Submitted:
14 February 2026
Posted:
17 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Physicochemical Properties of Tea Tree Essential Oils
2.1. Chemical Composition and Variability
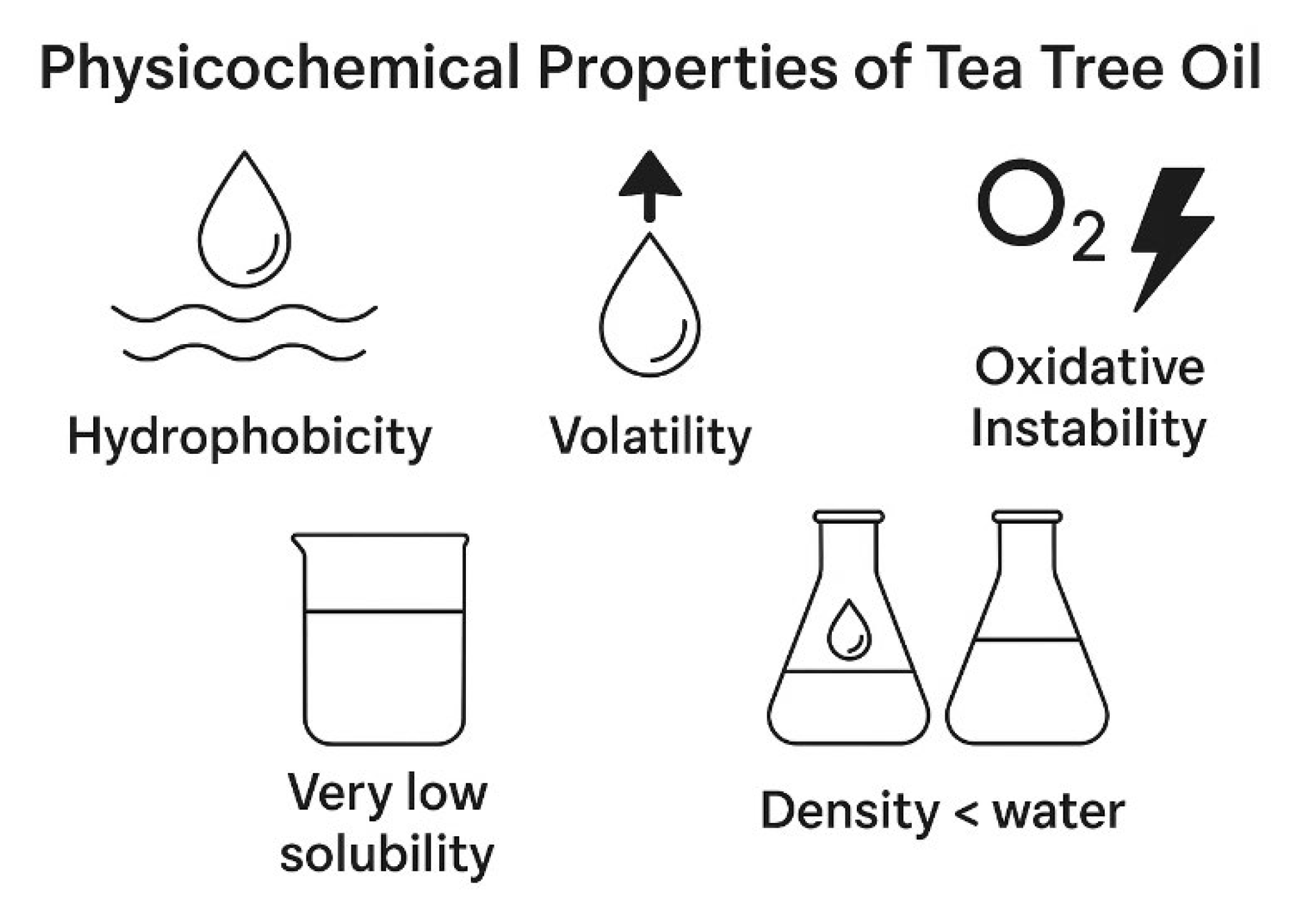
2.2. Physical Characteristics
2.3. Hydrophobicity and Solubility
2.4. Volatility and Aroma
2.5. Oxidative Stability
2.6. Interactions with Food Matrices
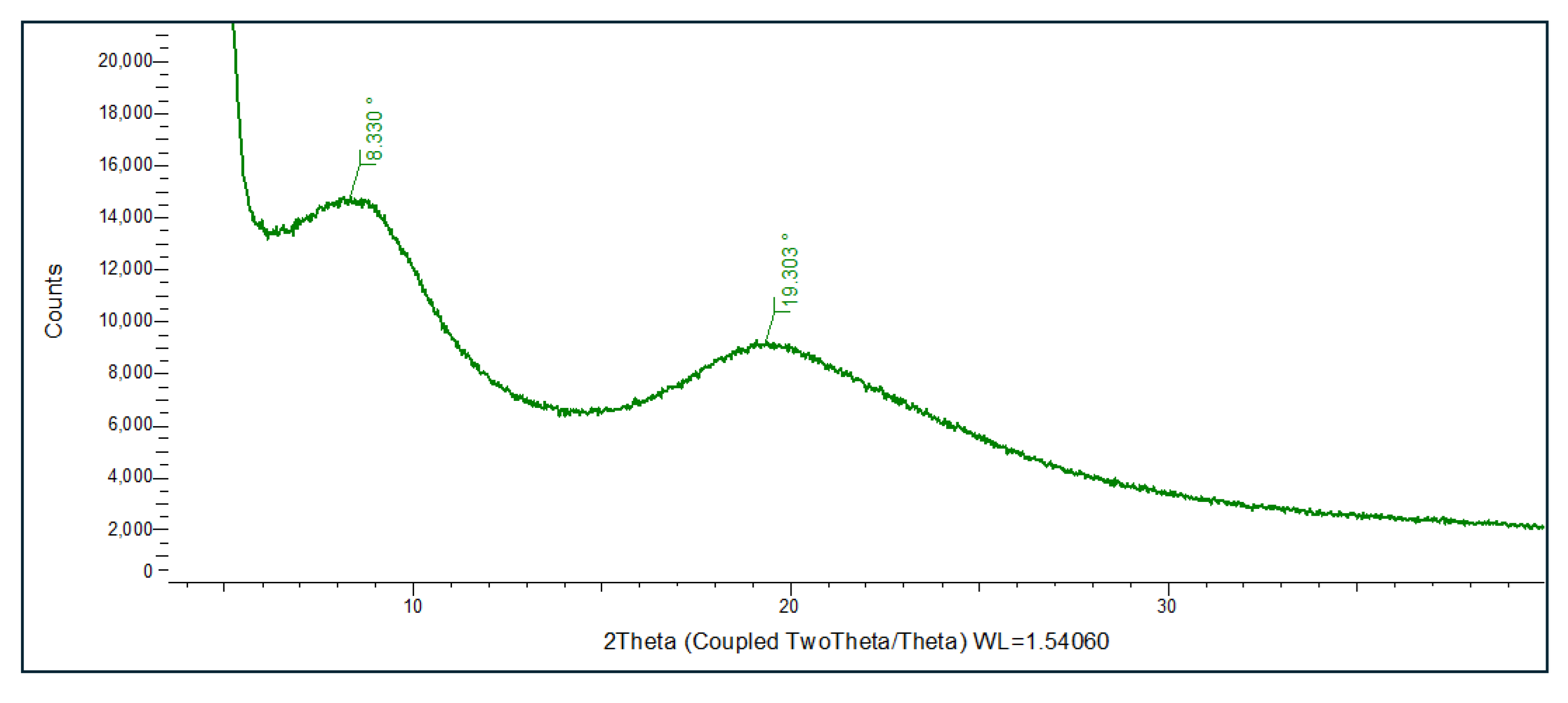
2.7. Analytical Methods for Characterization and Quality Assurance
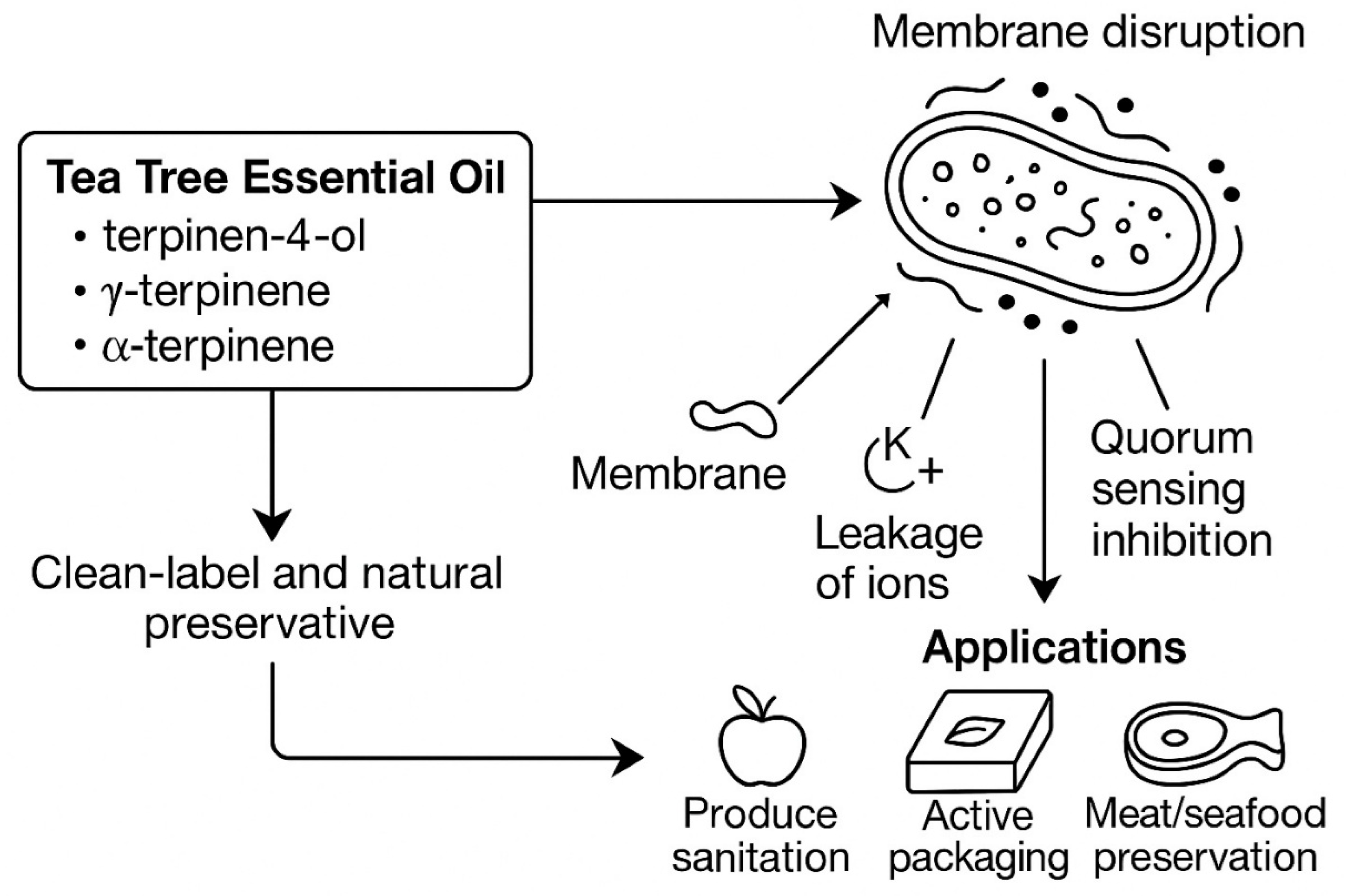
3. Antimicrobial and Antibiofilm Activities of Tea Tree Oils (Tto)
3.1. Antibacterial Activity Against Gram-Positive and Gram-Negative Bacteria
3.2. Antifungal Activity
3.3. Antiviral Activity
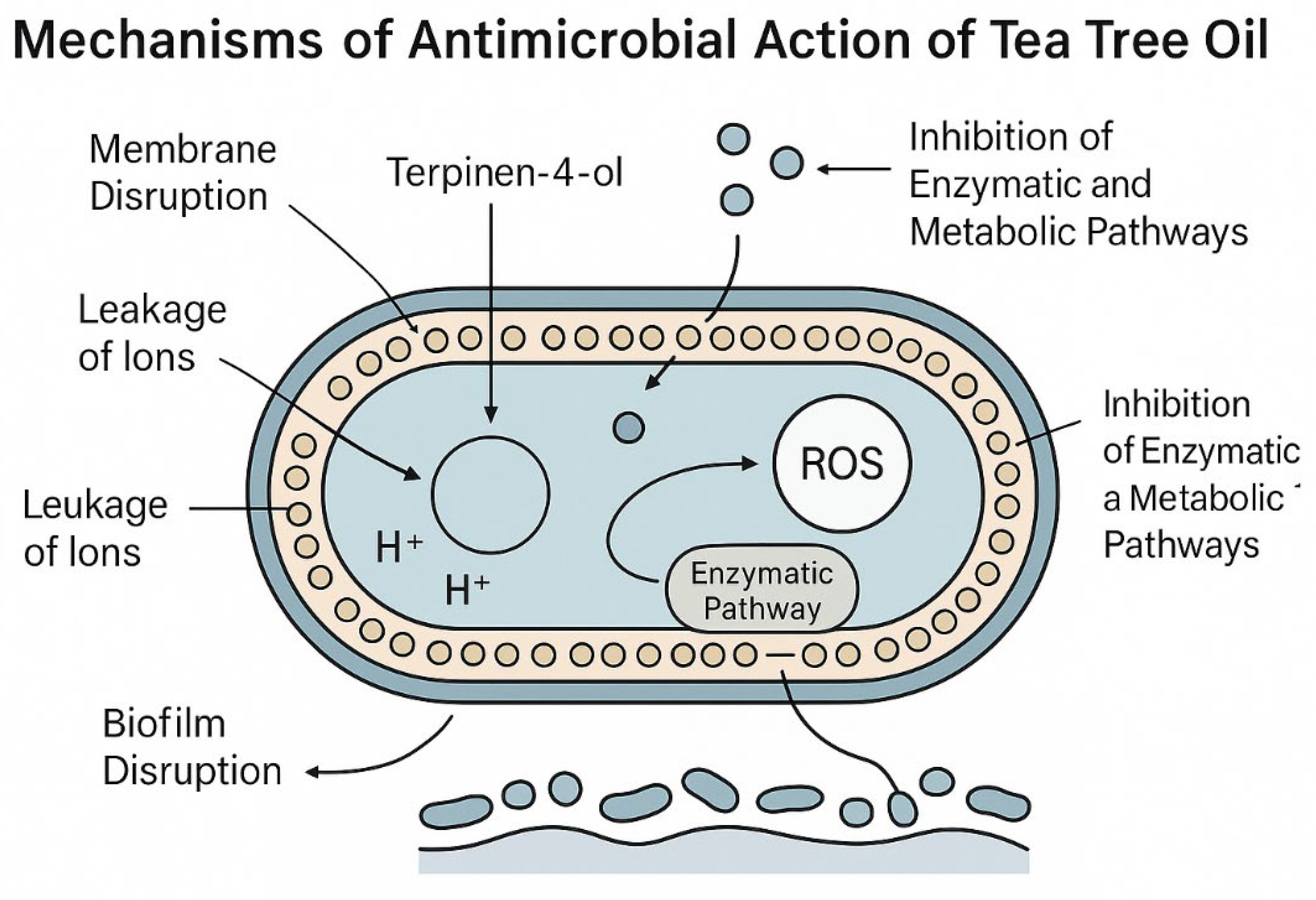
3.4. Mechanisms of Antimicrobial Action of Tto
3.4.1. Membrane Disruption and Cell Lysis
3.4.2. Inhibition of Respiration and ATP Synthesis
3.4.3. Quorum Sensing Interference and Antibiofilm Mechanisms
3.4.4. Antibiofilm Efficacy
3.4.5. Relevance to Food Safety
4. Applications of Tea Tree Essential Oils in Food Safety
4.1. Meat and Poultry Products
4.2. Dairy Products and Cheese
4.3. Seafood and Fish Products
4.4. Fruits, Vegetables, and Fresh Produce
4.4.1. Applications in Fruits
4.4.2. Applications in Vegetables and Fresh Cuts
4.5. Beverages and Liquid Food Systems
4.6. Delivery Systems for Tto in Food Applications
4.6.1. Delivery Systems for TTO in Food Applications
4.6.2. Edible Coatings and Films
4.6.3. Active Packaging Technologies
4.7. Industrial and Real-Word Case Studies
4.8. Oral and Functional Food Applications
5. Challenges, Safety Concerns, and Future Prospects of Tto in Food Safety Systems
5.1. Toxicity and Safety Concerns
5.2. Sensory and Organoleptic Impacts on Food
5.3. Regulatory Landscape
5.4. Technical Challenges in Food Applications
5.4.1. Volatility and Evaporation
5.4.2. Oxidation and Chemical Stability
5.4.3. Poor Water Solubility and Dispersion
5.4.4. Interactions with Food Matrices
5.4.5. Compatibility with Processing and Packaging Materials
5.4.6. Effect on Food Quality Parameters
5.5. Future Directions and Prospects
5.5.1. Novel Formulation and Delivery Systems
5.5.2. Synergistic Hurdle Approaches
5.5.3. Active and Smart Packaging Systems
5.5.4. Sustainability and Natural Sourcing Considerations
6. Outcomes and Focus Areas for Tto’s Future Use
7. Conclusions
Funding
Conflicts of Interest
References
- Afrokh, M.; El Mehrach, K.; Chatoui, K.; Bihi, M.A.; Sadki, H.; Zarrouk, A.; Tabyaoui, M.; Tahrouch, S. Quality criteria, chemical composition and antimicrobial activity of the essential oil of Mentha suaveolens Ehrh. Heliyon 2024, 10(7), e28125. [Google Scholar] [CrossRef] [PubMed]
- Aluyor, E.O.; Oboh, I.O. PRESERVATIVES Traditional Preservatives – Vegetable Oils. In Encyclopedia of Food Microbiology (Second Edition); 2014; pp. 137–140. [Google Scholar] [CrossRef]
- Amaral, R.A.; Pinto, C.A.; Lima, V.; Tavares, J.; Martins, A.P.; Fidalgo, L.G.; Silva, A.M.; Gil, M.M.; Teixeira, P.; Barbosa, J.; Barba, F.J.; Saraiva, J.A. Chemical-Based Methodologies to Extend the Shelf Life of Fresh Fish—A Review. Foods 2021, 10(10), 2300. [Google Scholar] [CrossRef] [PubMed]
- Amiri, A.; Mahmoodi, M.; Mo, H.; Kiasat, A.; Ramezani, Z. Fabrication the Antimicrobial Sachet by Encapsulation of Peppermint Essential Oil in Active Packaging of Strawberry Fruit. Journal of Food Processing and Preservation 2022, 46(5), 17181. [Google Scholar] [CrossRef]
- An, P.; Yang, X.; Yu, J.; Qi, J.; Ren, X.; Kong, Q. α-terpineol and terpene-4-ol, the critical components of tea tree oil, exert antifungal activities in vitro and in vivo against Aspergillus niger in grapes by inducing morphous damage and metabolic changes of fungus. Food Control 2018, 98, 42–53. [Google Scholar] [CrossRef]
- Avonto, C.; Wang, M.; Chittiboyina, A.G.; Vukmanovic, S.; Khan, I.A. Chemical stability and in chemico reactivity of 24 fragrance ingredients of concern for skin sensitization risk assessment. Toxicol In Vitro 2018, 46, 237–245. [Google Scholar] [CrossRef]
- Bagg, J.; Jackson, M.S.; Sweeney, M.P.; Ramage, G.; Davies, A.N. Susceptibility to Melaleuca alternifolia (tea tree) oil of yeasts isolated from the mouths of patients with advanced cancer. Oral Oncology 2006, 42(5), 487–492. [Google Scholar] [CrossRef]
- Bassolé, I.H.N.; Juliani, H.R. Essential Oils in Combination and Their Antimicrobial Properties. Molecules 2012, 17(4), 3989–4006. [Google Scholar] [CrossRef]
- Becerril, R.; Nerin, C.; Silva, F. Encapsulation Systems for Antimicrobial Food Packaging Components: An Update. Molecules 2020, 25(5), 1134. [Google Scholar] [CrossRef]
- Bekhof, A.S.M.W.; Van Hunsel, F.P.A.M.; van de Koppel, S.; Woerdenbag, H.J. Safety assessment and adverse drug reaction reporting of tea tree oil (Melaleuca aetheroleum). Phytotherapy Research 2023, 37, 1309–1318. [Google Scholar] [CrossRef]
- Borotova, P.; Galovičová, L.; Vukovic, N.; Vukic, M.; Tvrda, E.; Kačániová, M. Chemical and Biological Characterization of Melaleuca alternifolia Essential Oil. Plants 2022, 11(4), 558. [Google Scholar] [CrossRef]
- Burt, S. Essential oils: their antibacterial properties and potential applications in foods--a review. Int J Food Microbiol 2004, 94(3), 223–53. [Google Scholar] [CrossRef] [PubMed]
- Carson, C.F.; Hammer, K.A.; Riley, T.V. Melaleuca alternifolia (Tea Tree) Oil: a Review of Antimicrobial and Other Medicinal Properties. Clin Microbiol Rev 2006, 19(1), 50–62. [Google Scholar] [CrossRef] [PubMed]
- Cen, C.; Wang, X.; Li, H.; Miao, S.; Chen, J.; Wang, Y. Nano-Emulsification Potentiates Tea Tree Oil Bioactivity: High-Stability Formulation for Dual Antimicrobial and Antioxidant Food Preservation. Foods 2025, 14(19), 3405. [Google Scholar] [CrossRef] [PubMed]
- Cháfer, M.; Sánchez-González, L.; González-Martínez, Ch.; Chiralt, A. Fungal Decay and Shelf Life of Oranges Coated With Chitosan and Bergamot, Thyme, and Tea Tree Essential Oils. Journal of Food Science 2012, 77, 8–e187. [Google Scholar] [CrossRef]
- Chauhan, K.; Rao, A. Clean-Label Alternatives for Food Preservation: An Emerging Trend. Heliyon 2024, 10(1), e35815. [Google Scholar] [CrossRef]
- Cui, H.; Bai, M.; Li, C.; Liu, R.; Lin, L. Fabrication of chitosan nanofibers containing tea tree oil liposomes against Salmonella spp. in chicken. LWT 2018, 96, 671–678. [Google Scholar] [CrossRef]
- De Groot, A.C.; Schmidt, E. Tea tree oil: contact allergy and chemical composition. Contact Dermatitis 2016, 75(3), 129–43. [Google Scholar] [CrossRef]
- De Oliveira, I.; Santos-Buelga, C.; Aquino, Y.; Barros, L.; Heleno, S.A. New frontiers in the exploration of phenolic compounds and other bioactives as natural preservatives. Food Bioscience 2025, 68, 106571. [Google Scholar] [CrossRef]
- Deng, X.; Wei, Y.; Jiang, S.; Ye, J.; Chen, Y.; Xu, F.; Chen, J.; Shao, X. Recent advances in the application of tea tree oil in the storage of fruit and vegetables. Postharvest Biology and Technology 2025, 219, 113260. [Google Scholar] [CrossRef]
- De Sá Silva, C.; De Figueiredo, H.M.; Stamford, T.L.M.; Da Silva, L.H.M. Inhibition of Listeria monocytogenes by Melaleuca alternifolia (tea tree) essential oil in ground beef. International Journal of Food Microbiology 2019, 293, 79–86. [Google Scholar] [CrossRef]
- Didehdar, M.; Chegini, Z.; Shariati, A. Eugenol: A novel therapeutic agent for the inhibition of Candida species infection. Front Pharmacol 2022, 13, 872127. [Google Scholar] [CrossRef]
- Di Nunzio, M.; Valli, V.; Tomás-Cobos, L.; Tomás-Chisbert, T.; Murgui-Bosch, L.; Danesi, F.; Bordoni, A. Is cytotoxicity a determinant of the different in vitro and in vivo effects of bioactives? BMC Complement Altern Med. 2017, 17, 453. [Google Scholar] [CrossRef] [PubMed]
- Dong, X.; Bond, A.E.; Yang, L. Essential oil-incorporated carbon nanotubes filters for bacterial removal and inactivation. PLoS One 2019, 14(12), e0227220. [Google Scholar] [CrossRef] [PubMed]
- Dziagwa-Becker, M.; Oleszek, M. Is the Biopesticide from Tea Tree Oil an Effective and Low-Risk Alternative to Chemical Pesticides? A Critical Review. Molecules 2024, 29(14), 3248. [Google Scholar] [CrossRef] [PubMed]
- Elgendy, E.A.; Ali, S.A.M.; Zineldeen, D.H. Effect of local application of tea tree (Melaleuca alternifolia) oil gel on long pentraxin level used as an adjunctive treatment of chronic periodontitis: A randomized controlled clinical study. J Indian Soc Periodontol 2013, 17(4), 444–448. [Google Scholar] [CrossRef]
- English, M.; Okagu, O.D.; Stephens, K.; Goertzen, A.; Udenigwe, C. Flavour encapsulation: A comparative analysis of relevant techniques, physiochemical characterisation, stability, and food applications. Front Nutr 2023, 10, 1019211. [Google Scholar] [CrossRef]
- Espina, L.; Monfort, S.; Alvarez, I.; García-Gonzalo, D.; Pagán, R. Combination of pulsed electric fields, mild heat and essential oils as an alternative to the ultrapasteurization of liquid whole egg. Int J Food Microbiol 2014, 189, 119–25. [Google Scholar] [CrossRef]
- European Chemicals Agency. Melaleuca alternifolia, ext. 2025. [Google Scholar]
- Gao, F.; Zhou, H.; Shen, Z.; Zhu, G.; Hao, L.; Chen, H.; Xu, H.; Zhou, X. Long-lasting anti-bacterial activity and bacteriostatic mechanism of tea tree oil adsorbed on the amino-functionalized mesoporous silica-coated by PAA. Colloids and Surfaces B: Biointerfaces 2020, 188, 110784. [Google Scholar] [CrossRef]
- Garozzo, A.; Timpanaro, R.; Stivala, A.; Bisignano, G.; Castro, A. Activity of Melaleuca alternifolia (tea tree) oil on Influenza virus A/PR/8: Study on the mechanism of action. Antiviral Research 2011, 89(1), 83–8. [Google Scholar] [CrossRef]
- Giordani, C.; Molinari, A.; Toccacieli, L.; Calcabrini, A.; Stringaro, A.; Chistolini, P.; Arancia, G.; Diociaiuti, M. Interaction of Tea Tree Oil with Model and Cellular Membranes. Journal of Medicinal Chemistry 2006, 49(15), 4581–8. [Google Scholar] [CrossRef] [PubMed]
- Goncalves, D.D.C.; Ribeiro, W.R.; Goncalves, D.C.; Dian, V.S.; Xavier, A.D.S.; De Oliveira, A.A.; Menini, L. Use of Melaleuca alternifolia essential oil as an efficient strategy to extend the shelf life of banana fruits. Biochemical Systematics and Ecology 2023, 108, 104641. [Google Scholar] [CrossRef]
- Goñi, M.G.; Tomadoni, B.; Moreira, M.R.; Roura, S.I. Application of tea tree and clove essential oil on late development stages of Butterhead lettuce: Impact on microbiological quality. LWT – Food Science and Technology 2013, 54(1), 107–113. [Google Scholar] [CrossRef]
- González, L.S.; Cháfer, M.; Hernández, M.; Chiralt, A.; Gonzalez-Martinez, C. Antimicrobial activity of polysaccharide films containing essential oils. Food Control 2011, 22(8), 1302–1310. [Google Scholar] [CrossRef]
- Haba, E.; Bouhdid, S.; Torrego-Solana, N.; Marques, A.M.; Espuny, M.J.; Garcia-Celma, M.J.; Manresa, A. Rhamnolipids as emulsifying agents for essential oil formulations: Antimicrobial effect against Candida albicans and methicillin-resistant Staphylococcus aureus. International Journal of Pharmaceutics 2014, 476(1 – 2), 134–141. [Google Scholar] [CrossRef]
- Hammer, K.A.; Carson, C.F.; Riley, T.V. Antifungal activity of the components of Melaleuca alternifolia (tea tree) oil. Journal of Applied Microbiology 2003, 95(4), 853–60. [Google Scholar] [CrossRef]
- Hammer, K.A.; Carson, C.F.; Riley, T.V.; Nielsen, J.B. A review of the toxicity of Melaleuca alternifolia (tea tree) oil. Food Chem Toxicol 2006, 44(5), 616–25. [Google Scholar] [CrossRef]
- Hausen, B.M.; Reichling, J.; Harkenthal, M. Degradation products of monoterpenes are the sensitizing agents in tea tree oil. Am J Contact Dermat 1999, 10(2), 68–77. [Google Scholar] [CrossRef]
- Homer, L.E.; Leach, D.; Lea, D.; Lee, L.S.; Henry, R.J.; Baverstock, P.R. Natural variation in the essential oil content of Melaleuca alternifolia Cheel (Myrtaceae). Biochemical Systematics and Ecology 2000, 28(4), 367–382. [Google Scholar] [CrossRef]
- Iacovelli, F.; Romeo, A.; Lattanzio, P.; Ammendola, S.; Battistoni, A.; La Frazia, S.; Vindigni, G.; Unida, V.; Biocca, S.; Gaziano, R.; Divizia, M.; Falconi, M. Deciphering the Broad Antimicrobial Activity of Melaleuca alternifolia Tea Tree Oil by Combining Experimental and Computational Investigations. Int J Mol Sci 2023, 24(15), 12432. [Google Scholar] [CrossRef]
- International Organization for Standardization. (2007). ISO 4730:2004, Oil of Melaleuca, terpinen-4-ol type (Tea Tree oil).
- Jackson-Davis, A.; White, S.; Kassama, L.S.; Coleman, S.; Shaw, A.; Mendonca, A.; Cooper, B.; Thomas-Popo, E.; Gordon, K.; London, L. A Review of Regulatory Standards and Advances in Essential Oils as Antimicrobials in Foods. Journal of Food Protection 2023, 86(2), 100025. [Google Scholar] [CrossRef]
- Kairey, L.; Agnew, T.; Bowles, E.J.; Barkla, B.J.; Wardle, J.; Lauche, R. Efficacy and safety of Melaleuca alternifolia (tea tree) oil for human health—A systematic review of randomized controlled trials. Front Pharmacol 2023, 14, 1116077. [Google Scholar] [CrossRef]
- Kamau, P.G.; Cruz-Romero, M.C.; Alzate, P.C.; Morris, M.A.; Kerry, J.P. Active packaging containing natural antimicrobials as a potential and innovative technology to extend shelf-life of fish products – A review. Food Packaging and Shelf Life 2025, 49, 101500. [Google Scholar] [CrossRef]
- Karnwal, A.; Kumar, G.; Singh, R.; Selvaraj, M.; Malik, T.; Al Tawaha, A.R.M. Natural biopolymers in edible coatings: Applications in food preservation. Food Chemistry: X 2025, 25, 102171. [Google Scholar] [CrossRef] [PubMed]
- Kong, P.; Abe, J.P.; Masuo, S.; Enomae, T. Preparation and characterization of tea tree oil-β-cyclodextrin microcapsules with super-high encapsulation efficiency. Journal of Bioresources and Bioproducts 2023, 8(3), 224–234. [Google Scholar] [CrossRef]
- Kulik, E.; Lenkeit, K.; Meyer, J. Antimicrobial effects of tea tree oil (Melaleuca alternifolia) on oral microorganisms. Schweiz Monatsschr Zahnmed 2000, 110(11), 125–30. [Google Scholar] [PubMed]
- Kwieciński, J.; Eick, S.; Wójcik, K. Effects of tea tree (Melaleuca alternifolia) oil on Staphylococcus aureus in biofilms and stationary growth phase. International Journal of Antimicrobial Agents 2009, 33(4), 343–347. [Google Scholar] [CrossRef]
- Lai, H.; Chen, S.; Su, X.; Huang, X.; Zheng, Q.; Yang, M.; Shen, B.; Yue, P. Sponge-liked Silica Nanoporous Particles for Sustaining Release and Long-Term Antibacterial Activity of Natural Essential Oil. Molecules 2023, 28(2), 594. [Google Scholar] [CrossRef]
- Lam, N.S.; Long, X.X.; Su, X.; Lu, F. Melaleuca alternifolia (tea tree) oil and its monoterpene constituents in treating protozoan and helminthic infections. Biomedicine & Pharmacotherapy 2020, 130(12), 110624. [Google Scholar] [CrossRef]
- Larson, D.; Jacob, S.E. Tea tree oil. Dermatitis 2012, 23(1), 48–9. [Google Scholar] [CrossRef]
- Latorre, R.; Valerii, M.C.; Benati, M.; Lewis, R.E.; Spigarelli, R.; Bernacchi, A.; Lippi, G.; Spisni, E.; Gaibani, P. Lights and Shadows of Essential Oil-Derived Compounds: Antimicrobial and Anti-Inflammatory Properties of Eugenol, Thymol, Cinnamaldehyde, and Carvacrol. Curr. Issues Mol. Biol. 2025, 47(11), 915. [Google Scholar] [CrossRef] [PubMed]
- Lee, L.S.; Brooks, L.O.; Homer, L.E.; Rossetto, M.; Henry, R.J.; Baverstock, P.R. Geographic variation in the essential oils and morphology of natural populations of Melaleuca alternifolia (Myrtaceae). Biochemcial Systematics and Ecology 2002, 30(4), 343–360. [Google Scholar] [CrossRef]
- Leila, N.; Siavash, M.; Maryam, G.G.; Mohammad, M.S. Comparison of the effect of nanoemulsion and emulsion coating containing of Oliveria decumbens essential oil on bacteria inoculated into rainbow trout fillet. Electronic Journal of Food Processing and Preservation 2024, 16(1), 33–48. [Google Scholar]
- Li, W.; Zhang, J.; Chen, X.; Zhou, X.; Zhou, J.; Sun, H.; Wang, S.; Liu, Y. Organic nanoparticles incorporated starch/carboxymethylcellulose multifunctional coating film for efficient preservation of perishable products. Int J Biol Macromol 2024, 275(1), 133357. [Google Scholar] [CrossRef]
- Liao, Z.; Yeoh, Y.K.; Zhu, X.; Parumasivam, T.; Tan, T.C. Enhancing shelf life of refrigerated salmon: Synergistic roles of glutaric and azelaic acid with conventional preservatives. LWT 2025, 228, 118090. [Google Scholar] [CrossRef]
- Lin, G.; Chen, H.; Zhou, H.; Zhou, X.; Xu, H. Preparation of Tea Tree Oil/Poly(styrene-butyl methacrylate) Microspheres with Sustained Release and Anti-Bacterial Properties. Materials (Basel) 2018, 11(5), 710. [Google Scholar] [CrossRef]
- Liu, Y.; Ding, Y.; Wang, C.; Luo, J.; Yao, H.; Zhang, H.; Xu, L.; Niu, J. Study examines the use of tea tree oil in fruit and vegetable preservation. Food Chemistry 2025, 465(1), 142007. [Google Scholar] [CrossRef]
- Liu, Z.; Meng, R.; Zhao, X.; Shi, C.; Zhang, X.; Zhang, Y.; Guo, N. Inhibition effect of tea tree oil on Listeria monocytogenes growth and exotoxin proteins listeriolysin O and p60 secretion. Lett Appl Microbiol 2016, 63(6), 450–457. [Google Scholar] [CrossRef]
- Manzanelli, F.A.; Ravetti, S.; Brignone, S.; Garro, A.G.; Martinez, S.; Vallejo, M.G.; Palma, S.D. Enhancing the Functional Properties of Tea Tree Oil: In Vitro Antimicrobial Activity and Microencapsulation Strategy. Pharmaceutics 2023, 15(10), 2489. [Google Scholar] [CrossRef]
- Martins, M.A.R.; Silva, L.P.; Ferreira, O.; Schröder, B.; Coutinho, J.A.P.; Pinho, S.P. Terpenes solubility in water and their environmental distribution. Journal of Molecular Liquids 2017, 241, 996–1002. [Google Scholar] [CrossRef]
- Masyita, A.; Sari, R.M.; Astuti, A.D.; Yasir, B.; Rumata, N.R.; Emran, T.B.; Nainu, F.; Simal-Gandara, J. Terpenes and terpenoids as main bioactive compounds of essential oils, their roles in human health and potential application as natural food preservatives. Food Chem X 2022, 13, 100217. [Google Scholar] [CrossRef] [PubMed]
- Matussek, F.; Pavinatto, A.; Knospe, P.; Beuermann, S.; Sanfelice, R.C. Controlled Release of Tea Tree Oil from a Chitosan Matrix Containing Gold Nanoparticles. Polymers 2022, 14(18), 3808. [Google Scholar] [CrossRef] [PubMed]
- Mazzarrino, G.; Paparella, A.; Chaves-López, C.; Faberi, A.; Sergi, M.; Sigismondi, C.; Compagnone, D.; Serio, A. Salmonella enterica and Listeria monocytogenes inactivation dynamics after treatment with selected essential oils. Food Control 2015, 50, 794–803. [Google Scholar] [CrossRef]
- Moirangthem, S.; Patra, G.; Biswas, S.; Das, A.; Nath, S.; Verma, A.K.; Chatterjee, N.; Bandyopadhyay, S.; Nanda, P.K.; Sharma, G.; Das, A.K. Effect of Nutmeg (Myristica fragrans) and Tea Tree (Melaleuca alternifolia) Essential Oils on the Oxidative and Microbial Stability of Chicken Fillets During Refrigerated Storage. Foods 2024, 13(24), 4139. [Google Scholar] [CrossRef]
- Mondello, F.; Fontana, S.; Scaturro, M.; Girolamo, A.; Colone, M.; Stringaro, A.; Di Vito, M.; Ricci, M.L. Terpinen-4-ol, the Main Bioactive Component of Tea Tree Oil, as an Innovative Antimicrobial Agent against Legionella pneumophila. Pathogens 2022, 11(6), 682. [Google Scholar] [CrossRef]
- Mouatcho, J.; Tzortzakis, N.; Soundy, P.; Sivakumar, D. Bio-sanitation treatment using essential oils against E. coli O157:H7 on fresh lettuce. New Zealand Journal of Crop and Horticultural Science 2017, 45(3), 165–174. [Google Scholar] [CrossRef]
- Nguyen, H.L.; Moreira, R.G.; Castell-Perez, M.E. Antibacterial effectiveness of zeolitic imidazolate framework-8 (ZIF-8) nanoparticle solutions and its derivatives against Salmonella Typhimurium ATCC13311 on loose-leaf lettuce. Journal of Food Safety 2025, 45, 4. [Google Scholar] [CrossRef]
- Nguyen, L.; DeVico, B.; Mannan, M.; Chang, M.; Santacruz, C.R.; Siragusa, C.; Everhart, S.; Fazen, C.H. Tea Tree Essential Oil Kills Escherichia coli and Staphylococcus epidermidis Persisters. Biomolecules 2023, 13(9), 1404. [Google Scholar] [CrossRef]
- Nie, J.; Huang, Z.; Wen, L.; Li, H.; Xie, Q.; Wang, H.; Lai, Z.; Lin, C.; Jing, C. Association between exposure to terpene compounds and risk of metabolic syndrome: exploring the potential mediating role of inflammatory response. Front Endocrinol 2025, 16, 1551784. [Google Scholar] [CrossRef]
- PubChem. Essential oils, Melaleuca alternifolia. 2025. [Google Scholar]
- Puvača, N.; Milenkovic, J.; Coghill, T.G.; Bursic, V.; Petrovic, A.; Tanaskovic, S.; Pelic, M.; Pelic, D.L.; Miljkovic, T. Antimicrobial Activity of Selected Essential Oils against Selected Pathogenic Bacteria: In Vitro Study. Antibiotics (Basel) 2021, 10(5), 546. [Google Scholar] [CrossRef] [PubMed]
- Rahman, N.; Hashem, S.; Akther, S.; Jothi, J.S. Impact of various extraction methods on fatty acid profile, physicochemical properties, and nutritional quality index of Pangus fish oil. Food Sci Nutr 2023, 11(8), 4688–4699. [Google Scholar] [CrossRef] [PubMed]
- Ripari, F.; Cera, A.; Freda, M.; Zumbo, G.; Zara, F.; Vozza, I. Tea Tree Oil versus Chlorhexidine Mouthwash in Treatment of Gingivitis: A Pilot Randomized, Double Blinded Clinical Trial. Eur J Dent 2020, 14, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Rogawansamy, S.; Gaskin, S.; Taylor, M.; Pisaniello, D. An Evaluation of Antifungal Agents for the Treatment of Fungal Contamination in Indoor Air Environments. Int J Environ Res Public Health 2015, 12(6), 6319–6332. [Google Scholar] [CrossRef]
- Rytwo, G.; Zakai, R.; Wicklein, B. The Use of ATR-FTIR Spectroscopy for Quantification of Adsorbed Compounds. Journal of Spectroscopy 2015, 2015(1), 727595. [Google Scholar] [CrossRef]
- Sadekuzzaman, M.; Mizan, M.F.R.; Kim, H.S.; Yang, S.; Ha, S.D. Activity of thyme and tea tree essential oils against selected foodborne pathogens in biofilms on abiotic surfaces. LWT 2018, 89, 134–139. [Google Scholar] [CrossRef]
- Sadgrove, N.J.; Padilla-Gonzalez, G.F.; Phumthum, M. Fundamental Chemistry of Essential Oils and Volatile Organic Compounds, Methods of Analysis and Authentication. Plants (Basel) 2022, 11(6), 789. [Google Scholar] [CrossRef]
- Sánchez-González, L.; Vargas, M.; González-Martínez, C.; Chiralt, A.; Cháfer, M. Characterization of edible films based on hydroxypropylmethylcellulose and tea tree essential oil. Food Hydrocolloids 2009, 23(8), 2102–2109. [Google Scholar] [CrossRef]
- Sathiyaseelan, A.; Saravanakumar, K.; Mariadoss, A.V.A.; Chelliah, R.; Xiaowen, H.; Oh, D.H.; Wang, M.H. Chitosan-tea tree oil nanoemulsion and calcium chloride tailored edible coating increase the shelf life of fresh cut red bell pepper. Progress in Organic Coatings 2020, 151, 106010. [Google Scholar] [CrossRef]
- Schneider, G.; Steinbach, A.; Putics, A.; Solti-Hodovan, A.; Palkovics, T. Potential of Essential Oils in the Control of Listeria monocytogenes. Microorganisms Doi. 2023, 11(6), 1364. [Google Scholar] [CrossRef]
- Schnitzler, P.; Schön, K.; Reichling, J. Antiviral activity of Australian tea tree oil and eucalyptus oil against herpes simplex virus in cell culture. Pharmazie 2001, 56(4), 343–7. [Google Scholar]
- Scientific Committee on Consumer Safety. Scientific Opinion on Tea Tree Oil (CAS/EC No. 68647-73-4 /285-377-1). 2025. [Google Scholar]
- Sheikh, A.R.; Wu-Chen, R.A.; Matloob, A.; Mahmood, M.H.; Javed, M. Nanoencapsulation of volatile plant essential oils: a paradigm shift in food industry practices. Food Innovation and Advances 2024, 3(3), 305–319. [Google Scholar] [CrossRef]
- Singhania, A.; Sathe, S.; Ranka, R.; Godbole, S. Individual and Synergistic Effects of Tea Tree Oil and Neem Extract on Candida albicans Adhesion to Denture Soft Liner. Cureus 2022, 14(8), e27869. [Google Scholar] [CrossRef]
- Stoleru, E.; Brebu, M. Stabilization Techniques of Essential Oils by Incorporation into Biodegradable Polymeric Materials for Food Packaging. Molecules 2021, 26(20), 6307. [Google Scholar] [CrossRef] [PubMed]
- Sui, J.; Wang, N.; Wang, J.; Huang, X.; Wang, T.; Zhou, L.; Hao, H. Strategies for chiral separation: from racemate to enantiomer. Chem. Sci. 2023, 14, 11955–12003. [Google Scholar] [CrossRef] [PubMed]
- Szente, L.; Fenyvesi, E. Cyclodextrin-Enabled Polymer Composites for Packaging. Molecules 2018, 23(7), 1556. [Google Scholar] [CrossRef]
- Tian, Y.; Zhou, L.; Liu, J.; Yu, K.; Yu, W.; Jiang, H.; Zhong, J.; Zou, L.; Liu, W. Effect of sustained-release tea tree essential oil solid preservative on fresh-cut pineapple storage quality in modified atmospheres packaging. Food Chem 2023, 417, 135898. [Google Scholar] [CrossRef] [PubMed]
- Tighe, S.; Gao, Y.Y.; Tseng, S.C.G. Terpinen-4-ol is the Most Active Ingredient of Tea Tree Oil to Kill Demodex Mites. Translational Vision Science & Technology 2013, 2, 2. [Google Scholar] [CrossRef]
- Tsumoto, K.; Ejima, D.; Kumagai, I.; Arakawa, T. Practical considerations in refolding proteins from inclusion bodies. Protein Expression and Purification 2003, 28(1), 1–8. [Google Scholar] [CrossRef]
- Tuan, D.A.; Uyen, P.V.N.; Khuon, N.V.; Binh, L.A.; Masak, J. Innovative antifungal strategies: enhanced biofilm inhibition of Candida albicans by a modified tea tree oil formulation. Front Microbiol 2025, 15, 1518598. [Google Scholar] [CrossRef] [PubMed]
- Ul-Haq, I.; Khan, S.; Sohail, M.; Iqbal, M.J.; Awan, K.A.; Nayik, G.A. Chapter 20 - Tea tree essential oil. Essential Oils, Academic Press. 2023, pp. 479–500, https://www.ventos.com/index.php/en/producto/1349/TEA+TREE+OIL/223.. [CrossRef]
- Ventos. Tea tree oil; 2025. [Google Scholar]
- Wang, Y.; Wang, Y.; Wang, K.; Cheng, M.; Zhao, P.; Lu, J.; Xi, X.; Wang, X.; Han, X.; Wang, J. Evaluation of the postharvest quality of Agaricus bisporus packed using PVA/SG-based active packaging film containing tea tree essential oil. Food Measure 2024, 18, 4820–4831. [Google Scholar] [CrossRef]
- Wei, Y.; Wei, Y.; Xu, F.; Shao, X. The combined effects of tea tree oil and hot air treatment on the quality and sensory characteristics and decay of strawberry. Postharvest Biology and Technology 2018, 136, 139–144. [Google Scholar] [CrossRef]
- Westlake, J.R.; Tran, M.W.; Jiang, Y.; Zhang, X.; Burrows, A.D.; Xie, M. Biodegradable Active Packaging with Controlled Release: Principles, Progress, and Prospects. ACS Food Sci. Technol. 2022, 2(8), 1166–1183. [Google Scholar] [CrossRef]
- Whiley, H.; Gaskin, S.; Schroder, T.; Ross, K. Antifungal properties of essential oils for improvement of indoor air quality: a review. Reviews on Environmental Health 2017, 33(1), 63–76. [Google Scholar] [CrossRef]
- Wróblewska, M.; Szymańska, E.; Winnicka, K. The Influence of Tea Tree Oil on Antifungal Activity and Pharmaceutical Characteristics of Pluronic® F-127 Gel Formulations with Ketoconazole. Int J Mol Sci 2021, 22(21), 11326. [Google Scholar] [CrossRef]
- Yasin, M.; Younis, A.; Javed, T.; Akram, A.; Shabbir, R.; Ali, M.M.; Tahir, A.; El-Ballat, E.M.; Sheteiwy, M.; Sammour, R.H.; Hano, C.; Alhumaydhi, F.A.; El-Esawi, M.A. River Tea Tree Oil: Composition, Antimicrobial and Antioxidant Activities, and Potential Applications in Agriculture. Plants 2021, 10(10), 2105. [Google Scholar] [CrossRef]
- Zhao, M.L. Biofilms: Unveiling the Complexities of Microbial Communities in Diverse Environments. J Basic Clin Pharma 2024, 15(4), 371–372. [Google Scholar]
- Zhu, L.; Machmudah, S.; Wahyudiono; Kanda, H.; Goto, M. Reduced-Pressure Process for Fabricating Tea Tree Oil—Polyvinylpyrrolidone Electrospun Fibers. Polymers (Basel) 2022, 14(4), 743. [Google Scholar] [CrossRef]
- Zhu, Z.; Hu, J.; Zhong, Z. Preparation and characterization of long-term antibacterial and pH-responsive Polylactic acid/Octenyl succinic anhydride-chitosan @ tea tree oil microcapsules. International Journal of Biological Macromolecules 2022, 220, 1318–1328. [Google Scholar] [CrossRef]

| Food safety challenge | Applications of TTO | Role of TTO |
|---|---|---|
| Increasing antimicrobial resistance | Need for non-antibiotic interventions | Broad-spectrum activity with Gram+/Gram− and fungi |
| Consumer push for clean-label preservatives | Replacement of synthetic chemicals | Natural GRAS essential oil |
| Food-contact surface contamination | Biofilm persistence in industry | Strong antibiofilm disruption |
| Limited efficiency of washing/sanitizing | Chlorine/treatments insufficient | Vapor-phase and contact inactivation |
| Property | Typical value | Implication for food systems |
|---|---|---|
| Density | 0.885–0.906 g/mL | Floats on water and requires emulsification |
| Aqueous solubility | ~0.03% (300 mg/L) | Poor dispersion without formulation |
| Vapor pressure | ~2.1 kPa at 25 °C | High volatility → suitable for active packaging |
| Flash point | 56–60 °C | Must be handled carefully in processing |
| Dominant component | Terpinen-4-ol (35–48%) | Major contributor to antimicrobial action |
| Component | Relative abundance | Functional group | Contribution |
|---|---|---|---|
| Terpinen-4-ol | Highest | Terpenoid alcohol | Antibacterial, antibiofilm |
| γ-Terpinene | Moderate–high | Monoterpene | Oxidation-prone; aromatic changes |
| α-Terpinene | Moderate | Monoterpene | Supports membrane fluidization |
| 1,8-Cineole | 1–15% | Oxide | Aroma intensity, low antimicrobial |
| Microorganism | MIC (% v/v) | MBC (% v/v) | Sensitivity |
|---|---|---|---|
| S. aureus | 0.25–0.50 | 0.50–1.0 | High |
| L. monocytogenes | 0.05–0.25 | 0.25–0.50 | Very high |
| E. coli O157:H7 | 0.20–0.40 | 0.40–1.0 | Moderate |
| Salmonella spp. | 0.20–0.80 | 0.50–1.5 | Moderate |
| Candida albicans | 0.06–0.50 | 0.25–1.0 | High |
| Food Product & Microbial Issue | TTO Application & Formulation | Outcomes (Microbial Control & Shelf-life) | Sensory & Feasibility |
|---|---|---|---|
| Raw chicken fillets – general spoilage (bacteria, oxidation) | 1% TTO in marinade/dipping solution (lab-scale trial) | Decreased total viable counts; slowed spoilage, +7 days refrigerated shelf-life vs. control. Also decreased lipid oxidation (TBARS) over 9 days. | Maintained color and odor better than control (fewer off odors). Concluded as effective natural preservative for meat. |
| Raw chicken meat – Salmonella contamination | Chitosan nanofiber mat with TTO-loaded liposomes (active packaging) | ~5 log reduction of Salmonella on chicken within 4 days at 12–25 °C; prevented microbial recontamination, extending safety and shelf-life. | Minimal flavor impact: TTO nanofiber caused no noticeable sensory change in chicken. Demonstrated practicability for pathogen control. |
| Fresh lettuce (Butterhead) – field microflora & coliforms | Preharvest spray with TTO emulsion (single or repeated applications late in growth) | Decreased native mesophilic bacteria and coliforms at harvest and after storage. After 5 days @5 °C, treated lettuce had ~2 log10 lower total counts vs. untreated. | No significant differences in sensory quality (appearance, taste) vs. control after treatment. TTO did not adversely affect lettuce flavor. |
| Soft cheese (Feta, fresh Mozzarella) – Listeria, E. coli risk | Direct EO addition to cheese or brine (0.5–1% needed for activity) | High TTO concentrations in vitro inhibit L. monocytogenes and E. coli; however, efficacy drops in high-fat cheese. Thyme or clove EO often outperforms TTO against Listeria. | Sensory hurdle: 1% TTO imparted strong off flavors in Feta; panelists “disliked” TTO aroma in Fior di Latte cheese. Thus, TTO’s use in cheese is limited by flavor at effective doses. |
| Strawberries – postharvest spoilage (fungi, quality loss) | β-cyclodextrin/nano-clay microcapsules releasing TTO in package | TTO vapor slowed decay: treated berries stayed mold-free and firm ~3–6 days longer than control at 4 °C. Optimal dose (5 g microcapsules per 1.2 L) ⇒ least decay, lower weight loss, delayed ripening indices. | Maintained fruit appearance and nutrients better during storage. Controlled-release microcapsules prevented overpowering odor; berries’ aroma remained acceptable (no TTO off-taste noted). |
| Banana – anthracnose (fungal rot by Colletotrichum) | Edible coating/film: bilayer sodium alginate film with TTO nanoemulsion + TiO2 nanoparticles | Markedly suppressed anthracnose lesions. 3 µg/mL TTO in coating reduced rot severity and extended banana shelf-life; treated fruit had significantly less decay over 12–16 days vs. controls. | No significant sensory detriment reported. The alginate–TiO2 matrix slowed TTO release and blocked UV light, preserving fruit quality (firmness, color). Coating is food-grade and meets packaging safety norms. |
| Fresh salmon fillets – spoilage bacteria & oxidation | Electrospun chitosan nanofiber wrapped with encapsulated TTO (coating pad in package) | Lowered microbial loads (including Listeria, E. coli, S. aureus in tests) and slowed spoilage in cold storage. One study showed such EO nanofiber mats added ~6–7 days of shelf-life to fresh fish vs. normal ice storage. | TTO nanofiber inhibited fishy odor development; treated fillets maintained acceptable sensory quality longer. Active fiber is biodegradable (chitosan) and poses no direct residue on fish flesh. |
| Bread (sliced) – mold spoilage (Penicillium spp.) | Vapor-phase TTO in package headspace (experimental set-up) | Limited efficacy: TTO vapor only weakly inhibited P. citrinum and P. crustosum on bread; no meaningful delay of mold growth. (P. expansum even grew faster with TTO present). | Bread absorbed some TTO aroma, but doses high enough to suppress molds would likely cause off-flavors. TTO vapor alone is not effective for bread preservation. Other EOs (e.g., lemongrass, clove) show stronger antifungal effects in bakery products. |
| Organism | % Biofilm reduction | Concentration used | TTO effectivness |
|---|---|---|---|
| Staphylococcus aureus | 50–90% | 0.25–1% | Strong membrane disruption |
| E. coli | 20–60% | 0.5–1% | Strain-dependent |
| Pseudomonas aeruginosa | 10–40% | 1–2% | Highly resistant strain |
| Candida albicans | 40–80% | 0.5–1% | Matrix penetration effective |
| Food category | Application method | Effectiveness | Notes |
|---|---|---|---|
| Meat/poultry | Marinades, active films | +5–7 days shelf life | Flavor masking required |
| Seafood | Nano-fiber wrap, edible coatings | Strong suppression of spoilage bacteria | Minimal sensory change |
| Fresh produce | Washes, vapor-phase, coatings | 1–3 log reduction in microbes | Excellent antifungal |
| Dairy | Edible films, surface treatment | Limited due to flavor | Packaging preferable |
| Beverages | TTO nanoemulsions | Pathogen control | Sensory challenge |
| Advantages | Limitations |
|---|---|
| Broad antimicrobial spectrum | Strong odor, flavor |
| Natural, clean-label | Limited GRAS approval depending on country |
| Works in vapor and liquid phases | Highly volatile, oxidizes quickly |
| Compatible with nanocarriers | Difficult to disperse in water |
| Innovation | Research need | Expected benefit |
|---|---|---|
| Smart active packaging | pH/humidity-triggered release | Preventing spoilage events |
| Synergy blends | Nisin, carvacrol | Lower dose + better taste |
| Biopolymer–TTO composites | Stability optimization | GRAS-compliant packaging |
| Microfluidic nanoencapsulation | Narrow droplet size | Better antimicrobial delivery |
| Delivery system | Release profile | Stability | Sensory impact | Application |
|---|---|---|---|---|
| Nanoemulsion | Fast release | Moderate | Higher aroma | Beverages, washes |
| Microcapsules | Slow release | High | Low aroma | Fruits, fresh-cut produce |
| Edible films | Surface-controlled | High | Minimal | Produce, cheese, seafood |
| Active packaging | Vapor-release | High | Very low | Meat, produce |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




