1. Introduction
Tuberculosis (TB) is a significant problem in many developing countries, including South Africa. The World Health Organization (WHO) estimates that in 2022, there were 1.13 million deaths caused by TB worldwide, a slight decrease from TB deaths in 2022-2021, but this number is almost double the number of deaths caused by HIV/AIDS [
1]. The number of individuals who contracted the disease worldwide increased in 2022 to 10.6 million from 10.3 million, indicating an upward trend for the disease. South Africa is classified as one of the highest TB burden countries despite having achieved at least a 50% reduction in TB incidences between 2015 and 2022 [
1]. This highlights the need to aggressively search for new potent anti-TB drugs to achieve the 2035 End TB strategy.
Wild animals like baboons and chimpanzees consume various plants in the forest for nourishment and, at times, medicinal purposes [
2]. A recent study observed that wild chimpanzees consumed nutrient-poor plants or plant parts such as dead wood; this observation was followed by a collection of samples from plants that chimpanzees consumed with no nutritional value. These plants were tested for antibacterial activity, and the results of this study showed that some of the plants had strong antibacterial activity against
Staphylococcus aureus [
2]. These results indicate both the high level of intelligence and self-reliance of these animals.
It is widely documented that some plants worldwide have antimycobacterial activity with specific secondary metabolite groups associated with these antimycobacterial activities, and some medicinal plants showed activity against the extensively drug-resistant (XDR-TB) strains of
Mycobacterium tuberculosis [
3,
4,
5,
6]. In the Eastern Cape, locals also utilize various plants for medicinal purposes, with scientific research confirming that some of these plants possess antimycobacterial properties [
7]. However, identifying these plants for antimicrobial testing relies heavily on traditional knowledge handed down orally through generations. This method of transmission has sometimes resulted in the loss of knowledge about certain medicinal plants, particularly when the individual possessing this knowledge passed away before passing it on.
This study aims to analyse the phytochemical composition of wild baboon faecal matter and assess its antimycobacterial properties. Given that wild baboons, like their relatives, the chimpanzees, are likely to consume medicinal plants, identifying biologically active phytochemicals or even discovering novel compounds unknown to man in their faecal matter is possible.
Pharmacokinetics has demonstrated that a considerable portion of orally administered drugs is not absorbed into the bloodstream but excreted in the faecal matter [
8]. The inability of the gut to absorb all the orally administered drug concentration is due to various factors, such as efflux pumps on the intestinal membrane and the thick mucus layer, which form a restrictive barrier that drugs must first overcome to reach blood circulation [
9,
10]. Therefore, these factors increase our chances of discovering bioactive phytochemicals in the wild baboon faecal matter.
2. Results
2.1. Qualitative Phytochemical Screening
In this study, qualitative phytochemical screening was performed on the ethanol extract of the wild baboon faecal matter. Plant secondary metabolites, which belong to the following groups: tannins, alkaloids, glycosides, terpenoids and flavonoids, were detected in the ethanol extract, as displayed in
Table 1.
2.2. Gas Chromatography-Mass Spectrophotometer Analysis of Phytochemicals
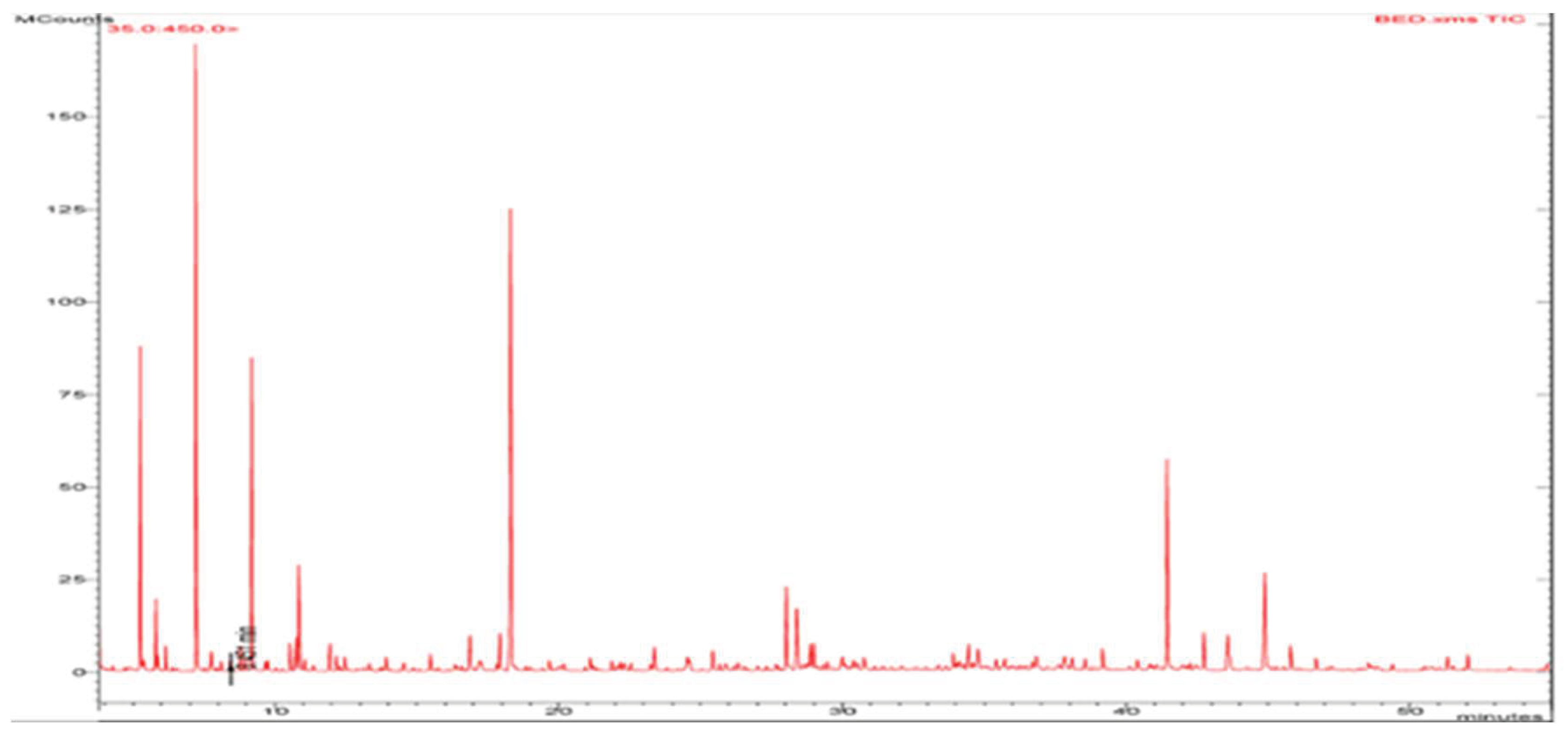
GC-MS analysis was performed on the Hydro-distillation extract of wild baboon faecal matter to identify the different types of plant secondary metabolites present. GC-MS chromatogram of the extract is shown in
Figure 1.
A total of 26 compounds were identified in the hydrodistillation extract of wild baboon fecal matter, as presented in
Table 2. The major constituents of the volatile oil were thialdine (21.029%), α-pinene (18.324%), 2-pentylfuran (10.446%), n-heptadecan-1-ol (9.178%), and hexyl chloroformate (7.114%).
2.3. Antimycobacterial Assay
The antimycobacterial activity of the wild baboon faecal extracts was assessed using the resazurin microtiter assay against
Mycobacterium smegmatis. The hydrodistillation extract (essential oil) exhibited a minimum inhibitory concentration (MIC) of 0.5 mg/mL, whereas the ethanol extract showed a higher MIC value of 2 mg/mL. Rifampicin, used as the positive control, confirmed assay validity, while DMSO showed no inhibitory effect. The microplate results following resazurin addition are presented in
Figure 2.
3. Discussion
Our study aimed to evaluate the presence of phytochemicals in the wild baboon faecal matter and their antimycobacterial activity. The first component of the study, phytochemical analysis, was performed with quantitative analysis and GC-MS. Quantitative analysis revealed the presence of tannins, alkaloids, glycosides, terpenoids and flavonoids in the wild baboon faecal matter. These results confirm our hypothesis that the gut does not absorb all ingested food or orally administered drugs due to the factors we listed above, and we are likely to find plant secondary metabolites in baboon faecal matter.
GCMS analysis revealed the presence of 26 compounds, including alpha-pinene, limonene eucalyptol, and n-heptadecanol. Studies performed on plant extracts with alpha-pinene, borneol, and limonene showed great antimicrobial activity [
14]. Another study showed that alpha-pinene has significant antimycobacterial activity with MIC of 8 µg/ml against
M. tuberculosis drug-sensitive strain H37Ra [
15]. Other compounds detected in high concentrations were 2-Pentylfuran and Eucalyptol. 2-Pentylfuran has been reported as a deterrent for spotted-wing drosophila insects and a safe alternative to toxic insecticides as it is an approved food additive [
16]. Eucalyptol is known to have positive effects on respiratory conditions, including anti-inflammatory and bronchodilatory effects. Multiple clinical trials have been conducted on individuals suffering from respiratory conditions like rhinosinusitis, bronchitis, asthma, and COPD, yielding positive outcomes [
17]. When eucalyptol was combined with citronellol and citronellal in an artificial mixture, the mixture showed over 90% inhibition of airborne TB [
18]. n-heptadecanol-1, another compound found in significant quantities in our essential oil extracts, has also been reported to have anti-inflammatory and antimicrobial activity [
19].
The second component of our study was to evaluate the antimycobacterial activity of the wild baboon faecal matter extracts. Our study shows that wild baboon faecal matter contains phytochemicals with antimycobacterial activity. This was demonstrated by essential Oils and ethanol extracts, which had MIC of 0,5 mg/ml and 2 mg/ml, respectively, against
M. smegmatis. Rifampicin was used as a positive control with MIC of 2,5 mg/mL and MBC of 5 mg/mL. A negative control DMSO showed no inhibition for both assays. The results obtained in this study are in agreement with those obtained by Boussamba-Digombou et al. (2022), whereby essential oils from different plant species showed significant inhibition with some species having MIC as little as 0,25 mg/mL and a mode MIC of 2 mg/mL against
M. smegmatis [
20]. The activity of the wild baboon faecal extracts could be attributed to the plant secondary metabolites identified in the GC-MS analysis, as some of the compounds have been reported in the literature to have anti-mycobacterium tuberculosis activity. Our study shows that phytochemicals excreted by wild baboons through the faecal matter may be unmodified and functional; if modified, the modification has not compromised their antimybacterial activity. Our study shows for the first time that wild baboon faecal matter is a valuable source of various antimycobacterial, antimicrobial and anti-inflammatory agents found in high concentrations.
4. Materials and Methods
4.1. Sample Collection and Processing
Wild baboon faecal matter was collected from Langeni forest, Mthatha, on the 5th of May 2024; this was done with assistance from a local hunter. Faecal matter was dried in an air-conditioned environment. Once dried, it was ground into powder using a pestle and mortar to make a fine powder and stored for later use.
4.2. Fecal Extract Preparation
Phytochemical extraction was conducted using two methods:
4.2.1. Ethanol Extraction
50g of the faecal powder was soaked in 500 ml of 70% ethanol, and the suspension was placed in a shaker at 80 rpm at room temperature for 72 hours. The extract was then filtered using a cotton wool ball followed by Whatman filter paper no. 2. The solvent was removed from the filtrate through vacuum distillation with a rotary evaporator set to 35 °C, and the dried filtrate was stored at -20 °C for later use.
4.2.2. Essential Oil Extraction
Hydro-distillation was performed by combining 205g of dried faecal powder with three litres of dH
2O in a 500 ml flask; the flask was heated in a Thermo Scientific heating mantle for four hours. The mixture was first heated at 100 °C for one hour, then reduced to 65 °C for the remaining three hours. The sample with essential oils was concentrated using a speed vacuum (Concentrator Plus, Eppendorf) to remove any remaining solvents and fully dry the samples. The vacuum concentrator was pre-warmed for 15 minutes at 60 °C, and the extracts were dried at 45 °C for one hour. The dried essential oils extract was transferred into the Eppendorf tubes covered with foil and stored at -20 °C for later use [
21].
4.3. Qualitative Phytochemical Screening
Qualitative phytochemical screening of ethanol extract of baboon faecal matter was performed according to Adil et al., 2024 [
22].
Alkaloids test: 100 mg of wild baboon faecal extract was combined with 20 ml of 2% H
2SO4 and then heated for a duration of 2 minutes in a water bath. After boiling, the liquid was filtered, and three drops of Dragendroff’s reagent were added. The presence of a red-orange precipitate confirmed the presence of alkaloids [
22].
Flavonoids: 100 mg of wild baboon faecal extract was dissolved in NaOH and mixed with a few drops of HCl. Changing the solution from yellow to colourless indicated the presence of flavonoids [
22].
Tannins: 50 mg of wild baboon faecal extract was added to 2 ml of distilled water, and the mixture was heated until boiling. The liquid was filtered, and a few drops of 10% Ferric chloride were added to the filtered liquid. A blackish-blue colour indicates the presence of gallic tannins, and a green-blackish colour indicates the presence of catechol tannins [
23].
Terpenoids test: 2 mL of chloroform and 3 mL of concentrated H
2SO4 were added to 200 mg of wild baboon faecal extract, creating a layered mixture. The red-brown interface suggested terpenoids were present [
22].
4.4. Gas Chromatography-Mass Spectrophotometer Analysis of Phytochemicals
Phytochemical analysis and quantification were performed according to Miya et al. (2021) [
24]. The analysis was conducted using a Bruker 450 Gas Chromatograph paired with a 300 MS/MS mass spectrometer system, operating in electron ionization (EI) mode at 70 eV. The instrument had an HP-5 MS fused silica capillary column featuring a 5% phenyl-methyl siloxane stationary phase. Column specifications included a length of 30 m, a diameter of 0.25 mm, and a film thickness of 0.25 µm. The initial column temperature was set at 50 °C and ramped up to 240 °C at 5 °C per minute, reaching a final temperature of 450 °C, with a total run time of 66.25 min. Helium was used as the carrier gas at a flow rate of 1.0 ml/min, with a split ratio of 100:1. The scan time was 78 min, covering a mass range from 35 to 450 amu. Compound identification was based on GC retention indices from the in-house library and in the literature [
25].
4.5. Antimycobacterial Assay
The Alamar Blue assay was used to assess the antimycobacterial activity of the wild baboon faecal extract, according to Agrawal et al., 2015 with minor modifications [
26]. Briefly, 100 µl of wild baboon faecal extract (4mg/ml), rifampicin control (20µg/ml), and DMSO (5%) were added to the first row of a 96-well plate, while 50 µl of 7H9 media filled the remaining wells. Serial two-fold dilutions were performed by mixing 50 µl compounds from the first row with media in each subsequent row until the final row. The
M. smegmatis strain was grown in replicates in 7H9 medium to an OD
600nm of 0.6, diluted 1000 times, and 50μl of the diluted culture was added to each well such that the final concentration of antibiotic in the first well came down to the desired concentration. The plate was sealed with parafilm to avoid drying of the cultures and incubated at 37 °C for three days under mild shaking (100 RPM) conditions. After three days of incubation, 30µl of sterile 0.2% resazurin dye was added to each well and incubated for 6 h, shaking at 100 rpm and photographed [
26]. Minimum Inhibitory Concentration was determined as the value of the first well showing no growth as indicated by resazurin dye staining. The experiments were performed in duplicates.
5. Conclusions
Our study shows for the first time that wild baboon faecal matter contains plant secondary metabolites with antimycobacterial activity and is a potential source of antimicrobial agents. This is significant in an error with increased antibiotic resistance and a great need for new, effective drugs. However, the concentration of the phytochemicals in the wild baboon faecal matter may vary depending on the season and the forest where the wild baboons are found; this needs to be investigated.
Author Contributions
Conceptualization, L.M.; methodology, N.M, L.O.M, and G.M.; software, G.U.M.; validation, L.M., N.M. G.A.M, and G.U.M; formal analysis, L.M., N.M. G.A.M, and G.U.M.; investigation, N.M and L.O.M.; resources, L.M and G.A.M.; data curation, L.M and N.M.; writing—original draft preparation, L.M and N.M.; writing—review and editing, L.M.; visualization, L.M.; supervision, L.M., N.M. G.A.M. M.S; project administration, L.M.; funding acquisition, L.M. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The Institutional Review Ethics Committee of Walter Sisulu University approved the study (Ethics approval number 107/2024 and 30 July 2024).
Informed Consent Statement
Not applicable.
Data Availability Statement
The original contributions presented in this study are included in the article/supplementary material. Further inquiries can be di-rected to the corresponding author.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- World Health Organization. Global Tuberculosis Report 2013; World Health Organization: Geneva, Switzerland, 2013. [Google Scholar]
- Freymann, E.; Carvalho, S.; Garbe, L.A.; Ghazhelia, D.D.; Hobaiter, C.; Huffman, M.A.; Muhumuza, G.; Schulz, L.; Sempebwa, D.; Wald, F.; et al. Pharmacological and behavioral investigation of putative self-medicative plants in Budongo chimpanzee diets. PLoS ONE 2024, 19, e0305219. [Google Scholar] [CrossRef]
- Newton, S.M.; Lau, C.; Gurcha, S.S.; Besra, G.S.; Wright, C.W. The evaluation of forty-three plant species for in vitro antimycobacterial activities; isolation of active constituents from Psoralea corylifolia and Sanguinaria canadensis. J. Ethnopharmacol. 2002, 79, 57–67. [Google Scholar] [CrossRef]
- Melendez, P.; Capriles, V. Antibacterial properties of tropical plants from Puerto Rico. Phytomedicine 2006, 13, 272–276. [Google Scholar] [CrossRef] [PubMed]
- Miryala, S.K.; Basu, S.; Naha, A.; Debroy, R.; Ramaiah, S.; Anbarasu, A.; Natarajan, S. Identification of bioactive natural compounds as efficient inhibitors against Mycobacterium tuberculosis protein-targets: A molecular docking and molecular dynamics simulation study. J. Mol. Liq. 2021, 341, 117340. [Google Scholar] [CrossRef]
- Veeramuthu, D.; Savarimuthu, I.; Khan, I.A.; Alodaini, H.A.; Hatamleh, A.A.; Antony, S. Antimycobacterial activity of plant compounds against extensively drug resistant (XDR-TB) Mycobacterium tuberculosis. J. King Saud Univ. Sci. 2024, 36, 103351. [Google Scholar] [CrossRef]
- Madikizela, B.; McGaw, L.J. Scientific rationale for traditional use of plants to treat tuberculosis in the eastern region of the OR Tambo district, South Africa. J. Ethnopharmacol. 2018, 224, 250–260. [Google Scholar] [CrossRef]
- Alagga, A.; Pellegrini, M.; Gupta, V. Drug Absorption. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2024. [Google Scholar]
- Azman, M.; Sabri, A.H.; Anjani, Q.K.; Mustaffa, M.F.; Hamid, K.A. Intestinal absorption study: Challenges and absorption enhancement strategies in improving oral drug delivery. Pharmaceuticals 2022, 15, 975. [Google Scholar] [CrossRef]
- Bansil, R.; Turner, B.S. Mucin structure, aggregation, physiological functions and biomedical applications. Curr. Opin. Colloid Interface Sci. 2006, 11, 164–170. [Google Scholar] [CrossRef]
- Tretyakov, K.V. Retention Data. In NIST Chemistry WebBook, NIST Standard Reference Database Number 69; Linstrom, P.J., Mallard, W.G., Eds.; National Institute of Standards and Technology: Gaithersburg, MD, USA, 2018. [Google Scholar] [CrossRef]
- Skaltsa, H.D.; Mavrommati, A.; Constantinidis, T. A chemotaxonomic investigation of volatile constituents in Stachys subsect. Swainsonianeae (Labiatae). Phytochemistry 2001, 57, 235–244. [Google Scholar] [CrossRef]
- Juliani, H.R.; Zygadlo, J.A.; Scrivanti, R.; de la Sota, E.; Simon, J.E. The essential oil of Anemia tomentosa (Savigny) Sw. var. anthriscifolia (Schrad.) Mickel. Flavour Fragr. J. 2004, 19, 541–543. [Google Scholar] [CrossRef]
- Poudel, A.; Dosoky, N.S.; Satyal, P.; Swor, K.; Setzer, W.N. Essential oil composition of Grindelia squarrosa from southern Idaho. Molecules 2023, 28, 3854. [Google Scholar] [CrossRef]
- Sieniawska, E.; Swatko-Ossor, M.; Sawicki, R.; Ginalska, G. Morphological changes in the overall Mycobacterium tuberculosis H37Ra cell shape and cytoplasm homogeneity due to Mutellina purpurea L. essential oil and its main constituents. Med. Princ. Pract. 2015, 24, 527–532. [Google Scholar] [CrossRef] [PubMed]
- Cha, D.H.; Roh, G.H.; Hesler, S.P.; Wallingford, A.; Stockton, D.G.; Park, S.K.; Loeb, G.M. 2-Pentylfuran: A novel repellent of Drosophila suzukii. Pest Manag. Sci. 2021, 77, 1757–1764. [Google Scholar] [CrossRef] [PubMed]
- Galan, D.M.; Ezeudu, N.E.; Garcia, J.; Geronimo, C.A.; Berry, N.M.; Malcolm, B.J. Eucalyptol (1,8-cineole): An underutilized ally in respiratory disorders? J. Essent. Oil Res. 2020, 32, 103–110. [Google Scholar] [CrossRef]
- Ramos Alvarenga, R.F.; Wan, B.; Inui, T.; Franzblau, S.G.; Pauli, G.F.; Jaki, B.U. Airborne antituberculosis activity of Eucalyptus citriodora essential oil. J. Nat. Prod. 2014, 77, 603–610. [Google Scholar] [CrossRef]
- Ugbogu, E.A.; Okoro, H.; Emmanuel, O.; Ugbogu, O.C.; Ekweogu, C.N.; Uche, M.; Dike, E.D.; Ijioma, S.N. Phytochemical characterization, anti-diarrhoeal, analgesic, anti-inflammatory activities and toxicity profile of Ananas comosus (L.) Merr. (pineapple) leaf in albino rats. J. Ethnopharmacol. 2024, 319, 117224. [Google Scholar] [CrossRef]
- Boussamba-Digombou, K.J.; Sandasi, M.; Kamatou, G.P.; van Vuuren, S.; Sawicki, R.; Fakhar, Z.; Viljoen, A.M. Investigating the antituberculosis activity of selected commercial essential oils and identification of active constituents using a biochemometrics approach and in silico modeling. Antibiotics 2022, 11, 948. [Google Scholar] [CrossRef]
- Elyemni, M.; Louaste, B.; Nechad, I.; Elkamli, T.; Bouia, A.; Taleb, M.; Chaouch, M.; Eloutassi, N. Extraction of essential oils of Rosmarinus officinalis L. by two different methods: Hydrodistillation and microwave-assisted hydrodistillation. Sci. World J. 2019, 2019, 3659432. [Google Scholar] [CrossRef]
- Adil, M.; Filimban, F.Z.; Ambrin; Quddoos, A.; Sher, A.A.; Naseer, M. Phytochemical screening, HPLC analysis, antimicrobial and antioxidant effect of Euphorbia parviflora L. (Euphorbiaceae Juss.). Sci. Rep. 2024, 14, 5627. [Google Scholar] [CrossRef]
- Nigussie, D.; Davey, G.; Legesse, B.A.; Fekadu, A.; Makonnen, E. Antibacterial activity of methanol extracts of the leaves of three medicinal plants against selected bacteria isolated from wounds of lymphoedema patients. BMC Complement. Med. Ther. 2021, 21, 2. [Google Scholar] [CrossRef]
- Miya, G.; Nyalambisa, M.; Oyedeji, O.; Gondwe, M.; Oyedeji, A. Chemical profiling, toxicity and anti-inflammatory activities of essential oils from three grapefruit cultivars from KwaZulu-Natal in South Africa. Molecules 2021, 26, 3387. [Google Scholar] [CrossRef]
- Adams, R.P. Identification of Essential Oils by Ion Trap Mass Spectroscopy; Academic Press: London, UK, 2012. [Google Scholar]
- Agrawal, P.; Miryala, S.; Varshney, U. Use of Mycobacterium smegmatis deficient in ADP-ribosyltransferase as surrogate for Mycobacterium tuberculosis in drug testing and mutation analysis. PLoS ONE 2015, 10, e0122076. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).