Submitted:
15 February 2026
Posted:
17 February 2026
You are already at the latest version
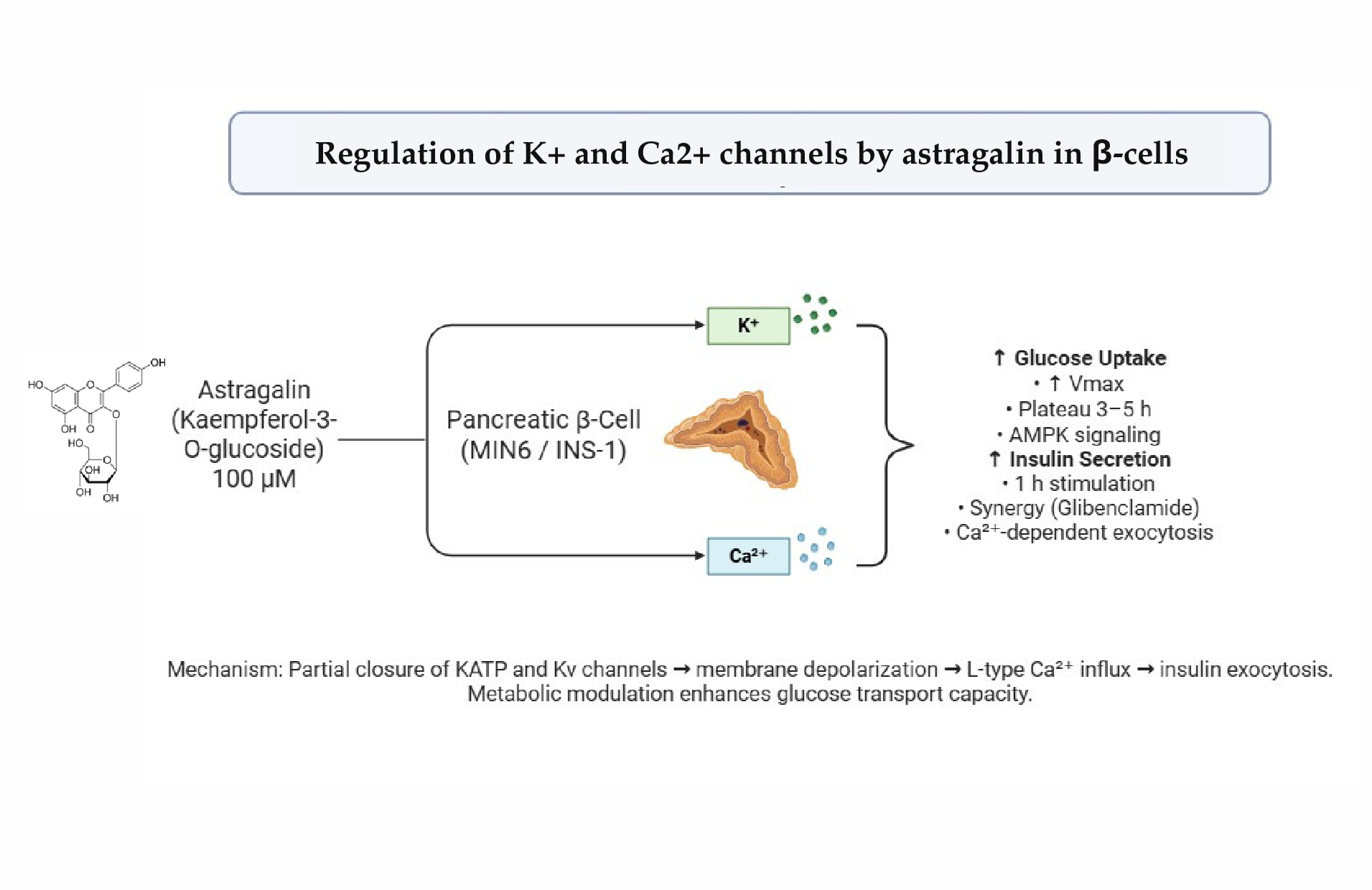
Abstract
Keywords:
1. Introduction
2. Results
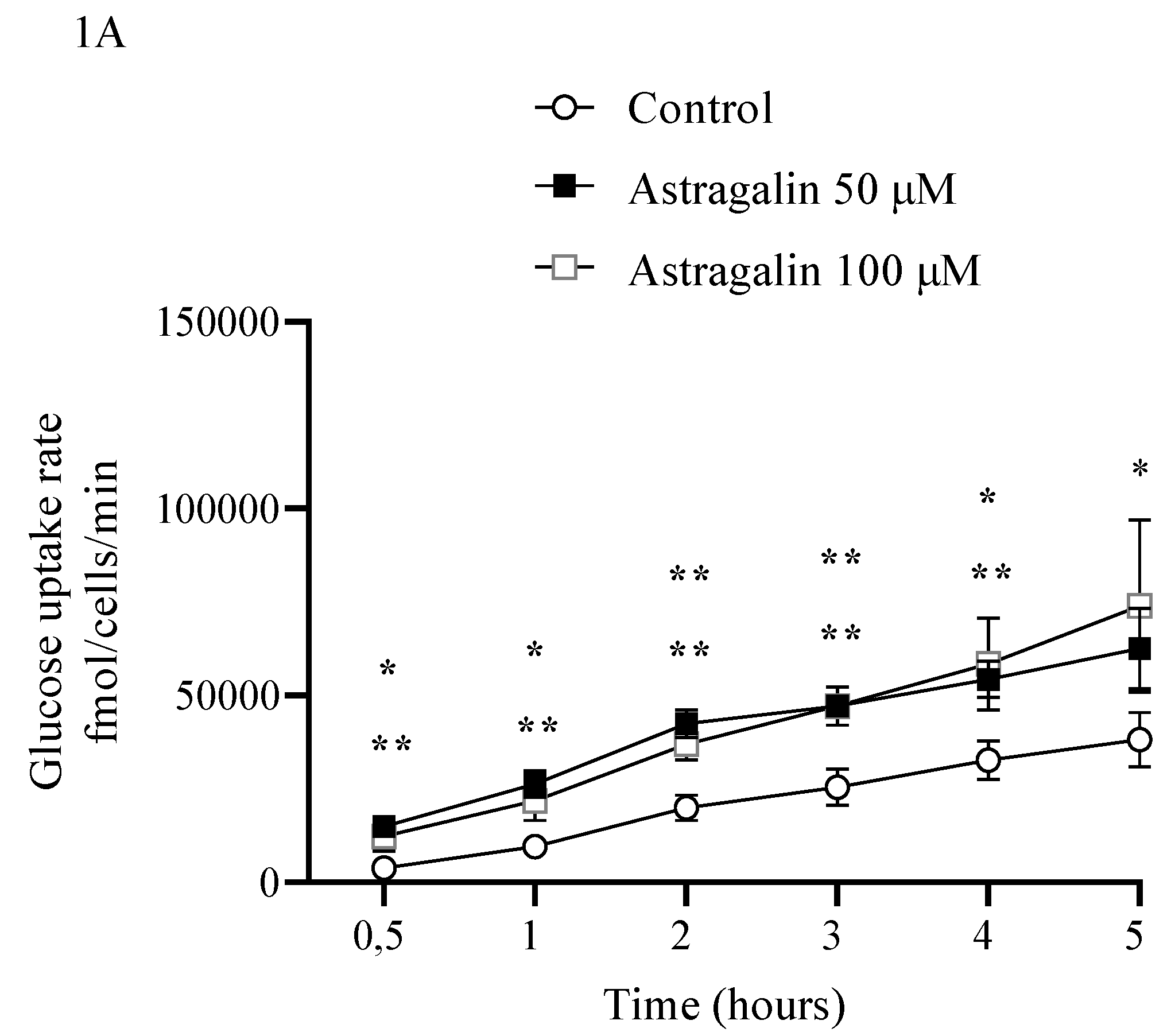
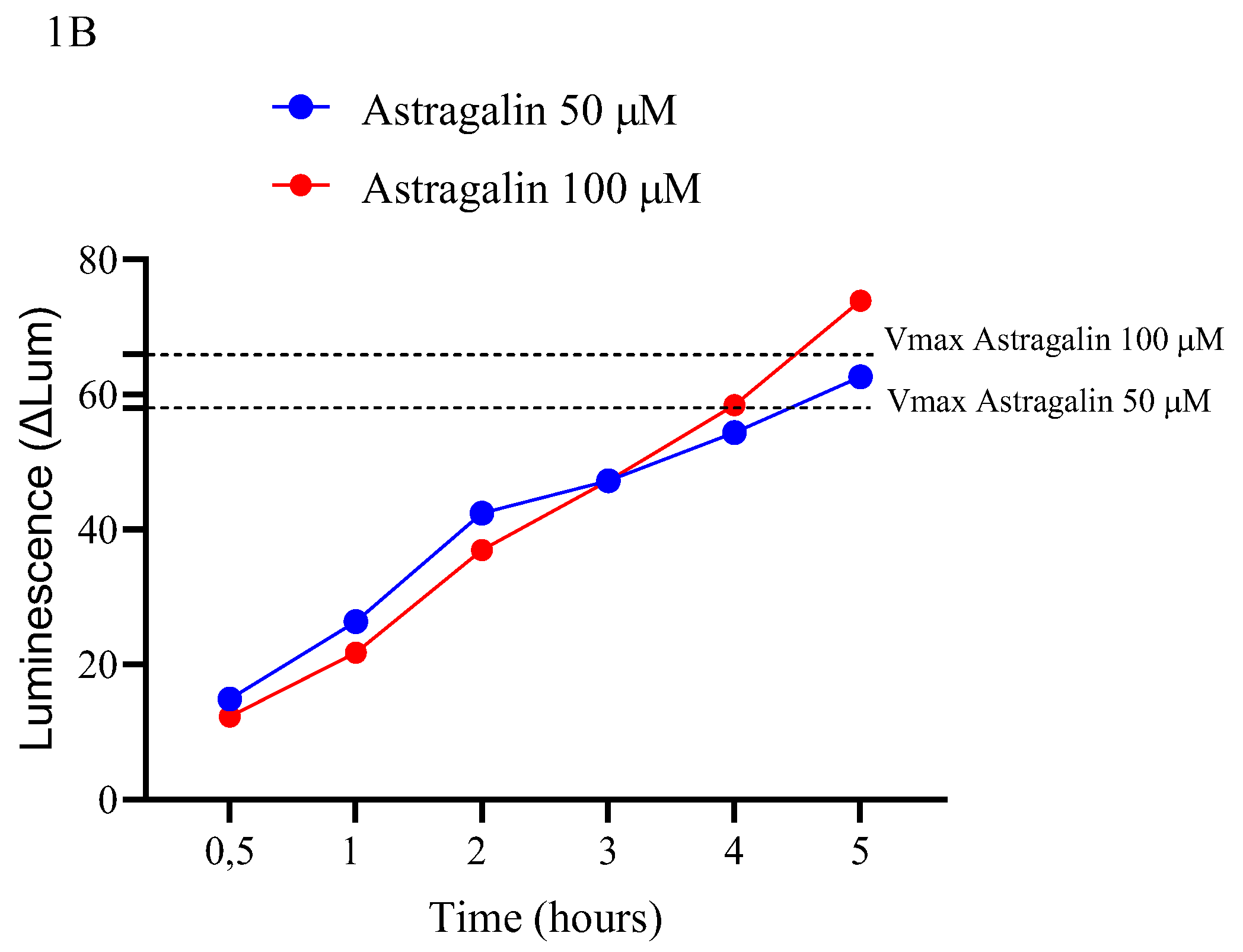
2.1. Glucose Uptake
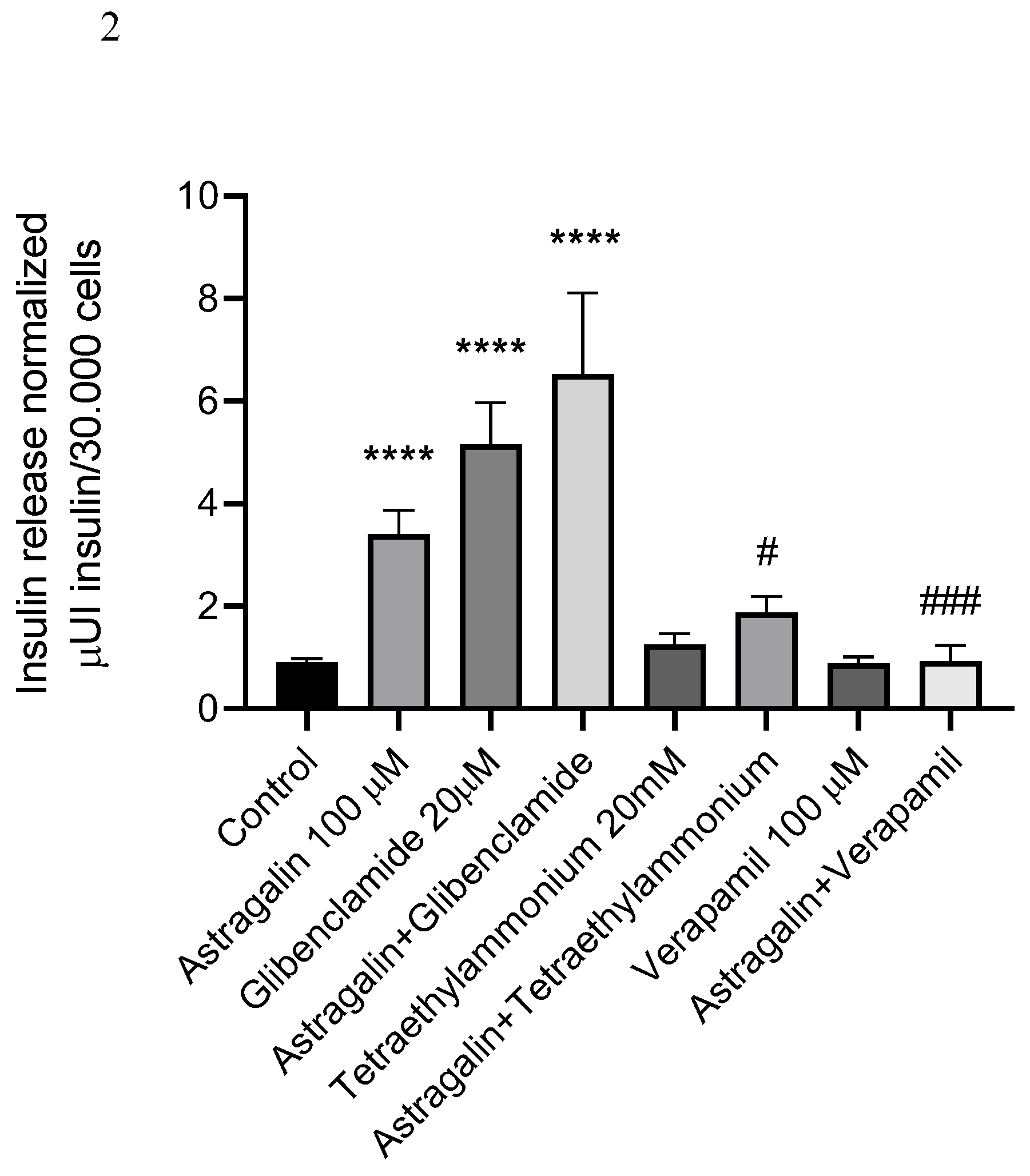
2.2. Insulin Secretion
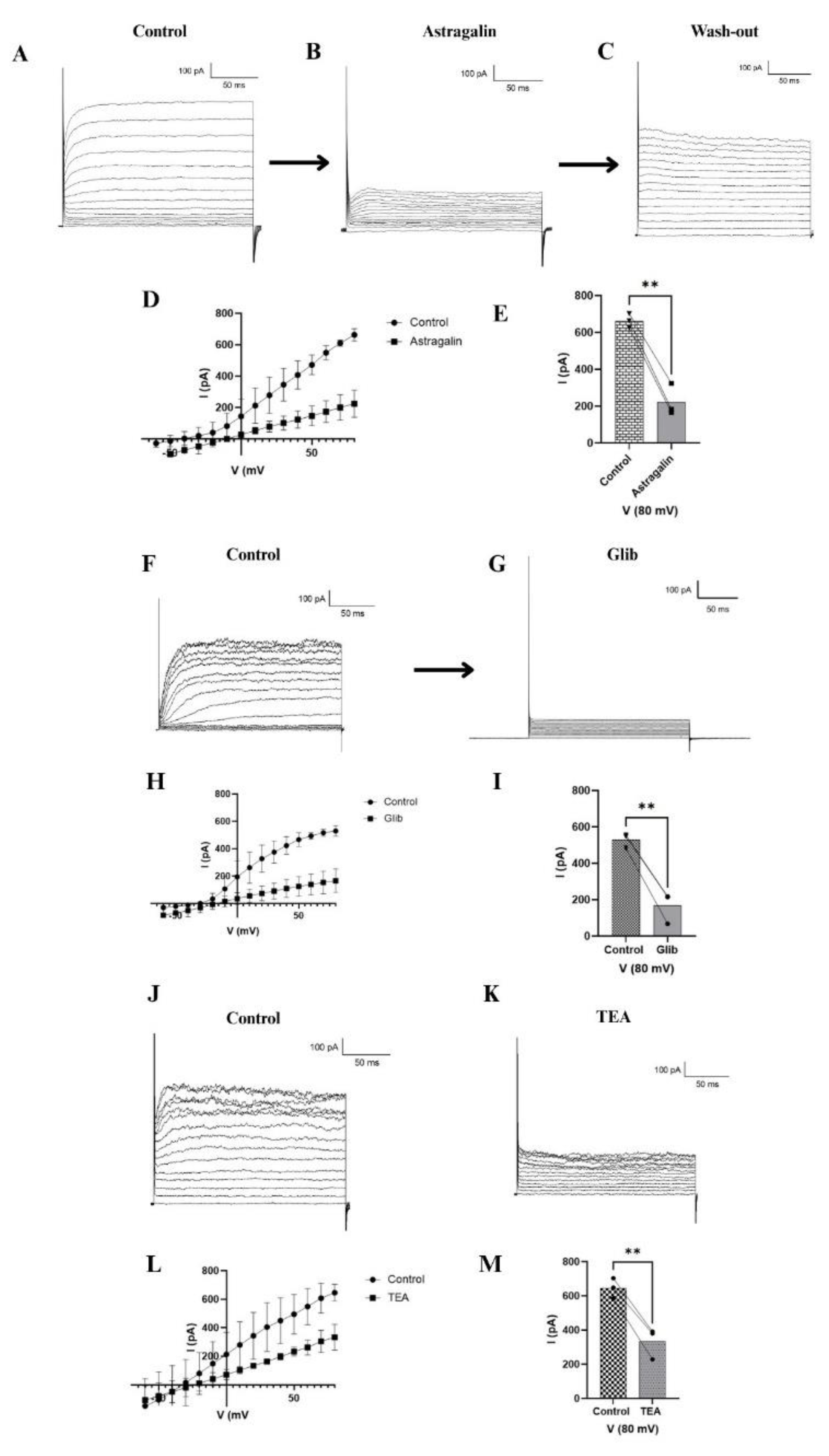
2.3. Electrophysiology (Patch Clamp) Results
3. Discussion
4. Materials and Methods
4.1. Chemical Compounds
4.2. Cell Culture
4.3. Glucose Uptake Assay
4.4. Insulin Secretion
4.5. Patch Clamp Technique
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AMPK | AMP-Activated Protein Kinase |
| ANOVA | Analysis of Variance |
| ATP | Adenosine Triphosphate |
| CaV1.2 | Voltage-Gated Calcium Channel Subtype 1.2 |
| DMEM | Dulbecco’s Modified Eagle Medium |
| DMSO | Dimethyl Sulfoxide |
| ELISA | Enzyme-Linked Immunosorbent Assay |
| FBS | Fetal Bovine Serum |
| GLUT2 | Glucose Transporter Type 2 |
| K⁺-ATP | ATP-Sensitive Potassium Channels |
| Kv | Voltage-Gated Potassium Channels |
| PKA | Protein Kinase A |
| PKC | Protein Kinase C |
| RPMI | Roswell Park Memorial Institute Medium |
| SERCA | Sarco/Endoplasmic Reticulum Ca²⁺-ATPase |
| T2DM | Type 2 Diabetes Mellitus |
| TEA | Tetraethylammonium |
| VDCC | Voltage-Dependent Calcium Channels |
| 2DG | 2-Deoxyglucose |
| 2DG6P | 2-Deoxyglucose-6-Phosphate |
| 2-NBDG | 2-(N-(7-Nitrobenz-2-oxa-1,3-diazol-4-yl)Amino)-2-Deoxyglucose |
References
- Punthakee, Z.; Goldenberg, R.; Katz, P. Definition, Classification and Diagnosis of Diabetes, Prediabetes and Metabolic Syndrome. Canadian Journal of Diabetes 2018, 42, S10–S15. [Google Scholar] [CrossRef]
- Martín, M.Á.; Ramos, S. Dietary Flavonoids and Insulin Signaling in Diabetes and Obesity. Cells 2021, 10, 1474. [Google Scholar] [CrossRef]
- Ranilla, L.G.; Kwon, Y.-I.; Apostolidis, E.; Shetty, K. Phenolic Compounds, Antioxidant Activity and in Vitro Inhibitory Potential against Key Enzymes Relevant for Hyperglycemia and Hypertension of Commonly Used Medicinal Plants, Herbs and Spices in Latin America. Bioresource Technology 2010, 101, 4676–4689. [Google Scholar] [CrossRef]
- Farzaei, F.; Morovati, M.R.; Farjadmand, F.; Farzaei, M.H. A Mechanistic Review on Medicinal Plants Used for Diabetes Mellitus in Traditional Persian Medicine. J Evid Based Complementary Altern Med 2017, 22, 944–955. [Google Scholar] [CrossRef]
- Behl, T.; Gupta, A.; Albratty, M.; Najmi, A.; Meraya, A.M.; Alhazmi, H.A.; Anwer, M.K.; Bhatia, S.; Bungau, S.G. Alkaloidal Phytoconstituents for Diabetes Management: Exploring the Unrevealed Potential. Molecules 2022, 27. [Google Scholar] [CrossRef]
- Cazarolli, L.; Zanatta, L.; Alberton, E.; Reis Bonorino Figueiredo, M.; Folador, P.; Damazio, R.; Pizzolatti, M.; Mena Barreto Silva, F. Flavonoids: Cellular and Molecular Mechanism of Action in Glucose Homeostasis. MRMC 2008, 8, 1032–1038. [Google Scholar] [CrossRef]
- Aragon Novoa, D.M.; Silva, F.R.M.B. The Role of Natural Products on Diabetes Mellitus Treatment; 2024. [Google Scholar] [CrossRef]
- Rey, D.; Fernandes, T.A.; Sulis, P.M.; Gonçalves, R.; Sepúlveda R, M.; Silva Frederico, M.J.; Aragon, M.; Ospina, L.F.; Costa, G.M.; Silva, F.R.M.B. Cellular Target of Isoquercetin from Passiflora Ligularis Juss for Glucose Uptake in Rat Soleus Muscle. Chemico-Biological Interactions 2020, 330, 109198. [Google Scholar] [CrossRef]
- Riaz, A.; Rasul, A.; Hussain, G.; Zahoor, M.K.; Jabeen, F.; Subhani, Z.; Younis, T.; Ali, M.; Sarfraz, I.; Selamoglu, Z. Astragalin: A Bioactive Phytochemical with Potential Therapeutic Activities. Adv Pharmacol Sci 2018, 2018, 9794625. [Google Scholar] [CrossRef]
- Li, Q.; Yang, Z.; Lu, H.; Liu, F.; Zhou, D.; Zou, Y. Astragalin Exerted Hypoglycemic Effect by Both Inhibiting α-Glucosidase and Modulating AMPK Signaling Pathway. Nutrients 2025, 17. [Google Scholar] [CrossRef]
- Rey, D.; Miranda Sulis, P.; Alves Fernandes, T.; Gonçalves, R.; Silva Frederico, M.J.; Costa, G.M.; Aragon, M.; Ospina, L.F.; Mena Barreto Silva, F.R. Astragalin Augments Basal Calcium Influx and Insulin Secretion in Rat Pancreatic Islets. Cell Calcium 2019, 80, 56–62. [Google Scholar] [CrossRef]
- Sun, M.; Ye, H.; Zheng, C.; Jin, Z.; Yuan, Y.; Weng, H. Astragalin Ameliorates Renal Injury in Diabetic Mice by Modulating Mitochondrial Quality Control via AMPK-Dependent PGC1α Pathway. Acta Pharmacol Sin 2023, 44, 1676–1686. [Google Scholar] [CrossRef]
- Niu, B.; Ma, L.; Yao, L.; Zhang, Y.; Su, H. HCV Affects KATP Channels through GnT-IVa-Mediated N-Glycosylation of GLUT2 on the Surface of Pancreatic β-Cells Leading to Impaired Insulin Secretion. Endocrine 2024, 84, 427–440. [Google Scholar] [CrossRef]
- Samario-Román, J.; Velasco, M.; Larqué, C.; Cárdenas-Vázquez, R.; Ortiz-Huidobro, R.I.; Hiriart, M. NGF Effects Promote the Maturation of Rat Pancreatic Beta Cells by Regulating GLUT2 Levels and Distribution, and Glucokinase Activity. PLOS ONE 2024, 19, e0303934. [Google Scholar] [CrossRef]
- Rorsman, P.; Ashcroft, F.M. Pancreatic β-Cell Electrical Activity and Insulin Secretion: Of Mice and Men. Physiological Reviews | American Physiological Society 2017, 98. [Google Scholar] [CrossRef]
- Ashcroft, F.M. KATP Channels and the Metabolic Regulation of Insulin Secretion in Health and Disease: The 2022 Banting Medal for Scientific Achievement Award Lecture. Diabetes 2023, 72, 693–702. [Google Scholar] [CrossRef]
- ElSheikh, A.; Shyng, S.-L. KATP Channel Mutations in Congenital Hyperinsulinism: Progress and Challenges towards Mechanism-Based Therapies. Front. Endocrinol. 2023, 14, 1161117. [Google Scholar] [CrossRef]
- Seino, S. Cell Signalling in Insulin Secretion: The Molecular Targets of ATP, cAMP and Sulfonylurea. Diabetologia 2012, 55, 2096–2108. [Google Scholar] [CrossRef]
- Velasco, M.; Díaz-García, C.M.; Larqué, C.; Hiriart, M. Modulation of Ionic Channels and Insulin Secretion by Drugs and Hormones in Pancreatic Beta Cells. Molecular Pharmacology 2016, 90, 341–357. [Google Scholar] [CrossRef]
- MacDonald, P.E.; Sewing, S.; Wang, J.; Joseph, J.W.; Smukler, S.R.; Sakellaropoulos, G.; Wang, J.; Saleh, M.C.; Chan, C.B.; Tsushima, R.G.; et al. Inhibition of Kv2.1 Voltage-Dependent K+Channels in Pancreatic β-Cells Enhances Glucose-Dependent Insulin Secretion*. Journal of Biological Chemistry 2002, 277, 44938–44945. [Google Scholar] [CrossRef]
- Yang, X.; Yang, Y. The Crucial Role of Potassium Ion Channels in Diabetes Mellitus and Its Complications: A Review. Channels 2025, 19, 2531949. [Google Scholar] [CrossRef]
- Tanaka, Y.; Farkhondeh, A.; Yang, W.; Ueno, H.; Noda, M.; Hirokawa, N. Kinesin-1 Mediates Proper ER Folding of the CaV1.2 Channel and Maintains Mouse Glucose Homeostasis. EMBO Rep 2024, 25, 4777–4802. [Google Scholar] [CrossRef]
- Nitert, M.D.; Nagorny, C.L.F.; Wendt, A.; Eliasson, L.; Mulder, H. CaV1.2 Rather than CaV1.3 Is Coupled to Glucose-Stimulated Insulin Secretion in INS-1 832/13 Cells. 2008. [Google Scholar] [CrossRef]
- York, N.W.; Yan, Z.; Osipovich, A.B.; Tate, A.; Patel, S.; Piston, D.W.; Magnuson, M.A.; Remedi, M.S.; Nichols, C.G. Loss of β-Cell KATP Reduces Ca2+ Sensitivity of Insulin Secretion and Trpm5 Expression. Diabetes 2024, 74, 376–383. [Google Scholar] [CrossRef]
- Proks, P.; Lippiat, J.D. Membrane Ion Channels and Diabetes. Current Pharmaceutical Design 2006, 12, 485–501. [Google Scholar] [CrossRef]
- Thompson, B.; Satin, L.S. Beta-Cell Ion Channels and Their Role in Regulating Insulin Secretion. Comprehensive Physiology 2021, 11, 1–21. [Google Scholar] [CrossRef]
- Trezza, A.; Cicaloni, V.; Porciatti, P.; Langella, A.; Fusi, F.; Saponara, S.; Spiga, O. From in Silico to in Vitro: A Trip to Reveal Flavonoid Binding on the Rattus Norvegicus Kir6.1 ATP-Sensitive Inward Rectifier Potassium Channel. PeerJ 2018, 6, e4680. [Google Scholar] [CrossRef]
- Nakashima, K.; Kanda, Y.; Hirokawa, Y.; Kawasaki, F.; Matsuki, M.; Kaku, K. MIN6 Is Not a Pure Beta Cell Line but a Mixed Cell Line with Other Pancreatic Endocrine Hormones. Endocr J 2009, 56, 45–53. [Google Scholar] [CrossRef]
- Mendes, C.P.; Postal, B.G.; Oliveira, G.T.C.; Castro, A.J.G.; Frederico, M.J.S.; Moraes, A.L.L.; Neuenfeldt, P.D.; Nunes, R.J.; Menegaz, D.; Silva, F.R.M.B. Insulin Stimulus-secretion Coupling Is Triggered by a Novel Thiazolidinedione/Sulfonylurea Hybrid in Rat Pancreatic Islets. Journal Cellular Physiology 2019, 234, 509–520. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.



