Submitted:
17 February 2026
Posted:
25 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
The Study Site


The Study Species and Experimental Rocks
The Experiments
3. Results
4. Discussion
4.1. Salinity as a Contextual Movement Cue Under Constrained Conditions
4.2. Constraints Imposed by Experimental Isolation
4.3. Modulating Effects of Rock Features and Water Depth
4.4. Scale Mismatch and Behavioural Interpretation
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| DBML | Discovery Bay Marine Lab |
Appendix A




References
- Denny, M. W. (1987). Life in the maelstrom: The biomechanics of wave-swept rocky shores. Trends in Ecology & Evolution, 2(3), 61–66. [CrossRef]
- Menge, B. A., & Sutherland, J. P. (1987). Community Regulation: Variation in Disturbance, Competition, and Predation in Relation to Environmental Stress and Recruitment. In Source: The American Naturalist (Vol. 130, Issue 5).
- Denny, M. W., & Paine, R. T. (1998). Celestial Mechanics, Sea-Level Changes, and Intertidal Ecology. In Reference: Biol. Bull (Vol. 194). [CrossRef]
- Davies, M. S., & Beckwith, P. (1999). Role of mucus trails and trail-following in the behaviour and nutrition of the periwinkle Littorina littorea. In Source: Marine Ecology Progress Series (Vol. 179). [CrossRef]
- Chapman, M. G. (2000). A comparative study of differences among species and patches of habitat on movements of three species of intertidal gastropods. In Journal of Experimental Marine Biology and Ecology (Vol. 244). [CrossRef]
- Little, C., Williams, G. A., & Trowbridge, C. D. (2009). The Biology of Rocky Shores. Oxford University PressOxford. [CrossRef]
- Thompson, D. The Online Guide to the Animals of Trinidad and Tobago: Cenchritis muricatus (Beaded Periwinkle). University of the West Indies, 2015. Available online: https://www.uwi.edu.
- Emson, R. H., Morritt, D., Andrews, E. B., & Young, C. M. (2002). Life on a hot dry beach: Behavioural, physiological, and ultrastructural adaptations of the littorinid gastropod Cenchritis (Tectarius) muricatus. Marine Biology, 140(4), 723–732. [CrossRef]
- Piovia-Scott, J. (2009). Effects of disturbance on Cenchritis muricatus (beaded periwinkle) populations on small islands in the Bahamas. In BULLETIN OF MARINE SCIENCE (Vol. 84, Issue 3).
- Rosewater, J. (1963). Resistance to desiccation in dormancy by Tectarius muricatus. Nautilus, 76, 111.
- Lima And, S. L., & Dill, L. M. (n.d.). Behavioral decisions made under the risk of predation: a review and prospectus.
- Brown, J. S., Laundre, J. W., & Gurung, M. (1999). The Ecology of Fear: Optimal Foraging, Game Theory, and Trophic Interactions. Journal of Mammalogy, 80(2), 385–399. [CrossRef]
- Helmuth, B., Broitman, B. R., Blanchette, C. A., Gilman, S., Halpin, P., Harley, C. D. G., O’Donnell, M. J., Hofmann, G. E., Menge, B., & Strickland, D. (2006). Mosaic patterns of thermal stress in the rocky intertidal zone: Implications for climate change. Ecological Monographs, 76(4), 461–479. [CrossRef]
- Underwood, A. J., & Chapman, M. G. (2003). Power, precaution, Type II error and sampling design in assessment of environmental impacts. Journal of Experimental Marine Biology and Ecology, 296(1), 49–70. [CrossRef]
- Resetarits, W. J. (2005). Habitat selection behaviour links local and regional scales in aquatic systems. Ecology Letters, 8(5), 480–486. [CrossRef]
- Moisez, E., & Seuront, L. (2020). Deciphering the known unknowns in the behavioural ecology of the intertidal gastropod Littorina littorea. Journal of Experimental Marine Biology and Ecology, 524. [CrossRef]
- Marković, M., Šoštarić, J., Kojić, A., Popović, B., Bubalo, A., Bošnjak, D., & Stanisavljević, A. (2022). Zinnia (Zinnia elegans L.) and Periwinkle (Catharanthus roseus (L.) G. Don) Responses to Salinity Stress. Water (Switzerland), 14(7). [CrossRef]
- Chapperon, C., & Seuront, L. (2011). Variability in the motion behaviour of intertidal gastropods: ecological and evolutionary perspectives. Journal of the Marine Biological Association of the United Kingdom, 91(1), 237–244. [CrossRef]
- Underwood, A. J. (2000). Experimental ecology of rocky intertidal habitats: what are we learning? In Journal of Experimental Marine Biology and Ecology (Vol. 250). [CrossRef]

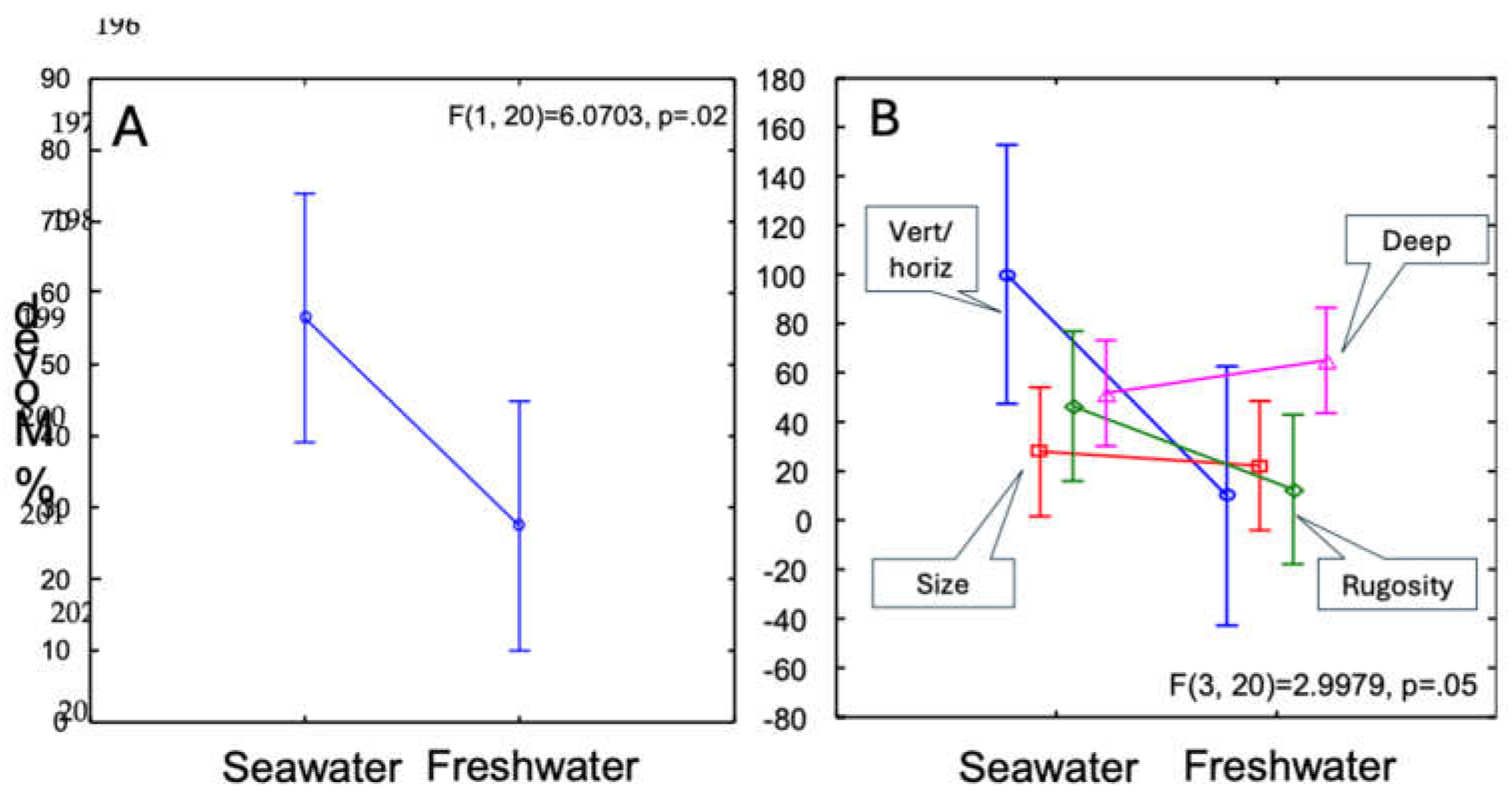
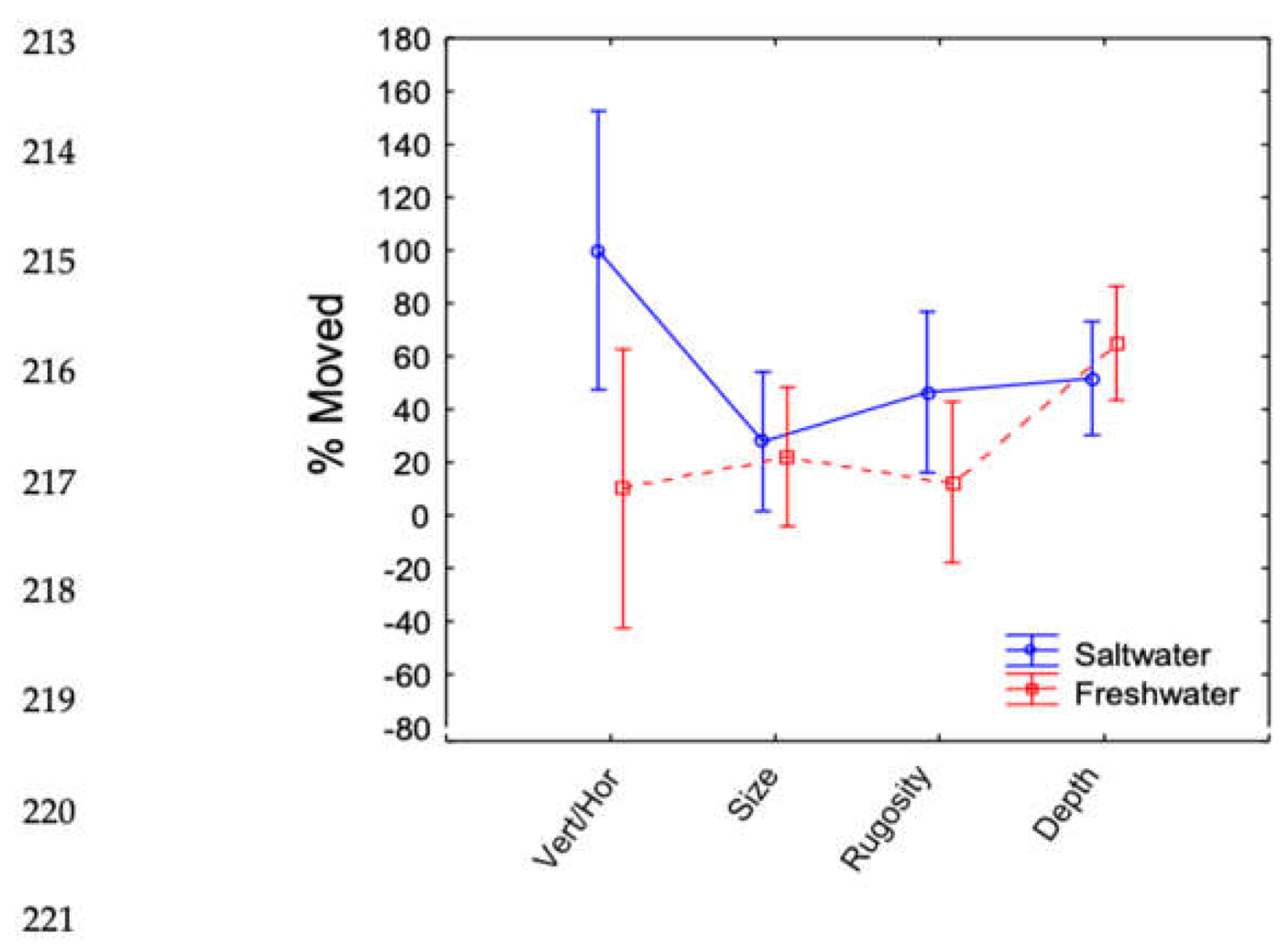
| SS | Degr. of Freedom | MS | F | P-value | |
| Intercept | 32184.04 | 1 | 32184.04 | 50.53258 | 0.000001 |
| Experiment | 6786.20 | 3 | 2262.07 | 3.55170 | 0.0329 |
| Fresh/Salt | 3866.14 | 1 | 3866.14 | 6.070 | 0.0229 |
| Experiment*Fresh/Salt | 5728.09 | 3 | 1909.36 | 2.998 | 0.0549 |
| Error | 12737.94 | 20 | 636.90 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




