Submitted:
15 February 2026
Posted:
16 February 2026
You are already at the latest version
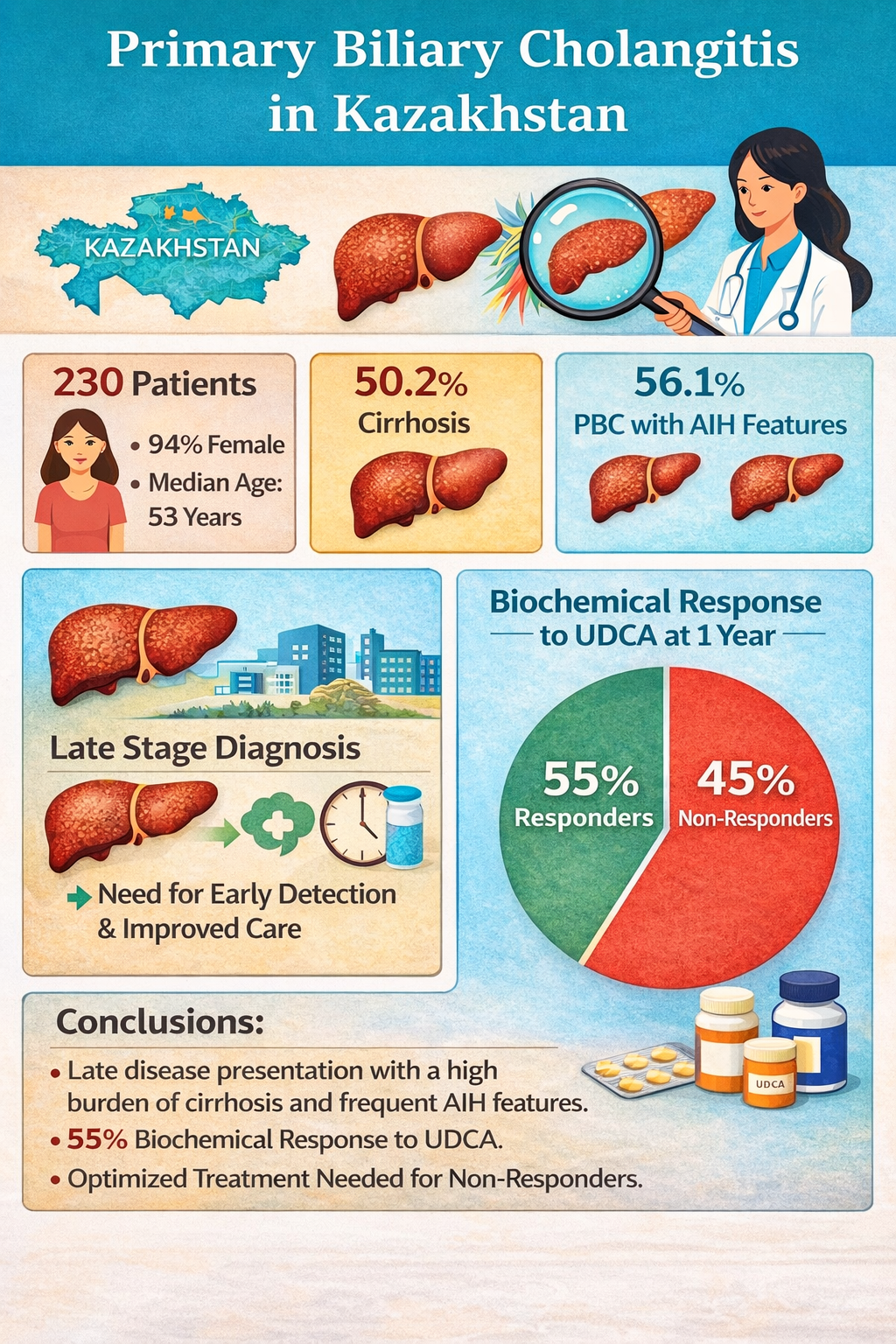
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. The Study Population
2.2. Data Collection
2.3. Ethics
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| PBC | Primary biliary cholangitis |
| PBC + AIH | Primary biliary cholangitis with autoimmune hepatitis features |
| UDCA | Ursodeoxycholic acid |
| AIH | Autoimmune hepatitis |
| AST | Aspartate aminotransferase |
| ALT | Alanine transaminase |
| ALP | Alkaline phosphatase |
| GGT | Gamma-glutamyl transferase |
| IQR | Interquartile range |
| SD | Standard deviation |
| AMA/AMA-M2 | Antimitochondrial M2 antibody |
| ANA | Antinuclear antibody |
| BMI | Body mass index |
References
- Trivella, J; John, BV; Levy, C. Primary biliary cholangitis: Epidemiology, prognosis, and treatment. Hepatol Commun. 2023, 7(6), e0179. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Tan, JJ; Chung, AH; Loo, JH; Quek, JWE; Sharma, S; Singh, CL; Yap, RXJ; Tay, WX; Smith, MK; Lytvyak, E; Mason, A; Montano-Loza, AJ; Wong, YJ. Global Epidemiology of Primary Biliary Cholangitis: An Updated Systematic Review and Meta-analysis. Clin Gastroenterol Hepatol Epub ahead of print. 2025, S1542-3565(25)00418-5. [Google Scholar] [CrossRef] [PubMed]
- Lv, T; Chen, S; Li, M; Zhang, D; Kong, Y; Jia, J. Regional variation and temporal trend of primary biliary cholangitis epidemiology: A systematic review and meta-analysis. J Gastroenterol Hepatol. 2021, 36(6), 1423–1434. [Google Scholar] [CrossRef] [PubMed]
- Gazda, J; Drazilova, S; Janicko, M; Jarcuska, P. The Epidemiology of Primary Biliary Cholangitis in European Countries: A Systematic Review and Meta-Analysis. Can J Gastroenterol Hepatol 2021, 2021, 9151525. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chalifoux, SL; Konyn, PG; Choi, G; Saab, S. Extrahepatic Manifestations of Primary Biliary Cholangitis. Gut Liver 2017, 11(6), 771–780. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- de Veer, RC; van Hooff, MCB; Werner, E; Beuers, U; Drenth, JPH; Cuperus, FJC; van Hoek, B; Veldt, BJ; Klemt-Kropp, M; van Meer, S; Verdonk, RC; Flink, HJ; Vrolijk, JM; Gevers, TJG; Ponsioen, CY; Ter Borg, MJ; Soufidi, K; Boersma, F; de Jonge, HJM; Wolfhagen, FHJ; Baak, LC; Onderwater, SL; van Bergeijk, JD; van Putten, PG; de Bruin, GJ; Adang, RPR; Aparicio-Pages, MN; de Boer, W; Borg, FT; van Soest, H; Janssen, HLA; Hansen, BE; Erler, NS; van der Meer, AJ. Dutch PBC Study Group. Incidence and prevalence of primary biliary cholangitis in the Netherlands - A nationwide cohort study. JHEP Rep. 2024, 6(8), 101132. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Marschall, HU; Henriksson, I; Lindberg, S; Söderdahl, F; Thuresson, M; Wahlin, S; Ludvigsson, JF. Incidence, prevalence, and outcome of primary biliary cholangitis in a nationwide Swedish population-based cohort. Sci Rep. 2019, 9(1), 11525. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Saparbay, J; Spatayev, J; Sharmenov, A; Aytbayev, S; Uristenova, A; Mukazhanov, A; Zhexembayev, A. Liver Transplantation: A 10-Year Low-Volume Transplant Center Experience in Kazakhstan. Ann Transplant. 2021, 26, e931786. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Melling, N; Hohenberger, W; Yedibela, S. Opisthorchiasis mimicking primary biliary cirrhosis as an indication for liver transplantation. J Hepatol. 2009, 50(5), 1057–9. [Google Scholar] [CrossRef] [PubMed]
- Saipiyeva, D; Askarov, M; Jafari, N; Zhankina, R; Heath, PR; Kozina, L; Boltanova, A; Omarbekov, A; Ilyassov, N; Tuganbekov, T; Mussin, NM; Kaliyev, AA; Sultangereyev, Y; Rahmanifar, F; Mahdipour, M; Bakhshalizadeh, S; Shirazi, R; Tanideh, N; Tamadon, A. Bone Marrow and Peripheral Blood Mononuclear Cell Phenotype Changes after Cultivation and Autologous Infusion in Patients with Primary Biliary Cholangitis. Iran J Med Sci. 2024, 49(9), 559–572. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Semenova, Y; Beyembetova, A; Shaisultanova, S; Asanova, A; Sailybayeva, A; Altynova, S; Pya, Y. Evaluation of liver transplantation services in Kazakhstan from 2012 to 2023. Sci Rep. 2024, 14(1), 9304. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Doszhan, A; Tostanovskaya, N; Shaimardanova, G; Kozina, L; Imambayeva, G; Nauryzbayeva, A. Primary Biliary Cholangitis with Associated Raynaud Syndrome Treatment Outcome: a Case Report. J CLIN MED KAZ. 2025, 22(5), 119–22. [Google Scholar] [CrossRef] [PubMed]
- Lindor, KD; Bowlus, CL; Boyer, J; Levy, C; Mayo, M. Primary Biliary Cholangitis: 2018 Practice Guidance from the American Association for the Study of Liver Diseases. Hepatology 2019, 69(1), 394–419. [Google Scholar] [CrossRef] [PubMed]
- Angelara, M; Papachristou, K; Papatheodoridi, M; Nasiri-Ansari, N; Karagiannakis, DS; Androutsakos, T. Primary biliary cholangitis. Treatment options in 2025. A narrative review. Front Immunol 2025, 16, 1698833. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Trivedi, PJ; Hirschfield, GM. Recent advances in clinical practice: epidemiology of autoimmune liver diseases. Gut 2021, 70(10), 1989–2003. [Google Scholar] [CrossRef] [PubMed]
- Mantaka, A; Koulentaki, M; Samonakis, D; Sifaki-Pistolla, D; Voumvouraki, A; Tzardi, M; Kouroumalis, E. Association of smoking with liver fibrosis and mortality in primary biliary cholangitis. Eur J Gastroenterol Hepatol 2018, 30(12), 1461–1469. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y; Shi, Y; Wu, R; Wang, X; Gao, X; Niu, J. Primary biliary cholangitis is more severe in previous hepatitis B virus infection patients. Eur J Gastroenterol Hepatol 2018, 30(6), 682–686. [Google Scholar] [CrossRef] [PubMed]
- European Association for the Study of the Liver. EASL Clinical Practice Guidelines: The diagnosis and management of patients with primary biliary cholangitis. J Hepatol. 2017, 67(1), 145–172. [Google Scholar] [CrossRef] [PubMed]
- Freedman, BL; Danford, CJ; Patwardhan, V; Bonder, A. Treatment of Overlap Syndromes in Autoimmune Liver Disease: A Systematic Review and Meta-Analysis. J Clin Med. 2020, 9(5), 1449. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Costa, V; Moreira, M; Costa, R; Fontes, J. Primary Biliary Cholangitis Overlap With Autoimmune Hepatitis? A Case Report. J Med Cases 2021, 12(4), 131–133. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Surdea-Blaga, T; Cărăguț, RL; Caraiani, C; Spârchez, Z; Al Hajjar, N; Dumitrașcu, DL. Overlap syndrome of autoimmune hepatitis and primary biliary cholangitis complicated with atypical hepatocellular carcinoma: a case report. J Med Case Rep. 2023, 17(1), 328. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Rodrigues Barbosa, B; Pereira, L; Campante, F; Pona, AP. Chronic Liver Disease, Not Everything Is What It Seems: Autoimmune Hepatitis/Primary Biliary Cholangitis Overlap Syndrome. Cureus 2024, 16(1), e51630. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sohal, A; Nikzad, N; Kowdley, KV. Overlap syndromes in autoimmune liver disease: a review. Transl Gastroenterol Hepatol 2025, 10, 33. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Jayabalan, D; Huang, Y; Calzadilla-Bertot, L; Janjua, M; de Boer, B; Joseph, J; Cheng, W; Hazeldine, S; Smith, BW; MacQuillan, GC; Wallace, MC; Garas, G; Adams, LA; Jeffrey, GP. Predictors of survival in autoimmune liver disease overlap syndromes. World J Hepatol. 2024, 16(9), 1269–1277. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Angelara, M; Papachristou, K; Papatheodoridi, M; Nasiri-Ansari, N; Karagiannakis, DS; Androutsakos, T. Primary biliary cholangitis. Treatment options in 2025. A narrative review. Front Immunol 2025, 16, 1698833. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- You, H; Duan, W; Li, S; Lv, T; Chen, S; Lu, L; Ma, X; Han, Y; Nan, Y; Xu, X; Duan, Z; Wei, L; Jia, J; Zhuang, H; Chinese Society of Hepatology; Chinese Medical Association. Guidelines on the Diagnosis and Management of Primary Biliary Cholangitis (2021). J Clin Transl Hepatol. 2023, 11(3), 736–746. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Yang, C; Guo, G; Li, B; Zheng, L; Sun, R; Wang, X; Deng, J; Jia, G; Zhou, X; Cui, L; Guo, C; Zhou, X; Leung, PSC; Gershwin, ME; Shang, Y; Han, Y. Prediction and evaluation of high-risk patients with primary biliary cholangitis receiving ursodeoxycholic acid therapy: an early criterion. Hepatol Int. 2023, 17(1), 237–248. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Gao, Y; Li, L; Li, B; Zhan, Y. Response Rate and Impact on Lipid Profiles of Obeticholic Acid Treatment for Patients with Primary Biliary Cholangitis: A Meta-Analysis. Can J Gastroenterol Hepatol 2021, 2021, 8829510. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
| Characteristics | Frequency | Percent (%) |
| Ethnicity | ||
| Asian | 210 | 91.30 |
| Other | 20 | 8.70 |
| Gender | ||
| Female | 216 | 93.91 |
| Male | 14 | 6.09 |
| Main diagnosis | ||
| PBC | 101 | 43.91 |
| PBC with AIH features | 129 | 56.09 |
| Smokers | 10 | 4.59 |
| Alcohol consumers | 5 | 2.29 |
| Cirrhosis, yes | 108 | 50.23 |
| Ascites, yes | 37 | 16.23 |
| Hepatic encephalopathy, yes | 56 | 24.45 |
| Oesophageal varices, yes | 68 | 29.82 |
| Hashimoto’s thyroiditis, yes | 30 | 13.04 |
| Sjögren’s syndrome, yes | 20 | 8.70 |
| Rheumatoid arthritis, yes | 11 | 4.78 |
| CREST syndrome/scleroderma, yes | 6 | 2.61 |
| Pruritus, yes | 147 | 64.76 |
| Fatigue, yes | 180 | 78.95 |
| Peripheral edema, yes | 37 | 16.37 |
| AMA/AMA-M2 | ||
| Negative | 46 | 21.60 |
| Positive | 167 | 78.40 |
| ANA | ||
| Negative | 114 | 56.44 |
| Positive | 88 | 43.56 |
| Treatment adherence | ||
| Adhered fully | 168 | 82.35 |
| Adhered with violations | 36 | 17.65 |
| Age, years | 55.5 (18-84) | 17* |
| Age at diagnosis, years | 53 (18-84) | 15* |
| BMI, kg/m2 | 23.82 (16.4-46) | 4.65* |
| Time to diagnosis, months | 16 (0-132) | 24* |
| Elastography, kPa | 3 (0-4) | 3* |
| ALT, U/L | 51.74 (8.09-641) | 59.9* |
| AST, U/L | 55.9 (9.7-288) | 50.5* |
| ALP, U/L | 270 (45-1815) | 296.6* |
| GGT, U/L | 151.1 (10-1521) | 208* |
| Ursodeoxycholic acid, mg | 1000 (500-2250) | 250* |
| FIB 4 | 2.03 (0.37-26.19) | 2.46* |
| De Ritis | 1 (0.36-16.33) | 0.51* |
| APRI | 0.80 (0.10-13.62) | 0.98* |
| Characteristics | PBC | PBC+ AIH features | p-value |
| Ethnicity | 0.9184 | ||
| Asian | 92 (91.09) | 118 (91.47) | |
| Other | 9 (8.91) | 11 (8.53) | |
| Gender | 0.9345 | ||
| female | 95 (94.06) | 121 (93.80) | |
| male | 6 (5.94) | 8 (6.20) | |
| Smoking | 0.2550 | ||
| no | 87 (95.60) | 121 (95.28) | |
| yes | 4 (4.40) | 6 (4.72) | |
| Alcohol consumption | 0.2350 | ||
| no | 91 (98.91) | 122 (96.83) | |
| yes | 1 (1.09) | 4 (3.17) | |
| Cirrhosis | 0.0012 | ||
| no | 57 (62.64) | 50 (40.32) | |
| yes | 34 (37.36) | 74 (59.68) | |
| Ascites | 0.9342 | ||
| no | 84 (84.00) | 107 (83.59) | |
| yes | 16 (16.00) | 21 (16.41) | |
| Hepatic encephalopathy | 0.0209 | ||
| no | 83 (83.00) | 90 (69.77) | |
| yes | 17 (17.00) | 39 (30.23) | |
| Esophageal varices | 0.0054 | ||
| no | 79 (79.80) | 81 (62.79) | |
| yes | 20 (20.20) | 48 (37.21) | |
| Hashimoto’s thyroiditis | 0.7445 | ||
| no | 87 (86.14) | 113 (87.60) | |
| yes | 14 (13.86) | 16 (12.40) | |
| Sjogren’s syndrome | 0.4006 | ||
| no | 94 (93.07) | 116 (89.92) | |
| yes | 7 (6.93) | 13 (10.08) | |
| Rheumatoid arthritis | 0.2406 | ||
| no | 96 (95.05) | 123 (95.35) | |
| yes | 5 (4.95) | 6 (4.65) | |
| Crest syndrome/scleroderma | 0.2888 | ||
| no | 99 (98.02) | 125 (96.90) | |
| yes | 2 (1.98) | 4 (3.10) | |
| Pruritus | 0.4766 | ||
| no | 32 (32.65) | 48 (37.21) | |
| yes | 66 (67.35) | 81 (62.79) | |
| Fatigue | 0.0909 | ||
| no | 26 (26.26) | 22 (17.05) | |
| yes | 73 (73.74) | 107 (82.95) | |
| Peripheral edema | 0.1587 | ||
| no | 85 (87.63) | 104 (80.62) | |
| yes | 12 (12.37) | 25 (19.38) | |
| АМА/АМА-М2 | 0.1434 | ||
| negative | 24 (26.37) | 22 (18.03) | |
| positive | 67 (73.63) | 100 (81.97) | |
| ANA | <0.0001 | ||
| negative | 70 (84.34) | 44 (36.97) | |
| positive | 13 (15.66) | 75 (63.03) | |
| Treatment adherence | 0.9827 | ||
| adhered full | 75 (82.42) | 93 (82.30) | |
| adhered with violations | 16 (17.58) | 20 (17.70) | |
| Age, years | 55 (20–75) | 56 (18–84) | 0.4091 |
| Age at diagnosis, years | 53.5 (20–75) | 53 (18–84) | 0.6808 |
| BMI, kg/m2 | 23.70 (16.44–34.14) | 23.95 (16.49–46.00) | 0.6579 |
| Time to diagnosis, months | 16 (0–132) | 16 (0–132) | 0.7074 |
| Elastography, kPa | 2 (0–4) | 4 (0–4) | 0.0004 |
| ALT, U/L | 46.99 (8.09–418) | 52.8 (9–641) | 0.1206 |
| AST, U/L | 47.22 (9.7–288) | 61.72 (15.2–282) | 0.0451 |
| ALP, U/L | 265.55 (45–955) | 275 (65–1815) | 0.6764 |
| GGT, U/L | 164.7 (12.56–1086) | 137 (10–1521) | 0.896 |
| Ursodeoxycholic acid dose, mg | 1000 (500–2250) | 1000 (500–1500) | 0.7931 |
| FIB-4 | 1.80 (0.38–17.68) | 2.14 (0.37–26.19) | 0.1522 |
| DE RITIS | 0.99 (0.36–16.33) | 1.03 (0.39–3.83) | 0.5575 |
| APRI | 0.64 (0.10–7.98) | 0.88 (0.14–13.63) | 0.0542 |
| Response Criteria |
Overall response at 1year |
Response at 1 year | ||||
| PBC | PBC with AIH features | |||||
| Available data (n) | Cohort Response n (%) | Available data (n) | Cohort Response n (%) | Available data (n) | Cohort Response n (%) | |
| Paris-1 | 208 | 116 (55.77) | 89 | 51 (57.30) | 119 | 65 (54.62) |
| Barcelona | 216 | 119 (55.09) | 91 | 49 (53.85) | 125 | 70 (56.00) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




