Submitted:
14 February 2026
Posted:
26 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
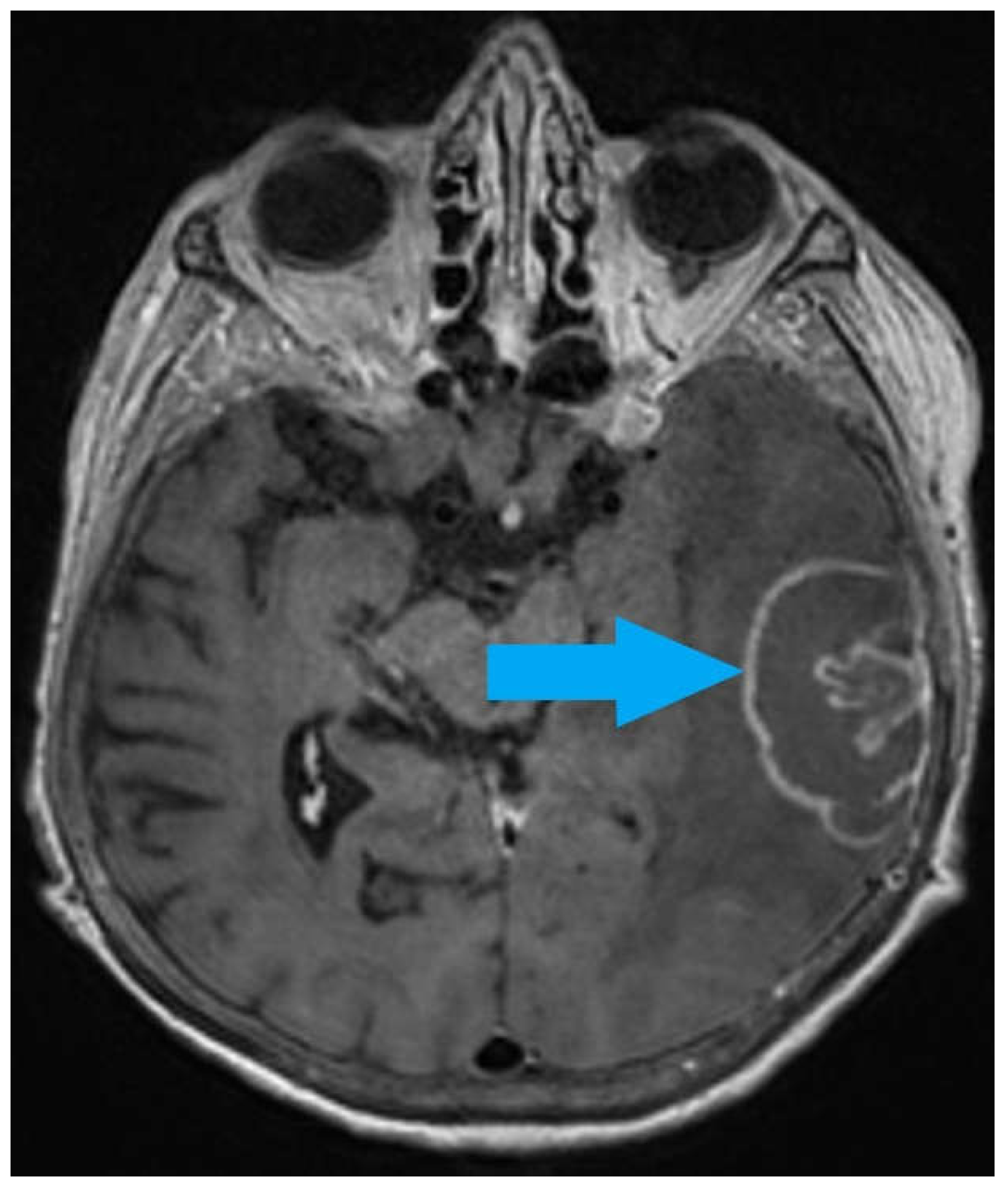
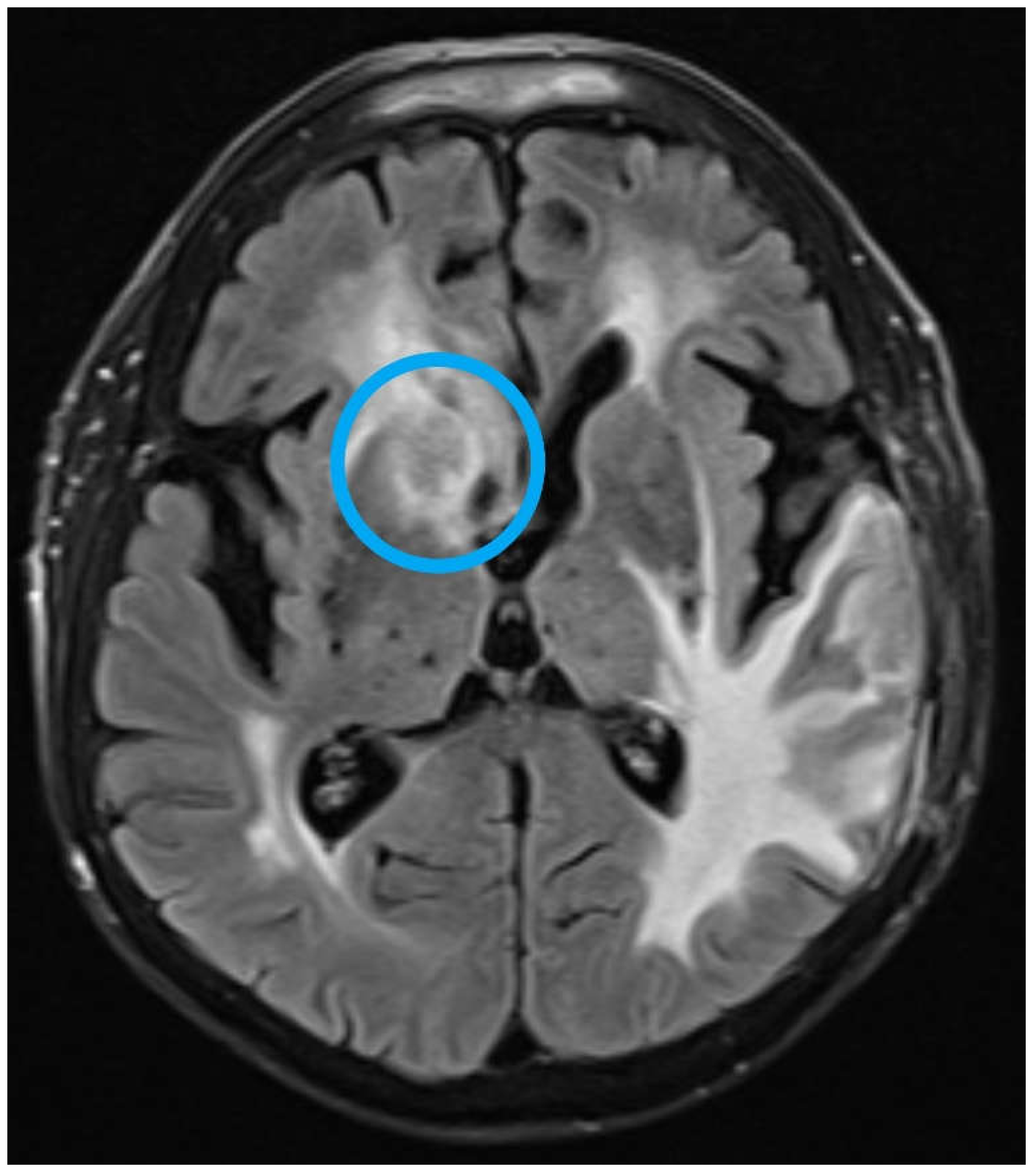
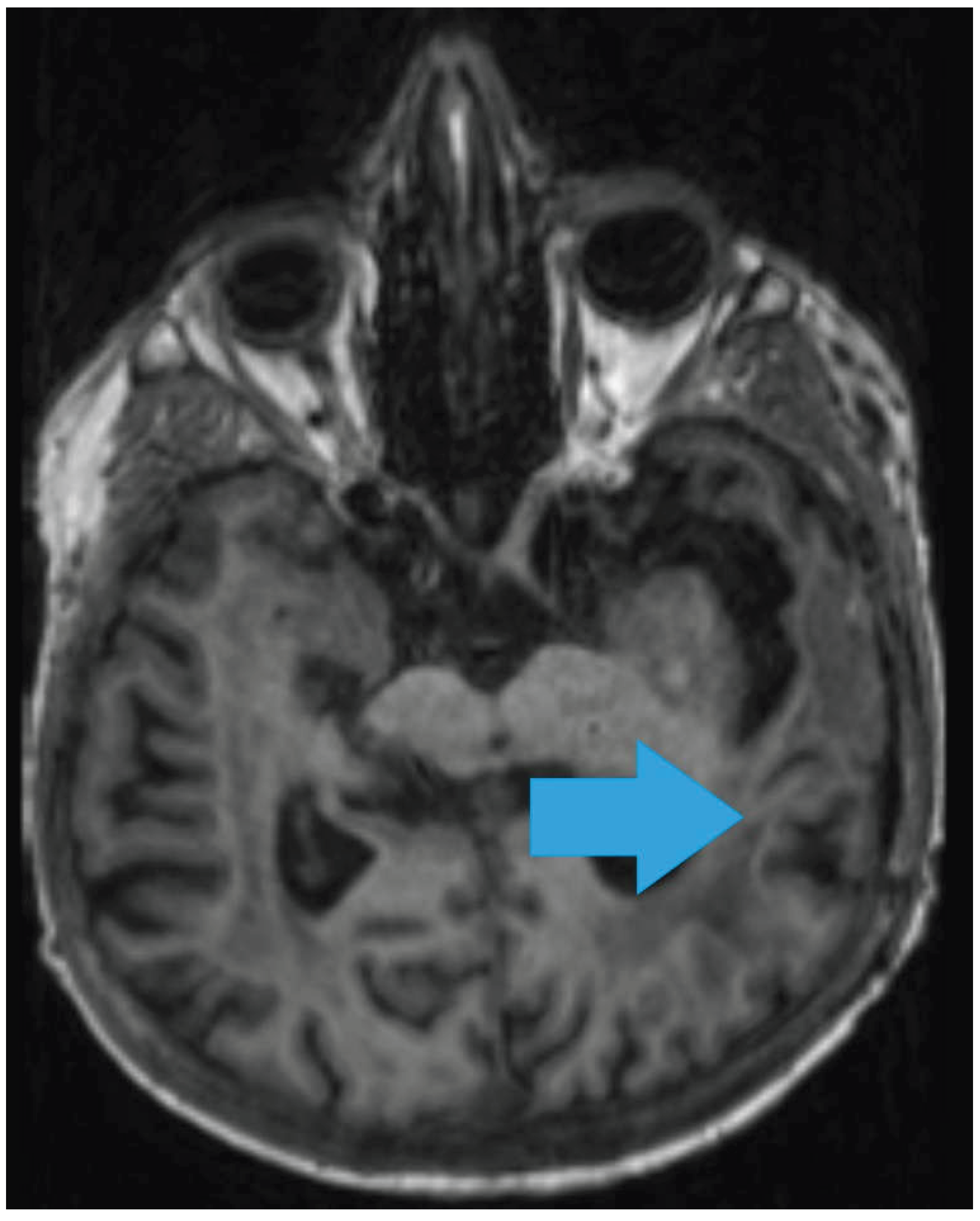
2. Case Report
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sawinski, D.; Blumberg, E.A. Infection in Renal Transplant Recipients. In Chronic Kidney Disease, Dialysis, and Transplantation; Gilbert, S.J., Weiner, D.E., Eds.; Elsevier: Philadelphia, PA, USA, 2019; pp. 621–638.e6. [Google Scholar]
- Nambiar, P.; Silibovsky, R.; Belden, K.A. Infection in Kidney Transplantation. In Contemporary Kidney Transplantation; Gabardi, S., Ed.; Springer: Cham, Switzerland, 2018; pp. 307–327. [Google Scholar]
- Yadav, P.; Khalil, S.; Mirdha, B.R. Molecular appraisal of intestinal parasitic infection in transplant recipients. Indian J. Med. Res. 2016, 144, 258–263. [Google Scholar] [CrossRef]
- Valar, C.; Keitel, E.; Dal Prá, R.L.; Gnatta, D.; Santos, A.F.; Bianco, P.D.; Sukiennik, T.C.; Pegas, K.L.; Bittar, A.E.; Oliveira, K.T.; Garcia, V.D. Parasitic infection in renal transplant recipients. Transplant Proc. 2007, 39, 460–462. [Google Scholar] [CrossRef] [PubMed]
- Smith, N.C.; Goulart, C.; Hayward, J.A.; Kupz, A.; Miller, C.M.; van Dooren, G.G. Control of human toxoplasmosis. Int. J. Parasitol. 2021, 51, 95–121. [Google Scholar] [CrossRef] [PubMed]
- Robert-Gangneux, F.; Dardé, M.L. Epidemiology of and diagnostic strategies for toxoplasmosis. Clin. Microbiol. Rev. 2012, 25, 264–296. [Google Scholar] [CrossRef] [PubMed]
- Elsheikha, H.M.; Khan, N.A. Protozoa traversal of the blood-brain barrier to invade the central nervous system. FEMS Microbiol. Rev. 2010, 34, 532–553. [Google Scholar] [CrossRef]
- Martino, R.; Bretagne, S. Toxoplasmosis in hematopoietic stem cell transplant recipients. Clin. Infect. Dis. 2000, 31, 1188–1195. [Google Scholar] [CrossRef]
- Elsheikha, H.M.; Marra, C.M.; Zhu, X.Q. Epidemiology, Pathophysiology, Diagnosis, and Management of Cerebral Toxoplasmosis. Clin. Microbiol. Rev. 2021, 34, e00115-19. [Google Scholar] [CrossRef]
- Human organ transplantation in Europe; an overview. Report from the General Health and Consumer Protection Public Health and Risk Assessment Directorate; European Commission: Brussels, Belgium, 2003; p. 120. Available online: http://ec.europa.eu/health/ph_threats/human_substance/documents/organ_survey.pdf (accessed on 25 January 2026).
- Luft, B.J.; Remington, J.S. Toxoplasmic encephalitis in AIDS. Clin. Infect. Dis. 1992, 15, 211–222. [Google Scholar] [CrossRef]
- Porter, S.B.; Sande, M.A. Toxoplasmosis of the central nervous system in the acquired immunodeficiency syndrome. N. Engl. J. Med. 1992, 327, 1643–1648. [Google Scholar] [CrossRef]
- Montoya, J.G.; Liesenfeld, O. Toxoplasmosis. Lancet 2004, 363, 1965–1976. [Google Scholar] [CrossRef]
- Derouin, F.; Pelloux, H.; et al. Toxoplasmosis in organ transplant recipients. Clin. Infect. Dis. 1997, 24, 625–634. [Google Scholar]
- Martina, M.N.; Cervera, C.; et al. Toxoplasmosis after solid organ transplantation. Transpl. Int. 2010, 23, 845–851. [Google Scholar]
- Robert-Gangneux, F.; Belaz, S. Toxoplasmosis in transplant recipients, Europe, 2010–2014. Euro Surveill. 2015, 20, 210–218. [Google Scholar] [CrossRef] [PubMed]
- Luft, B.J.; Hafner, R.; et al. Toxoplasmic encephalitis in patients with the acquired immunodeficiency syndrome. N. Engl. J. Med. 1993, 329, 995–1000. [Google Scholar] [CrossRef]
- Maschke, M.; Kastrup, O.; et al. Opportunistic infections of the central nervous system. Lancet Neurol. 2004, 3, 485–498. [Google Scholar]
- Post, M.J.; Chan, J.C.; et al. Central nervous system toxoplasmosis in the era of highly active antiretroviral therapy. AJNR Am. J. Neuroradiol. 2001, 22, 1349–1357. [Google Scholar]
- Kotton, C.N.; Fishman, J.A. Infectious complications of transplantation. Am. J. Transplant. 2013, 13 (Suppl. 4), 280–303. [Google Scholar]
- Luft, B.J.; Chua, A.; et al. Treatment of toxoplasmic encephalitis with pyrimethamine and sulfadiazine. N. Engl. J. Med. 1993, 329, 995–1000. [Google Scholar] [CrossRef]
- Hughes, W.T.; Leoung, G.S.; et al. Comparison of pyrimethamine–sulfadiazine and trimethoprim–sulfamethoxazole for treatment of toxoplasmic encephalitis. N. Engl. J. Med. 1990, 323, 776–782. [Google Scholar]
- Derouin, F.; Pelloux, H.; et al. Prevention of toxoplasmosis in transplant recipients. Clin. Infect. Dis. 2008, 46, 167–175. [Google Scholar]
- Martínez, J.; Torre-Cisneros, J.; et al. Toxoplasmosis after kidney transplantation: clinical features and outcomes. Transpl. Infect. Dis. 2011, 13, 1–7. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



