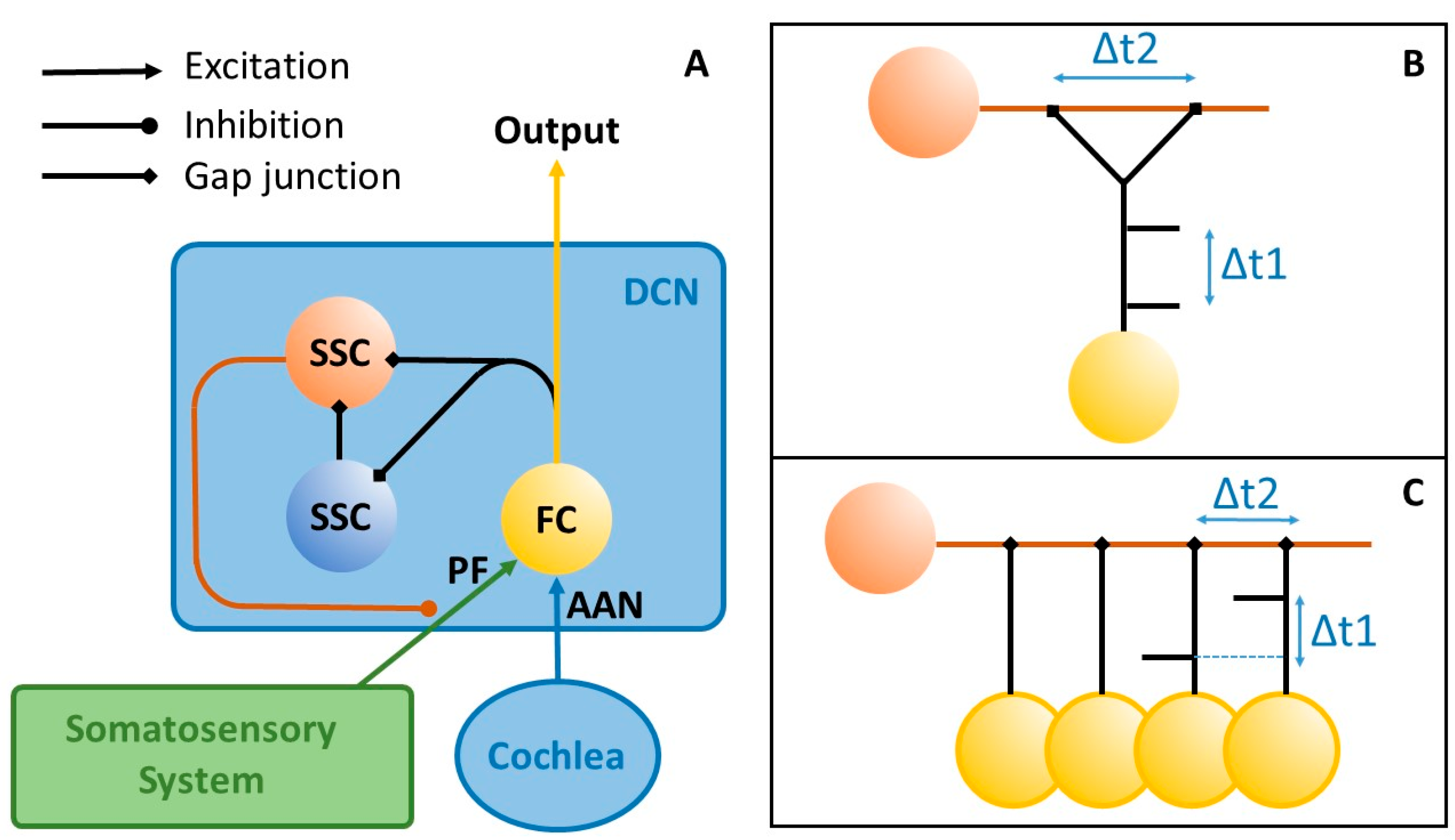
2. Materials and Methods
3. Results
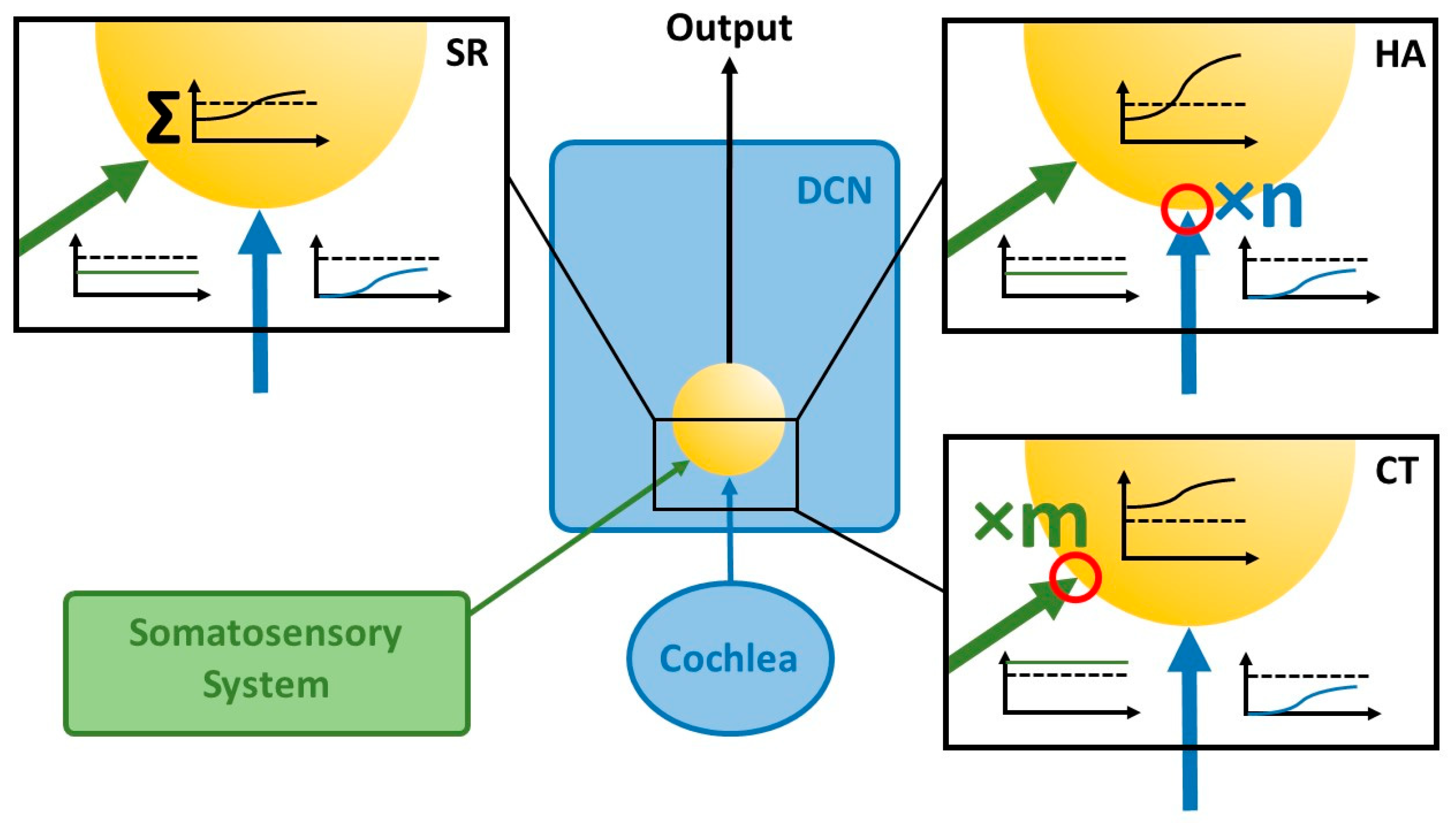
3.1. Development of Chronic Tinnitus
3.2. Development of Hyperacusis
4. Discussion
5. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AAN | auditory afferent nerve |
| ACo | autocorrelation |
| CR | conditioned response |
| CS | conditioned stimulus |
| CT | chronic tinnitus |
| DCN | dorsal cochlear nucleus |
| FC | fusiform cells |
| HA | hyperacusis |
| LTD | Long term depression |
| LTP | long term potentiation |
| NMDA | N-methyl-D-aspartate |
| PF | parallel fiber |
| SR | stochastic resonance |
| SSC | superficial stellate cell |
| UR | unconditioned response |
| US | unconditioned stimulus |
References
- Prengel, J.; Dobel, C.; Guntinas-Lichius, O. Tinnitus. Laryngo-Rhino-Otologie 2023, 102, 132-145. [CrossRef]
- Boecking, B.; von Sass, J.; Sieveking, A.; Schaefer, C.; Brueggemann, P.; Rose, M.; Mazurek, B. Tinnitus-related distress and pain perceptions in patients with chronic tinnitus–Do psychological factors constitute a link? PloS one 2020, 15, e0234807. [CrossRef]
- Biswas, R.; Lugo, A.; Akeroyd, M.A.; Schlee, W.; Gallus, S.; Hall, D.A. Tinnitus prevalence in Europe: a multi-country cross-sectional population study. The Lancet Regional Health - Europe 2022, 12, 100250. [CrossRef]
- Neff, P.; Simões, J.; Psatha, S.; Nyamaa, A.; Boecking, B.; Rausch, L.; Dettling-Papargyris, J.; Funk, C.; Brueggemann, P.; Mazurek, B. The impact of tinnitus distress on cognition. Scientific reports 2021, 11, 1-9. [CrossRef]
- Cederroth, C.R.; Lugo, A.; Edvall, N.K.; Lazar, A.; Lopez-Escamez, J.-A.; Bulla, J.; Uhlen, I.; Hoare, D.J.; Baguley, D.M.; Canlon, B. Association between hyperacusis and tinnitus. Journal of Clinical Medicine 2020, 9, 2412. [CrossRef]
- Pienkowski, M. Rationale and efficacy of sound therapies for tinnitus and hyperacusis. Neuroscience 2019, 407, 120-134. [CrossRef]
- Bigras, C.; Villatte, B.; Duda, V.; Hébert, S. The electrophysiological markers of hyperacusis: a scoping review. International journal of audiology 2023, 62, 489-499. [CrossRef]
- Baguley, D.M. Hyperacusis: an overview. In Proceedings of the Seminars in Hearing, 2014; pp. 074-083.
- Schilling, A.; Sedley, W.; Gerum, R.; Metzner, C.; Tziridis, K.; Maier, A.; Schulze, H.; Zeng, F.-G.; Friston, K.J.; Krauss, P. Predictive coding and stochastic resonance as fundamental principles of auditory phantom perception. Brain 2023. [CrossRef]
- Schaette, R.; McAlpine, D. Tinnitus with a normal audiogram: physiological evidence for hidden hearing loss and computational model. J Neurosci 2011, 31, 13452-13457. [CrossRef]
- Noreña, A.J.; Chery-Croze, S. Enriched acoustic environment rescales auditory sensitivity. Neuroreport 2007, 18, 1251-1255. [CrossRef]
- Knipper, M.; Van Dijk, P.; Nunes, I.; Ruttiger, L.; Zimmermann, U. Advances in the neurobiology of hearing disorders: recent developments regarding the basis of tinnitus and hyperacusis. Prog Neurobiol 2013, 111, 17-33. [CrossRef]
- Rauschecker, J.P. The Frontostriatal Gating Model of Tinnitus. In Textbook of Tinnitus; Springer: 2024; pp. 221-230.
- Sedley, W.; Friston, K.J.; Gander, P.E.; Kumar, S.; Griffiths, T.D. An integrative tinnitus model based on sensory precision. Trends in neurosciences 2016, 39, 799-812. [CrossRef]
- Schilling, A.; Krauss, P. The Bayesian brain: world models and conscious dimensions of auditory phantom perception. 2024, 132, 317-318. [CrossRef]
- Yasoda-Mohan, A.; Chen, F.; Ó Sé, C.; Allard, R.; Ost, J.; Vanneste, S. Phantom perception as a Bayesian inference problem: a pilot study. Journal of Neurophysiology 2024, 131, 1311-1327. [CrossRef]
- Zeng, F.-G. An active loudness model suggesting tinnitus as increased central noise and hyperacusis as increased nonlinear gain. Hearing research 2013, 295, 172-179. [CrossRef]
- Kaltenbach, J.A. The dorsal cochlear nucleus as a contributor to tinnitus: mechanisms underlying the induction of hyperactivity. Progress in brain research 2007, 166, 89-106. [CrossRef]
- Krauss, P.; Schilling, A.; Tziridis, K.; Schulze, H. Models of tinnitus development: From cochlea to cortex. HNO 2019. [CrossRef]
- Tziridis, K.; Forster, J.; Buchheidt-Dorfler, I.; Krauss, P.; Schilling, A.; Wendler, O.; Sterna, E.; Schulze, H. Tinnitus development is associated with synaptopathy of inner hair cells in Mongolian gerbils. Eur J Neurosci 2021, 54, 4768-4780. [CrossRef]
- Kujawa, S.G.; Liberman, M.C. Adding insult to injury: cochlear nerve degeneration after "temporary" noise-induced hearing loss. J Neurosci 2009, 29, 14077-14085. [CrossRef]
- Kaltenbach, J.A.; Afman, C.E. Hyperactivity in the dorsal cochlear nucleus after intense sound exposure and its resemblance to tone-evoked activity: a physiological model for tinnitus. Hearing research 2000, 140, 165-172. [CrossRef]
- Hofmeier, B.; Wertz, J.; Refat, F.; Hinrichs, P.; Saemisch, J.; Singer, W.; Rüttiger, L.; Klose, U.; Knipper, M.; Wolpert, S. Functional biomarkers that distinguish between tinnitus with and without hyperacusis. Clinical and Translational Medicine 2021, 11, e378. [CrossRef]
- Koops, E.; van Dijk, P. Hyperacusis in tinnitus patients relates to enlarged subcortical and cortical responses to sound except at the tinnitus frequency. Hearing research 2021, 401, 108158. [CrossRef]
- Almond, L.M.; Patel, K.; Rejali, D. Transient auditory dysfunction: a description and study of prevalence. Ear, Nose & Throat Journal 2013, 92, 352-356. [CrossRef]
- Turrigiano, G.G. Homeostatic plasticity in neuronal networks: the more things change, the more they stay the same. Trends in neurosciences 1999, 22, 221-227. [CrossRef]
- Zenke, F.; Gerstner, W.; Ganguli, S. The temporal paradox of Hebbian learning and homeostatic plasticity. Current opinion in neurobiology 2017, 43, 166-176. [CrossRef]
- Krauss, P.; Tziridis, K.; Metzner, C.; Schilling, A.; Hoppe, U.; Schulze, H. Stochastic Resonance Controlled Upregulation of Internal Noise after Hearing Loss as a Putative Cause of Tinnitus-Related Neuronal Hyperactivity. Front Neurosci 2016, 10, 597. [CrossRef]
- Schulze, H.; Schilling, A.; Krauss, P.; Tziridis, K. The Erlangen model of tinnitus development-New perspective and treatment strategy. HNO 2023, 71, 662-668. [CrossRef]
- Shore, S.E.; Zhou, J. Somatosensory influence on the cochlear nucleus and beyond. Hearing research 2006, 216, 90-99. [CrossRef]
- Krauss, P.; Metzner, C.; Schilling, A.; Schutz, C.; Tziridis, K.; Fabry, B.; Schulze, H. Adaptive stochastic resonance for unknown and variable input signals. Sci Rep 2017, 7, 2450. [CrossRef]
- Licklider, J.C.R. A duplex theory of pitch perception. Experientia 1951, 7, 128-134. [CrossRef]
- Apostolides, P.F.; Trussell, L.O. Superficial stellate cells of the dorsal cochlear nucleus. Frontiers in Neural Circuits 2014, 8, 63. [CrossRef]
- Balmer, T.S.; Trussell, L.O. Descending axonal projections from the inferior colliculus target nearly all excitatory and inhibitory cell types of the dorsal cochlear nucleus. Journal of Neuroscience 2022, 42, 3381-3393. [CrossRef]
- Trussell, L.O.; Oertel, D. Microcircuits of the dorsal cochlear nucleus. In The Mammalian Auditory Pathways: Synaptic Organization and Microcircuits; Springer: 2018; pp. 73-99.
- Tzounopoulos, T.; Rubio, M.E.; Keen, J.E.; Trussell, L.O. Coactivation of pre-and postsynaptic signaling mechanisms determines cell-specific spike-timing-dependent plasticity. Neuron 2007, 54, 291-301. [CrossRef]
- Wu, Q.-W.; Tang, Z.-Q. Focusing on the emerging role of kainate receptors in the Dorsal Cochlear Nucleus (DCN) and cerebellum. International Journal of Molecular Sciences 2023, 24, 1718. [CrossRef]
- Wever, E.G.; Bray, C.W. The perception of low tones and the resonance-volley theory. The Journal of Psychology 1937, 3, 101-114. [CrossRef]
- Larkum, M.E.; Rioult, M.G.; Luscher, H.-R. Propagation of action potentials in the dendrites of neurons from rat spinal cord slice cultures. Journal of neurophysiology 1996, 75, 154-170. [CrossRef]
- Blackstad, T.; Osen, K.; Mugnaini, E. Pyramidal neurones of the dorsal cochlear nucleus: a Golgi and computer reconstruction study in cat. Neuroscience 1984, 13, 827-854. [CrossRef]
- Bi, G.-q.; Poo, M.-m. Synaptic modification by correlated activity: Hebb's postulate revisited. Annual review of neuroscience 2001, 24, 139-166. [CrossRef]
- Oertel, D.; Young, E.D. What's a cerebellar circuit doing in the auditory system? Trends in neurosciences 2004, 27, 104-110. [CrossRef]
- Knipper, M.; Mazurek, B.; van Dijk, P.; Schulze, H. Too Blind to See the Elephant? Why Neuroscientists Ought to Be Interested in Tinnitus. J Assoc Res Otolaryngol 2021, 22, 609-621. [CrossRef]
- Refat, F.; Wertz, J.; Hinrichs, P.; Klose, U.; Samy, H.; Abdelkader, R.M.; Saemisch, J.; Hofmeier, B.; Singer, W.; Rüttiger, L. Co-occurrence of hyperacusis accelerates with tinnitus burden over time and requires medical care. Frontiers in Neurology 2021, 12, 627522. [CrossRef]
- Vielsmeier, V.; Santiago Stiel, R.; Kwok, P.; Langguth, B.; Schecklmann, M. From acute to chronic tinnitus: pilot data on predictors and progression. Frontiers in Neurology 2020, 11, 997. [CrossRef]
- Wake, N.; Shiramatsu, T.I.; Takahashi, H. Map plasticity following noise exposure in auditory cortex of rats: implications for disentangling neural correlates of tinnitus and hyperacusis. Frontiers in Neuroscience 2024, 18, 1385942. [CrossRef]
- Shore, S.E.; Koehler, S.; Oldakowski, M.; Hughes, L.F.; Syed, S. Dorsal cochlear nucleus responses to somatosensory stimulation are enhanced after noise-induced hearing loss. European Journal of Neuroscience 2008, 27, 155-168. [CrossRef]
- Tagoe, T.; Deeping, D.; Hamann, M. Saturation of long-term potentiation in the dorsal cochlear nucleus and its pharmacological reversal in an experimental model of tinnitus. Experimental Neurology 2017, 292, 1-10. [CrossRef]
- Suckfüll, M.; Althaus, M.; Ellers-Lenz, B.; Gebauer, A.; Görtelmeyer, R.; Jastreboff, P.J.; Moebius, H.J.; Rosenberg, T.; Russ, H.; Wirth, Y. A randomized, double-blind, placebo-controlled clinical trial to evaluate the efficacy and safety of neramexane in patients with moderate to severe subjective tinnitus. BMC Ear, Nose and Throat Disorders 2011, 11, 1. [CrossRef]
- Salembier, L.; De Ridder, D.; Van de Heyning, P. The use of flupirtine in treatment of tinnitus. Acta Oto-Laryngologica 2006, 126, 93-95. [CrossRef]
- Figueiredo, R.R.; Langguth, B.; de Oliveira, P.M.; de Azevedo, A.A. Tinnitus treatment with memantine. Otolaryngology—Head and Neck Surgery 2008, 138, 492-496. [CrossRef]
- Sedley, W.; Alter, K.; Gander, P.E.; Berger, J.; Griffiths, T.D. Exposing pathological sensory predictions in tinnitus using auditory intensity deviant evoked responses. Journal of Neuroscience 2019, 39, 10096-10103. [CrossRef]
- Rauschecker, J.P.; May, E.S.; Maudoux, A.; Ploner, M. Frontostriatal gating of tinnitus and chronic pain. Trends in cognitive sciences 2015, 19, 567-578. [CrossRef]
- Jastreboff, P.J. Tinnitus retraining therapy. Progress in brain research 2007, 166, 415-423. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



